Currently, there is no consensus as to the optimal method for measuring tumor length or percentage of cancer on a core when there are two or more foci of prostate cancer in a single core separated by benign intervening glands and stroma.2 Some urologic pathologists, including our group, measure discontinuous foci of cancer as if they were a single continuous focus. The rationale is that these discontinuous foci are undoubtedly the same cancer going in and out of the plane of section. Others choose to add the measurements of the individual separate foci of cancer, ignoring the extent of the intervening benign prostate tissue. In a recent study from our group, Karram et al.21 demonstrated that for prostate cancer in which the needle biopsy grade is representative of the entire tumor, quantifying cancer extent on biopsy by measuring discontinuous cancer on biopsy from one end to the other as opposed to “collapsing” the cancer by subtracting out the intervening benign prostate tissue correlates better with organ-confined disease and risk of positive margins.
PREDICTION OF POSTTREATMENT PROGRESSION. Several studies have demonstrated that percent of positive cores factored in with biopsy Gleason score, PSA, and clinical stage predict progression following radical prostatectomy.22–25 Other biopsy tumor measurements that have been reported in these studies to correlate with progression following surgery are (a) percent total needle biopsy specimen with cancer (total length of cancer/total length of cores), (b) greatest percentage of cancer on a given core, and (c) percent positive cores from the most involved side (i.e., left or right). Similarly, percent of positive cores have been shown to independently predict recurrence following either brachytherapy or external beam radiotherapy after factoring in PSA and biopsy Gleason score.26–30
PREDICTION OF PROSTATECTOMY TUMOR VOLUME AND POTENTIALLY INSIGNIFICANT TUMOR. Multiple studies have demonstrated that measurements of cancer on the needle biopsy correlate with radical prostatectomy tumor volume.15,31–35 These include linear extent of cancer, percent of cancer, bilateral tumor, greatest percent of cancer, and number of positive cores. However, all of these studies emphasize that the correlation is very weak. The importance of preoperatively attempting to predict tumor volume is to predict which men harbor potentially insignificant cancers that could be treated expectantly with watchful waiting.36 Typically, finding limited cancer on needle biopsy, by itself, is not sufficiently predictive of insignificant cancer without factoring in other clinical and other pathologic information (reviewed in Anast et al.).37–39 For example, Guzzo et al.40 reported that if there was less than 5% of Gleason score less than or equal to 6 cancer on sextant biopsy, only 37% of radical prostatectomies had less than 5% cancer. The same limited amount of cancer on more extended biopsy was more predictive with 73% having less than 5% cancer in the prostatectomy. Zackrisson et al.41 found that on sextant biopsies, if the total cancer length on biopsy was less than 3 mm, only one-third of the corresponding radical prostatectomy specimens contained less than 0.5 cc of tumor. One could only predict with 95% confidence that there was more than 0.5 cc tumor if the total tumor length on biopsy was more than 10 mm. Multiple studies have demonstrated that a limited extent (<3 mm) of cancer on biopsy does not necessarily predict “insignificant” amounts of tumor in the entire prostate.31,36,42–45 One feasible and rational approach would be to have pathologists report the number of cores containing cancer, as well as one other system quantifying tumor extent. This was formally studied by Bismar et al.,12 where multiple measurements of tumor volume on needle biopsy were tested to predict radical prostatectomy tumor volume. They concluded that several measurements of needle biopsy tumor volume (as opposed to only one) provided maximal information on prostate cancer size.12 In their study, they found that number of positive cores and total tumor length along with PSA best predicted whole gland tumor volume. At our institution, the number of cores containing cancer is reported along with the percentage of cancer present on each involved core. Calculating the percent of each core involved by cancer is based on a visual estimate of the length of the cancer involvement divided by the length of the core. An example of how we report our needle biopsy findings is as follows: “Adenocarcinoma of the prostate, Gleason score 3 + 3 = 6, involving three cores (10%, 15%, 30%).” In cases where the cores are fragmented and difficult to assess, we state that the specimen is fragmented and give an estimate as to the percentage of the entire slide involved by cancer. Occasionally, there will be scattered small foci of cancer occupying, for example, 80% of the length of the core, yet only 5% of the total core volume. Merely reporting such a case as showing 80% involvement by cancer may be misleading, because one would expect to see extensive cancer on the biopsy. On the other hand, such a case should be distinguished from one with only a single minute focus of cancer involving 5% of the core. An example of how we report such a case is as follows: “Scattered small foci of adenocarcinoma, Gleason score 3 + 3 = 6, discontinuously involving 80% of the length of one core.” We would predict that such a tumor would not be insignificant and would need definitive therapy in contrast to some cases with only a single minute focus of cancer. A recent report demonstrated that quantifying discontinuous foci of cancer on needle biopsy by measuring from one end of the cancer to the other with inclusion of intervening benign tissue in the measurement correlated better with radical prostatectomy tumor volume, as opposed to “collapsing the cancer” and ignoring intervening benign prostate tissue.46
PREDICTION OF EXTRAPROSTATIC EXTENSION INTO NEUROVASCULAR BUNDLE: IMPLICATIONS FOR POTENCY-PRESERVING SURGERY. The critical decision the urologist must make either before or during radical prostatectomy is whether to spare the neurovascular bundle(s) (NVB[s]) to preserve potency or to resect the bundle(s) in cases where there is a high risk of extraprostatic extension (EPE) in the region of the NVB. How to select the right patients for nerve-sparing radical prostatectomy is still controversial. One option that might be more reliable than various preoperative algorithms would be to assess surgical margins in the region of the NVB by intraoperative frozen sections. There have been conflicting studies as to the utility of performing frozen sections for this purpose, because the technique has significant false-positive and false-negative rates.47–49 Another method of predicting NVB invasion relies on intraoperative visual and tactile assessment at the time of surgery. We have demonstrated that a very experienced urologist can do so with excellent reliability.50 However, such skills and subjective evaluations are not easily transferable to other less experienced urologists. Thus, the most widely used method in predicting EPE to decide whether to spare the NVB(s) relies on various preoperative algorithms. The most complete models utilize needle biopsy cancer tumor volume, grade, and PSA values. Most of these nomograms report the probability of EPE somewhere in the prostate, not specifically within the area of the NVB. Graefen et al.51 reported the presence in a lobe of more than two positive cores containing Gleason grade 4/5 cancer or the presence of more than two positive cores in a lobe with a PSA value more than 10 ng/mL regardless of the Gleason score able to predict side-specific EPE. The Steuber et al.52 model to predict side-specific EPE included PSA, clinical stage, and Gleason score and percent cancer in the ipsilateral biopsy specimen. Ohori et al.53 developed a nomogram to predict side-specific EPE factoring in PSA and the following side-specific variables of digital rectal exam (DRE), maximum Gleason score, percent positive cores, and length of cancer/total length of cores. There have been two studies that specifically defined preoperative parameters to identify patients with a low likelihood of side-specific EPE in the region of the NVB who could safely undergo nerve-sparing prostatectomy without compromising cancer control. Shah et al.54 showed that PSA, clinical stage, ipsilateral Gleason score, and tumor volume in the needle biopsies were statistically significant by multivariate analysis for predicting side-specific EPE in the NVB region. Our group at Hopkins found in a study of 2,660 cases that PSA of 10 or more, side-specific Gleason score of 7 or more, abnormal DRE, more than one-third of side-specific cores with tumor, and more than 20% average percent involvement of each positive core per side were independent predictors of NVB penetration.55 If no more than one of the cited adverse features was present, urologists could predict with 90% or greater accuracy that a man would be an ideal candidate for nerve-sparing radical prostatectomy. Perineural invasion (PNI) on needle biopsy was not independently predictive of NVB EPE in the Hopkins study, although D’Amico56 found that when the NVB was resected as a result of PNI on biopsy, the positive margin rate was decreased as opposed to if the NVB was spared and left within the patient. Rubin et al.57 reported that only 24% of urologists use the presence of PNI on biopsy to determine whether to resect the NVB, although surgeons who do more radical prostatectomies were more likely to consider PNI important.57
LOCATION OF POSITIVE BIOPSY CORES
Some urologists hesitate to submit routine sextant needle biopsies as six separate cores in six separate containers and instead submit them as left- and right-sided cores. There are several advantages to submitting cores by separate sextant site (Table 8.2). Evidence to support lumping the cores together is provided by a study reporting that the positive predictive value of an individual positive core for the location of EPE was not sufficient to guide the surgical decision to spare or excise an NVB.58 However, biopsy core location is of potentially critical importance in the 5% to 10% of biopsies diagnosed as atypical and suspicious for cancer. We advocate the precise labeling of the initial biopsies to localize the sites of an initial atypical diagnosis and to direct the location of repeat biopsies, because increased sampling of the initial atypical site and adjacent ipsilateral and adjacent contralateral sites will increase the yield of cancer detection on repeat biopsy.59

Several studies have demonstrated that the location of a positive biopsy core is predictive of adverse findings at radical prostatectomy. Badalament et al.3 reported that the percentage of cancer in the biopsies from the base and apex correlated with EPE and positive margins, respectively. Similarly, Koh et al.17 reported that percent of cancer at the base on biopsy along with clinical stage, Gleason score, and PSA predicted seminal vesicle invasion. The presence of cancer in multiple sextant sites is predictive of the presence of multifocal rather than solitary cancer at radical prostatectomy; however, these differences do not correlate with pathologic stage or margin positivity.60 Bilateral cancer in most studies correlates with EPE and total tumor volume.15,33
The submission of needle biopsy specimens in separate containers may lead to much larger pathology charges, although the payments for the pathologist’s services are less than the charges. If this is an issue, it is important for urologists and pathologists to come up with alternative strategies that result in agreeable charges while preserving the maximum possible predictive value for biopsy specimens. One option is to put the biopsies from the left gland and the biopsies from the right gland in separate containers. Biopsy cores from the mid and basal areas can be marked with different colors of dye, with the apex unstained, such that the specimens could be submitted together with their sextant origin preserved. A third option would be to dye only biopsies from one side of the gland (i.e., left), with three separate containers (apex, mid, base) submitted to the pathologist. With these options, the pathologist would still be able to identify the site of the atypical focus, and the patient would only be charged for either two or three parts to a case. It has also been demonstrated that with increased numbers of cores per part, there is increased core fragmentation, which can impact in some cases accurate Gleason grading and quantification of number of positive cores, such that a maximum of two cores per cassette is recommended.61
NEEDLE BIOPSY PERINEURAL INVASION
PNI is defined as the presence of prostate cancer tracking along or around a nerve (eFigs. 8.1 and 8.2). The role of prostate needle biopsy PNI in treatment planning has been a source of considerable debate. PNI has been demonstrated to be one of the major mechanisms of extension of prostate cancer from the prostatic parenchyma to the periprostatic soft tissue.62 Whether PNI extensive enough to be sampled on needle biopsy signals an increased risk of EPE of cancer is controversial (reviewed in Bismar et al.).12 In many studies of PNI, assessment of EPE was performed on only partially submitted prostates such that it was most likely underrecognized. Most studies show PNI on needle biopsy to be predictive of EPE on univariate analysis but not independently predictive once other preoperative clinical and pathologic features are factored in. Other studies show PNI to be predictive of EPE also in multivariate analyses, whereas an equal number finds PNI not be predictive even in univariate analysis (Table 8.3).
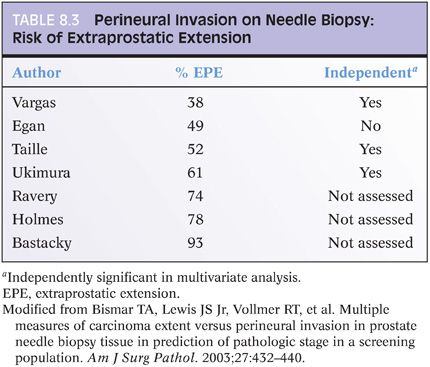
The data as to whether PNI on needle biopsy predicts progression after radiotherapy and surgery is also contradictory and experienced surgeons are almost equally divided as to whether PNI on biopsies influence their treatment decisions.57 In a recent study from our institution, Loeb et al.63 evaluated the relationship of PNI on prostate biopsy and radical prostatectomy outcomes in a contemporary series of 1,256 radical prostatectomies performed by one urologist. On multivariate analysis, PNI was significantly associated with EPE and seminal vesicle invasion. Biochemical progression was more likely in patients with PNI; however, PNI was not a significant independent predictor of biochemical progression on multivariate analysis. Furthermore, the study found that bilateral nerve-sparing surgery did not compromise the oncologic outcomes for patients with PNI on biopsy.63 Harnden et al.64 in a systematic review of the importance of PNI on needle biopsy following radiotherapy or surgery concluded that the weight of the evidence in 21 studies is that this finding is a significant prognostic indicator, particularly in specific patient subgroups defined by serum PSA and Gleason scores. Over two-thirds of the studies using external beam radiotherapy but none using brachytherapy showed prognostic significance for PNI. Difficulty with interpreting studies included (a) varying biopsy techniques (needle angle, location of sampling, number of cores) can affect the type and amount of prostatic tissue sampled and the number of nerves present; (b) number of levels histologically examined can vary; (c) in some studies, data taken from reports where recording of PNI may not be uniformly performed; (d) whether any nerves are present in the specimen is often not taken into account; (e) different pathologists can have interobserver variation in diagnosis of PNI; and (f) pathologists are not blinded to outcome. As some studies have found PNI prognostically useful, some radiotherapists favor the use of external beam radiotherapy over brachytherapy (interstitial seed therapy) in order to treat cases with PNI and a potentially higher risk of cancer being exterior to the prostate. Given that PNI is readily identifiable in most cases, that it is prognostic in some studies although the data is conflicting, and that it is uncertain what factors an individual clinician may consider in treatment decisions, it is the opinion of these authors that it is reasonable for pathologists to note its presence on the biopsy pathology report.
The role of PNI in prostate cancer patients who are candidates for active surveillance has been recently evaluated by our group. Assessing a large cohort (313 cases) of patients who met the biopsy criteria for active surveillance, and elected to undergo immediate radical prostatectomy, the study found that despite a greater extent of cancer on biopsy, cases with and those without PNI on biopsy showed no significant difference in surgical margin involvement or organ-confined disease. Based on the latter findings, in our institution, patients with PNI who meet criteria for active surveillance are not excluded from this treatment option.65
Vascular invasion is rarely seen on needle biopsy and its significance on biopsy has not been studied, although typically, it is seen in the setting of other adverse histopathologic features (eFigs. 8.3 and 8.4). As vascular invasion at radical prostatectomy is an independent predictor of progression, its finding on needle biopsy is likely to be associated with a relatively poor prognosis.
USE OF NOMOGRAMS
Various nomograms have been developed to predict pathologic stage and postradical prostatectomy progression.66–71 These nomograms use preoperative variables such as Gleason score; clinical stage; serum PSA; and in a more recent study, the extent of cancer on biopsy to predict the risk of extraprostatic disease, seminal vesicle invasion, and lymph node metastases. Nomograms using the same preoperative variables have also been formulated to predict the risk and the outcome after radiotherapy.72 The most widely used of these are the Partin tables and Kattan nomograms, which are used by urologists, radiotherapists, oncologists, and patients to predict pathologic stage.71 The validity of these tables in part rests on accurate Gleason scoring, which is dealt with in the next chapter.
DIRECT STAGING ON NEEDLE BIOPSY
Skeletal muscle fibers admix within the normal prostate, especially distally (apically) and anteriorly. Recognition of this finding is important for two reasons. First, nonneoplastic prostate glands may be seen admixed with skeletal muscle fibers occasionally in both transurethral resection (TUR) material as well as on needle biopsy (Fig. 3.5) and should not be diagnosed as prostate carcinoma. Also, the finding of adenocarcinoma of the prostate admixed with skeletal muscle fibers is not diagnostic of EPE by carcinoma (Fig. 8.1, eFig. 8.5).73 Most patients with limited Gleason score 6 cancer involvement of skeletal muscle on biopsy have organ-confined disease and negative margins.73
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


