43
Beta-Lactam & Other Cell Wall- & Membrane-Active Antibiotics
CASE STUDY
A 55-year-old man is brought to the local hospital emergency department by ambulance. His wife reports that he had been in his normal state of health until 3 days ago when he developed a fever and a productive cough. During the last 24 hours he has complained of a headache and is increasingly confused. His wife reports that his medical history is significant only for hypertension, for which he takes hydrochlorothiazide and lisinopril, and that he is allergic to amoxicillin. She says that he developed a rash many years ago when prescribed amoxicillin for bronchitis. In the emergency department, the man is febrile (38.7°C [101.7°F]), hypotensive (90/54 mm Hg), tachypneic (36/min), and tachycardic (110/min). He has no signs of meningismus but is oriented only to person. A stat chest x-ray shows a left lower lung consolidation consistent with pneumonia. The plan is to start empiric antibiotics and perform a lumbar puncture to rule out bacterial meningitis. What antibiotic regimen should be prescribed to treat both pneumonia and meningitis? Does the history of amoxicillin rash affect the antibiotic choice? Why or why not?
 BETA-LACTAM COMPOUNDS
BETA-LACTAM COMPOUNDS
PENICILLINS
The penicillins share features of chemistry, mechanism of action, pharmacology, and immunologic characteristics with cephalosporins, monobactams, carbapenems, and β-lactamase inhibitors. All are β-lactam compounds, so named because of their four-membered lactam ring.
Chemistry
All penicillins have the basic structure shown in Figure 43–1. A thiazolidine ring (A) is attached to a β-lactam ring (B) that carries a secondary amino group (RNH–). Substituents (R; examples shown in Figure 43–2) can be attached to the amino group. Structural integrity of the 6-aminopenicillanic acid nucleus (rings A plus B) is essential for the biologic activity of these compounds. Hydrolysis of the β-lactam ring by bacterial β-lactamases yields penicilloic acid, which lacks antibacterial activity.
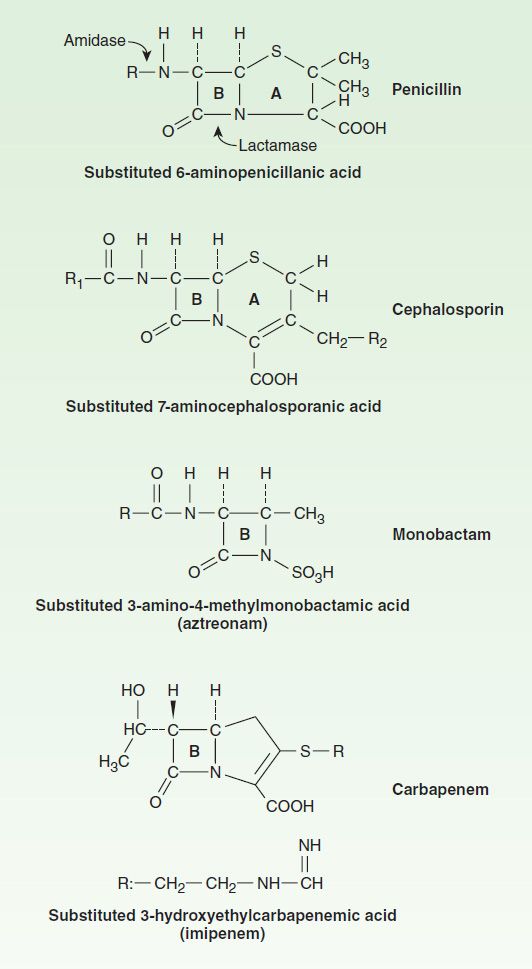
FIGURE 43–1 Core structures of four β-lactam antibiotic families. The ring marked B in each structure is the β-lactam ring. The penicillins are susceptible to bacterial metabolism and inactivation by amidases and lactamases at the points shown. Note that the carbapenems have a different stereochemical configuration in the lactam ring that imparts resistance to most common β lactamases. Substituents for the penicillin and cephalosporin families are shown in Figures 43–2 and 43–6, respectively.
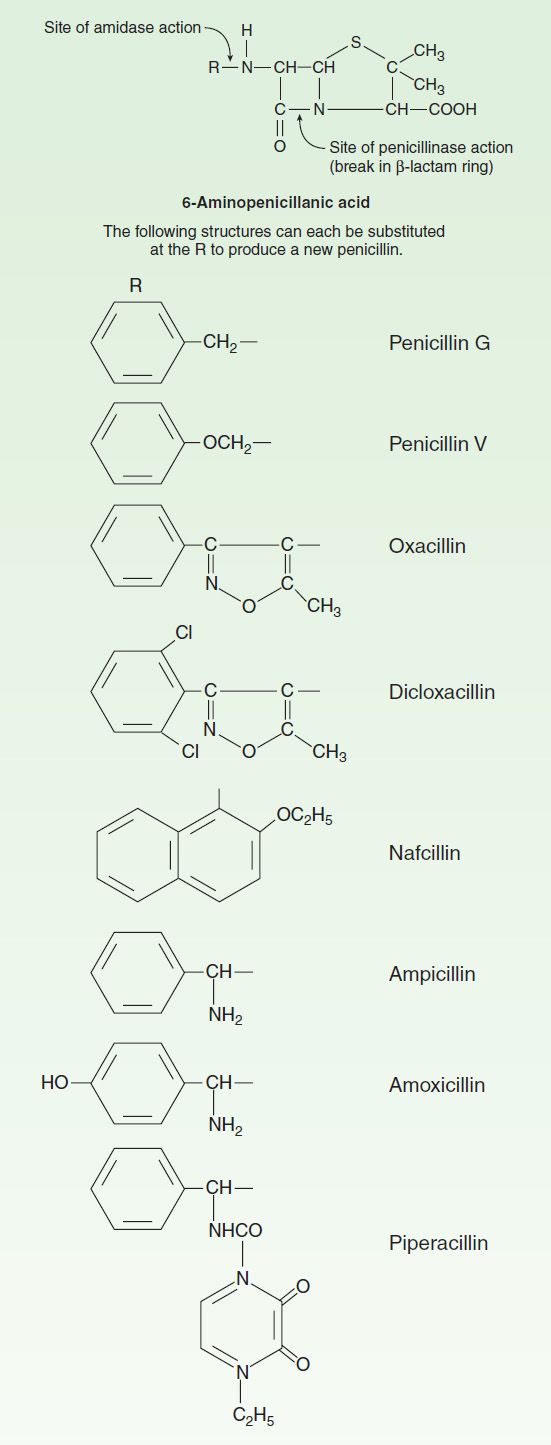
FIGURE 43–2 Side chains of some penicillins (R groups).
A. Classification
Substituents of the 6-aminopenicillanic acid moiety determine the essential pharmacologic and antibacterial properties of the resulting molecules. Penicillins can be assigned to one of three groups (below). Within each of these groups are compounds that are relatively stable to gastric acid and suitable for oral administration, eg, penicillin V, dicloxacillin, and amoxicillin. The side chains of some representatives of each group are shown in Figure 43–2, with a few distinguishing characteristics.
1. Penicillins (eg, penicillin G)—These have greatest activity against gram-positive organisms, gram-negative cocci, and non-β-lactamase-producing anaerobes. However, they have little activity against gram-negative rods, and they are susceptible to hydrolysis by β-lactamases.
2. Antistaphylococcal penicillins (eg, nafcillin)—These penicillins are resistant to staphylococcal β-lactamases. They are active against staphylococci and streptococci but not against enterococci, anaerobic bacteria, and gram-negative cocci and rods.
3. Extended-spectrum penicillins (aminopenicillins and antipseudomonal penicillins)—These drugs retain the antibacterial spectrum of penicillin and have improved activity against gram-negative organisms. Like penicillin, however, they are relatively susceptible to hydrolysis by β-lactamases.
B. Penicillin Units and Formulations
The activity of penicillin G was originally defined in units. Crystalline sodium penicillin G contains approximately 1600 units per mg (1 unit = 0.6 mcg; 1 million units of penicillin = 0.6 g). Semisynthetic penicillins are prescribed by weight rather than units. The minimum inhibitory concentration (MIC) of any penicillin (or other antimicrobial) is usually given in mcg/mL. Most penicillins are formulated as the sodium or potassium salt of the free acid. Potassium penicillin G contains about 1.7 mEq of K+ per million units of penicillin (2.8 mEq/g). Nafcillin contains Na+, 2.8 mEq/g. Procaine salts and benzathine salts of penicillin G provide repository forms for intramuscular injection. In dry crystalline form, penicillin salts are stable for years at 4°C. Solutions lose their activity rapidly (eg, 24 hours at 20°C) and must be prepared fresh for administration.
Mechanism of Action
Penicillins, like all β-lactam antibiotics, inhibit bacterial growth by interfering with the transpeptidation reaction of bacterial cell wall synthesis. The cell wall is a rigid outer layer that completely surrounds the cytoplasmic membrane (Figure 43–3), maintains cell shape and integrity, and prevents cell lysis from high osmotic pressure. The cell wall is composed of a complex, cross-linked polymer of polysaccharides and polypeptides, peptidoglycan (also known as murein or mucopeptide). The polysaccharide contains alternating amino sugars, N-acetylglucosamine and N-acetylmuramic acid (Figure 43–4). A five-amino-acid peptide is linked to the N-acetylmuramic acid sugar. This peptide terminates in D-alanyl-D-alanine. Penicillin-binding protein (PBP, an enzyme) removes the terminal alanine in the process of forming a cross-link with a nearby peptide. Cross-links give the cell wall its structural rigidity. Beta-lactam antibiotics, structural analogs of the natural D-Ala-D-Ala substrate, covalently bind to the active site of PBPs. This binding inhibits the transpeptidation reaction (Figure 43–5) and halts peptidoglycan synthesis, and the cell dies. The exact mechanism of cell death is not completely understood, but autolysins and disruption of cell wall morphogenesis are involved. Beta-lactam antibiotics kill bacterial cells only when they are actively growing and synthesizing cell wall.
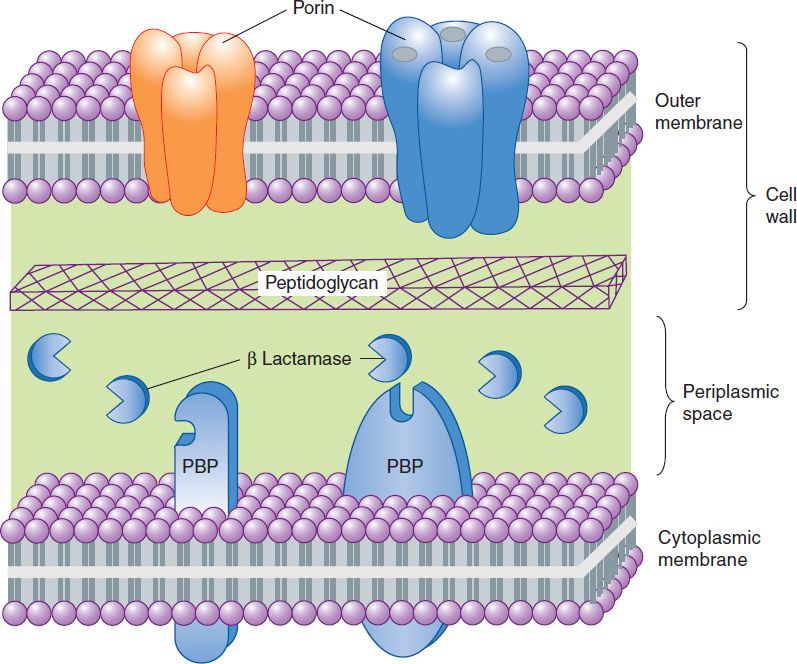
FIGURE 43–3 A highly simplified diagram of the cell envelope of a gram-negative bacterium. The outer membrane, a lipid bilayer, is present in gram-negative but not gram-positive organisms. It is penetrated by porins, proteins that form channels providing hydrophilic access to the cytoplasmic membrane. The peptidoglycan layer is unique to bacteria and is much thicker in gram-positive organisms than in gram–negative ones. Together, the outer membrane and the peptidoglycan layer constitute the cell wall. Penicillin-binding proteins (PBPs) are membrane proteins that cross-link peptidoglycan. Beta lactamases, if present, reside in the periplasmic space or on the outer surface of the cytoplasmic membrane, where they may destroy β-lactam antibiotics that penetrate the outer membrane.
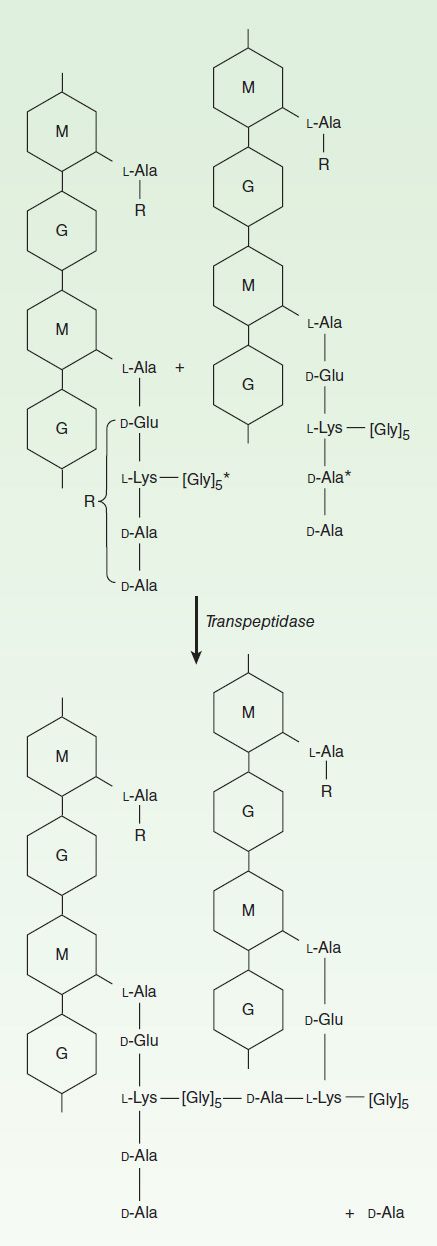
FIGURE 43–4 The transpeptidation reaction in Staphylococcus aureus that is inhibited by β-lactam antibiotics. The cell wall of gram-positive bacteria is made up of long peptidoglycan polymer chains consisting of the alternating aminohexoses N-acetylglucosamine (G) and N-acetylmuramic acid (M) with pentapeptide side chains linked (in S aureus) by pentaglycine bridges. The exact composition of the side chains varies among species. The diagram illustrates small segments of two such polymer chains and their amino acid side chains. These linear polymers must be cross-linked by transpeptidation of the side chains at the points indicated by the asterisk to achieve the strength necessary for cell viability.
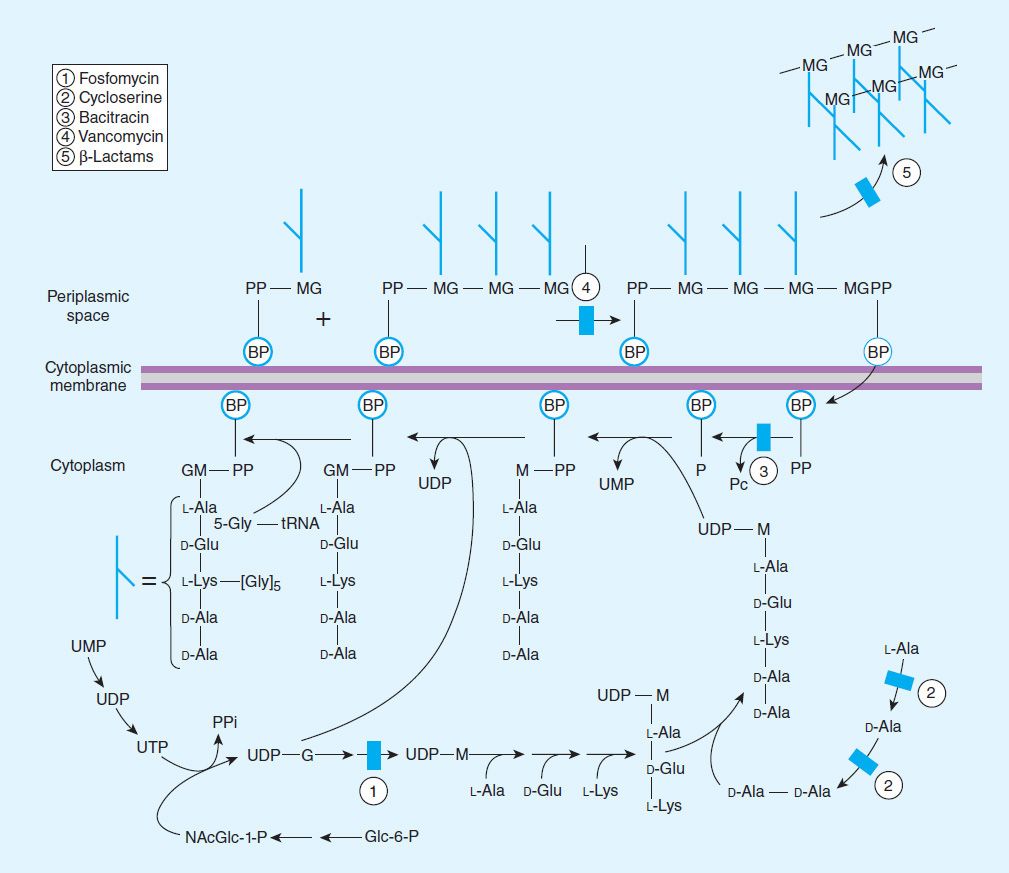
FIGURE 43–5 The biosynthesis of cell wall peptidoglycan, showing the sites of action of five antibiotics (shaded bars; 1 = fosfomycin, 2 = cycloserine, 3 = bacitracin, 4 = vancomycin, 5 = β-lactam antibiotics). Bactoprenol (BP) is the lipid membrane carrier that transports building blocks across the cytoplasmic membrane; M, N-acetylmuramic acid; Glc, glucose; NAcGlc or G, N-acetylglucosamine.
Resistance
Resistance to penicillins and other β-lactams is due to one of four general mechanisms: (1) inactivation of antibiotic by β-lactamase, (2) modification of target PBPs, (3) impaired penetration of drug to target PBPs, and (4) antibiotic efflux. Beta-lactamase production is the most common mechanism of resistance. Hundreds of different β-lactamases have been identified. Some, such as those produced by Staphylococcus aureus, Haemophilus influenzae, and Escherichia coli, are relatively narrow in substrate specificity, preferring penicillins to cephalosporins. Other β-lactamases, eg, AmpC β-lactamase produced by Pseudomonas aeruginosa and Enterobacter sp, and extended–spectrum β-lactamases (ESBLs), hydrolyze both cephalosporins and penicillins. Carbapenems are highly resistant to hydrolysis by penicillinases and cephalosporinases, but they are hydrolyzed by metallo-β lactamase and carbapenemases.
Altered target PBPs are the basis of methicillin resistance in staphylococci and of penicillin resistance in pneumococci and enterococci. These resistant organisms produce PBPs that have low affinity for binding β-lactam antibiotics, and consequently, they are not inhibited except at relatively high, often clinically unachievable, drug concentrations.
Resistance due to impaired penetration of antibiotic to target PBPs occurs only in gram-negative species because of the impermeable outer membrane of their cell wall, which is absent in gram-positive bacteria. Beta-lactam antibiotics cross the outer membrane and enter gram-negative organisms via outer membrane protein channels called porins. Absence of the proper channel or down-regulation of its production can greatly impair drug entry into the cell. Poor penetration alone is usually not sufficient to confer resistance because enough antibiotic eventually enters the cell to inhibit growth. However, this barrier can become important in the presence of a β-lactamase, even a relatively inactive one, as long as it can hydrolyze drug faster than it enters the cell. Gram-negative organisms also may produce an efflux pump, which consists of cytoplasmic and periplasmic protein components that efficiently transport some β-lactam antibiotics from the periplasm back across the cell wall outer membrane.
Pharmacokinetics
Absorption of orally administered drug differs greatly for different penicillins, depending in part on their acid stability and protein binding. Gastrointestinal absorption of nafcillin is erratic, so it is not suitable for oral administration. Dicloxacillin, ampicillin, and amoxicillin are acid-stable and relatively well absorbed, producing serum concentrations in the range of 4–8 mcg/mL after a 500-mg oral dose. Absorption of most oral penicillins (amoxicillin being an exception) is impaired by food, and the drugs should be administered at least 1–2 hours before or after a meal.
Intravenous administration of penicillin G is preferred to the intramuscular route because of irritation and local pain from intramuscular injection of large doses. Serum concentrations 30 minutes after an intravenous injection of 1 g of penicillin G (equivalent to approximately 1.6 million units) are 20–50 mcg/mL. Only a fraction of the total drug in serum is present as free drug, the concentration of which is determined by protein binding. Highly protein-bound penicillins (eg, nafcillin) generally achieve lower free-drug concentrations in serum than less protein-bound penicillins (eg, penicillin G or ampicillin). Protein binding becomes clinically relevant when the protein-bound percentage is approximately 95% or more. Penicillins are widely distributed in body fluids and tissues with a few exceptions. They are polar molecules, so intracellular concentrations are well below those found in extracellular fluids.
Benzathine and procaine penicillins are formulated to delay absorption, resulting in prolonged blood and tissue concentrations. A single intramuscular injection of 1.2 million units of benzathine penicillin maintains serum levels above 0.02 mcg/mL for 10 days, sufficient to treat β-hemolytic streptococcal infection. After 3 weeks, levels still exceed 0.003 mcg/mL, which is enough to prevent β-hemolytic streptococcal infection. A 600,000 unit dose of procaine penicillin yields peak concentrations of 1–2 mcg/mL and clinically useful concentrations for 12–24 hours after a single intramuscular injection.
Penicillin concentrations in most tissues are equal to those in serum. Penicillin is also excreted into sputum and breast milk to levels 3–15% of those in the serum. Penetration into the eye, the prostate, and the central nervous system is poor. However, with active inflammation of the meninges, as in bacterial meningitis, penicillin concentrations of 1–5 mcg/mL can be achieved with a daily parenteral dose of 18–24 million units. These concentrations are sufficient to kill susceptible strains of pneumococci and meningococci.
Penicillin is rapidly excreted by the kidneys; small amounts are excreted by other routes. Tubular secretion accounts for about 90% of renal excretion, and glomerular filtration accounts for the remainder. The normal half-life of penicillin G is approximately 30 minutes; in renal failure, it may be as long as 10 hours. Ampicillin and the extended-spectrum penicillins are secreted more slowly than penicillin G and have half-lives of 1 hour. For penicillins that are cleared by the kidney, the dose must be adjusted according to renal function, with approximately one fourth to one third the normal dose being administered if creatinine clearance is 10 mL/min or less (Table 43–1).
TABLE 43–1 Guidelines for dosing of some commonly used penicillins.
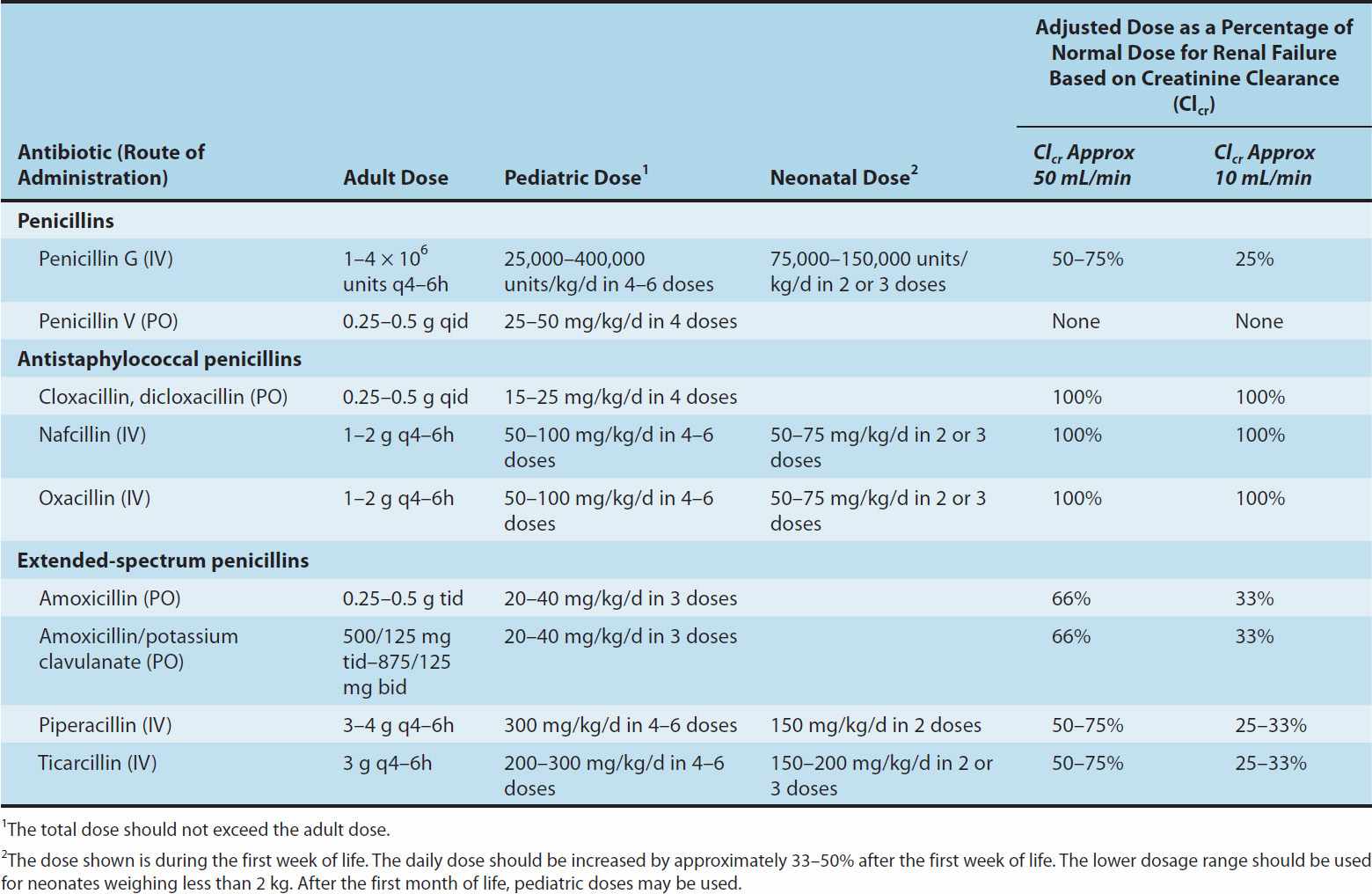
Nafcillin is primarily cleared by biliary excretion. Oxacillin, dicloxacillin, and cloxacillin are eliminated by both the kidney and biliary excretion; no dosage adjustment is required for these drugs in renal failure. Because clearance of penicillins is less efficient in the newborn, doses adjusted for weight alone result in higher systemic concentrations for longer periods than in the adult.
Clinical Uses
Except for amoxicillin, oral penicillins should be given 1–2 hours before or after a meal; they should not be given with food to minimize binding to food proteins and acid inactivation. Amoxicillin may be given without regard to meals. Blood levels of all penicillins can be raised by simultaneous administration of probenecid, 0.5 g (10 mg/kg in children) every 6 hours orally, which impairs renal tubular secretion of weak acids such as β-lactam compounds. Penicillins should never be used for viral infections and should be prescribed only when there is reasonable suspicion of, or documented infection with, susceptible organisms.
A. Penicillin
Penicillin G is a drug of choice for infections caused by streptococci, meningococci, some enterococci, penicillin-susceptible pneumococci, non-β-lactamase-producing staphylococci, Treponema pallidum and certain other spirochetes, some Clostridium species, Actinomyces and certain other gram-positive rods, and non-β-lactamase-producing gram-negative anaerobic organisms. Depending on the organism, the site, and the severity of infection, effective doses range between 4 and 24 million units per day administered intravenously in four to six divided doses. High-dose penicillin G can also be given as a continuous intravenous infusion.
Penicillin V, the oral form of penicillin, is indicated only in minor infections because of its relatively poor bioavailability, the need for dosing four times a day, and its narrow antibacterial spectrum. Amoxicillin (see below) is often used instead.
Benzathine penicillin and procaine penicillin G for intramuscular injection yield low but prolonged drug levels. A single intramuscular injection of benzathine penicillin, 1.2 million units, is effective treatment for β-hemolytic streptococcal pharyngitis; given intramuscularly once every 3–4 weeks, it prevents reinfection. Benzathine penicillin G, 2.4 million units intramuscularly once a week for 1–3 weeks, is effective in the treatment of syphilis. Procaine penicillin G was once a commonly used treatment for uncomplicated pneumococcal pneumonia and gonorrhea; however, it is rarely used now because many gonococcal strains are penicillin-resistant, and many pneumococci require higher doses of penicillin G or the use of more potent β-lactams.
B. Penicillins Resistant to Staphylococcal Beta Lactamase (Methicillin, Nafcillin, and Isoxazolyl Penicillins)
These semisynthetic penicillins are indicated for infections caused by β-lactamase-producing staphylococci, although penicillin susceptible strains of streptococci and pneumococci are also susceptible to these agents. Listeria monocytogenes, enterococci, and methicillin-resistant strains of staphylococci are resistant. In recent years the empirical use of these drugs has decreased substantially because of increasing rates of methicillin-resistance in staphylococci. However, for infections caused by methicillin-susceptible and penicillin-resistant strains of staphylococci, these are considered the drugs of choice.
An isoxazolyl penicillin such as cloxacillin or dicloxacillin, 0.25–0.5 g orally every 4–6 hours (15–25 mg/kg/d for children), is suitable for treatment of mild to moderate localized staphylococcal infections. They are relatively acid-stable and have reasonable bioavailability. However, food interferes with absorption, and the drugs should be administered 1 hour before or after meals.
Methicillin, the first antistaphylococcal penicillin to be developed, is no longer used clinically due to high rates of adverse effects. Oxacillin and nafcillin, 8–12 g/d, given by intermittent intravenous infusion of 1–2 g every 4–6 hours (50–100 mg/kg/d for children), are considered the drugs of choice for serious systemic staphylococcal infections.
C. Extended-Spectrum Penicillins (Aminopenicillins, Carboxypenicillins, and Ureidopenicillins)
These drugs have greater activity than penicillin against gram-negative bacteria because of their enhanced ability to penetrate the gram-negative outer membrane. Like penicillin G, they are inactivated by many β lactamases.
The aminopenicillins, ampicillin and amoxicillin, have very similar spectrums of activity, but amoxicillin is better absorbed orally. Amoxicillin, 250–500 mg three times daily, is equivalent to the same amount of ampicillin given four times daily. Amoxicillin is given orally to treat urinary tract infections, sinusitis, otitis, and lower respiratory tract infections. Ampicillin and amoxicillin are the most active of the oral β-lactam antibiotics against pneumococci with elevated MICs to penicillin and are the preferred β-lactam antibiotics for treating infections suspected to be caused by these strains. Ampicillin (but not amoxicillin) is effective for shigellosis. Ampicillin, at dosages of 4–12 g/d intravenously, is useful for treating serious infections caused by susceptible organisms, including anaerobes, enterococci, L monocytogenes, and β-lactamase-negative strains of gram-negative cocci and bacilli such as E coli, and Salmonella sp. Non-β-lactamase-producing strains of H influenzae are generally susceptible, but strains that are resistant because of altered PBPs are emerging. Due to production of β lactamases by gram-negative bacilli, ampicillin can no longer be used for empirical therapy of urinary tract infections and typhoid fever. Ampicillin is not active against Klebsiella sp, Enterobacter sp, P aeruginosa, Citrobacter sp, Serratia marcescens, indole-positive proteus species, and other gram-negative aerobes that are commonly encountered in hospital-acquired infections. These organisms intrinsically produce β lactamases that inactivate ampicillin.
Carbenicillin, the first antipseudomonal carboxypenicillin, is no longer used in the USA, because there are more active, better tolerated alternatives. A carboxypenicillin with activity similar to that of carbenicillin is ticarcillin. It is less active than ampicillin against enterococci. The ureidopenicillins, piperacillin, mezlocillin, and azlocillin, are also active against selected gram-negative bacilli, such as Klebsiella pneumoniae. Although supportive clinical data are lacking for superiority of combination therapy over single-drug therapy, because of the propensity of P aeruginosa to develop resistance during treatment, an antipseudomonal penicillin is sometimes used in combination with an aminoglycoside or fluoroquinolone for pseudomonal infections outside the urinary tract.
Ampicillin, amoxicillin, ticarcillin, and piperacillin are also available in combination with one of several β-lactamase inhibitors: clavulanic acid, sulbactam, or tazobactam. The addition of a β-lactamase inhibitor extends the activity of these penicillins to include β-lactamase-producing strains of S aureus as well as some β-lactamase-producing gram-negative bacteria (see Beta-Lactamase Inhibitors).
Adverse Reactions
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


