61
Dermatologic Pharmacology
CASE STUDY
A 22-year-old woman presents with a complaint of worsening psoriasis. She has a strong family history of the disease and has had lesions on her scalp and elbows for several years. She recently noted new lesions developing on her knees and the soles of her feet. She has been using topical over-the-counter hydrocortisone cream but admits that this treatment does not seem to help. What therapeutic options are available for the treatment of this chronic disease?
Diseases of the skin offer special opportunities to the clinician. In particular, the topical administration route is especially appropriate for skin diseases, although some dermatologic diseases respond as well or better to drugs administered systemically.
The general pharmacokinetic principles governing the use of drugs applied to the skin are the same as those involved in other routes of administration (see Chapters 1 and 3). Although often depicted as a simple three-layered structure, human skin is a complex series of diffusion barriers (Figure 61–1). Quantitation of the flux of drugs and drug vehicles through these barriers is the basis for pharmacokinetic analysis of dermatologic therapy, and techniques for making such measurements are rapidly increasing in number and sensitivity.
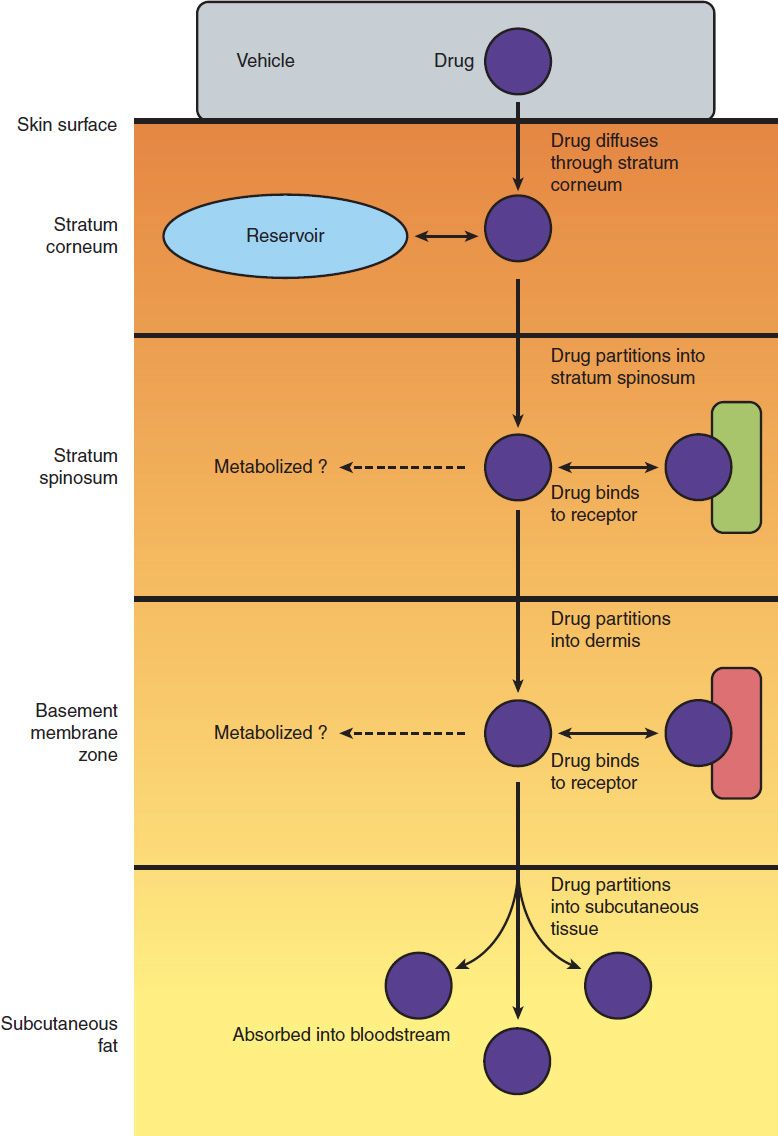
FIGURE 61–1 Schematic diagram of percutaneous absorption. (Redrawn from Orkin M, Maibach HI, Dahl MV: Dermatology. Appleton & Lange, 1991.)
Major variables that determine pharmacologic response to drugs applied to the skin include the following:
1. Regional variation in drug penetration: For example, the scrotum, face, axilla, and scalp are far more permeable than the forearm and may require less drug for equivalent effect.
2. Concentration gradient: Increasing the concentration gradient increases the mass of drug transferred per unit time, just as in the case of diffusion across other barriers (see Chapter 1). Thus, resistance to topical corticosteroids can sometimes be overcome by use of higher concentrations of drug.
3. Dosing schedule: Because of its physical properties, the skin acts as a reservoir for many drugs. As a result, the “local half-life” may be long enough to permit once-daily application of drugs with short systemic half-lives. For example, once-daily application of corticosteroids appears to be just as effective as multiple applications in many conditions.
4. Vehicles and occlusion: An appropriate vehicle maximizes the ability of the drug to penetrate the outer layers of the skin. In addition, through their physical properties (moistening or drying effects), vehicles may themselves have important therapeutic effects. Occlusion (application of a plastic wrap to hold the drug and its vehicle in close contact with the skin) is extremely effective in maximizing efficacy.
 REACTIONS TO DERMATOLOGIC MEDICATIONS
REACTIONS TO DERMATOLOGIC MEDICATIONS
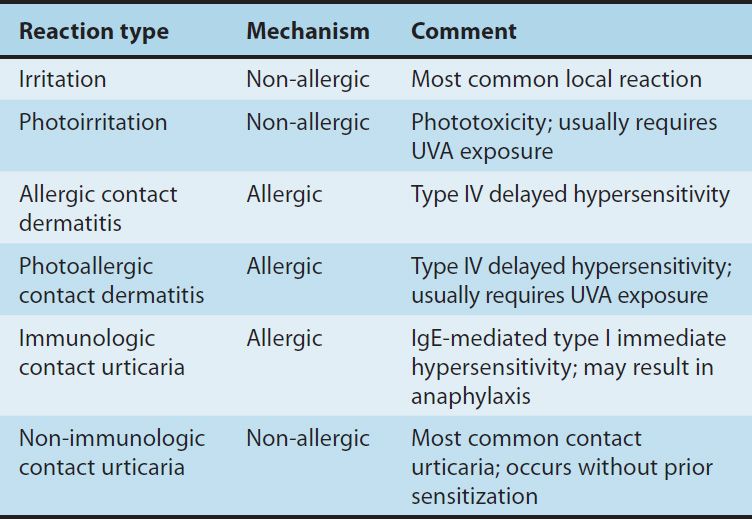
The skin reacts to many systemic medications with a variety of symptom-generating responses. In addition, some dermatologic medications themselves cause skin reactions. The major types of reactions are summarized in Table 61–1.
TABLE 61–1 Local cutaneous reactions to topical medications.
 DERMATOLOGIC VEHICLES
DERMATOLOGIC VEHICLES
Topical medications usually consist of active ingredients incorporated in a vehicle that facilitates cutaneous application. Important considerations in vehicle selection include the solubility of the active agent in the vehicle; the rate of release of the agent from the vehicle; the ability of the vehicle to hydrate the stratum corneum, thus enhancing penetration; the stability of the therapeutic agent in the vehicle; and interactions, chemical and physical, of the vehicle, stratum corneum, and active agent.
Depending upon the vehicle, dermatologic formulations may be classified as tinctures, wet dressings, lotions, gels, aerosols, powders, pastes, creams, foams, and ointments. The ability of the vehicle to retard evaporation from the surface of the skin increases in this series, being least in tinctures and wet dressings and greatest in ointments. In general, acute inflammation with oozing, vesiculation, and crusting is best treated with drying preparations such as tinctures, wet dressings, and lotions, whereas chronic inflammation with xerosis, scaling, and lichenification is best treated with more lubricating preparations such as creams and ointments. Tinctures, lotions, gels, foams, and aerosols are convenient for application to the scalp and hairy areas. Emulsified vanishing-type creams may be used in intertriginous areas without causing maceration.
Emulsifying agents provide homogeneous, stable preparations when mixtures of immiscible liquids such as oil-in-water creams are compounded. Some patients develop irritation from these agents. Substituting a preparation that does not contain them or using one containing a lower concentration may resolve the problem.
 ANTIBACTERIAL AGENTS
ANTIBACTERIAL AGENTS
TOPICAL ANTIBACTERIAL PREPARATIONS
Topical antibacterial agents may be useful in preventing infections in clean wounds, in the early treatment of infected dermatoses and wounds, in reducing colonization of the nares by staphylococci, in axillary deodorization, and in the management of acne vulgaris. The efficacy of antibiotics in these topical applications is not uniform. The general pharmacology of the antimicrobial drugs is discussed in Chapters 43–51.
Some topical anti-infectives contain corticosteroids in addition to antibiotics. There is no convincing evidence that topical corticosteroids inhibit the antibacterial effect of antibiotics when the two are incorporated in the same preparation. In the treatment of secondarily infected dermatoses, which are usually colonized with streptococci, staphylococci, or both, combination therapy may prove superior to corticosteroid therapy alone. Antibiotic-corticosteroid combinations may be useful in treating diaper dermatitis, otitis externa, and impetiginized eczema.
The selection of a particular antibiotic depends upon the diagnosis and, when appropriate, in vitro culture and sensitivity studies of clinical samples. The pathogens isolated from most infected dermatoses are group A β-hemolytic streptococci, Staphylococcus aureus, or both. The pathogens present in surgical wounds will be those resident in the environment. Information about regional patterns of drug resistance is therefore important in selecting a therapeutic agent. Prepackaged topical antibacterial preparations that contain multiple antibiotics are available in fixed dosages well above the therapeutic threshold. These formulations offer the advantages of efficacy in mixed infections, broader coverage for infections due to undetermined pathogens, and delayed microbial resistance to any single component antibiotic.
BACITRACIN & GRAMICIDIN
Bacitracin and gramicidin are peptide antibiotics, active against gram-positive organisms such as streptococci, pneumococci, and staphylococci. In addition, most anaerobic cocci, neisseriae, tetanus bacilli, and diphtheria bacilli are sensitive. Bacitracin is compounded in an ointment base alone or in combination with neomycin, polymyxin B, or both. The use of bacitracin in the anterior nares may temporarily decrease colonization by pathogenic staphylococci. Microbial resistance may develop following prolonged use. Bacitracin-induced contact urticaria syndrome, including anaphylaxis, occurs rarely. Allergic contact dermatitis occurs frequently, and immunologic allergic contact urticaria rarely. Bacitracin is poorly absorbed through the skin, so systemic toxicity is rare.
Gramicidin is available only for topical use, in combination with other antibiotics such as neomycin, polymyxin, bacitracin, and nystatin. Systemic toxicity limits this drug to topical use. The incidence of sensitization following topical application is exceedingly low in therapeutic concentrations.
MUPIROCIN
Mupirocin (pseudomonic acid A) is structurally unrelated to other currently available topical antibacterial agents. Most gram-positive aerobic bacteria, including methicillin-resistant S aureus (MRSA), are sensitive to mupirocin (see Chapter 50). It is effective in the treatment of impetigo caused by S aureus and group A β-hemolytic streptococci.
Intranasal mupirocin ointment for eliminating nasal carriage of S aureus may be associated with irritation of mucous membranes caused by the polyethylene glycol vehicle. Mupirocin is not appreciably absorbed systemically after topical application to intact skin.
RETAPAMULIN
Retapamulin is a semisynthetic pleromutilin derivative effective in the treatment of uncomplicated superficial skin infection caused by group A β-hemolytic streptococci and S aureus, excluding MRSA. Topical retapamulin 1% ointment is indicated for use in adult and pediatric patients, 9 months or older, for the treatment of impetigo. Recommended treatment regimen is twice-daily application for 5 days. Retapamulin is well tolerated with only occasional local irritation of the treatment site. To date only four cases of allergic contact dermatitis have been reported.
POLYMYXIN B SULFATE
Polymyxin B is a peptide antibiotic effective against gram-negative organisms, including Pseudomonas aeruginosa, Escherichia coli, enterobacter, and klebsiella. Most strains of proteus and serratia are resistant, as are all gram-positive organisms. Topical preparations may be compounded in either a solution or ointment base. Numerous prepackaged antibiotic combinations containing polymyxin B are available. Detectable serum concentrations are difficult to achieve from topical application, but the total daily dose applied to denuded skin or open wounds should not exceed 200 mg in order to reduce the likelihood of neurotoxicity and nephrotoxicity. Allergic contact dermatitis to topically applied polymyxin B sulfate is uncommon.
NEOMYCIN & GENTAMICIN
Neomycin and gentamicin are aminoglycoside antibiotics active against gram-negative organisms, including E coli, proteus, klebsiella, and enterobacter. Gentamicin generally shows greater activity against P aeruginosa than neomycin. Gentamicin is also more active against staphylococci and group A β-hemolytic streptococci. Widespread topical use of gentamicin, especially in a hospital environment, should be avoided to slow the appearance of gentamicin-resistant organisms.
Neomycin is available in numerous topical formulations, both alone and in combination with polymyxin, bacitracin, and other antibiotics. It is also available as a sterile powder for topical use. Gentamicin is available as an ointment or cream.
Topical application of neomycin rarely results in detectable serum concentrations. However, in the case of gentamicin, serum concentrations of 1–18 mcg/mL are possible if the drug is applied in a water-miscible preparation to large areas of denuded skin, as in burned patients. Both drugs are water-soluble and are excreted primarily in the urine. Renal failure may permit the accumulation of these antibiotics, with possible nephrotoxicity, neurotoxicity, and ototoxicity.
Neomycin frequently causes allergic contact dermatitis, particularly if applied to eczematous dermatoses or if compounded in an ointment vehicle. When sensitization occurs, cross-sensitivity to streptomycin, kanamycin, paromomycin, and gentamicin is possible.
TOPICAL ANTIBIOTICS IN ACNE
Several systemic antibiotics that have traditionally been used in the treatment of acne vulgaris have been shown to be effective when applied topically. Currently, four antibiotics are so utilized: clindamycin phosphate, erythromycin base, metronidazole, and sulfacetamide. The effectiveness of topical therapy is less than that achieved by systemic administration of the same antibiotic. Therefore, topical therapy is generally suitable only in mild to moderate cases of inflammatory acne.
Clindamycin
Clindamycin has in vitro activity against Propionibacterium acnes; this has been postulated as the mechanism of its beneficial effect in acne therapy. Approximately 10% of an applied dose is absorbed, and rare cases of bloody diarrhea and pseudomembranous colitis have been reported following topical application. The hydroalcoholic vehicle and foam formulation (Evoclin) may cause drying and irritation of the skin, with complaints of burning and stinging. The water-based gel and lotion formulations are well tolerated and less likely to cause irritation. Allergic contact dermatitis is uncommon. Clindamycin is also available in fixed-combination topical gels with benzoyl peroxide (Acanya, BenzaClin, Duac), and with tretinoin (Ziana).
Erythromycin
In topical preparations, erythromycin base rather than a salt is used to facilitate penetration. The mechanism of action of topical erythromycin in inflammatory acne vulgaris is unknown but is presumed to be due to its inhibitory effects on P acnes. One of the possible complications of topical therapy is the development of antibiotic-resistant strains of organisms, including staphylococci. If this occurs in association with a clinical infection, topical erythromycin should be discontinued and appropriate systemic antibiotic therapy started. Adverse local reactions to erythromycin solution may include a burning sensation at the time of application and drying and irritation of the skin. The topical water-based gel is less drying and may be better tolerated. Allergic contact dermatitis is uncommon. Erythromycin is also available in a fixed combination preparation with benzoyl peroxide (Benzamycin) for topical treatment of acne vulgaris.
Metronidazole
Topical metronidazole is effective in the treatment of rosacea. The mechanism of action is unknown, but it may relate to the inhibitory effects of metronidazole on Demodex brevis; alternatively, the drug may act as an anti-inflammatory agent by direct effect on neutrophil cellular function. Oral metronidazole has been shown to be a carcinogen in susceptible rodent species, and topical use during pregnancy and by nursing mothers and children is therefore not recommended.
Adverse local effects of the water-based gel formulation (MetroGel) include dryness, burning, and stinging. Less drying formulations may be better tolerated (MetroCream, MetroLotion, and Noritate cream). Caution should be exercised when applying metronidazole near the eyes to avoid excessive tearing.
Sodium Sulfacetamide
Topical sulfacetamide is available alone as a 10% lotion (Klaron) and as a 10% wash (Ovace), and in several preparations in combination with sulfur for the treatment of acne vulgaris and acne rosacea. The mechanism of action is thought to be inhibition of P acnes by competitive inhibition of p-aminobenzoic acid utilization. Approximately 4% of topically applied sulfacetamide is absorbed percutaneously, and its use is therefore contraindicated in patients having a known hypersensitivity to sulfonamides.
Dapsone
Topical dapsone is available as a 5% gel (Aczone) for the treatment of acne vulgaris. The mechanism of action is unknown. Topical use in patients with glucose-6-phosphate dehydrogenase (G6PD) deficiency has not been shown to cause clinically relevant hemolysis or anemia. However, a slight decrease in hemoglobin concentration was noted in patients with G6PD deficiency, suggestive of mild hemolysis. To date, serious adverse reactions associated with oral dapsone use as delineated in Chapter 47 have not been reported with topical use. Adverse local side effects include mild dryness, redness, oiliness, and skin peeling. Application of dapsone gel followed by benzoyl peroxide may result in a temporary yellow discoloration of the skin and hair.
 ANTIFUNGAL AGENTS
ANTIFUNGAL AGENTS
The treatment of superficial fungal infections caused by dermatophytic fungi may be accomplished (1) with topical antifungal agents, eg, clotrimazole, miconazole, econazole, ketoconazole, oxiconazole, sulconazole, sertaconazole, ciclopirox olamine, naftifine, terbinafine, butenafine, and tolnaftate; or (2) with orally administered agents, ie, griseofulvin, terbinafine, fluconazole, and itraconazole. Their mechanisms of action are described in Chapter 48. Superficial infections caused by candida species may be treated with topical applications of clotrimazole, miconazole, econazole, ketoconazole, oxiconazole, ciclopirox olamine, nystatin, or amphotericin B.
TOPICAL ANTIFUNGAL PREPARATIONS
TOPICAL AZOLE DERIVATIVES
The topical imidazoles, which currently include clotrimazole, econazole, ketoconazole, miconazole, oxiconazole, sulconazole, and sertaconazole, have a wide range of activity against dermatophytes (epidermophyton, microsporum, and trichophyton) and yeasts, including Candida albicans and Pityrosporum orbiculare (see Chapter 48).
Miconazole (Monistat, Micatin) is available for topical application as a cream or lotion and as vaginal cream or suppositories for use in vulvovaginal candidiasis. Clotrimazole (Lotrimin, Mycelex) is available for topical application to the skin as a cream or lotion and as vaginal cream and tablets for use in vulvovaginal candidiasis. Econazole (Spectazole) is available as a cream for topical application. Oxiconazole (Oxistat) is available as a cream and lotion for topical use. Ketoconazole (Nizoral) is available as a cream for topical treatment of dermatophytosis and candidiasis and as a shampoo or foam for the treatment of seborrheic dermatitis. Sulconazole (Exelderm) is available as a cream or solution. Sertaconazole (Ertaczo) is available as a cream. Topical antifungal-corticosteroid fixed combinations have been introduced on the basis of providing more rapid symptomatic improvement than an antifungal agent alone. Clotrimazole-betamethasone dipropionate cream (Lotrisone) is one such combination.
Once- or twice-daily application to the affected area will generally result in clearing of superficial dermatophyte infections in 2–3 weeks, although the medication should be continued until eradication of the organism is confirmed. Paronychial and intertriginous candidiasis can be treated effectively by any of these agents when applied three or four times daily. Seborrheic dermatitis should be treated with twice-daily applications of ketoconazole until clinical clearing is obtained.
Adverse local reactions to the imidazoles may include stinging, pruritus, erythema, and local irritation. Allergic contact dermatitis is uncommon.
CICLOPIROX OLAMINE
Ciclopirox olamine is a synthetic broad-spectrum antimycotic agent with inhibitory activity against dermatophytes, candida species, and P orbiculare. This agent appears to inhibit the uptake of precursors of macromolecular synthesis; the site of action is probably the fungal cell membrane.
Pharmacokinetic studies indicate that 1–2% of the dose is absorbed when applied as a solution on the back under an occlusive dressing. Ciclopirox olamine is available as a 1% cream and lotion (Loprox) for the topical treatment of dermatomycosis, candidiasis, and tinea versicolor. The incidence of adverse reactions has been low. Pruritus and worsening of clinical disease have been reported. The potential for allergic contact dermatitis is small.
Topical 8% ciclopirox olamine (Penlac nail lacquer) has been approved for the treatment of mild to moderate onychomycosis of fingernails and toenails. Although well tolerated with minimal side effects, the overall cure rates in clinical trials are less than 12%.
ALLYLAMINES: NAFTIFINE & TERBINAFINE
Naftifine hydrochloride and terbinafine (Lamisil) are allylamines that are highly active against dermatophytes but less active against yeasts. The antifungal activity derives from selective inhibition of squalene epoxidase, a key enzyme for the synthesis of ergosterol (see Figure 48–1).
They are available as 1% creams and other forms for the topical treatment of dermatophytosis, to be applied on a twice-daily dosing schedule. Adverse reactions include local irritation, burning sensation, and erythema. Contact with mucous membranes should be avoided.
BUTENAFINE
Butenafine hydrochloride (Mentax) is a benzylamine that is structurally related to the allylamines. As with the allylamines, butenafine inhibits the epoxidation of squalene, thus blocking the synthesis of ergosterol, an essential component of fungal cell membranes. Butenafine is available as a 1% cream to be applied once daily for the treatment of superficial dermatophytosis.
TOLNAFTATE
Tolnaftate is a synthetic antifungal compound that is effective topically against dermatophyte infections caused by epidermophyton, microsporum, and trichophyton. It is also active against P orbiculare but not against candida.
Tolnaftate (Aftate, Tinactin) is available as a cream, solution, powder, or powder aerosol for application twice daily to infected areas. Recurrences following cessation of therapy are common, and infections of the palms, soles, and nails are usually unresponsive to tolnaftate alone. The powder or powder aerosol may be used chronically following initial treatment in patients susceptible to tinea infections. Tolnaftate is generally well tolerated and rarely causes irritation or allergic contact dermatitis.
NYSTATIN & AMPHOTERICIN B
Nystatin and amphotericin B are useful in the topical therapy of C albicans infections but ineffective against dermatophytes. Nystatin is limited to topical treatment of cutaneous and mucosal candida infections because of its narrow spectrum and negligible absorption from the gastrointestinal tract following oral administration. Amphotericin B has a broader antifungal spectrum and is used intravenously in the treatment of many systemic mycoses (see Chapter 48) and to a lesser extent in the treatment of cutaneous candida infections.
The recommended dosage for topical preparations of nystatin in treating paronychial and intertriginous candidiasis is application two or three times a day. Oral candidiasis (thrush) is treated by holding 5 mL (infants, 2 mL) of nystatin oral suspension in the mouth for several minutes four times daily before swallowing. An alternative therapy for thrush is to retain a vaginal tablet in the mouth until dissolved four times daily. Recurrent or recalcitrant perianal, vaginal, vulvar, and diaper area candidiasis may respond to oral nystatin, 0.5–1 million units in adults (100,000 units in children) four times daily, in addition to local therapy. Vulvovaginal candidiasis may be treated by insertion of 1 vaginal tablet twice daily for 14 days, then nightly for an additional 14–21 days.
Amphotericin B (Fungizone) is available for topical use in cream and lotion form. The recommended dosage in the treatment of paronychial and intertriginous candidiasis is application two to four times daily to the affected area.
Adverse effects associated with oral administration of nystatin include mild nausea, diarrhea, and occasional vomiting. Topical application is nonirritating, and allergic contact hypersensitivity is exceedingly uncommon. Topical amphotericin B is well tolerated and only occasionally locally irritating. The drug may cause a temporary yellow staining of the skin, especially when the cream vehicle is used.
ORAL ANTIFUNGAL AGENTS
ORAL AZOLE DERIVATIVES
Azole derivatives currently available for oral treatment of candida and dermatophyte infections include fluconazole (Diflucan) and itraconazole (Sporanox). As discussed in Chapter 48, imidazole derivatives act by affecting the permeability of the cell membrane of sensitive cells through alterations of the biosynthesis of lipids, especially sterols, in the fungal cell.
Fluconazole and itraconazole are effective in the therapy of cutaneous infections caused by epidermophyton, microsporum, and trichophyton species as well as candida. Tinea versicolor is responsive to short courses of oral azoles.
Fluconazole is well absorbed following oral administration, with a plasma half-life of 30 hours. In view of this long half-life, daily doses of 100 mg are sufficient to treat mucocutaneous candidiasis; alternate-day doses are sufficient for dermatophyte infections. The plasma half-life of itraconazole is similar to that of fluconazole, and detectable therapeutic concentrations remain in the stratum corneum for up to 28 days following termination of therapy. Itraconazole is effective for the treatment of onychomycosis in a dosage of 200 mg daily taken with food to ensure maximum absorption for 3 consecutive months. Recent reports of heart failure in patients receiving itraconazole for onychomycosis have resulted in recommendations that it not be given for treatment of onychomycosis in patients with ventricular dysfunction. Additionally, routine evaluation of hepatic function is recommended for patients receiving itraconazole for onychomycosis.
Administration of oral azoles with midazolam or triazolam has resulted in elevated plasma concentrations and may potentiate and prolong hypnotic and sedative effects of these agents. Administration with HMG-CoA reductase inhibitors has been shown to cause a significant risk of rhabdomyolysis. Therefore, administration of the oral azoles with midazolam, triazolam, or HMG-CoA inhibitors is contraindicated.
GRISEOFULVIN
Griseofulvin is effective orally against dermatophyte infections caused by epidermophyton, microsporum, and trichophyton. It is ineffective against candida and P orbiculare. Griseofulvin’s mechanism of antifungal action is not fully understood, but it is active only against growing cells.
Following the oral administration of 1 g of micronized griseofulvin, drug can be detected in the stratum corneum 4–8 hours later. Reducing the particle size of the medication greatly increases absorption of the drug. Formulations that contain the smallest particle size are labeled “ultramicronized.” Ultramicronized griseofulvin achieves bioequivalent plasma levels with half the dose of micronized drug. In addition, solubilizing griseofulvin in polyethylene glycol enhances absorption even further. Micronized griseofulvin is available as 250 mg and 500 mg tablets, and ultramicronized drug is available as 125 mg, 165 mg, 250 mg, and 330 mg tablets and as 250 mg capsules.
The usual adult dosage of the micronized (“microsize”) form of the drug is 500 mg daily in single or divided doses with meals; occasionally, 1 g/d is indicated in the treatment of recalcitrant infections. The pediatric dosage is 10 mg/kg of body weight daily in single or divided doses with meals. An oral suspension is available for use in children.
Griseofulvin is most effective in treating tinea infections of the scalp and glabrous (nonhairy) skin. In general, infections of the scalp respond to treatment in 4–6 weeks, and infections of glabrous skin will respond in 3–4 weeks. Dermatophyte infections of the nails respond only to prolonged administration of griseofulvin. Fingernails may respond to 6 months of therapy, whereas toenails are quite recalcitrant to treatment and may require 8–18 months of therapy; relapse almost invariably occurs.
Adverse effects seen with griseofulvin therapy include headaches, nausea, vomiting, diarrhea, photosensitivity, peripheral neuritis, and occasionally mental confusion. Griseofulvin is derived from a penicillium mold, and cross-sensitivity with penicillin may occur. It is contraindicated in patients with porphyria or hepatic failure or those who have had hypersensitivity reactions to it in the past. Its safety in pregnant patients has not been established. Leukopenia and proteinuria have occasionally been reported. Therefore, in patients undergoing prolonged therapy, routine evaluation of the hepatic, renal, and hematopoietic systems is advisable. Coumarin anticoagulant activity may be altered by griseofulvin, and anticoagulant dosage may require adjustment.
TERBINAFINE
Terbinafine (described above) is quite effective given orally for the treatment of onychomycosis. Recommended oral dosage is 250 mg daily for 6 weeks for fingernail infections and 12 weeks for toenail infections. Patients receiving terbinafine for onychomycosis should be monitored closely with periodic laboratory evaluations for possible hepatic dysfunction.
 TOPICAL ANTIVIRAL AGENTS
TOPICAL ANTIVIRAL AGENTS
ACYCLOVIR, VALACYCLOVIR, PENCICLOVIR, & FAMCICLOVIR
Acyclovir, valacyclovir, penciclovir, and famciclovir are synthetic guanine analogs with inhibitory activity against members of the herpesvirus family, including herpes simplex types 1 and 2. Their mechanism of action, indications, and usage in the treatment of cutaneous infections are discussed in Chapter 49.
Topical acyclovir (Zovirax) is available as a 5% ointment; topical penciclovir (Denavir), as a 1% cream for the treatment of recurrent orolabial herpes simplex virus infection in immunocompetent adults. Adverse local reactions to acyclovir and penciclovir may include pruritus and mild pain with transient stinging or burning.
 IMMUNOMODULATORS
IMMUNOMODULATORS
IMIQUIMOD
Imiquimod is available as 5% cream (Aldara) for the treatment of external genital and perianal warts in adults, actinic keratoses on the face and scalp, and biopsy-proven primary basal cell carcinomas on the trunk, neck, and extremities. A lower 3.75% concentration cream (Zyclara) is available for the treatment of face and scalp actinic keratoses. The mechanism of its action is thought to be related to imiquimod’s ability to stimulate peripheral mononuclear cells to release interferon alpha and to stimulate macrophages to produce interleukins-1, -6, and -8, and tumor necrosis factor-α (TNF-α).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


