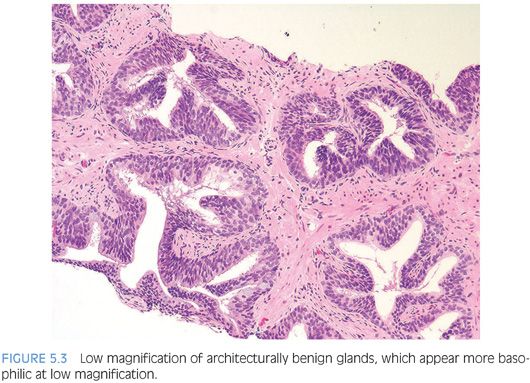
Low-grade PIN consists of preexisting benign prostate glands with minimal epithelial proliferation in terms of nuclear stratification, where nuclei are minimally enlarged without prominent nucleoli (Figs. 5.3 and 5.4, eFigs. 5.1 to 5.19). If one has to hunt at 40× lens in a gland for a rare cell with prominent nucleoli, then the case should not be diagnosed as high-grade PIN. It may represent low-grade PIN, but for the reasons described in the following case should merely be signed out as benign prostate tissue without mentioning PIN.

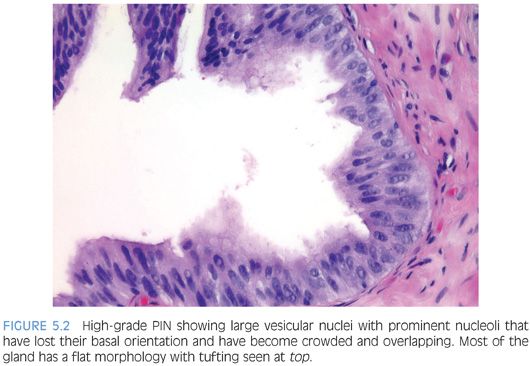
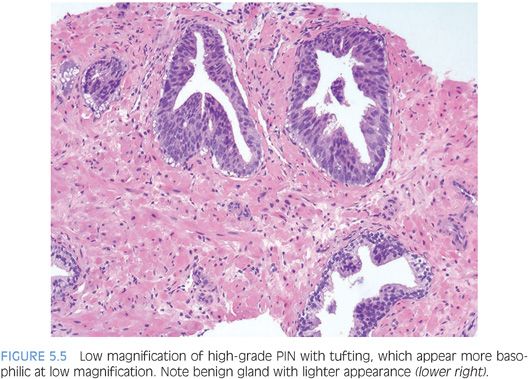
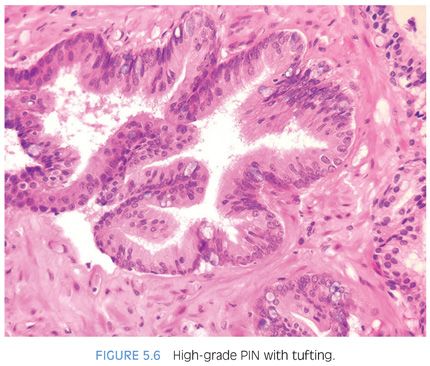
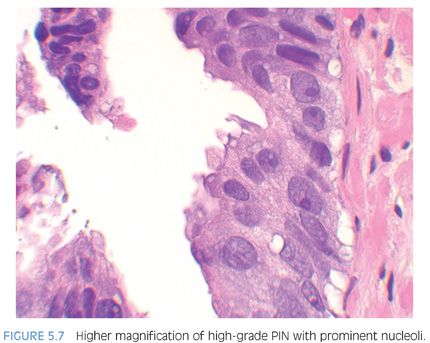
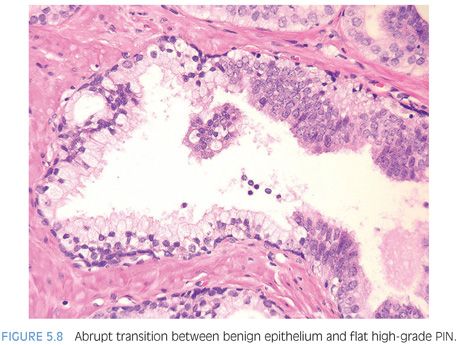
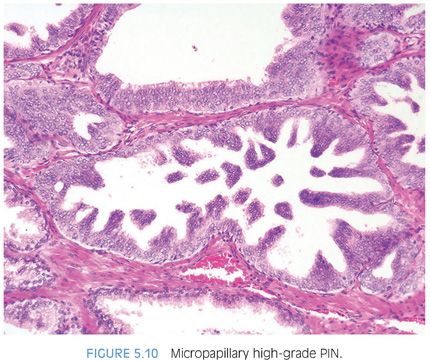
Although high-grade PIN is characterized by nuclear atypia, there is often accompanying architectural abnormalities. At low magnification, basophilic glands that are separated by a modest amount of stroma and have a normal overall architectural pattern characterize high-grade PIN (Fig. 5.5). These glands resemble benign glands in that they are large, branch, and typically have papillary and undulating luminal surfaces. (Fig. 5.6, eFigs. 5.20 and 5.21). Their basophilic appearance is due to a combination of features including nuclear enlargement, hyperchromasia, overlapping, and amphophilic cytoplasm (Fig. 5.7, eFig. 5.22). Flat high-grade PIN is characterized by nuclear atypia without significant epithelial hyperplasia (Fig. 5.8). The basal cell layer may or not be visible and the demarcation between atypical and normal nuclei is frequently abrupt. With more hyperplastic forms of high-grade PIN, nuclei become more piled up into tufts and can develop micropapillary projections (Figs. 5.9 and 5.10). These micropapillary projections are similar to those seen with micropapillary intraductal carcinoma of the breast, in that they are composed of tall epithelial buds, typically lacking fibrovascular cores.
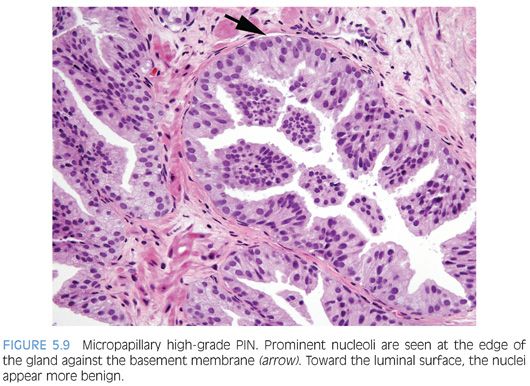
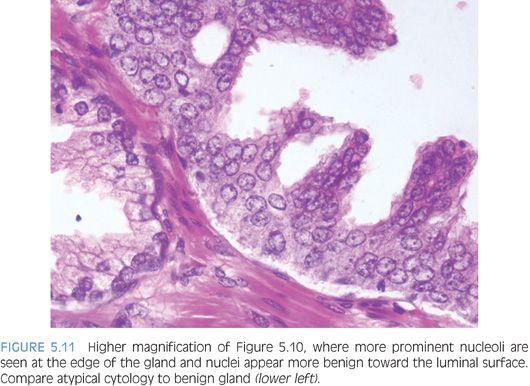
An interesting phenomenon in high-grade PIN is that nuclei toward the center of the gland tend to have more bland cytology, as compared to the nuclei peripherally located up against the basement membrane (Fig. 5.11, eFig. 5.23). Small cell–like change in the center of glands with high-grade PIN is an uncommon finding, which may be a more pronounced manifestation of this phenomenon where there is an abrupt transition between mostly centrally located populations of small cells with bland nuclei and scant cytoplasm and large more typical PIN cells with enlarged nuclei and prominent nucleoli at the periphery.5 The grade of PIN is assigned based on assessment of the nuclei peripherally located up against the basement membrane (eFigs. 5.24 to 5.82).
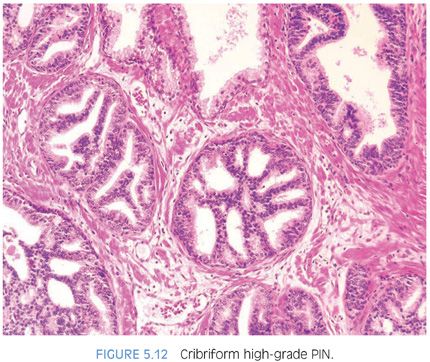
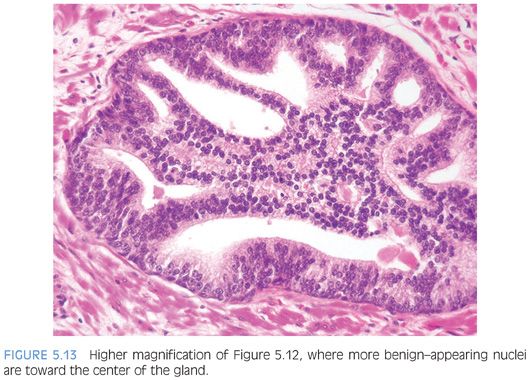
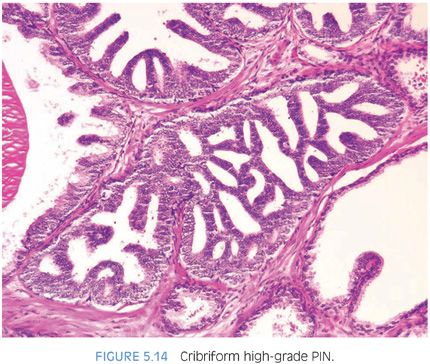
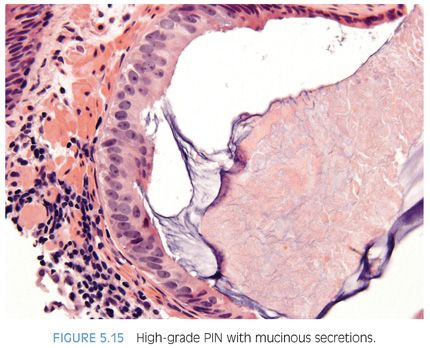
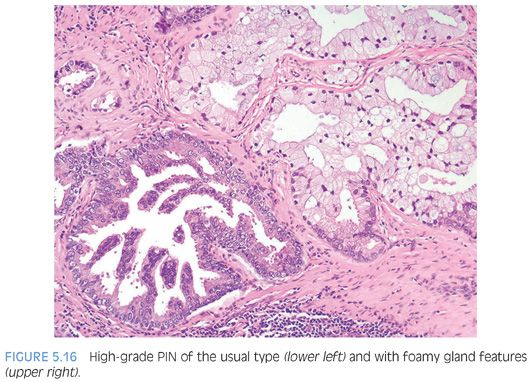
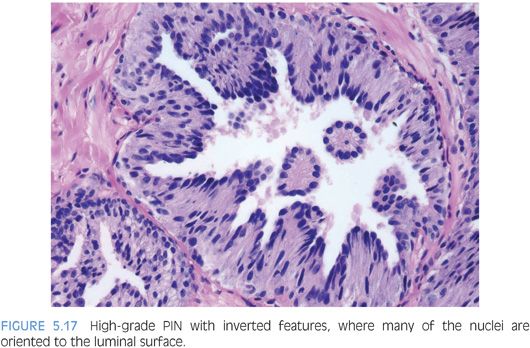
With further epithelial hyperplasia, more complex architectural patterns appear such as Roman bridge and cribriform formation (Figs. 5.12 to 5.14). The various patterns of PIN have been designated as flat (eFigs. 5.24 to 5.29), tufting (eFigs. 5.30 to 5.38), micropapillary (eFigs. 5.39 to 5.52, 5.80 to 5.82), and cribriform (eFigs. 5.53 to 5.67).6 Unusual subtypes of high-grade PIN include PIN with signet-ring features, small cell neuroendocrine PIN, PIN with mucinous features (Fig. 5.15, eFigs. 5.68 to 5.73), foamy PIN (Fig. 5.16, eFigs. 5.20, 5.74 to 5.79), PIN with inverted nuclei (Fig. 5.17, eFig. 5.21), and desquamating apoptotic PIN.5,7–11
EVIDENCE LINKING PROSTATIC INTRAEPITHELIAL NEOPLASIA TO CANCER
Comparing prostates with carcinoma to those without carcinoma, there is an increase in the size and number of high-grade PIN foci, in addition to an increased incidence of high-grade PIN.12,13 Also, with increasing amounts of high-grade PIN, there are a greater number of multifocal carcinomas. This observation follows if high-grade PIN is a precursor to some carcinomas, since with more precursor lesion, one would expect that there would be more early carcinomas. The finding of zones of high-grade PIN from which there appears to be budding off glands of carcinoma is further histologic evidence that high-grade PIN is a precursor to some prostate carcinomas. McNeal14 has designated these foci as “transitive glands,” although most other investigators prefer the term high-grade PIN with microinvasive carcinoma. Several studies have also noted an increase of high-grade PIN in the peripheral zone of the prostate, corresponding to the site of origin for most adenocarcinomas of the prostate.15 Also, the expression of various biomarkers in high-grade PIN is either (a) the same in high-grade PIN and carcinoma as opposed to benign prostate tissue or (b) intermediate between benign prostate tissue and carcinoma.12,15,16 All these findings would be expected if high-grade PIN is a precursor lesion to carcinoma of the prostate.
It has been shown that high-grade PIN is more closely related to peripheral, as opposed to transition zone cancers. Intermediate- or high-grade cancers are also more likely to be associated with high-grade PIN, compared to low-grade cancer. This weaker association of high-grade PIN to low-grade transition zone carcinomas is also supported by the histologic differences of high-grade PIN and transition zone carcinomas.17 Centrally located low-grade adenocarcinomas tend to have bland cytology, often lacking nuclear enlargement or nucleoli in contrast to high-grade PIN. Peripherally located intermediate-grade carcinomas often have identical cytologic features to those of high-grade PIN.
MIMICKERS OF PIN
Central Zone Histology
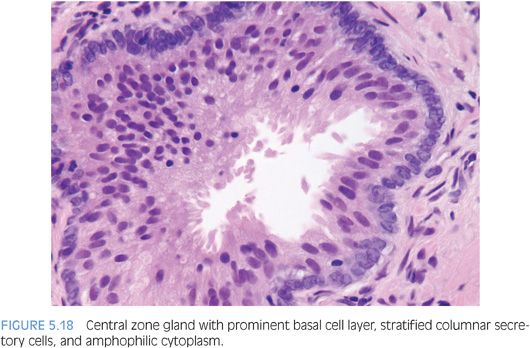
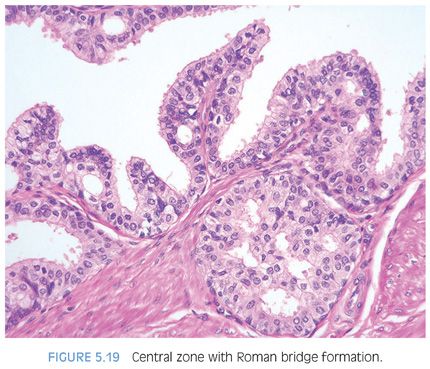
Glands within the central zone up at the base of the prostate are complex and large with numerous papillary infoldings and often are lined by tall pseudostratified epithelium with eosinophilic cytoplasm (eFigs. 5.83 to 5.90). Occasionally, a prominent basal cell layer surrounds these glands, whereas with high-grade PIN, the basal cell layer is typically indistinct (Fig. 5.18). Central zone glands are frequently overdiagnosed as high-grade PIN because their nuclei are piled up and they may be arranged in Roman bridge and cribriform glandular patterns.18 However, within these central zone glands, nuclei stream parallel to the glandular bridges, in contrast to the more rigid bridges seen in high-grade PIN (Fig. 5.19). Most important, central zone glands are distinguished from high-grade PIN by their lack of cytologic atypia.
Clear Cell Cribriform Hyperplasia
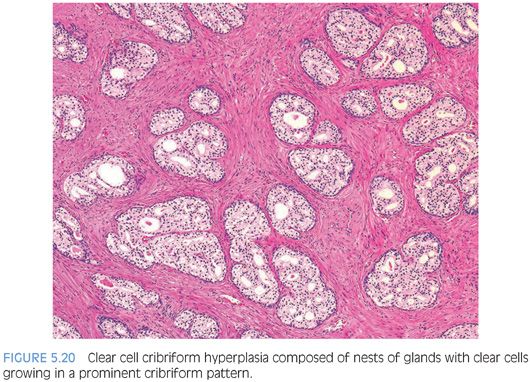
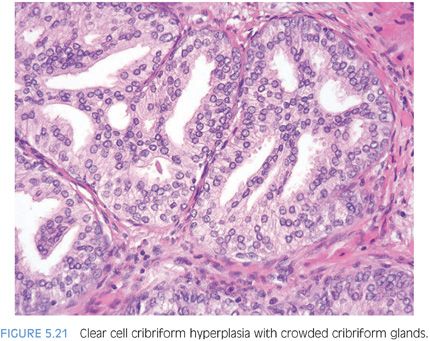
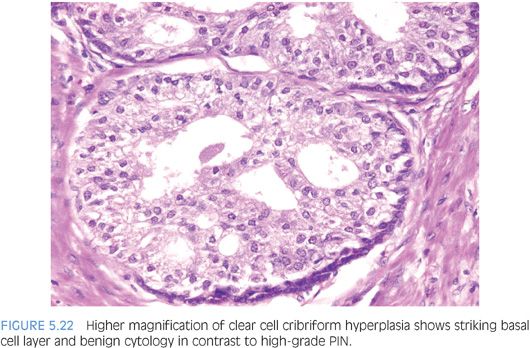
Clear cell cribriform hyperplasia consists of crowded cribriform glands, with clear cytoplasm sometimes growing as a nodule and in other instances more diffusely (Figs. 5.20 and 5.21, eFig. 5.91).19 The key distinguishing feature of clear cell cribriform hyperplasia from high-grade PIN is the lack of nuclear atypia. Furthermore, within a nodule of clear cell cribriform hyperplasia, at least some of the cribriform glands show a strikingly evident basal cell layer, which as noted earlier is not typically seen in high-grade PIN (Fig. 5.22). Immunostaining with antibodies to basal cells cannot distinguish between the two entities, since both have a patchy basal cell layer. Clear cell cribriform hyperplasia is typically located in the transition zone, whereas high-grade PIN predominates in the peripheral zone.
Basal Cell Hyperplasia
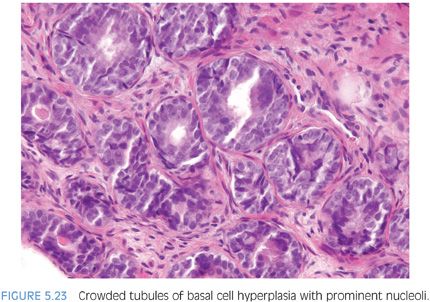
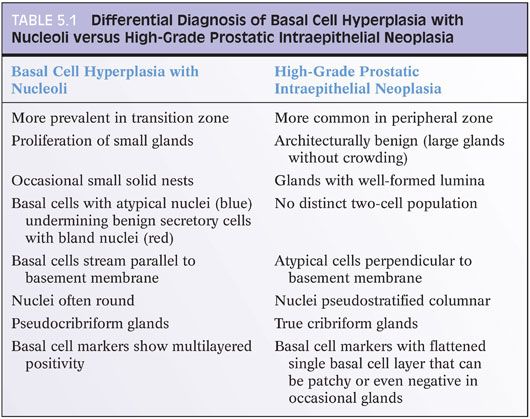
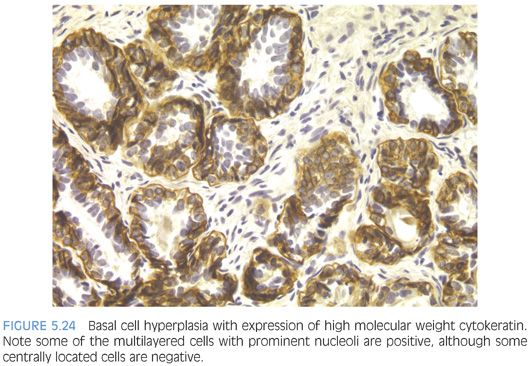
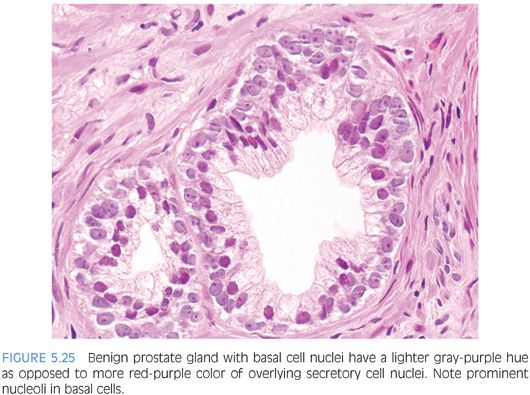
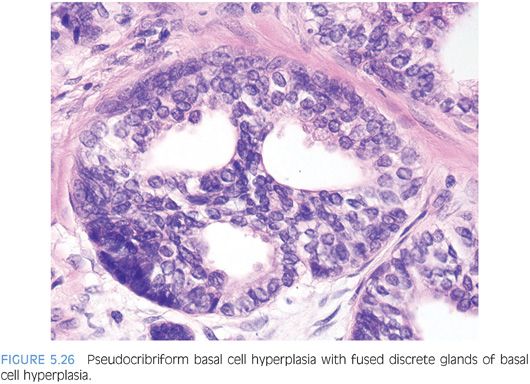
Otherwise typical basal cell hyperplasia may show prominent nucleoli along with mitotic activity20,21 (Fig. 5.23, eFigs. 5.92 to 5.103). Because of the prominent nucleoli, these lesions may be mistaken for high-grade PIN. Although occasionally, the distinction between these two entities may be difficult, usually they are distinct (Table 5.1). There is a proliferation of small round crowded glands in basal cell hyperplasia, whereas in high-grade PIN, the atypical nuclei fill preexisting larger benign glands that are separated from each other by a greater amount of stroma. The nuclei in basal cell hyperplasia tend to be round and at times form small solid basaloid nests. In contrast, the nuclei in high-grade PIN tend to be more pseudostratified and columnar and do not occlude the glandular lumina. Within areas of basal cell hyperplasia, atypical basal cells can be seen undermining overlying benign-appearing secretory cells. The basal cells in these foci tend to have a streaming morphology parallel to the basement membrane. High-grade PIN has full thickness cytologic atypia, with the nuclei oriented perpendicular to the basement membrane. In cases of full thickness basal cell hyperplasia, where an overlying secretory cell layer may not be apparent, the luminal cytoplasm is atrophic, whereas in high-grade PIN, the luminal cells have apical cytoplasm. An additional difference between the two entities is that most cases of basal cell hyperplasia are found in transurethral resection of the prostate (TURP) specimens, indicating growth in the transition zone, in contrast to high-grade PIN’s preferential location in the periphery of the prostate. Occasionally, when there are only a few glands to evaluate, such as on needle biopsy, immunohistochemical stains are needed to distinguish the two. Basal cell hyperplasia reveals high molecular weight cytokeratin or p63 positivity in multilayered nuclei, although in some cases, the more centrally located cells are not immunoreactive22 (Fig. 5.24, eFig. 5.104). In high-grade PIN, high molecular weight cytokeratin or p63 labels only flattened cytologically benign basal cells beneath the negatively stained atypical cells of PIN. Basal cells in benign glands, even when not proliferative, can also have prominent nucleoli and be mistaken for high-grade PIN (Fig. 5.25, eFigs. 5.105 to 5.109). In some institutions’ material, basal cell nuclei have a blue-gray appearance, in contrast to the red-violet hue of secretory cell nuclei. Basal cell hyperplasia may also be cribriform, further mimicking high-grade PIN (Fig 5.26, eFigs. 5.110 to 5.113). Whereas cribriform high-grade PIN glands represent a single glandular unit with punched out lumina, many of the glands within a focus of cribriform basal cell hyperplasia appeared as fused individual basal cell hyperplasia glands (pseudocribriform). The use of basal cell immunohistochemistry can help in difficult cases. Cribriform basal cell hyperplasia shows multilayered staining of the basal cells in some of the glands and a continuous layer of immunoreactivity. Cribriform high-grade PIN demonstrates an interrupted immunoreactive single cell layer of basal cells.
Acinar (Usual) Adenocarcinoma
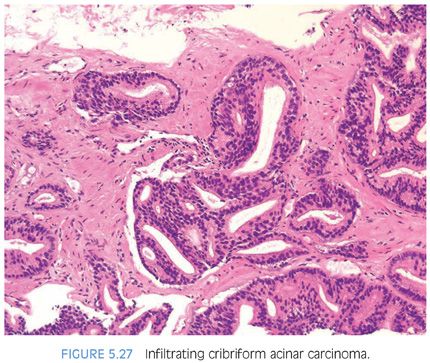
In some cases, it is difficult to distinguish cribriform high-grade PIN from cribriform Gleason pattern 3 adenocarcinoma.23 Although the distinction between cribriform Gleason pattern 3 cancer and cribriform high-grade PIN may be difficult, from a diagnostic standpoint, this is usually not critical. Almost always, when there are atypical cribriform glands, they are accompanied by small atypical infiltrating glands where the diagnosis of infiltrating tumor can be made (Fig. 5.27). Only when cytologically atypical cribriform glands are so large, back-to-back, or outside of the prostate that they are inconsistent with cribriform PIN should infiltrating cribriform carcinoma be diagnosed on hematoxylin and eosin (H&E)–stained sections in the absence of small atypical infiltrating glands (eFigs. 5.15 to 5.23). Immunohistochemistry with antibodies to basal cell markers can be used in difficult cases to differentiate these two entities. In the setting of numerous atypical cribriform glands, a negative reaction in all of the glands is diagnostic of carcinoma; positive staining, even if patchy, verifies the lesion as cribriform PIN. If there are only a few cribriform glands, negative for basal cell markers, it is not diagnostic of carcinoma. This results from the patchy nature with which basal cell markers label high-grade PIN and the recognition that even benign glands may occasionally not be labeled with these antibodies.22 When there is only one or a few small cribriform glands on needle biopsy without small glands of infiltrating carcinoma, these cases in general are not diagnostic of infiltrating carcinoma. Instead, the diagnosis is “focus of atypical cribriform glands” with a comment that “the distinction between cribriform high-grade PIN and cribriform carcinoma cannot be made with certainty, and a repeat biopsy is recommended” (eFigs. 5.124 to 5.132). This finding is associated with a higher association of cancer on repeat biopsy (50%) than the finding of high-grade PIN on a biopsy.24
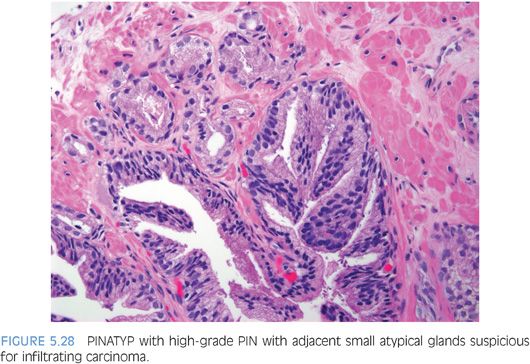
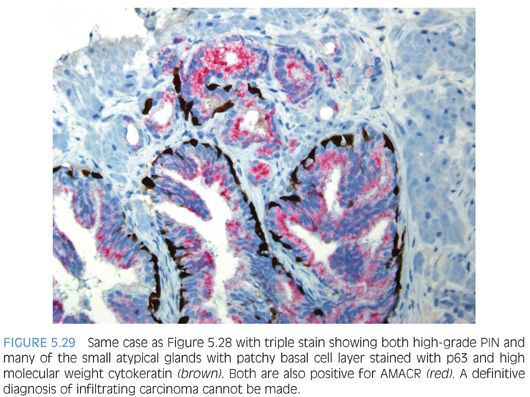
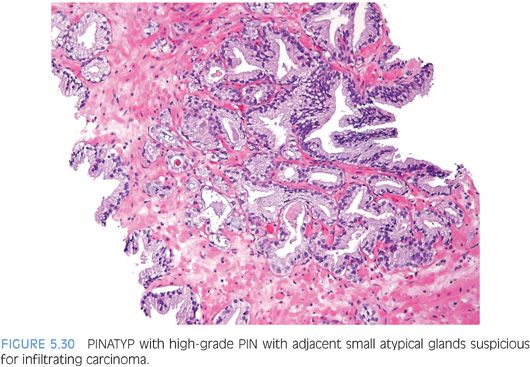
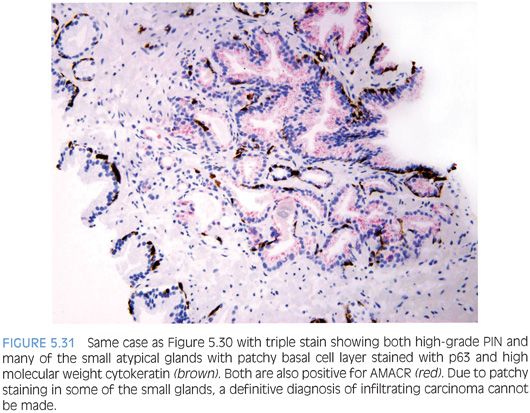
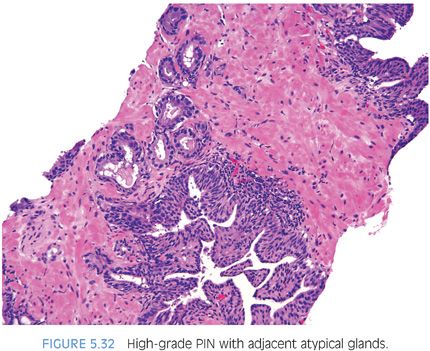
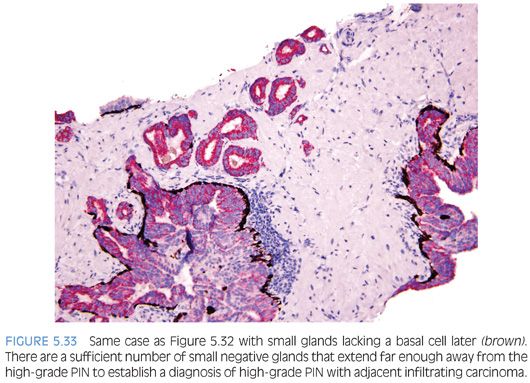
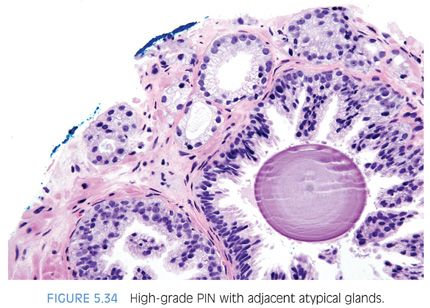
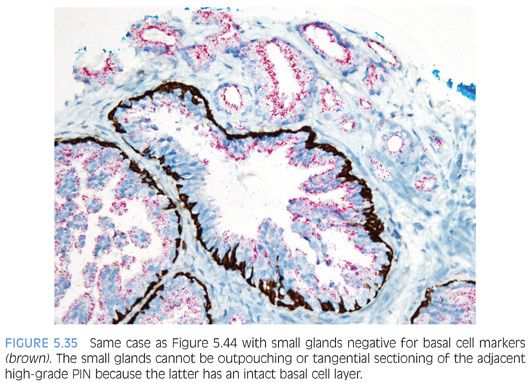
The other more common scenario where it is difficult to distinguish acinar adenocarcinoma from high-grade PIN is when there are a few atypical glands immediately adjacent to high-grade PIN.23 The differential diagnosis is whether these small glands represent tangential sectioning or outpouching off of the high-grade PIN glands or a small focus of carcinoma adjacent to the high-grade PIN (Figs. 5.28 to 5.31, eFigs. 5.133 to 5.149). We refer to these foci at PINATYP. A diagnosis of carcinoma can be rendered only if the small atypical glands are too numerous or too far away from the high-grade PIN glands to represent outpouching or tangential sectioning from the PIN glands (Figs. 5.32 and 5.33, eFigs. 5.150 to 5.159). In cases of PINATYP, the lack of basal cells in the small atypical glands can be construed as evidence that these glands represent infiltrating cancer only if there are more than a few such glands. As high-grade PIN glands can have discontinuous basal cells, one can envision tangential sections off PIN glands in which all cells would appear negative for basal cell markers, such that a few negative small atypical glands adjacent to PIN is not diagnostic of cancer.25 If the PIN gland has a continuous basal cell later, then one can more confidently diagnose adjacent small focus of carcinoma (Figs. 5.34 and 5.35). Some cases may have the appearance of PINATYP yet will be entirely negative for basal cell markers; these foci may be diagnostic of cancer if there are many glands that are not immunoreactive. One may also see classic high-grade PIN where some of the glands show the expected patchy basal cell layer and a few identical glands are negative for the basal cell markers; these cases we would still diagnose as high-grade PIN. Racemase does not differentiate between high-grade PIN and cancer because both typically express this antigen.26,27 Immunohistochemical expression of ERG in high-grade PIN has been reported in 5% to 16% of cases, whereas it is seen in approximately 50% of prostate cancers, such that ERG expression cannot be used to discriminate between the two entities.28,29
Ductal Adenocarcinoma
A difficult distinction is between high-grade PIN and ductal adenocarcinoma of the prostate (see Chapter 11) (Table 5.2).30–32 Ductal adenocarcinomas are aggressive tumors, often of advanced pathologic stage, and associated with a poor prognosis. Their distinction from cribriform PIN is critical. There are several features that distinguish these two lesions. Ductal adenocarcinomas are often centrally located in the periurethral region and sampled on TURP (eFigs. 5.160 and 5.161). PIN is uncommonly found within the periurethral region and infrequently seen on TURP. Ductal adenocarcinomas often contain true papillary fronds with well-established fibrovascular cores, whereas high-grade PIN more frequently reveals micropapillary fronds with tall columns of epithelium without fibrovascular stalks (eFig. 5.162). Ductal adenocarcinomas frequently contain comedonecrosis, which may be extensive (eFig. 161). High-grade PIN lacks comedonecrosis. Finally, ductal adenocarcinomas may consist of very large and/or back-to-back glands, whereas glands involved by PIN are of the size and distribution of benign glands (eFig. 5.163). The use of basal cell markers in this differential diagnosis may be problematic, as both high-grade PIN and ductal adenocarcinoma may display a patchy basal cell layer. However, absence of a basal cell layer in numerous glands rules out PIN.33
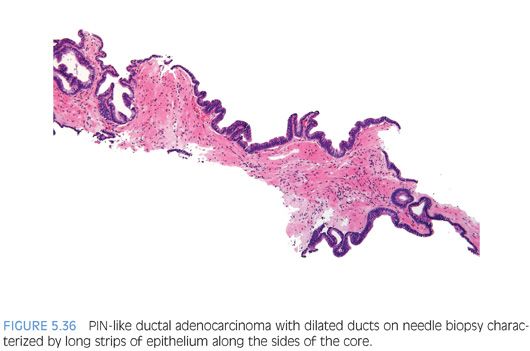
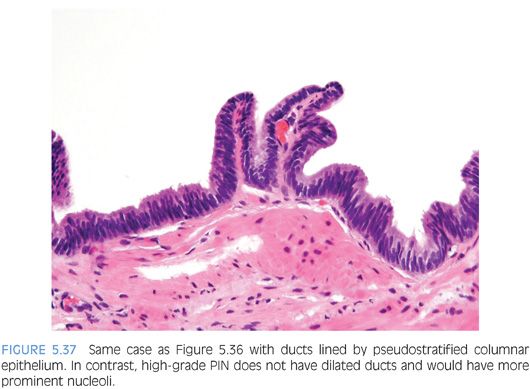
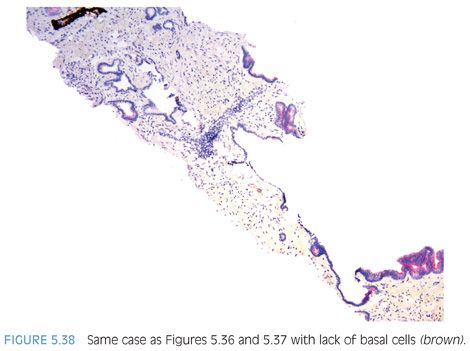
Although the most common forms of ductal adenocarcinoma mimic cribriform and micropapillary high-grade PIN, ductal adenocarcinoma may be composed of simple glands lined by stratified columnar epithelium with cytologic and architectural features of flat and tufting high-grade PIN. These PIN-like ductal cancers are distinguished from high-grade PIN either because the atypical glands are too crowded to represent high-grade PIN or there are too many atypical glands that are negative for basal cell markers to be consistent with high-grade PIN.34,35 Additional differences are in PIN-like ductal adenocarcinoma, many of the glands are lined by flat epithelium (an uncommon pattern in high-grade PIN) and the glands are often cystically dilated (Figs. 5.36 to 5.38, eFigs. 5.164 to 5.169) (see Chapter 11).
LOW-GRADE PROSTATIC INTRAEPITHELIAL NEOPLASIA: RISK OF CANCER ON A REBIOPSY
Low-grade PIN should not be documented as a finding in pathology reports for several reasons. First, there is a lack of reproducibility in its diagnosis even by uropathologists.23 McNeal’s36 quotation summarizes his consideration of low-grade PIN: “There is not a sharp line of demarcation between grade 1 dysplasia (i.e., low-grade PIN) and mild degrees of deviation from normal histology.” More important, there does not appear to be a higher risk of cancer following a diagnosis of low-grade PIN on a biopsy as compared to that following a benign diagnosis on a biopsy.37 If a diagnosis of low-grade PIN is rendered on a needle biopsy pathology report, it may lead to multiple unnecessary repeat biopsies, which result not only in added costs to the health care system but also to potential patient morbidity and concern.
HIGH-GRADE PROSTATIC INTRAEPITHELIAL NEOPLASIA ON BIOPSY: INCIDENCE, RISK OF CANCER ON A REBIOPSY, AND A REBIOPSY STRATEGY
High-Grade Prostatic Intraepithelial Neoplasia: Incidence on a Biopsy
There is marked variation within the literature on the incidence of high-grade PIN on needle biopsy, ranging from 0% to 24.6%.37 The mean reported incidence of high-grade PIN on needle biopsy is 7.6%, with a median value of 5.2%. There is no consistent trend in the incidence of high-grade PIN on biopsy as it relates to the type of practice setting (i.e., community hospital, commercial laboratory, academic institution). Although one might expect that greater sampling of the prostate would increase the likelihood of identifying high-grade PIN on needle biopsy, there does not appear to be a relationship between the number of cores sampled and the incidence of high-grade PIN on needle biopsy. There is also no trend for the reported incidence of high-grade PIN over time.
There are several potential explanations for the observed variation in the incidence of high-grade PIN. The hallmark distinguishing low- and high-grade PIN is the presence of prominent nucleoli. Beyond this relatively vague definition, there is much latitude for interpretation. There are no standard criteria defining how prominent or how frequent the nucleoli must be before they are sufficient to warrant a diagnosis of high-grade PIN; as noted at the beginning of this chapter, we use nucleoli visible with the 20× lens as the threshold for diagnosing high-grade PIN. In a survey of urologic pathologists, the majority diagnosed high-grade PIN if any visible nucleoli were present.38 However, a third of urologic pathologists required nucleoli in at least 10% of the cells in the gland. Allam et al.39 demonstrated that some general pathologists reviewing a prostate biopsy considered nucleoli were sufficiently prominent to render a diagnosis of high-grade PIN, whereas others looking at the same case felt that the nucleoli were not large enough. Another problem noted by Allam was that while partial involvement of the gland was sufficient for the diagnosis of high-grade PIN for some of the observers, for others it was not. Even within partially involved glands, the number of nucleoli sufficient for the diagnosis of high-grade PIN varied among observers. In another study, 75% of cases diagnosed as high-grade PIN by outside pathologists sent to a genitourinary pathology expert at the request of either the patient or urologist were confirmed as high-grade PIN.40
Technical factors relating to the processing of needle biopsy specimens can also contribute to the reported variability in the incidence of high-grade PIN on biopsy. In several studies with higher incidences of high-grade PIN, nonstandard fixatives were used to preserve specimens, which tend to enhance nuclear detail and nucleolar prominence. Technical factors can also potentially explain lower incidences of high-grade PIN reported in the literature. In cases with suboptimal microtomy, sections are thick, resulting in increased uptake of dyes used to stain the tissue that can obscure fine nuclear detail. The resulting difficulty in visualizing nucleoli would lead to a lower reported incidence of high-grade PIN.
Although one study has reported that African American men have a higher incidence of high-grade PIN than Caucasian men, this by itself is unlikely an explanation for the marked variation seen in the literature.41 An incidence of high-grade PIN between 4% and 8% is a reasonable figure with which a pathologist can benchmark his or her practice. Incidences markedly lower or higher raise the question of either underdiagnosing or overdiagnosing high-grade PIN on needle biopsy.
High-Grade Prostatic Intraepithelial Neoplasia on Biopsy: Overall Risk of Cancer
The most important clinical aspect of high-grade PIN is the risk of cancer for a patient who has been diagnosed with high-grade PIN on needle biopsy. The initial studies evaluating this question in the early 1990s on relatively few cases reported a high percentage of cancer following the diagnosis of high-grade PIN, with most studies citing the risk of cancer to be around 50%.37 Analyzing more contemporary data published in the year 2000 or later, 23 studies report that the median risk of cancer following a diagnosis of high-grade PIN on biopsy is only 21%.37
In order to assess the significance of high-grade PIN on needle biopsy, it is not enough to know the risk of cancer on rebiopsy. However, one must compare the risk of cancer on repeat biopsy following a high-grade PIN diagnosis to the risk of cancer on repeat biopsy following a benign diagnosis. It is widely recognized that even with more extensive sampling of the prostate, a certain percentage of cancers will remain undetected due to sampling error. The median risk of finding cancer in a repeat biopsy following a benign diagnosis is 19%,37 which is not appreciably different than the risk following a diagnosis of high-grade PIN. Of nine publications that have examined in the same study the risk of cancer on rebiopsy following a needle biopsy diagnosis of high-grade PIN to that following a benign diagnosis, seven showed no statistically significant difference. Subsequent studies have in general shown similar findings, including a study using saturation biopsy.42,43
High-Grade Prostatic Intraepithelial Neoplasia on Biopsy: Risk of Cancer Stratified by Clinical Predictors
As several studies have shown that high-grade PIN by itself does not elevate serum prostate-specific antigen (PSA) levels, one might hypothesize that an elevated serum PSA level in a man with high-grade PIN is more likely the result of an associated carcinoma.44,45 However, most studies have found that serum PSA levels are not predictive of cancer on rebiopsy.37 Several studies have also examined PSA velocity, free to total PSA, and PSA density with most showing no correlation with risk of cancer on rebiopsy. The results of digital rectal exam, transrectal ultrasound, age, and family history of prostate cancer are also not influential in predicting which men with high-grade PIN on needle biopsy will have carcinoma on rebiopsy.37 In summary, there does not appear to be any clinical parameter that helps to identify men with high-grade PIN on a needle biopsy who are more likely to have cancer on rebiopsy.
High-Grade Prostatic Intraepithelial Neoplasia on a Biopsy: Risk of Cancer Stratified by Pathologic Predictors
In contemporary studies, the one feature that has correlated with an increased risk of cancer on rebiopsy is the number of cores involved with high-grade PIN. In the largest study by Merrimen et al.,46,47 there was approximately a 30% chance of cancer on rebiopsy following the diagnosis of high-grade PIN in 2 or more cores. Contemporary smaller studies have also reported an increased risk of cancer on rebiopsy with increased numbers of involved cores. Some of these studies have used a higher cutoff in terms of the number of cores with high-grade PIN (i.e., ≥3 or ≥4) where 2 or more cores with high-grade PIN may not have been statistically significantly associated with an increased risk of subsequent cancer due to the smaller number of cases.48–50 Other studies have found an increased risk of cancer with bilateral high-grade PIN or increased proportion of the cores involved by high-grade PIN.51,52
Most studies have not found that the morphology of high-grade PIN (flat vs. tufting vs. micropapillary vs. cribriform) can predict which high-grade PIN lesions are at greater risk of being associated with carcinoma on repeat biopsy.53–56 A caveat to the previous conclusion is that pathologists must use strict criteria to differentiate cribriform high-grade PIN and cribriform carcinoma.
Racemase (alpha-methylacyl coenzyme A racemase [AMACR]) is upregulated both in carcinoma and high-grade PIN and not in benign prostate tissue.26 In one study utilizing radical prostatectomy specimens, high-grade PIN lesions adjacent to carcinoma had more AMACR overexpression (56%) than high-grade PIN lesions away from cancer (14%).27 Further studies are needed to determine whether AMACR expression of high-grade PIN lesions on needle biopsy could help predict which patients are more likely to have cancer.
There are conflicting studies as to the relation of ERG on high-grade PIN biopsies and the subsequent risk of cancer. In the study by He et al.28 from the Cleveland Clinic, positive ERG immunohistochemical expression was not associated with an increased cancer detection in subsequent repeat biopsies. In the other study to assess this marker, a greater number of patients (56 of 59, 94.9%) with an ERG rearrangements rate of 1.6% or greater on initial biopsy were diagnosed with prostate cancer during repeat biopsy follow-ups as compared with those (5 of 103, 4.9%) with an ERG rearrangements rate of less than 1.6% (P <0.001) using fluorescent in situ hybridization (FISH). Although there was a significant positive correlation between the ERG rearrangement rate by FISH and the ERG protein expression, the authors did not state that using ERG immunohistochemistry was useful to predict which PIN lesions on biopsy were associated with cancer on rebiopsy.57
High-Grade Prostatic Intraepithelial Neoplasia on Biopsy: Risk of Cancer Stratified by Number of Cores Sampled
A major contributing factor to the decreased incidence of cancer following a diagnosis of high-grade PIN on needle biopsy in the contemporary era relates to increased needle core biopsy sampling. Relatively poor sampling (i.e., sextant biopsies) on the initial biopsy misses associated cancers resulting in only high-grade PIN on the initial biopsy. With rebiopsy, some of these initially missed cancers are detected, yielding a high risk of cancer following a sextant needle biopsy diagnosis of high-grade PIN. Sampling more extensively on the initial biopsy detects many associated cancers, such that when only high-grade PIN is found, they often truly represent isolated high-grade PIN; therefore, rebiopsy even with good sampling does not detect many additional cancers.58
High-Grade Prostatic Intraepithelial Neoplasia on Biopsy: Repeat Biopsy Strategy and Technique
Recommendations for following men with high-grade PIN on biopsy have varied widely. It is the recommendation of these reviewers that men do not need a routine repeat needle biopsy within the first year following the diagnosis of a single core with high-grade PIN in the absence of other clinical indicators of cancer. Unifocal high-grade PIN is not associated with an increased risk of cancer on rebiopsy that is much different than men with a benign diagnosis on the initial biopsy. If there are 2 or more cores with high-grade PIN, the risk of cancer is sufficiently high (30% to 40%), justifying rebiopsy within 6 months.
Studies from New York University Medical Center have raised the question of whether repeat biopsy should be performed several years following a high-grade PIN diagnosis because men with high-grade PIN have a continued risk of developing prostate cancer during long-term follow-up.59–61 However, these studies did not report the number of cores with high-grade PIN. Data from Cleveland Clinic reported estimated cancer rates of 3.6%, 12.5%, and 22.4% for patients with a benign diagnosis; 4.4%, 14.7%, and 26.1% for patients with unifocal high-grade PIN; and 9.1%, 29.0%, and 47.8% for patients with multifocal high-grade PIN, at 1, 3, and 5 years, respectively.52 At 3 and 5 years’ follow-up after the initial biopsy, there were no significant differences in the risk of cancer between an initial benign diagnosis and a diagnosis of unifocal high-grade PIN. It is therefore questionable whether men with a single core with high-grade PIN ever need a repeat biopsy.
A limited number of studies have evaluated where cancer is found on rebiopsy. The risk of cancer being found in the contralateral lobe to where high-grade PIN is initially diagnosed ranges in studies from 10% to 40% with an average of 30%. The risk of finding cancer in the same sextant site where high-grade PIN was initially found is reported to be on average 55%.62–65 In one of these studies, there was a 74% probability of cancer on repeat biopsy being in the same sextant site as the initial high-grade PIN and an 89% risk of cancer being in the same and adjacent sextant sites as the initial high-grade PIN.64 If cancer was randomly distributed throughout the prostate in men with high-grade PIN on needle biopsy, one would expect the risk of cancer in any given sextant site to be one-sixth (16.7%). Based on these data, it is recommended that if rebiopsy is performed for high-grade PIN, sampling should be proportionally more in the region of the original high-grade PIN site and in adjacent sites, although the entire prostate should be sampled.
High-Grade Prostatic Intraepithelial Neoplasia: Significance on Transurethral Resection of the Prostate Material
The significance of finding high-grade PIN on TURP is more controversial. Whereas two studies have found that high-grade PIN on TURP places an individual at higher risk for the subsequent detection of cancer, a long-term study from Norway demonstrated no association between the presence of high-grade PIN on TURP and the incidence of subsequent cancer.66–68 In a younger man with high-grade PIN on TURP, we would recommend that needle biopsies be performed to rule out a peripheral zone cancer. In an older man without elevated serum PSA levels, clinical follow-up is probably sufficient. When high-grade PIN is found on TURP, some pathologists recommend sectioning deeper into the corresponding block and most pathologists recommend processing the entire specimen.67
High-Grade Prostatic Intraepithelial Neoplasia on Biopsy: Findings at Subsequent Radical Prostatectomy
Men who are found to have high-grade PIN then cancer on repeat biopsy needle who subsequently undergo radical prostatectomy have more favorable pathologic stage than cases where cancer is diagnosed on the first biopsy. Tumor size at radical prostatectomy is also small in cases where the initial diagnosis is high-grade PIN followed by cancer on repeat needle biopsy. These findings likely reflect cancers associated with high-grade PIN, in which the cancers were missed on the initial biopsy as a result of smaller size.69
INTRADUCTAL CARCINOMA OF THE PROSTATE
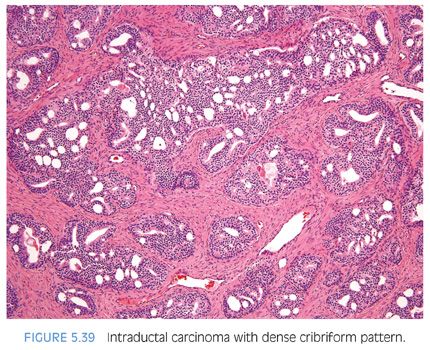
Intraductal carcinoma of the prostate (IDC-P) in radical prostatectomy specimens is described as an atypical glandular lesion that spans the entire lumen of prostatic ducts or acini while the normal architecture of ducts or acini is still maintained (Figs. 5.39 and 5.40).70–74 Rarely, IDC-P may be identified on biopsy material in the absence of infiltrating carcinoma. Our definition of IDC-P on needle biopsy was derived to identify objective morphologic criteria that either architecturally or cytologically clearly exceed those seen in high-grade PIN (Table 5.3) 75 (Figs. 5.41 to 5.49, eFigs. 5.170 to 5.182). A dense cribriform pattern was one where there were overtly more solid than luminal areas (i.e., ratio of solid to luminal areas >70%). IDC-P may also rarely show small cell–like change, which may also be seen in high-grade PIN and infiltrating carcinoma (Fig. 5.50).5 IDC-P on prostate biopsies is frequently associated with high-grade cancer and poor prognostic parameters at radical prostatectomy. In the largest study, of 21 radical prostatectomies, there was extraprostatic extension without seminal vesicle invasion in 38% and seminal vesicle invasion in 13% of cases. Organ-confined disease was present in 38% of cases and intraductal carcinoma without identifiable invasive cancer was seen in 10%. Average Gleason score was 7.9.76 These findings support prior studies that IDC-P represents an advanced stage of tumor progression with intraductal spread of tumor in most cases. However, in some cases, IDC-P is precursor distinct and more likely to be associated with aggressive prostate cancer than high-grade PIN.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


