1 The need for antimicrobial stewardship
In testimony to a US government committee in June 2010, the Infectious Diseases Society of America (IDSA) stated that
Most commonly used antibiotics cost only a few dollars for a typical course of treatment… (and) a single course of antibiotics has the potential to protect and preserve many quality years of life for many people. No other type of medicine can claim such an achievement at such a price.
These statements were made as part of the argument presented by the IDSA to promote both antibiotic research and appropriate use ( ‘stewardship ’) of antibiotics.
The wider point being made was that antibiotics have, since their discovery in the 1940s, revolutionized medicine, and many of the procedures that are taken for granted now —transplantation, cancer treatment, the care of premature babies and several forms of surgery—would be impossible without them. Yet, unfortunately, largely because they have been taken for granted, the antibiotics we already possess are becoming less effective as a result of bacterial resistance, and the prospects for producing new antibiotics currently look bleak. It was recognized right from the start of the antibiotic era that bacteria had the potential to develop resistance to antimicrobial drugs, but it was quite some time before the perception of antibiotic resistance changed from one in which it was regarded as unusual to one where it was expected; in other words, a recognition that long-term efficacy was the exception, and resistance was the rule.
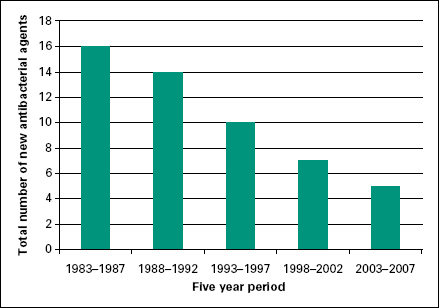
As the 20th century drew to a close the emergence of the antibiotic-resistant pathogens described in Chapter 13 brought with it both the spectre of untreatable infections where the organisms responsible were resistant to all available agents and a growing sense of urgency to take steps to preserve the usefulness of the antibiotics we currently have. The situation was exacerbated by a reduction in the number of new antibiotics coming into clinical use.Figure 15.1 shows that this number has continued to decline steadily for the last 20 years—a trend that would be difficult to reverse in the short term simply because several of the major international pharmaceutical companies are moving out of antimicrobials as a research and development area. Unfortunately, antibiotics have, in a sense, become victims of their own success: the more effective an antimicrobial is, the shorter the likely duration of treatment, so the lower the payback to the company that developed it. Many courses of antibiotic treatment last for a week or less, so the sales accruing from them are far inferior to those from drugs treating chronic conditions like diabetes and hypertension. This fact, together with (1) increasing pressure to use antibiotics sparingly anyway, (2) the expectation that the drug will ultimately become less effective due to resistance and (3) difficulties in establishing clinical trials for antibiotics that satisfy the US Food and Drugs Administration (FDA) criteria, all combined to create a climate in which antibiotics became an unattractive commercial proposition (except in the case of HIV/AIDS therapies that have to be taken throughout the patient’ s life).
Figure 15.1 Number of new antibacterial agents licensed for use by the FDA, 1983–2007. Spellberg (2007). Reproduced with permission of the University of Chicago Press.
Certainly the problem of growing antibiotic resistance had been recognized for some time before the end of the 20th century and policies designed to improve the quality of antibiotic prescribing and restrict resistance in hospitals became progressively more common in Europe and North America from the 1970s onwards. However, the increasing frequency in the new millennium of infections due to the so-called ‘ESKAPE’ pathogens( Enterococcus faecium, Staphylococcus aureus, Klebsiella species, Acinetobacter baumannii, Pseudomonas aeruginosa and extended spectrum β-lactamase-producing strains of Escherichia coli and Enterobacter species) together with the drying up of the pipeline of new antibiotics from the pharmaceutical industry has further increased the tempo of measures to preserve what is increasingly being seen as a precious, and perhaps irreplaceable, resource that society has a duty to pass on to future generations rather than squander. These measures include the ‘10 × 20 ’ initiative to establish an international research effort to develop 10 new antibiotics by the year 2020, and the STAAR (Strategies to Address Antimicrobial Resistance) Act, which is, at the time of writing, being considered by the US Congress.
The cost of antibiotic resistance should, of course, be measured primarily in terms of the suffering that results from the failure of antibiotics to cure infections against which they were formerly effective. The antibiotic-resistant pathogens responsible for these infections can arise both in the home or the hospital environment, but it is in the latter that they are, by far, more common and problematic. Hospital-acquired infections account for a substantial number of deaths each year and the treatment of such infections is time-consuming, difficult and costly. In its testimony to Congress as part of the consultation process accompanying the STAAR Act, the IDSA stated that antibiotic-resistant infections acquired within hospitals were responsible for 90 000 deaths each year in the USA and cost the healthcare system between $21 and $34 billion annually. There are less obvious consequences of resistance too: when the first-line drugs cease to be effective it is sometimes necessary to revert to alternatives that are more toxic.Acinetobacter infections are a good example of this situation because the organism is naturally multidrug resistant and the incidence of isolates resistant to all first-line antibiotics has risen in the USA from 5% to 40% in 10 years, so now colistin, a drug that became virtually obsolete in the 1960s because of the significant risk of kidney damage is, for many patients, the most likely antibiotic choice.
There is widespread agreement that greater use of antibiotics predisposes to the development of resistance. The strength of the link between use and resistance varies from one antibiotic to another, but for many antibiotics the connection is irrefutable. However, the situation is far from simple: there is substantial evidence, for example, that heavy use of one antibiotic may be a risk factor for the acquisition of infections by organisms resistant to other, unrelated antibiotics—heavy cephalosporin use has been shown to increase the risk of vancomycinresistant enterococci, and fluoroquinolone use has been associated with the prevalence of meticillin-resistant Staph. aureus (MRSA). The selective pressure created by the use of one antibiotic will often select for resistance in others because plasmids within the bacterial cell may carry resistance genes for multiple antibiotics from different chemical groups. If, for example, an organism possessed a plasmid with genes for both rifampicin and gentamicin resistance, constant exposure to gentamicin would represent a selective pressure that afforded an advantage to that organism so, not only would the incidence of isolates with gentamicin resistance be expected to rise, but so too would the incidence of rifampicin resistant isolates.
Arguments for curtailing antibiotic use in order to restrict resistance development have been supported by audits and surveys of antibiotic prescribing and costs. It has been estimated that up to 50% of antibiotic prescribing is either inappropriate (wrong drug, duration, dose, etc.) or unnecessary (not required at all). Antibiotic consumption varies widely throughout Europe: a 2005 survey showed that France, for example, used three times as much antibiotics per head of population as the Netherlands, but this difference was not justified by higher infection rates or better cure rates, so logic suggests that part of the antibiotic use in France was unnecessary. Inappropriate prescribing and consumption, together with the fact that antibiotics can represent up to 30% of a hospital pharmacy budget, have provided further impetus for measures designed to achieve more prudent prescribing. Such thinking is not new, however; it was the rationale for the introduction of antibiotic policies in the 1970s and 1980s which, in addition to setting out the general standards for safe and appropriate prescribing, advised on the selection of antibiotics for specific infections, for special situations like surgical prophylaxis and for the treatment of specific groups of patients, e.g. the newborn, those with poor kidney function and the immunocompromised. What has changed since then is the recognition of the need for a much broader approach to the problem —in other words, the need for a comprehensive antimicrobial stewardship strategy that incorporates, but also extends, the policies formulated in the last century.
There is not a universally accepted definition of, or agreement upon, what constitutes an antimicrobial stewardship programme. For most people the term applies particularly, or even exclusively, to the manner in which antibiotics are used and distributed in hospitals. However, some see it in a much broader sense as a range of initiatives which, together, impact upon antibiotic resistance but are not necessarily even confined to antibiotic-related practices in the hospital or the home. The great majority of the annual global production of antibiotics is not used in the treatment of human or animal infection anyway. Most of the antibiotic output of the international pharmaceutical industry is used as a food additive to increase weight gain in cattle, pigs and poultry —some estimates put this proportion as high as 80%—and yet more is used in plant production, but this fraction is ill-d efined. Although antibiotics that are used to treat human infections have been banned as growth promoters in Europe for many years it is still a common practice in many countries, and even the legitimate veterinary use of antibiotics is considerable: the total volume of antibiotics used in the UK for agricultural purposes in 2007 was 387 tonnes. Curtailment of the use of antibiotics for growth promotion—which many see as inappropriate and a likely contributor to resistance development—together with better-targeted and-promoted use of vaccines (that would reduce the need for antibiotics), better diagnostic agents which would more rapidly and accurately identify the infecting organism and so inform the selection of the best antibiotic, better epidemiological data and computer analysis to provide early warnings of resistance trends, and changes in other medical practices like the early removal of catheters and cannulas which are, themselves, a means by which pathogens can enter the body, might all be seen as part of a stewardship programme. But from the perspective of controlling the incidence and spread of antibiotic-resistant organisms within a hospital it cannot be over-emphasized that a comprehensive infection control programme is of paramount importance (see Chapter 16) and the best results are achieved when data from antibiotic stewardship and infection control can be linked and analysed together.
It is generally accepted that the principal goals of a stewardship programme are to:
• Improve patient outcomes
• Lessen the risk of adverse effects
• Reduce resistance levels, or at least slow the rate of resistance development
• Improve cost-effectiveness.
Following the international financial crisis that began in 2007 it is likely, at least in many European countries, that the last of these goals will receive particular attention, so it is worth emphasizing that stewardship programmes can be self-financing. Although there is an initial start-up cost, there is substantial evidence that this is rapidly recovered by cost savings resulting from reductions in antimicrobial use that some reports have estimated to vary from 22 to 36%.
2 Components of antimicrobial stewardship programmes
There are no nationally or internationally accepted guidelines on the structure of a stewardship programme, so they vary from country to country and even from one hospital to another within a geographical region. Indeed, even the titles vary, so terms like ‘good antimicrobial practice programmes ’, ‘antibiotic management programmes’ and ‘antibiotic control programmes’have all been used. There is, however, a measure of agreement on the personnel comprising an antimicrobial stewardship team. They are multidisciplinary and usually consist of:
• An infectious diseases physician
• An clinical pharmacist with infectious diseases training
• A medical microbiologist
• An infection control professional
• A hospital epidemiologist
• An information technology specialist.
In the USA the IDSA and the Society for Heathcare Epidemiology of America (SHEA) set forth stewardship guidelines in 2007 that have been widely adopted both in the USA and elsewhere; but in Europe the situation is less uniform. Here, an expert group reported in 2000 that little was known about which stewardship measures were employed in Europe and which were optimal. As a consequence, the European Commission Concerted Action Antibiotic Resistance Prevention and Control (ARPAC) survey of stewardship practices in 263 hospitals through 32 European countries was undertaken, and it reported in 2009.Table 15.1 lists both the components of the USguidelines and the six key indicators of stewardship that were the subject of the ARPAC survey.
Table 15.1 The components of antimicrobial stewardship programmes
ISDA, Infectious Diseases Society of America; SHEA, Society for Healthcare Epidemiology of America.
| Core strategies and elements of a stewardship programme as recommended by the ISDA and SHEA | Key indicators of stewardship as identified by the ARPAC survey |
| Two core strategies (not necessarily mutually exclusive): | The hospital had: |
| Prospective audit with intervention and feedback | An antibiotic committee |
| Formulary restriction and preauthorization | A written antibiotic policy |
| Elements supplementing these strategies: | A written antibiotic formulary |
| Education | A formulary that included a restricted antibiotic list |
| Guidelines and clinical pathways | A drugs and therapeutic committee |
| Antimicrobial order forms | A strategic management goal of improving prescribing |
| De-escalation of therapy | |
| Parenteral to oral conversion | |
| Dose optimization | |
| Antimicrobial cycling | |
| Combination therapy |
Before considering in more detail the elements of a stewardship programme, it is worth explaining the meaning of some of the terms used in Table 15.1. In this context a prospective audit means a review of the future delivery of healthcare to ensure that best practice is being carried out, so a prospective audit with intervention and feedback is a process in which the use of antibiotics is monitored and suggestions made for improvement, where necessary,while the course of treatment is still in progress. The feedback element is the provision of information to the prescriber about the drug in question, e.g. local resistance patterns and dose information based upon pharmacokinetic data.
Most hospitals have a drugs and therapeutics committee (DTC; sometimes also known as a pharmacy and therapeutics committee) whose function is to evaluate drugs for inclusion in a hospital formulary on the basis of their efficacy, toxicity and cost. The intention is to avoid the unnecessary stocking of multiple drugs from the same class which have similar or identical properties; few hospitals stock every available aminoglycoside or third generation cephalosporin, for example. An antibiotics committee would be a subgroup of the DTC with responsibility for selecting the drugs for inclusion in the antibiotics formulary. The strategy of formulary restriction and preauthorization could limit the availability of certain antibiotics simply by excluding them from the formulary (although they could usually be obtained in exceptional circumstances if required). The preauthorization element would simply be a requirement to seek the recommendation (or even written approval) of senior clinical staff when prescribing antibiotics in a restricted category—usually those drugs whose effectiveness needs to be preserved because of their value in treating infections for which few drugs are effective, e.g. linezolid or daptomycin, or antibiotics which may predispose to other problems, e.g. clindamycin because of its link to Clostridium difficile outbreaks.
The ARPAC stewardship indicator that the hospital should have a strategic management goal of improving prescribing has important implications for the provision of resources and granting the authority to implement decisions. Establishing a stewardship programme costs money: staff time must be available for (1) the preparation of the antibiotics policy document and formulary, (2) intervention and feedback, and (3) monitoring of antibiotic use and resistance patterns. It is easier to gain approval for the necessary budget allocation if there is a publically stated management goal of improving prescribing. Where preauthorization for selected antibiotics is a part of the programme, the greater the authority bestowed on the person(s) making the recommendations, the greater the effectiveness of the policy has been shown to be.
3 The effectiveness of stewardship strategies
How effective a particular action is will be determined by the parameter used to measure it. In the case of stewardship programmes almost all of the individual components of the two IDSA/SHEA strategies (Table 15.1) can be shown to reduce resistance development, adverse effects or costs, but there are few actions that result in favourable changes in all three criteria.
Both of the core strategies of prospective audit and formulary restriction are active measures and, as such, are more effective than passive ones. Prospective audit/ intervention has been shown to reduce inappropriate use of antibiotics, achieve cost savings and, in some cases, restricted the isolation of particular antibiotic-resistant organisms. Formulary restriction/preauthorization has similarly been shown to reduce antibiotic consumption, and this may be immediate and significant, but its long-term impact on restricting resistance development is not proven. Education of patients to dissuade them from pressurizing prescribers for antibiotics—particularly for colds and flu or other viral infections —is now well established, but education of prescribers (conference presentations, teaching sessions, e-mail alerts and bulletins) is a passive approach which, by itself, has been shown to have only a marginal and short-term impact. Clinical path-ways (also known as critical care pathways or care maps) are intended to reduce the variability both in the quality of care and in patient outcomes by the adoption of defined, standardized and sequenced procedures for patients with specific conditions —in this context, infections. The use of a clinical pathway that includes a specified antibiotic regimen in the treatment of community-acquired pneumonia, for example, has been shown to be capable of reducing the duration of hospital stay and duration of antibiotic therapy, and there is strong evidence that practice guidelines and clinical path-ways incorporating local resistance patterns can generally improve antimicrobial utilization.
Four other elements of the IDSA/SHEA that have been shown to afford clear benefits are the use of antimicrobial order forms, de-escalation of therapy, parenteral to oral conversion and optimized dosing. Order forms are particularly useful when antibiotics are prescribed for prophylactic purposes to reduce the incidence of infection following surgery. In this situation there is a tendency to continue the course of treatment for an unnecessarily long period after the operation, and studies have shown that order forms with a default stop date have diminished drug consumption with no adverse effect. When hospital treatment for an infection begins, it is often the case that the organism responsible has not been identified, so initial treatment is empirical or ‘blind ’ and, in order to maximize the probability of inhibiting the pathogen, it may involve the use of either a broad-spectrum antibiotic, or of two or more different drugs. Once the organism has been identified it is good practice to replace broad-spectrum antibiotics (or redundant components of a combination) with a drug having more specific activity, because the continued unnecessary use of broad-spectrum therapy contributes to selection of resistant pathogens. Converting from intravenous to oral therapy affords a benefit primarily in terms of cost reduction but may also permit earlier removal of intravenous lines which facilitate the establishment of infections by skin pathogens like Staph. epidermidis. Oral antibiotics are usually cheaper than intravenous ones, quite simply because the use of the latter is largely restricted to hospitals anyway, so their manufacturers have to recover the development costs from lower lifetime sales. Optimized dosing is, as the name implies, modifying the antibiotic dose to suit an individual patient’ s circumstances. Factors that may influence the dose, and hence the effectiveness of the therapy, include the patient ’ s physical characteristics (weight, age, immune status, renal function), the site of infection and the pharmacokinetics of the drug which determine its access to, and concentration at, that infection site.
Two procedures that have been advocated as means of restricting resistance development are cycling of antibiotics and routinely using them in combination, particularly for infections with a long time-course of treatment. However, there is little evidence to support the use of routine cycling —the planned replacement of one specific antibiotic, or category of antibiotics, with another at predetermined intervals—as a means of controlling resistance. True cycling involves the return into use of the original antibiotic after a specified time, and although the first switch may lead to a reduced incidence of resistance to the first drug, some studies have shown that when it is subsequently reintroduced the original resistance level is quickly restored. Using antibiotics in combination as a means of restricting resistance development is well established and of undoubted benefit in the treatment of both tuberculosis, for which the duration of therapy is typically 6 months, and in HIV/AIDS which requires life-long treatment. During such long time periods the number of pathogen replication cycles is so large, and in hese two examples the mutation frequency is so high, that the probability of a resistant mutant arising and being selected as the predominant strain at the site of infection is significant, so using two or more agents with different modes of action is both logical and effective. However, the same logic has been applied to other, relatively short-term, infections without clear evidence of benefit.
4 Monitoring of antibiotic resistance
To get the most out of a newly implemented steward-ship programme (or a change to an existing one) it is necessary to have the means by which to measure its effect. It is, therefore, necessary to plan in advance what parameters will be measured and what will be the baseline data against which the changes will be judged. Some of the parameters might include, but are not restricted to:
• Antibiotic consumption and costs, both in total and by specific drug class
• Costs associated with prescribing potentially toxic antibiotics, e.g. gentamicin and vancomycin blood level monitoring
• Rates of resistance to specific antibiotics by problem pathogens
• Pharmacy interventions to advise on inappropriate antibiotic use
• The incidence of hospital-acquired infections.
One of the problems that has dogged reviewers trying to assess the extent of the benefits of stewardship programmes is that in many of the cases reported in the medical literature multiple changes to an established programme have been introduced together, or they have overlapped in time so that evaluating the contribution of each change has been difficult. It is, therefore, worthwhile deciding in advance when a new policy or practice will be implemented and when its effect will be assessed. The input of information technology specialists and hospital epidemiologists to a stewardship management team becomes important since they, together, can decide how the data will be recorded and analysed to best effect.
The more sophisticated antibiotic control and information systems do not simply record data on antibiotic consumption, cost and resistance, but are capable of relating infection control data to antibiotic use and would be expected to draw attention to situations where a change in use of a particular antibiotic was associated ith increasing isolation of a particular pathogen. Such n association does not, of course, mean that one caused the other, but it does raise staff awareness of that potential. Computer-assisted surveillance of hospital-acquired infection (HAI) has been shown, in some cases, to be more effective than manual monitoring and reporting; as long ago as 1986 one study reported that 90% of antibiotic-resistant HAIs were detected by computer compared to 76% manually. There is also the potential to improve antibiotic prescribing by minimizing the risk of adverse effects when information systems provide patient-specific warnings on allergies, immune and renal functions and the potential for interaction with the patient ’ s other drugs.
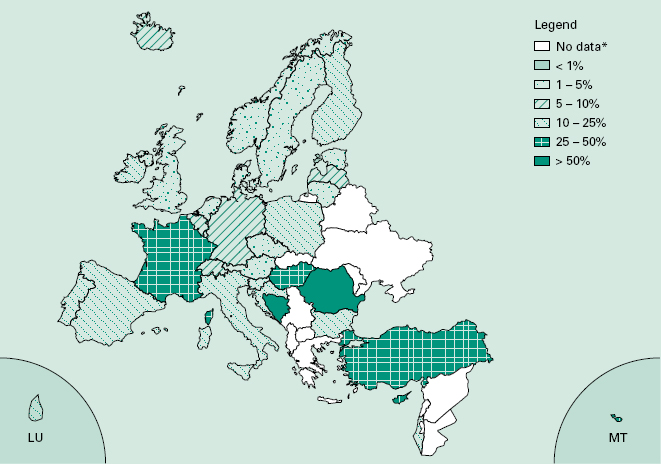
It is not surprising, perhaps, that there are marked differences in antibiotic resistance patterns from one country to another. This is illustrated in Figure 15.2, which shows that penicillin resistance in Streptococcus pneumoniae can vary from less than 5% in some European countries to more than 50% in others. Smaller, but nevertheless significant, variations may also arise between different regions of a single country; Figure 15.3 shows the corresponding Strep. pneumoniae data for the UK from the national Heath Protection Agency. However, data on local resistance patterns are of paramount importance and well-structured monitoring programmes should be capable of identifying unforeseen consequences of changes in antibiotic use such as that arising when a preapproval policy for cephalosporins was introduced in a New York hospital in an attempt to control cephalosporin resistance in Klebsiella species. The policy did achieve a 71% reduction in ceftazidime-resistant Klebsiella isolated in intensive care units, but monitoring revealed a concomitant rise in imipenem use and a 69% increase in imipenem-resistant Ps. aeruginosa that was attributed largely to the preapproval policy.
Figure 15.2 Incidence of penicillin-resistant Strep. pneumoniae in Europe, 2008. (From the European Antimicrobial Resistance Surveillance System; reproduced with permission.)