1 Introduction
Disinfectants, antiseptics and preservatives are chemical agents that have the ability to destroy or inhibit the growth of microorganisms. They play a major role in medical and health care but also have widespread use in the management of livestock, the environment, paints and coatings, plastics, pharmaceutical, food and beverage manufacture, textiles, the catering industry and consumer products. The term biocide is increasingly used to describe this group of chemical agents and this term is used widely throughout European Union (EU) directives.
1.1 European Union regulation
Regulation of biocides continues to develop within the EU and many other countries wherein guidance is defined for both manufacturers and users. Specific guidance documents agreed between the EU Commission services and the competent authorities of the Member States include, importantly, the Biocidal Products Directive 98/8/EC and the related directives: Medicinal Products for Human Use Directive 2001/83/EC and the Veterinary Medicinal Products Directive 2001/82/EC. Additionally, ‘agricultural pesticides’, are regulated by the Plant Protection Products Directive 91/414/EC.
The term ‘biocidal product’ (98/8/EC) encompasses active substances and preparations containing one or more active substances, put up in the form in which they are supplied to the user, intended to destroy, deter, render harmless, prevent the action of, or otherwise exert a controlling effect on any harmful organism by chemical or biological means.
The European Commission has proposed a new Regulation of the European Parliament and of the Council concerning the placing on the market and use of biocidal products. The proposed European Regulation will, over a period of time, revise and replace the current regulatory framework for the marketing and use of biocidal products contained in the Biocidal Products Directive (BPD) 98/8/EC (as amended). The BPD is currently transposed into domestic UK law by the Biocidal Products Regulations (BPR) 2001 ([SI 2001/880] as amended). The UK Government Health and Safety Executive (HSE) engages in negotiations with all 27 Member States of the EU, the European Commission and the European Parliament in the development of a new directly-acting EU law. The new draft regulation which is scheduled to come into effect on 1 January 2013 proposes several important changes to the current approach to dealing with biocides, including:
• Extending the scope of the regime to cover treated articles and materials containing biocides
• Adopting a Community authorization scheme for certain types of products
• Requiring mandatory data-sharing of some animal testing data
• Reducing the burden of data collection requirements
• Harmonizing fee structures across member states
1.2 Definitions
1.2.1 Disinfectant and disinfection
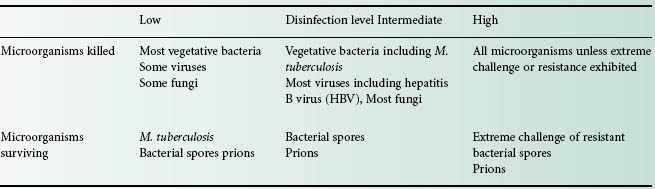
Disinfection is the process of removing microorganisms, including pathogens, from the surfaces of inanimate objects. The British Standards Institution (BSI) further defines disinfection as not necessarily killing all microorganisms but reducing them to a level acceptable for a defined purpose, for example, a level that is harmful neither to health nor to the quality of perishable goods. Chemical disinfectants are capable of different levels of action (Table 19.1). The term high-level disinfection indicates destruction of all microorganisms but not necessarily bacterial spores; intermediate-level disinfection indicates destruction of all vegetative bacteria including Mycobacterium tuberculosis but may exclude some resistant viruses and fungi and implies little or no sporicidal activity; low-level disinfection can destroy most vegetative bacteria, fungi and viruses, but this will not include spores and some of the more resistant microorganisms. Some high-level disinfectants have good sporicidal activity and are described as liquid chemical sterilants or chemosterilants to indicate that they can effect a complete kill of all microorganisms, as in sterilization. In defining each of these disinfection l evels the activity and outcome is determined by correct use of the disinfectant product in regard to concentration, time of contact and prevailing environmental conditions as described in subsequent sections of this chapter.
Table 19.1 Levels of disinfection attainable when products are used according to manufacturer’s instructions
1.2.2 Antiseptic and antisepsis
Antisepsis is defined as destruction or inhibition of microorganisms on living tissues having the effect of limiting or preventing the harmful results of infection. It is not a synonym for disinfection (BSI). The chemicals used are applied to skin and mucous membranes, so as well as having adequate antimicrobial activity they must not be toxic or irritating for these tissues. Antiseptics are mostly used to reduce the microbial population on the skin before surgery or on the hands to help prevent spread of infection by this route. Antiseptics are sometimes formulated as products containing significantly lower concentrations of agents used for disinfection.
1.2.3 Preservative and preservation
Preservatives are included in pharmaceutical and many other types of formulations, both to prevent microbial spoilage of the product and to minimize the risk to the consumer of acquiring an infection when the preparation is administered. Preservatives must be able to limit proliferation of microorganisms that may be introduced unavoidably into non-sterile products such as oral and topical medications during their manufacture and use. In sterile products, where multiuse preparations remain available, preservatives should kill any microbial contaminants introduced inadvertently during use. It is essential that a preservative is not toxic in relation to the intended route of administration of the preserved preparation. Preservatives tend to be employed at very low concentrations and consequently levels of antimicrobial action also tend to be of a lower order than for disinfect-ants or antiseptics. This is illustrated by the European Pharmacopoeia requirements for preservative efficacy where a degree of bactericidal activity is necessary, although this should be obtained within a few hours or over several days of microbial challenge depending on the type of product to be preserved. Other terms are considered in Chapter 18.
1.3 Economic aspects
The international antimicrobial chemical market, particularly in disinfectants, is expected to grow significantly over the coming years, on the basis of concerns about bacterial and other pathogenic threats and the increasing emphasis on hygiene in the home and work-place. The turnover of the US antimicrobial chemical industry is estimated to be in the region of $2 billion per annum. Key disinfectant products in use contain aldehydes, iodophors, nitrogen compounds (quaternary ammonium compounds and amine compounds), organometallics, organosulphurs, chloroisocyanurates and phenolics. There are around 250 chemicals that have been identified as active components of microbicidal products in the EU.
The aim of this chapter is to introduce the range of chemicals in common use and to indicate their activities and applications.
2 Factors affecting choice of antimicrobial agent
Choice of the most appropriate antimicrobial compound for a particular purpose depends on many factors and the key parameters are described.
2.1 Properties of the chemical agent
The process of killing or inhibiting the growth of microorganisms using an antimicrobial agent is basically that of a chemical reaction and the rate and extent of this reaction will be influenced by concentration of agent, temperature, pH and formulation. The influence of these factors on activity is considered in Chapter 18, and is referred to in discussing the individual agents in section 3. Tissue toxicity influences whether a chemical can be used as an antiseptic or preservative, and this limits the range of agents for these applications or necessitates the use of lower concentrations of the agent. This is discussed further in section 2.5.
2.2 Microbiological challenge
The types of microorganism present and the levels of microbial contamination (the bioburden) both have a significant effect on the outcome of treatment. If the bioburden is high, long exposure times and/or higher concentrations of antimicrobial may be required. Microorganisms vary in their sensitivity to the action of chemical agents. Some organisms merit attention either because of their resistance to disinfection (for further discussion see Chapter 20) or because of their significance in cross-infection or nosocomial (hospital-acquired) infections. Of particular concern is the significant increase in resistance to disinfectants resulting from microbial growth in biofilm form rather than free suspension (see Chapter 8). Microbial biofilms form readily on available surfaces, posing a serious problem for hospital infection control committees in advising suitable disinfectants for use in such situations.
The efficacy of an antimicrobial agent must be investigated by appropriate capacity, challenge and in-use tests to ensure that a standard is obtained which is appropriate to the intended use (Chapter 18). In practice, it is not usually possible to know which organisms are present on the articles being treated. Thus, it is necessary to categorize agents according to their antimicrobial activity and for the user to be aware of the level of antimicrobial action required in a particular situation (see Table 19.1).
2.2.1 Vegetative bacteria
At in-use concentrations, chemicals used for disinfection should be capable of killing bacteria and other organisms expected in that environment within a defined contact period. This includes ‘problem’ organisms such as methicillin-resistant Staphylococcus aureus (MRSA), vancomycin-resistant enterococci (VRE) and species of Listeria, Campylobacter and Legionella. Antiseptics and preservatives are also expected to have a broad spectrum of antimicrobial activity but at their in-use concentrations, after exerting an initial biocidal (killing) effect, their main function may be biostatic (inhibitory). Gram-negative bacilli, which are a major causes of nosocomial infections, are often more resistant than Gram-positive species. Pseudomonas aeruginosa, an opportunistic pathogen (see also Chapter 7), has gained a reputation as the most resistant of the Gram-negative organisms. However, problems mainly arise when a number of additional factors such as heavily soiled articles or diluted or degraded disinfectant solutions are employed.
2.2.2 Mycobacterium tuberculosis
M. tuberculosis (the tubercle bacillus) and other mycobacteria are resistant to many bactericides. Resistance is either (1) intrinsic, mainly due to reduced cellular permeability or (2) acquired, due to mutation or the acquisition of plasmids (Chapter 13). Tuberculosis remains an important public health hazard, and indeed the annual number of tuberculosis cases is rising in many countries. The greatest risk of acquiring infection is from the undiagnosed patient. Equipment used for respiratory investigations can become contaminated with mycobacteria if the patient is a carrier of this organism. It is important to be able to disinfect the equipment to a safe level to prevent transmission of infection to other patients (Table 19.2).
Table 19.2 Antibacterial activity of commonly used disinfectants and antiseptics
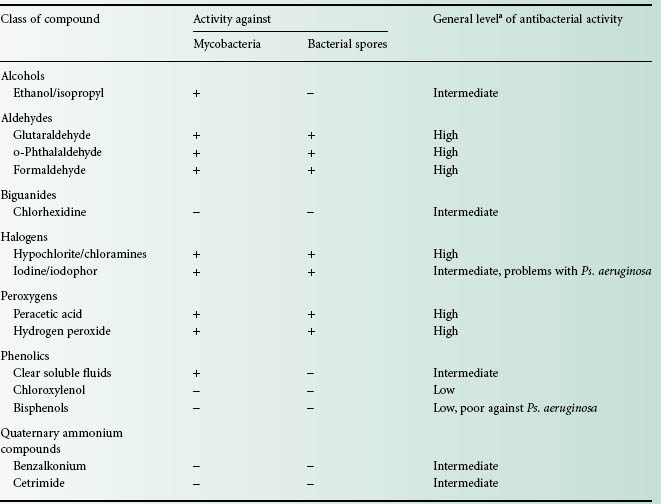
aActivity expected per manufacturer’s instructions and will depend on environmental conditions and bioburden.
2.2.3 Bacterial spores
Bacterial spores can exhibit significant resistance to even the most active chemical disinfectant treatment. The majority of disinfectants have no useful sporicidal action in a pharmaceutical context, which relates to disinfection of materials, instruments and environments that are likely to be contaminated by the spore-forming genera Bacillus and Clostridium. However, certain aldehydes, halogens and peroxygen compounds display very good activity under controlled conditions and are sometimes used as an alternative to physical methods for sterilization of heat-sensitive equipment. In these circumstances, correct usage of the agent is of paramount importance, as safety margins are lower in comparison with physical methods of sterilization (Chapter 21).
Clostridium difficile is a particularly problematic contaminant in hospital environments, resulting in high levels of morbidity and mortality. In addition to stringent hand-washing, meticulous environmental disinfection procedures must be in place, e.g. using solutions of 5.25–6.15% sodium hypochlorite for routine disinfection. When high-level disinfection of Cl. difficile is required, 2% glutaraldehyde, 0.55% o-phthalaldehyde and 0.35% peracetic acid are effective.
The antibacterial activity of some disinfectants and antiseptics is summarized in Table 19.2.
2.2.4 Fungi
The vegetative fungal form is often as sensitive as vegetative bacteria to chemical antimicrobial agents. Fungal spores (conidia and chlamydospores; see Chapter 4) may be more resistant, but this resistance is of much lesser magnitude than that exhibited by bacterial spores. The ability to rapidly destroy pathogenic fungi such as the important nosocomial pathogen Candida albicans, filamentous fungi such as Trichophyton mentagrophytes, and spores of common spoilage moulds such as Aspergillus niger is put to advantage in many applications of use. Many disinfectants have good activity against these fungi (Table 19.3). In addition, ethanol (70%) is rapid and reliable against Candida species.
Table 19.3 Antifungal activity of disinfectants and antiseptics
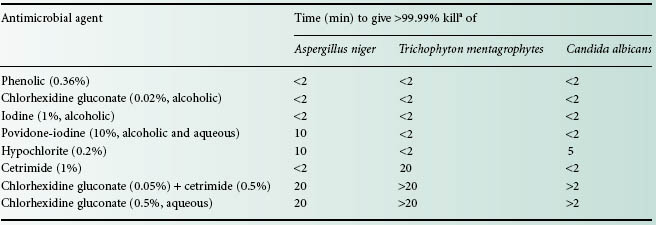
aInitial viable counts c. 1 × 106/ml in suspension test.
2.2.5 Viruses
Susceptibility of viruses to antimicrobial agents can depend on whether the viruses possess a lipid envelope. Non-lipid viruses are frequently more resistant to disinfectants and it is also likely that such viruses cannot be readily categorized with respect to their sensitivities to antimicrobial agents. These viruses are responsible for many nosocomial infections, e.g. rotaviruses, picornaviruses and adenoviruses (see Chapter5) and it may be necessary to select an antiseptic or disinfectant to suit specific circumstances. Certain viruses, such as Ebola and Marburg, which cause haemorrhagic fevers, are highly infectious and their safe destruction by disinfectants is of paramount importance. Hepatitis A is an enterovirus considered to be one of the most resistant viruses to disinfection.
There is much concern for the safety of personnel handling articles contaminated with pathogenic viruses such as hepatitis B virus (HBV) and HIV. Disinfectants must be able to treat rapidly and reliably accidental spills of blood, body fluids or secretions from HIV-infected patients. Such spills may contain levels of HIV as high as 104 infectious units/ml. Fortunately, HIV is inactivated by most chemicals at in-use concentrations. However, the recommendation is to use high-level disinfectants (see Table 19.2) for decontamination of HIV-or HBV-infected reusable medical equipment. For patient care areas, cleaning and disinfection with intermediate-level disinfectants is satisfactory. Flooding with a liquid germicide is required only when large spills of cultured or concentrated infectious agents have to be dealt with.
The World Health Organization (WHO) and epidemiologists in many countries track outbreaks of influenza, especially in relation to potential epidemic and pandemic situations arising. The H1N1 outbreak in 2009 generated considerable concern. As an influenza A virus, however, it is susceptible to a large number of disinfectant products when they are used on hard, non-porous surfaces that may be contaminated. Although no research has been conducted on the susceptibility of 2009 H1N1 influenza virus to chlorine and other disinfectants in swimming pools and spas, studies have demonstrated that free chlorine levels of 1–3 mg/L (1–3 ppm) are adequate to disinfect avian H5N1 influenza virus.
2.2.6 Protozoa
Acanthamoeba spp. can cause acanthamoeba keratitis with associated corneal scarring and loss of vision in wearers of soft contact lenses. The cysts of this protozoan present a particular problem in respect of lens disinfection. The chlorine-generating systems in use are generally inadequate. Polyaminopropyl biguanide with or without chlorhexidine (0.003%) and polyhexamethylene biguanide (0.0005%) both show ability as an acanthamoebicide in combating 103 levels of cysts. Hydrogen peroxide-based disinfection is considered completely reliable and consistent in producing an acanthamoebicidal effect.
2.2.7 Prions
Prions are generally considered to be the infectious agents most resistant to chemical disinfectants and sterilization processes; strictly speaking, however, they are not microorganisms because they have no cellular structure nor do they contain nucleic acids. As small proteinaceous infectious particles they are a unique class of infectious agent causing spongiform encephalopathies such as bovine spongiform encephalopathy (BSE) in cattle and Creutzfeldt–Jakob disease (CJD) in humans. There is considerable concern about the transmission of these agents from infected animals or patients. Risk of infectivity is highest in brain, spinal cord and eye tissues. Prions are considered resistant to most disinfectant procedures. For heat-resistant medical instruments that come into contact with high infectivity tissues or high-risk contacts, immersion in sodium hydroxide (1 M) or sodium hypochlorite (20 000 ppm available chlorine) for 1 hour is advised in WHO guidelines and this must be followed by further treatment including autoclaving, cleaning and routine sterilization. Recently, a formulation of 0.2% sodium docecyl sulphate, 0.3% NaOH in 20% n-propanol, has achieved potent decontamination of steel carriers contaminated with PrPTSE, the biochemical marker for prion infectivity, from 263K scrapie hamsters (5.5 log10 units reduction) or patients with sporadic or variant Creutzfeldt–Jacob disease. No low-temperature sterilization technology is effective.
2.3 Intended application
The intended application of the antimicrobial agent, whether for preservation, antisepsis or disinfection, will influence its selection and also affect its performance. For example, in medicinal preparations the ingredients in the formulation may antagonize preservative activity. The risk to the patient will depend on whether the antimicrobial is in close contact with a break in the skin or mucous membranes or is introduced into a sterile area of the body.
In disinfection of instruments, the chemicals used must not adversely affect the instruments, e.g. cause corrosion of metals, affect clarity or integrity of lenses, or change the texture of synthetic polymers. Many materials such as fabrics, rubber and plastics are capable of adsorbing certain disinfectants, e.g. quaternary ammonium compounds (QACs) are adsorbed by fabrics, while phenolics are adsorbed by rubber, the consequence of this being a reduction in the concentration of active compound. A disinfectant can only exert its effect if it is in contact with the item being treated. Therefore, access to all parts of an instrument or piece of equipment is essential. For small items, total immersion in the disinfectant must also be ensured.
2.4 Environmental factors
Organic matter can have a drastic effect on antimicrobial capacity either by adsorption or chemical inactivation, thus reducing the concentration of active agent in solution or by acting as a barrier to the penetration of the disinfectant. Blood, body fluids, pus, milk, food residues or colloidal proteins, even when present in small amounts, all reduce the effectiveness of antimicrobial agents to varying degrees, and some are seriously affected. In their normal habitats, microorganisms have a tendency to adhere to surfaces and are thus less accessible to the chemical agent. Some organisms are specific to certain environments and their destruction will be of paramount importance in the selection of a suitable agent, e.g. Legionella in cooling towers and non-potable water supply systems, Listeria in the dairy and food industry and HBV in blood-contaminated articles.
Dried organic deposits may inhibit penetration of the chemical agent. Where possible, objects to be disinfected should be thoroughly cleaned. The presence of ions in water can also affect activity of antimicrobial agents, thus water for testing biocidal activity can be made artificially ‘hard’ by addition of ions.
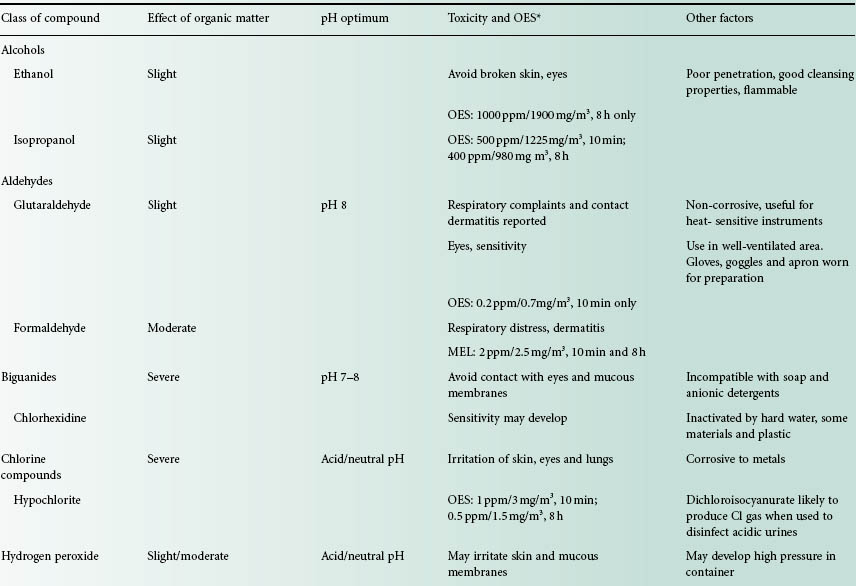
These factors can have very significant effects on activity and are summarized in Table 19.4.
Table 19.4 Properties of commonly used disinfectants and antiseptics
MEL, maximum exposure limit; OES: occupational exposure standard; QAC, quaternary ammonium compound.
MELs are the time-weighted average upper limits of a substance permitted in the breathing zone of a person. OES levels are the maximum concentrations of a substance in air to which individuals may be exposed during their working life and at which present knowledge indicates there will be no ill effects. Often similar to 8 h MEL levels.
(MELs have now been replaced by workplace exposure limits, WELs.) Source: HSE (2007).
2.5 Toxicity of the agent
In choosing an antimicrobial agent for a particular application some consideration must be given to its toxicity. Increasing concern for health and safety is reflected in the Control of Substances Hazardous to Health (COSHH) Regulations that specify the precautions required in handling toxic or potentially toxic agents. In respect of disinfectants these regulations affect, particularly, the use of phenolics, formaldehyde and glutaraldehyde. Toxic volatile substances, in general, should be kept in covered containers to reduce the level of exposure to irritant vapour and they should be used with an extractor facility. Limits governing the exposure of individuals to such substances are now listed, e.g. 0.7 mg/m3 (0.2 ppm) glutaraldehyde for both short-and long-term exposure. Many disinfectants including the aldehydes, glutaraldehyde less so than formaldehyde, may affect the eyes, skin (causing contact dermatitis) and induce respiratory distress. Face protection and impermeable nitrile rubber gloves should be worn when using these agents. Table 19.4 lists the toxicity of many of the disinfectants in use and other concerns of toxicity are described below for individual agents.
The COSHH Regulations specify certain disinfectants that contain active substances not supported under the BPD that had to be phased out by 2006. Specified disinfection procedures applied to laboratories in relation to spills and routine use state that certain phenolic agents (including 2,4,6-trichlorophenol and xylenol) can no longer be employed in disinfectant products.
Because of the historically high number of occupational asthma cases caused by glutaraldehyde (an alkylating agent) products in chemical disinfection of endoscopes, an HSE report (2007) sought alternatives to this agent. The report recommended the preferential use of an oxidizing agent such as a chlorine-based or peroxygen-based product rather than a product containing an alkylating agent. However, it was recognized that consideration must be given to incompatibility of disinfectants with endoscope construction materials in some cases (Table 19.5).
Table 19.5 HSE Recommendations (2007) for endoscopy disinfection
HSE, Health and Safety Executive; COSHH, Control of Substances Hazardous to Health.
Based on COSHH Essentials (HSE 2009).
| Disinfectant agent | COSHH Essentials Hazard Group | COSHH Essentials Control Approach |
| Chlorine | A (low hazard) | 1 (general ventilation) |
| Peroxygen | A (low hazard) | 1 (general ventilation) |
| Peracetic acid | C (medium hazard) | 3 (containment) |
| o-Phthalaldehyde | C (medium hazard) | 3 (containment) |
| Glutaraldehyde | E (special case) | 4 (special case) |
In all situations where the atmosphere of a workplace is likely to be contaminated by disinfectant, sampling and analysis of the atmosphere may need to be carried out on a periodic basis with a frequency determined by conditions.
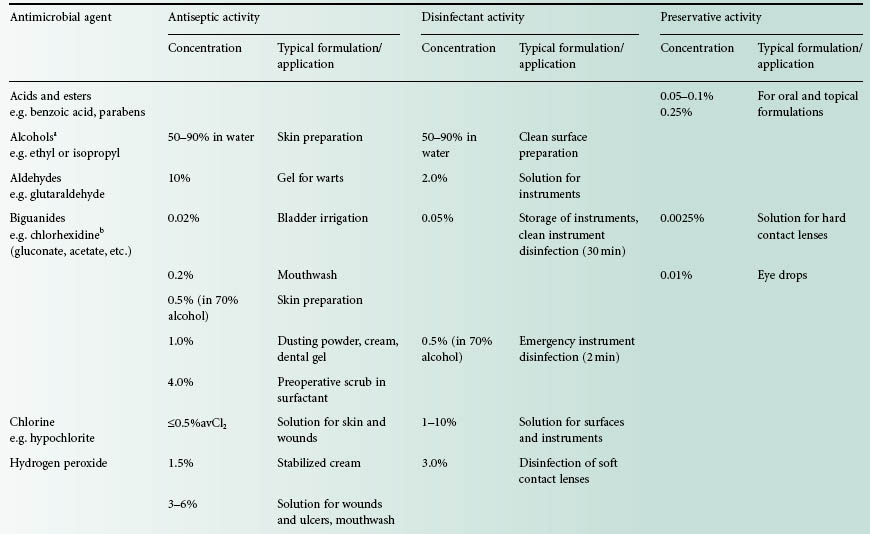
The following section presents, in alphabetical order by chemical grouping, the agents most often employed for disinfection, antisepsis and preservation. This information is summarized in Table 19.6.
Table 19.6 Examples of the main antimicrobial groups as antiseptics, disinfectants and preservatives
QAC, quaternary ammonium compound.