12
Vasodilators & the Treatment of Angina Pectoris
CASE STUDY
A 52-year-old man presents with a history of recent onset chest discomfort when jogging or swimming vigorously. The pain is substernal and radiates to his jaw but disappears after 10–15 minutes of rest. He has a history of hyperlipidemia (total cholesterol level of 245 mg/dL and low-density lipoprotein [LDL] of 160 mg/dL one year ago) and admits that he has not been following the recommended diet. His father survived a “heart attack” at age 55 and an uncle died of some cardiac disease at age 60. On physical examination, the patient’s blood pressure is 145/90 mm Hg and his heart rate is 80 bpm. There are no other significant physical findings and an electrocardiogram is normal except for slight left ventricular hypertrophy. Assuming that a diagnosis of stable effort angina is correct, what medical treatment should be implemented?
Ischemic heart disease is one of the most common cardiovascular diseases in developed countries, and angina pectoris is the most common condition involving tissue ischemia in which vasodilator drugs are used. The name angina pectoris denotes chest pain caused by accumulation of metabolites resulting from myocardial ischemia. The organic nitrates, eg, nitroglycerin, are the mainstay of therapy for the immediate relief of angina. Another group of vasodilators, the calcium channel blockers, is also important, especially for prophylaxis, and β blockers, which are not vasodilators, are also useful in prophylaxis. Several newer groups of drugs are under investigation, including drugs that alter myocardial metabolism and selective cardiac rate inhibitors.
By far the most common cause of angina is atheromatous obstruction of the large coronary vessels (coronary artery disease, CAD). Inadequate blood flow in the presence of CAD results in effort angina, also known as classic angina. However, transient spasm of localized portions of these vessels, which is usually associated with underlying atheromas, can also cause significant myocardial ischemia and pain (vasospastic or variant angina). Variant angina is also called Prinzmetal angina.
The primary cause of angina pectoris is an imbalance between the oxygen requirement of the heart and the oxygen supplied to it via the coronary vessels. In effort angina, the imbalance occurs when the myocardial oxygen requirement increases, especially during exercise, and coronary blood flow does not increase proportionately. The resulting ischemia usually leads to pain. In fact, coronary flow reserve is frequently impaired in such patients because of endothelial dysfunction, which is associated with impaired vasodilation. As a result, ischemia may occur at a lower level of myocardial oxygen demand. In some individuals, the ischemia is not always accompanied by pain, resulting in “silent” or “ambulatory” ischemia. In variant angina, oxygen delivery decreases as a result of reversible coronary vasospasm.
Unstable angina, an acute coronary syndrome, is said to be present when episodes of angina occur at rest and when there is an increase in the severity, frequency, and duration of chest pain in patients with previously stable angina. Unstable angina is caused by episodes of increased epicardial coronary artery resistance or small platelet clots occurring in the vicinity of an atherosclerotic plaque. In most cases, formation of labile partially occlusive thrombi at the site of a fissured or ulcerated plaque is the mechanism for reduction in flow. Inflammation may be a risk factor, because patients taking tumor necrosis factor inhibitors appear to have a lower risk of myocardial infarction. The course and the prognosis of unstable angina are variable, but this subset of acute coronary syndrome is associated with a high risk of myocardial infarction and death and is considered a medical emergency.
In theory, the imbalance between oxygen delivery and myocardial oxygen demand can be corrected by decreasing oxygen demand or by increasing delivery (by increasing coronary flow). In effort angina, oxygen demand can be reduced by decreasing cardiac work or, according to some studies, by shifting myocardial metabolism to substrates that require less oxygen per unit of adenosine triphosphate (ATP) produced. In variant angina, on the other hand, spasm of coronary vessels can be reversed by nitrate or calcium channel-blocking vasodilators. Lipid-lowering drugs, especially the “statins,” have become extremely important in the long-term treatment of atherosclerotic disease (see Chapter 35). In unstable angina, vigorous measures are taken to achieve both—increase oxygen delivery (by medical or physical interventions), and decrease oxygen demand.
PATHOPHYSIOLOGY OF ANGINA
Determinants of Myocardial Oxygen Demand
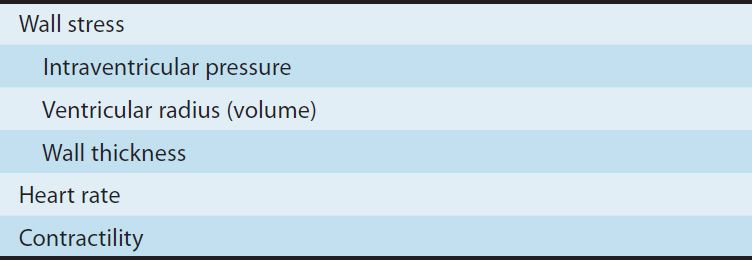
The major determinants of myocardial oxygen requirement are listed in Table 12–1. The effects of arterial blood pressure and venous pressure are mediated through their effects on myocardial wall stress. As a consequence of its continuous activity, the heart’s oxygen needs are relatively high, and it extracts approximately 75% of the available oxygen even in the absence of stress. The myocardial oxygen requirement increases when there is an increase in heart rate, contractility, arterial pressure, or ventricular volume. These hemodynamic alterations frequently occur during physical exercise and sympathetic discharge, which often precipitate angina in patients with obstructive coronary artery disease.
TABLE 12–1 Determinants of myocardial oxygen consumption.
Drugs that reduce cardiac size, rate, or force reduce cardiac oxygen demand. Thus, vasodilators, β blockers, and calcium blockers have predictable benefits in angina. A small, late component of sodium current helps to maintain the long plateau and prolong the calcium current of myocardial action potentials. Drugs that block this late sodium current can indirectly reduce calcium influx and consequently reduce cardiac contractile force. The heart favors fatty acids as a substrate for energy production. However, oxidation of fatty acids requires more oxygen per unit of ATP generated than oxidation of carbohydrates. Therefore, drugs that shift myocardial metabolism toward greater use of glucose (fatty acid oxidation inhibitors) have the potential, at least in theory, to reduce the oxygen demand without altering hemodynamics.
Determinants of Coronary Blood Flow & Myocardial Oxygen Supply
In the normal heart, increased demand for oxygen is met by augmenting coronary blood flow. Because coronary flow drops to negligible values during systole, coronary blood flow is directly related to the aortic diastolic pressure and the duration of diastole. Therefore, the duration of diastole becomes a limiting factor for myocardial perfusion during tachycardia. Coronary blood flow is inversely proportional to coronary vascular resistance. Resistance is determined mainly by intrinsic factors, including metabolic products and autonomic activity, and can be modified—in normal coronary vessels—by various pharmacologic agents. Damage to the endothelium of coronary vessels has been shown to alter their ability to dilate and to increase coronary vascular resistance.
Determinants of Vascular Tone
Peripheral arteriolar and venous tone (smooth muscle tension) both play a role in determining myocardial wall stress (Table 12–1). Arteriolar tone directly controls peripheral vascular resistance and thus arterial blood pressure. In systole, intraventricular pressure must exceed aortic pressure to eject blood; arterial blood pressure thus determines the systolic wall stress in an important way. Venous tone determines the capacity of the venous circulation and controls the amount of blood sequestered in the venous system versus the amount returned to the heart. Venous tone thereby determines the diastolic wall stress.
The regulation of smooth muscle contraction and relaxation is shown schematically in Figure 12–1. The mechanisms of action of the major types of vasodilators are listed in Table 11–3. As shown in Figures 12–1 and 12–2, drugs may relax vascular smooth muscle in several ways:
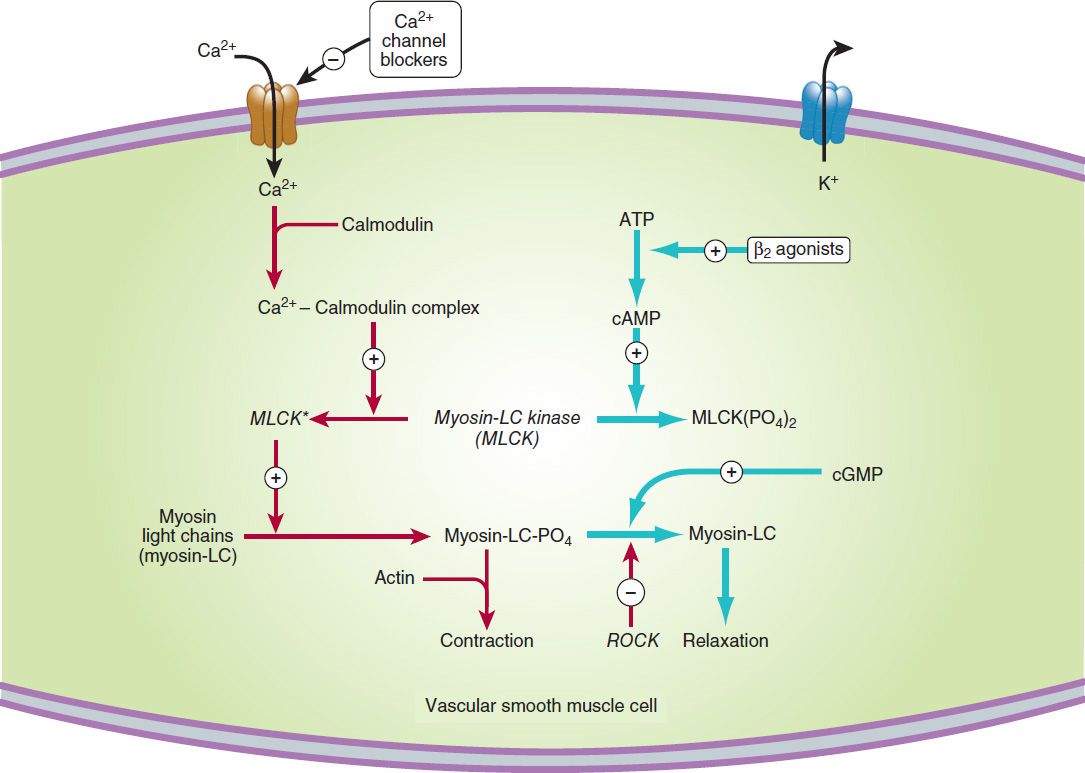
FIGURE 12–1 A simplified diagram of smooth muscle contraction and the site of action of calcium channel-blocking drugs. Contraction is triggered (red arrows) by influx of calcium (which can be blocked by calcium channel blockers) through transmembrane calcium channels. The calcium combines with calmodulin to form a complex that converts the enzyme myosin light-chain kinase to its active form (MLCK*). The latter phosphorylates the myosin light chains, thereby initiating the interaction of myosin with actin. Other proteins, including calponin and caldesmon (not shown), inhibit the ATPase activity of myosin during the relaxation of smooth muscle. Interaction with the Ca2+-calmodulin complex reduces their interaction with myosin during the contraction cycle. Beta2 agonists (and other substances that increase cAMP) may cause relaxation in smooth muscle (blue arrows) by accelerating the inactivation of MLCK and by facilitating the expulsion of calcium from the cell (not shown). cGMP facilitates relaxation by the mechanism shown in Figure 12–2. ROCK, Rho kinase.
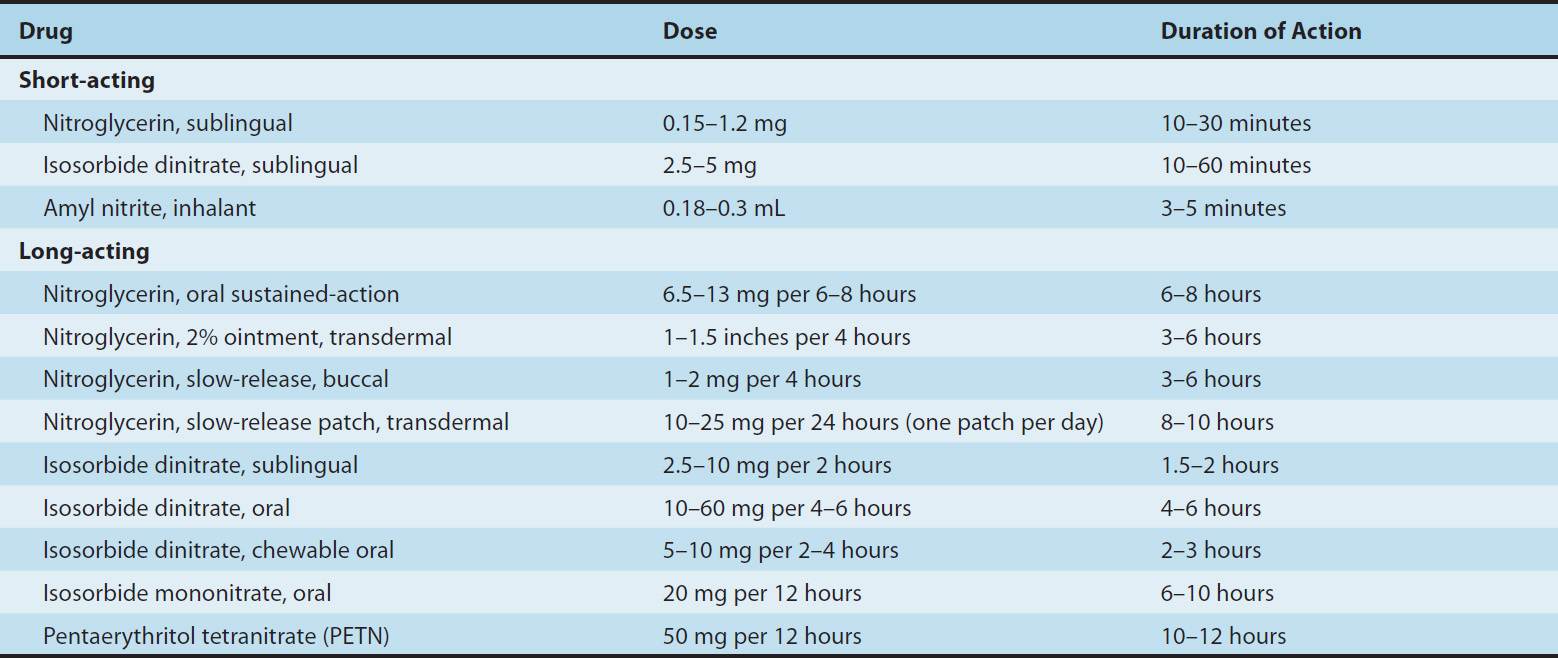
TABLE 12–3 Nitrate and nitrite drugs used in the treatment of angina.
1. Increasing cGMP: cGMP facilitates the dephosphorylation of myosin light chains, preventing the interaction of myosin with actin. Nitric oxide is an effective activator of soluble guanylyl cyclase and acts mainly through this mechanism. Important molecular donors of nitric oxide include nitroprusside (see Chapters 11 and 19) and the organic nitrates used in angina. Atherosclerotic disease may diminish endogenous endothelial NO synthesis, thus making the vascular smooth muscle more dependent upon exogenous sources of NO.
2. Decreasing intracellular Ca2+: Calcium channel blockers predictably cause vasodilation because they reduce intracellular Ca2+, a major modulator of the activation of myosin light chain kinase (Figure 12–1) in smooth muscle. Beta blockers and calcium channel blockers also reduce Ca2+ influx in cardiac muscle fibers, thereby reducing rate, contractility, and oxygen requirement under most circumstances.
3. Stabilizing or preventing depolarization of the vascular smooth muscle cell membrane: The membrane potential of excitable cells is stabilized near the resting potential by increasing potassium permeability. cGMP may increase permeability of Ca2+-activated K+ channels. Potassium channel openers, such as minoxidil sulfate (see Chapter 11) increase the permeability of K+ channels, probably ATP-dependent K+ channels. Certain agents used elsewhere and under investigation in the United States (eg, nicorandil) may act, in part, by this mechanism.
4. Increasing cAMP in vascular smooth muscle cells: As shown in Figure 12–1, an increase in cAMP increases the rate of inactivation of myosin light chain kinase, the enzyme responsible for triggering the interaction of actin with myosin in these cells. This appears to be the mechanism of vasodilation caused by β2 agonists, drugs that are not used in angina (because they cause too much cardiac stimulation), and by fenoldopam, a D1 agonist used in hypertensive emergencies.
 BASIC PHARMACOLOGY OF DRUGS USED TO TREAT ANGINA
BASIC PHARMACOLOGY OF DRUGS USED TO TREAT ANGINA
Drug Action in Angina
The three drug groups traditionally used in angina (organic nitrates, calcium channel blockers, and β blockers) decrease myocardial oxygen requirement by decreasing the determinants of oxygen demand (heart rate, blood pressure, and contractility). Nitrates usually also cause a beneficial decrease in ventricular volume. In some patients, the nitrates and the calcium channel blockers may cause a redistribution of coronary flow and increase oxygen delivery to ischemic tissue. In variant angina, these two drug groups also increase myocardial oxygen delivery by reversing coronary artery spasm. Two newer drugs, ranolazine and ivabradine, are discussed later.
NITRATES & NITRITES
Chemistry
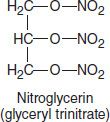
These agents are simple nitric and nitrous acid esters of polyalcohols. Nitroglycerin may be considered the prototype of the group. Although nitroglycerin is used in the manufacture of dynamite, the systemic formulations used in medicine are not explosive. The conventional sublingual tablet form of nitroglycerin may lose potency when stored as a result of volatilization and adsorption to plastic surfaces. Therefore, it should be kept in tightly closed glass containers. Nitroglycerin is not sensitive to light.
All therapeutically active agents in the nitrate group appear to have identical mechanisms of action and similar toxicities, although susceptibility to tolerance may vary. Therefore, pharmacokinetic factors govern the choice of agent and mode of therapy when using the nitrates.
Pharmacokinetics
The liver contains a high-capacity organic nitrate reductase that removes nitrate groups in a stepwise fashion from the parent molecule and ultimately inactivates the drug. Therefore, oral bioavailability of the traditional organic nitrates (eg, nitroglycerin and isosorbide dinitrate) is low (typically <10–20%). For this reason, the sublingual route, which avoids the first-pass effect, is preferred for achieving a therapeutic blood level rapidly. Nitroglycerin and isosorbide dinitrate both are absorbed efficiently by this route and reach therapeutic blood levels within a few minutes. However, the total dose administered by this route must be limited to avoid excessive effect; therefore, the total duration of effect is brief (15–30 minutes). When much longer duration of action is needed, oral preparations can be given that contain an amount of drug sufficient to result in sustained systemic blood levels of the parent drug plus active metabolites. Pentaerythritol (PETN) is another organic nitrate that is promoted for oral use as a “long-acting” nitrate (>6 hours). Other routes of administration available for nitroglycerin include transdermal and buccal absorption from slow-release preparations (described below).
Amyl nitrite and related nitrites are highly volatile liquids. Amyl nitrite is available in fragile glass ampules packaged in a protective cloth covering. The ampule can be crushed with the fingers, resulting in rapid release of vapors inhalable through the cloth covering. The inhalation route provides very rapid absorption and, like the sublingual route, avoids the hepatic first-pass effect. Because of its unpleasant odor and short duration of action, amyl nitrite is now obsolete for angina.
Once absorbed, the unchanged nitrate compounds have half-lives of only 2–8 minutes. The partially denitrated metabolites have much longer half-lives (up to 3 hours). Of the nitroglycerin metabolites (two dinitroglycerins and two mononitro forms), the 1,2-dinitro derivative has significant vasodilator efficacy and probably provides most of the therapeutic effect of orally administered nitroglycerin. The 5-mononitrate metabolite of isosorbide dinitrate is an active metabolite of the latter drug and is available for oral use as isosorbide mononitrate. It has a bioavailability of 100%.
Excretion, primarily in the form of glucuronide derivatives of the denitrated metabolites, is largely by way of the kidney.
Pharmacodynamics
A. Mechanism of Action in Smooth Muscle
After more than a century of study, the mechanism of action of nitroglycerin is still not fully understood. There is general agreement that the drug must be bioactivated with the release of nitric oxide. Unlike nitroprusside and some other direct nitric oxide donors, nitroglycerin activation requires enzymatic action. Nitroglycerin can be denitrated by glutathione S-transferase in smooth muscle and other cells. A mitochondrial enzyme, aldehyde dehydrogenase isoform 2 (ALDH2) and possibly isoform 3 (ALDH3), appears to be key in the activation and release of nitric oxide from nitroglycerin and pentaerythritol tetranitrate. Different enzymes may be involved in the denitration of isosorbide dinitrate and mononitrate. Free nitrite ion is released, which is then converted to nitric oxide (see Chapter 19). Nitric oxide (probably complexed with cysteine) combines with the heme group of soluble guanylyl cyclase, activating that enzyme and causing an increase in cGMP. As shown in Figure 12–2, formation of cGMP represents a first step toward smooth muscle relaxation. The production of prostaglandin E or prostacyclin (PGI2) and membrane hyperpolarization may also be involved. There is no evidence that autonomic receptors are involved in the primary nitrate response. However, autonomic reflex responses, evoked when hypotensive doses are given, are common. As described in the following text, tolerance is an important consideration in the use of nitrates. Although tolerance may be caused in part by a decrease in tissue sulfhydryl groups, eg, on cysteine, tolerance can be only partially prevented or reversed with a sulfhydryl-regenerating agent. Increased generation of oxygen free radicals during nitrate therapy may be another important mechanism of tolerance. Recent evidence suggests that diminished availability of calcitonin gene-related peptide (CGRP, a potent vasodilator) is also associated with nitrate tolerance.
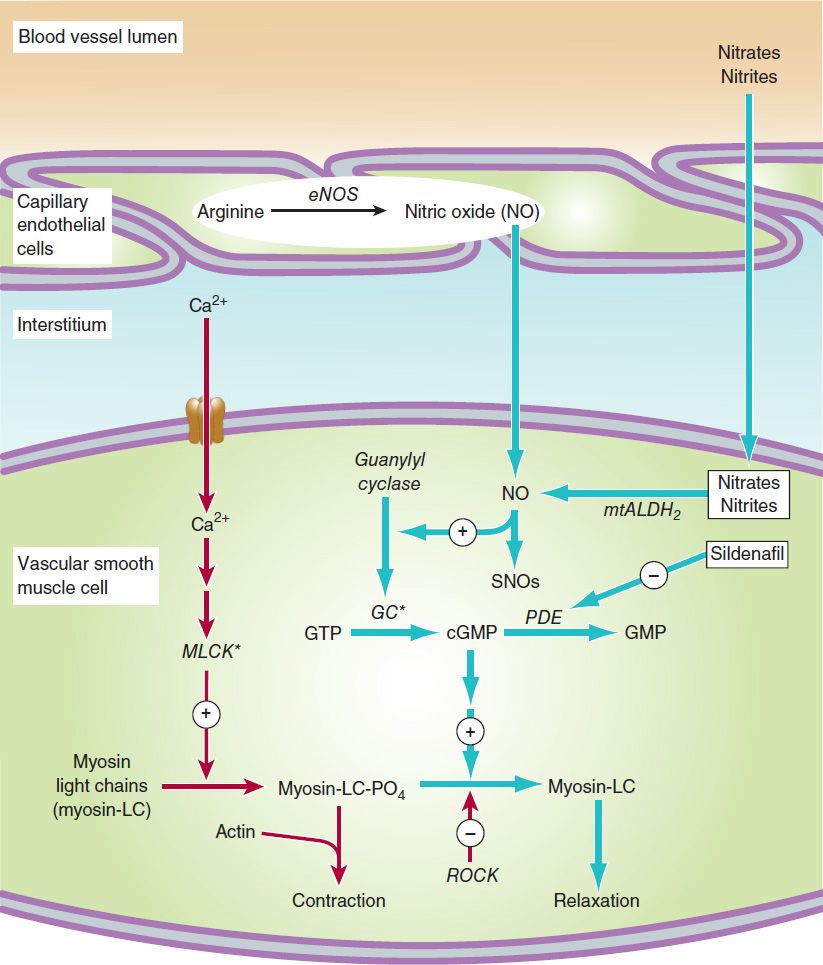
FIGURE 12–2 Mechanism of action of nitrates, nitrites, and other substances that increase the concentration of nitric oxide (NO) in vascular smooth muscle cells. Steps leading to relaxation are shown with blue arrows. MLCK*, activated myosin light-chain kinase (see Figure 12–1). Nitrosothiols (SNOs) appear to have non-cGMP-dependent effects on potassium channels and Ca2+-ATPase. GC*, activated guanylyl cyclase; PDE, phosphodiesterase; eNOS, endothelial nitric oxide synthase; mtALDH2, mitochondrial aldehyde dehydrogenase-2; ROCK, Rho kinase.
Nicorandil and several other antianginal agents not available in the United States appear to combine the activity of nitric oxide release with a direct potassium channel-opening action, thus providing an additional mechanism for causing vasodilation.
B. Organ System Effects
Nitroglycerin relaxes all types of smooth muscle regardless of the cause of the preexisting muscle tone (Figure 12–3). It has practically no direct effect on cardiac or skeletal muscle.
1. Vascular smooth muscle—All segments of the vascular system from large arteries through large veins relax in response to nitroglycerin. Most evidence suggests a gradient of response, with veins responding at the lowest concentrations, arteries at slightly higher ones. The epicardial coronary arteries are sensitive, but concentric atheromas can prevent significant dilation. On the other hand, eccentric lesions permit an increase in flow when nitrates relax the smooth muscle on the side away from the lesion. Arterioles and precapillary sphincters are dilated least, partly because of reflex responses and partly because different vessels vary in their ability to release nitric oxide from the drug.
A primary direct result of an effective dose of nitroglycerin is marked relaxation of veins with increased venous capacitance and decreased ventricular preload. Pulmonary vascular pressures and heart size are significantly reduced. In the absence of heart failure, cardiac output is reduced. Because venous capacitance is increased, orthostatic hypotension may be marked and syncope can result. Dilation of large epicardial coronary arteries may improve oxygen delivery in the presence of eccentric atheromas or collateral vessels. Temporal artery pulsations and a throbbing headache associated with meningeal artery pulsations are common effects of nitroglycerin and amyl nitrite. In heart failure, preload is often abnormally high; the nitrates and other vasodilators, by reducing preload, may have a beneficial effect on cardiac output in this condition (see Chapter 13).
The indirect effects of nitroglycerin consist of those compensatory responses evoked by baroreceptors and hormonal mechanisms responding to decreased arterial pressure (see Figure 6–7); this often results in tachycardia and increased cardiac contractility. Retention of salt and water may also be significant, especially with intermediate- and long-acting nitrates. These compensatory responses contribute to the development of tolerance.
In normal subjects without coronary disease, nitroglycerin can induce a significant, if transient, increase in total coronary blood flow. In contrast, there is no evidence that total coronary flow is increased in patients with angina due to atherosclerotic obstructive coronary artery disease. However, some studies suggest that redistribution of coronary flow from normal to ischemic regions may play a role in nitroglycerin’s therapeutic effect. Nitroglycerin also exerts a weak negative inotropic effect on the heart via nitric oxide.
2. Other smooth muscle organs—Relaxation of smooth muscle of the bronchi, gastrointestinal tract (including biliary system), and genitourinary tract has been demonstrated experimentally. Because of their brief duration, these actions of the nitrates are rarely of any clinical value. During recent decades, the use of amyl nitrite and isobutyl nitrite (not nitrates) by inhalation as recreational (sex-enhancing) drugs has become popular with some segments of the population. Nitrites readily release nitric oxide in erectile tissue as well as vascular smooth muscle and activate guanylyl cyclase. The resulting increase in cGMP causes dephosphorylation of myosin light chains and relaxation (Figure 12–2), which enhances erection. The pharmacologic approach to erectile dysfunction is discussed in the Box: Drugs Used in the Treatment of Erectile Dysfunction.
3. Action on platelets—Nitric oxide released from nitroglycerin stimulates guanylyl cyclase in platelets as in smooth muscle. The increase in cGMP that results is responsible for a decrease in platelet aggregation. Unfortunately, recent prospective trials have established no survival benefit when nitroglycerin is used in acute myocardial infarction. In contrast, intravenous nitroglycerin may be of value in unstable angina, in part through its action on platelets.
4. Other effects—Nitrite ion (not nitrate) reacts with hemoglobin (which contains ferrous iron) to produce methemoglobin (which contains ferric iron). Because methemoglobin has a very low affinity for oxygen, large doses of nitrites can result in pseudocyanosis, tissue hypoxia, and death. Fortunately, the plasma level of nitrite resulting from even large doses of organic and inorganic nitrates is too low to cause significant methemoglobinemia in adults. In nursing infants, the intestinal flora is capable of converting significant amounts of inorganic nitrate, eg, from well water, to nitrite ion. In addition, sodium nitrite is used as a curing agent for meats, eg, corned beef. Thus, inadvertent exposure to large amounts of nitrite ion can occur and may produce serious toxicity.
One therapeutic application of this otherwise toxic effect of nitrite has been discovered. Cyanide poisoning results from complexing of cytochrome iron by the CN– ion. Methemoglobin iron has a very high affinity for CN–; thus, administration of sodium nitrite (NaNO2) soon after cyanide exposure regenerates active cytochrome. The cyanmethemoglobin produced can be further detoxified by the intravenous administration of sodium thiosulfate (Na2S2O3); this results in formation of thiocyanate ion (SCN–), a less toxic ion that is readily excreted. Methemoglobinemia, if excessive, can be treated by giving methylene blue intravenously. This antidotal procedure is now being replaced by hydroxocobalamin, a form of vitamin B12, which also has a very high affinity for cyanide and combines with it to generate another form of vitamin B12.
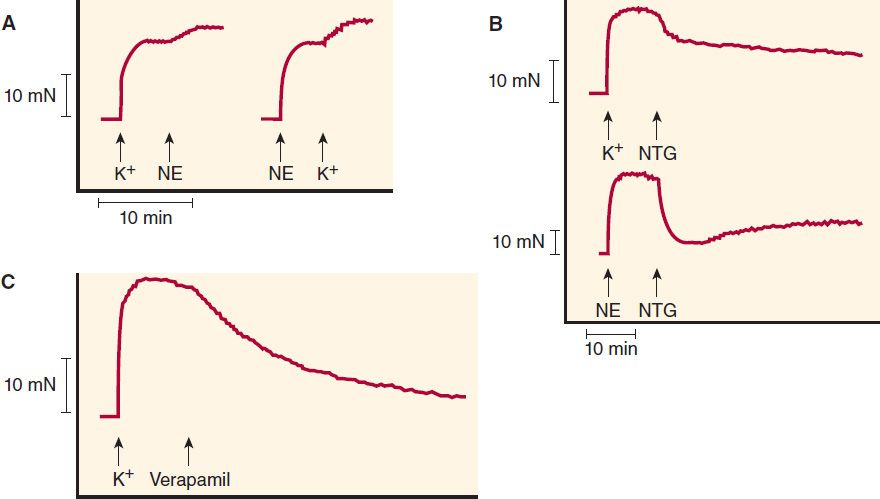
FIGURE 12–3 Effects of vasodilators on contractions of human vein segments studied in vitro. A
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


