1 Introduction
The worldwide use of antimicrobial drugs continues to rise; in 2005 these agents accounted for an expenditure of approximately £ 26 billion. In the UK, prescribing in general practice accounts for approximately 90% of all antibiotics and largely involves oral and topical agents. Hospital use accounts for the remaining 10% of antibiotic prescribing, with a much heavier use of injectable agents. Although this chapter is concerned with the clinical use of antimicrobial drugs, it should be remembered that these agents are also extensively used in veterinary practice and, to a diminishing extent, in animal husbandry as growth promoters. In humans the therapeutic use of anti-infectives has revolutionized the management of most bacterial infections, many parasitic and fungal diseases and, with the availability of aciclovir and a growing number of antiretroviral agents (see Chapters 5 and 11), selected herpesvirus infections and HIV infection, respectively. Although originally used for the treatment of established bacterial infections, antibiotics have proved useful in the prevention of infection in various high-risk circumstances; this applies especially to patients undergoing various surgical procedures where perioperative antibiotics have significantly reduced postoperative infectious complications.
The advantages of effective antimicrobial chemotherapy are selfevident, but this has led to a significant problem in ensuring that they are always appropriately used. Prescribers face a dilemma: initial antimicrobial therapy must be effective against all likely infective organisms for the individual presentation, but excessive use of broad-spectrum agents contributes to the development and selection of drug-resistant organisms. Hence, antiinfectives are the only class of drug where inappropriate use in one patient can jeopardize the efficacy of treatment in other individuals.
Examples of inappropriate antimicrobial use include prescribing in situations where antibiotics are either ineffective, such as viral infections, or where the selected agent, its dose, route of administration or duration of use are inappropriate. Of particular concern is the unnecessarily prolonged use of antibiotics for surgical prophylaxis. Apart from encouraging superinfection by drug-resistant organisms, prolonged use is wasteful of health resources and unnecessarily increases the risk of adverse drug reactions. Thus, it is essential that the clinical use of these agents be based on a clear understanding of the principles that have evolved to ensure safe, yet effective, prescribing.
Further information about the properties of antimicrobial agents described in this chapter can be found in Chapter 11.
2 Principles of use of antimicrobial drugs
2.1 Susceptibility of infecting organisms
Drug selection should be based on knowledge of its activity against infecting microorganisms. Selected organisms may be predictably susceptible to a particular agent, and laboratory testing is therefore rarely performed. For example, Streptococcus pyogenes is uniformly sensitive to penicillin. In contrast, the susceptibility of many Gramnegative enteric bacteria is less predictable and laboratory guidance is essential for safe prescribing. The susceptibility of common bacterial pathogens and widely prescribed antibiotics is summarized in Table 14.1. It can be seen that, although certain bacteria are susceptible in vitro to a particular agent, use of that drug may be inappropriate, either on pharmacological grounds or because other less toxic agents are preferred.
Table 14.1 Sensitivity of selected bacteria to common antibacterial agents
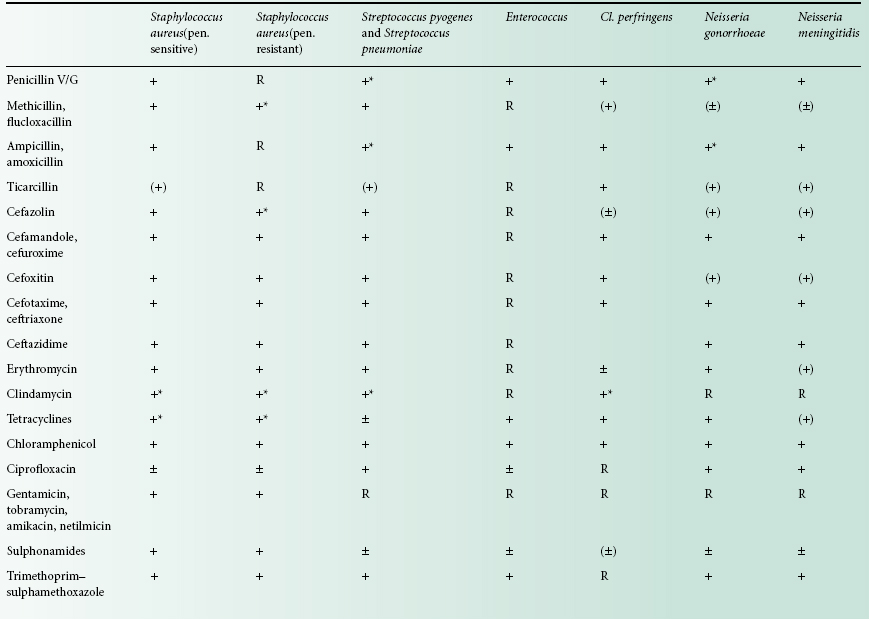

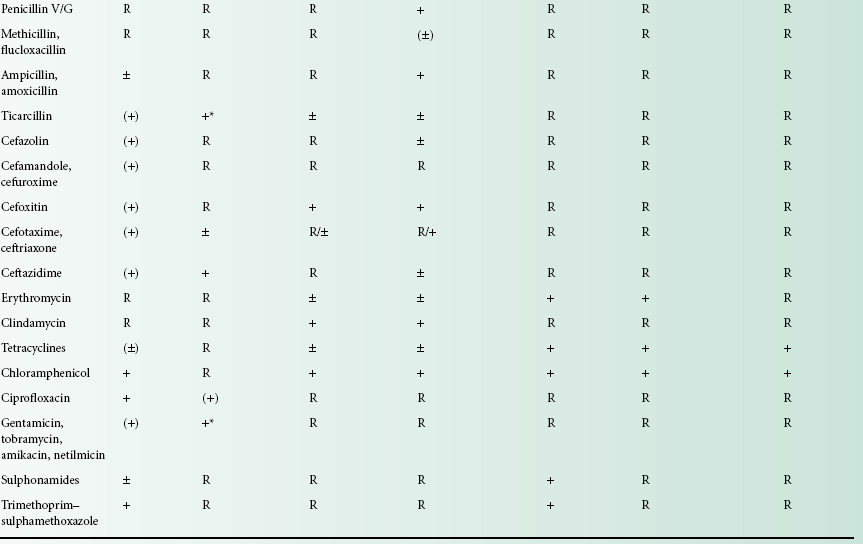
+, Sensitive; R, resistant; ±, some strains resistant; (), not appropriate therapy; *, rare strains resistant.
2.2 Host factors
In vitro susceptibility testing does not always predict clinical outcome. Host factors play an important part in determining outcome and this applies particularly to circulating and tissue phagocytic activity. Infections can progress rapidly in patients suffering from either an absolute or functional deficiency of phagocytic cells. This applies particularly to those suffering from various haematological malignancies, such as the acute leukaemias, where phagocyte function is impaired both by the disease and also by the use of potent cytotoxic drugs which destroy healthy, as well as malignant, white cells. Under these circumstances it is essential to select agents that are bactericidal, as bacteriostatic drugs, such as the tetracyclines or sulphonamides, rely on host phagocytic activity to clear bacteria. Widely used bactericidal agents include the aminoglycosides, broad-spectrum penicillins, the cephalosporins and quinolones (see Chapter 11).
In some infections the pathogenic organisms are located intracellularly within phagocytic cells and therefore remain relatively protected from drugs that penetrate cells poorly, such as the penicillins and cephalosporins. In contrast, erythromycin, rifampicin and the fluoroquinolones readily penetrate phagocytic cells. Legionnaires’ disease is an example of an intracellular infection and is treated with erythromycin with or without rifampicin.
2.3 Pharmacological factors
Clinical efficacy is also dependent on achieving satisfactory drug concentrations at the site of the infection; this is influenced by the standard pharmacological factors of absorption, distribution, metabolism and excretion. If an oral agent is selected, gastrointestinal absorption should be satisfactory. However, it may be impaired by factors such as the presence of food, drug interactions (including chelation), or impaired gastrointestinal function either as a result of surgical resection or malabsorptive states. Although effective, oral absorption may be inappropriate in patients who are vomiting or have undergone recent surgery; under these circumstances a parenteral agent will be required and has the advantage of providing rapidly effective drug concentrations.
Antibiotic selection also varies according to the anatomical site of infection. Lipid solubility is of importance in relation to drug distribution. For example, the aminoglycosides are poorly lipid-soluble and although achieving therapeutic concentrations within the extracellular fluid compartment, penetrate the cerebrospinal fluid (CSF) poorly. Likewise the presence of inflammation may affect drug penetration into the tissues. In the presence of meningeal inflammation, β-lactam agents achieve satisfactory concentrations within the CSF, but as the inflammatory response subsides drug concentrations fall. Hence it is essential to maintain sufficient dosaging throughout the treatment of bacterial meningitis. Other agents such as chloramphenicol are little affected by the presence or absence of meningeal inflammation.
Therapeutic drug concentrations within the bile duct and gallbladder are dependent on biliary excretion. In the presence of biliary disease, such as gallstones or chronic inflammation, the drug concentration may fail to reach therapeutic levels. In contrast, drugs that are excreted primarily via the liver or kidneys may require reduced dosaging in the presence of impaired renal or hepatic function. The malfunction of excretory organs may not only risk toxicity from drug accumulation, but will also reduce urinary concentration of drugs excreted primarily by glomerular filtration. This applies to the aminoglycosides and the urinary antiseptics nalidixic acid and nitrofurantoin, where therapeutic failure of urinary tract infections may complicate severe renal failure.
2.4 Drug resistance
Drug resistance may be a natural or an acquired characteristic of a microorganism. This may result from impaired cell wall or cell envelope penetration, enzymatic inactivation, altered binding sites or active extrusion from the cell as a result of efflux mechanisms (Chapter 13). Acquired drug resistance may result from mutation, adaptation or gene transfer. Spontaneous mutations occur at low frequency, as in the case of Mycobacterium tuberculosis where a minority population of organisms is resistant to isoniazid. In this situation the use of isoniazid alone will eventually result in overgrowth by this subpopulation of resistant organisms.
Genetic resistance may be chromosomal or transferable on transposons or plasmids. Plasmidmediated resistance has been increasingly recognized among Gram negative enteric pathogens. By the process of conjugation (Chapter 13), resistance plasmids may be transferred between bacteria of the same and different species and also different genera. Such resistance can code for multiple antibiotic resistance. For example, the penicillins, cephalosporins, chloramphenicol and the aminoglycosides are all subject to enzymatic inactivation, which may be plasmid mediated. Knowledge of the local epidemiology of resistant pathogens within a hospital, and especially within highdependency areas such as intensive care and haemodialysis units, is invaluable in guiding appropriate drug selection.
2.4.1 Multidrug resistance
In recent years multidrug resistance has increased among certain pathogens. These include Staphylococcus aureus, enterococci and M. tuberculosis. Staph. aureus resistant to meticillin is known as meticillin-resistant Staph. aureus (MRSA). These strains are resistant to many antibiotics and have been responsible for major epidemics worldwide, usually in hospitals where they affect patients in high dependency units such as intensive care units, burns units and cardiothoracic units. MRSA have the ability to colonize staff and patients and to spread readily among them. Several epidemic strains are currently circulating in the UK. The glycopeptides vancomycin or teicoplanin and the oxazolidinone linezolid are the currently recommended agents for treating patients infected with these organisms. Newer agents such as daptomycin and tigecycline are also active against MRSA and are increasingly used in the hospital setting; meanwhile, some strains retain sensitivity to older agents such as tetracyclines, rifampicin and clindamycin, and combinations of these agents are sometimes used, but only following full analysis of microbiological sensitivities.
Another serious resistance problem is that of drugresistant enterococci. These include Enterococcus faecalis and, in particular, E. faecium. Resistance to the glycopeptides has again been a problem among patients in highdependency units. Four different phenotypes are recognized (VanA, VanB, VanC and VanD). The VanA phenotype is resistant to both glycopeptides, while the others are sensitive to teicoplanin but demonstrate high (VanB) or intermediate (VanC) resistance to vancomycin; VanD resistance has only recently been described and remains uncommon. Those fully resistant to the glycopeptides are increasing in frequency and causing great concern as they are essentially resistant to almost all antibiotics.
Extended-spectrum β-lactamase (ESBL) producing Gramnegative organisms are an increasing problem in hospitals, and occasionally seen as a cause of urinary tract infection in primary care; ESBLs can hydrolyse most cephalosporins and penicillins, limiting therapeutic options to carbapenems or aminoglycosides. ESBLs can be chromosomally mediated (e.g. P seudomonas spp., Citrobacter spp.) or plasmidmediated (e.g. K lebsiellaspp.), the latter often being implicated in hospital outbreaks. Furthermore, carbapenem resistance, via metallo β-lactamase, has been seen in outbreak strains of Pseudomonas aeruginosa and Acinetobacter baumannii; in such situations, potentially toxic agents such as colistin are often the only effective choice.
Tuberculosis is on the increase after decades in which the incidence had been steadily falling. Drug-resistant strains have emerged largely among inadequately treated or noncompliant patients. These include the homeless, alcoholic, intravenous drug misusing, HIVpositive and immigrant populations. Resistance patterns vary but increasingly include rifampicin and isoniazid. Furthermore, outbreaks of multidrug-resistant tuberculosis have been increasingly reported from a number of hospital centres in the USA and more recently Europe, including the UK. These infections have occasionally spread to healthcare workers and are giving rise to considerable concern.
The underlying mechanisms of resistance are considered in Chapter 13.
2.5 Drug combinations
Antibiotics are generally used alone, but may on occasion be prescribed in combination. Combining two antibiotics may result in synergism, indifference or antagonism. In the case of synergism, microbial inhibition is achieved at concentrations below that for each agent alone and may prove advantageous in treating relatively insusceptible infections such as enterococcal endocarditis, where a combination of penicillin and gentamicin is synergistically active. Another advantage of synergistic combinations is that it may enable the use of toxic agents where dose reductions are possible. For example, meningitis caused by the fungus Cryptococcus neoformans responds to an abbreviated course of amphotericin B when it is combined with 5-flucytosine, thereby reducing the risk of toxicity from amphotericin B.
Combined drug use is occasionally recommended to prevent resistance emerging during treatment. For example, treatment may fail when fusidic acid is used alone to treat Staph. aureus infections, because resistant strains develop rapidly; this is prevented by combining fusidic acid with flucloxacillin. Likewise, tuberculosis is initially treated with a minimum of three agents, such as rifampicin, isoniazid and pyrazinamide; again drug resistance is prevented, which may result if either agent is used alone.
The most common reason for using combined therapy is in the treatment of confirmed or suspected mixed infections where a single agent alone will fail to cover all pathogenic organisms. This is the case in serious abdominal sepsis where mixed aerobic and anaerobic infections are common and the use of metronidazole in combination with either an aminoglycoside or a broad spectrum cephalosporin is essential. Finally, drugs are used in combination in patients who are seriously ill and about whom uncertainty exists concerning the microbiological nature of their infection. This initial ‘blind therapy ’ frequently includes a broad spectrum penicillin or cephalosporin in combination with an aminoglycoside. The regimen should be modified in the light of subsequent microbiological information.
2.6 Adverse reactions
Regrettably, all chemotherapeutic agents have the potential to produce adverse reactions with varying degrees of frequency and severity, and these include hypersensitivity reactions and toxic effects. These may be dose related and predictable in a patient with a history of hypersensitivity or a previous toxic reaction to a drug or its chemical analogues. However, many adverse events are idiosyncratic and therefore unpredictable.
Hypersensitivity reactions range in severity from fatal anaphylaxis, in which there is widespread tissue oedema, airway obstruction and cardiovascular collapse, to minor and reversible hypersensitivity reactions such as skin eruptions and drug fever. Such reactions are more likely in those with a history of hypersensitivity to the drug, and are more frequent in patients with previous allergic diseases such as childhood eczema or asthma. It is important to question patients closely concerning hypersensitivity reactions before prescribing, as it precludes the use of all compounds within a class, such as the sulphonamides or tetracyclines, while cephalosporins and carbapenems should be used only with caution in patients who are allergic to penicillin, because these agents are structurally related. They should be avoided entirely in those who have had a previous severe hypersensitivity reaction to penicillin.
Drug toxicity is often dose-related and may affect a variety of organs or tissues. For example, the aminoglycosides are both nephrotoxic and ototoxic to varying degrees; therefore, dosaging should be individualized and the serum assayed, especially where renal function is abnormal, to avoid toxic effects and non-therapeutic drug concentrations. An example of dose related toxicity is chloramphenicol-induced bone marrow suppression. Chloramphenicol interferes with the normal maturation of bone marrow stem cells and high concentrations may result in a steady fall in circulating red and white cells and also platelets. This effect is generally reversible with dose reduction or drug withdrawal. This dose-related toxic reaction of chloramphenicol should be contrasted with idiosyncratic bone marrow toxicity which is unrelated to dose and occurs at a much lower frequency of approximately 1:40000 and is frequently irreversible, ending fatally. Toxic effects may also be genetically determined. For example, peripheral neuropathy may occur in those who are slow acetylators of isoniazid, while haemolysis occurs in those deficient in the red cell enzyme glucose 6 phosphate dehydrogenase, when treated with sulphonamides, primaquine, quinolones or nitrofurantoin.
2.7 Superinfection
Anti-infective drugs not only affect the invading organism undergoing treatment but also have an impact on the normal bacterial flora, especially of the skin and mucous membranes. This may result in microbial overgrowth of resistant organisms with subsequent superinfection. One example is the common occurrence of oral or vaginal candidiasis in patients treated with broad-spectrum agents such as ampicillin or tetracycline. A more serious example is the development of pseudomembranous colitis from the overgrowth of toxin producing strains of Clostridium difficile present in the bowel flora following the use of clindamycin or broad-spectrum antibiotics, though any antimicrobial can precipitate this condition. C. difficile associated diarrhoea is managed by drug withdrawal and oral vancomycin, or oral/intravenous metronidazole. Intravenous immunoglogbulin is occasionally used in severe cases, and rarely, colectomy (excision of part or whole of the colon) may be necessary. Once established, C.difficile infection is transmissible, particularly in the hospital setting; isolation of symptomatic patients and strict observation of hygiene practices (e.g. hand washing) are therefore key in preventing outbreaks.
2.8 Chemoprophylaxis
An increasingly important use of antimicrobial agents is that of infection prevention, especially in relationship to surgery. Infection remains one of the most important complications of many surgical procedures, and the recognition that perioperative antibiotics are effective and safe in preventing this complication has proved a major advance in surgery. The principles that underlie the chemoprophylactic use of antibacterials relate to the predictability of infection for a particular surgical procedure, in terms of its occurrence, microbial aetiology and susceptibility to antibiotics. Therapeutic drug concentrations present at the operative site at the time of surgery rapidly reduce the number of potentially infectious organisms and prevent wound sepsis. If prophylaxis is delayed to the postoperative period, then efficacy is markedly impaired. It is important that chemoprophylaxis be limited to the perioperative period, the first dose being administered approximately 1 hour before surgery for injectable agents; for many procedures and operative sites, a single dose is now considered sufficient. Prolonging chemoprophylaxis beyond this period is not cost effective and increases the risk of adverse drug reactions and superinfection. One of the best examples of the efficacy of surgical prophylaxis is in the area of largebowel surgery. Before the widespread use of chemoprophylaxis, postoperative infection rates for colectomy were often 30% or higher; these have now been reduced to around 5%.
Chemoprophylaxis has been extended to other surgical procedures where the risk of infection may be low but its occurrence has serious consequences. This is especially true for the implantation of prosthetic joints or heart valves. These are major surgical procedures and although infection may be infrequent its consequences are serious and on balance the use of chemoprophylaxis is cost-effective.
Examples of chemoprophylaxis in the non-surgical arena include the prevention of pneumococcal infection with penicillin V in asplenia or patients with sicklecell disease, and the prevention of secondary cases of meningococcal meningitis with rifampicin or ciprofloxacin among household contacts of an index case.
The choice of antimicrobial chemotherapy is initially dependent on the clinical diagnosis. In some circumstances the clinical diagnosis implies a microbiological diagnosis which may dictate specific therapy. For example, typhoid fever is caused by Salmonella enterica serovar Typhi, which is generally sensitive to cotrimoxazole, ceftriaxone and ciprofloxacin. However, for many infections, establishing a clinical diagnosis implies a range of possible microbiological causes and requires laboratory confirmation from samples collected, preferably before antibiotic therapy is begun. Laboratory isolation and susceptibility testing of the causative agent establish the diagnosis with certainty and make drug selection more rational. However, in many circumstances, especially in general practice, microbiological documentation of an infection is not possible. Hence knowledge of the usual microbiological cause of a particular infection and its susceptibility to antimicrobial agents is essential for effective drug prescribing. The following section explores a selection of the problems associated with antimicrobial drug prescribing for a range of clinical conditions.
3.1 Respiratory tract infections
Infections of the respiratory tract are among the commonest of infections, and account for much consultation in general practice and a high percentage of acute hospital admissions. They are divided into infections of the upper respiratory tract, involving the ears, throat, nasal sinuses and the trachea, and the lower respiratory tract (LRT), where they affect the airways, lungs and pleura.
3.1.1 Upper respiratory tract infections
Acute pharyngitis presents a diagnostic and therapeutic dilemma. The majority of sore throats are caused by a variety of viruses; fewer than 20% are bacterial and hence potentially responsive to antibiotic therapy. However, antibiotics are widely prescribed and this reflects the difficulty in discriminating streptococcal from nonstreptococcal infections clinically in the absence of microbiological documentation. Nonetheless, Strep. pyogenes is the most important bacterial pathogen and this responds to oral penicillin. However, up to 10 days’ treatment is required for its eradication from the throat. This requirement causes problems with compliance as symptomatic improvement generally occurs within 2–3 days.
Although viral infections are important causes of both otitis media and sinusitis, they are generally self limiting. Bacterial infections may complicate viral illnesses, and are also primary causes of ear and sinus infections. Streptococcus pneumoniae and Haemophilus influenzae are the commonest bacterial pathogens. Amoxicillin is widely prescribed for these infections as it is microbiologically active, penetrates the middle ear and sinuses, is well tolerated and has proved effective.
3.1.2 Lower respiratory tract infections
Infections of the LRT include pneumonia, lung abscess, bronchitis, bronchiectasis and infective complications of cystic fibrosis. Each presents a specific diagnostic and therapeutic challenge, which reflects the variety of pathogens involved and the frequent difficulties in establishing an accurate microbial diagnosis. The laboratory diagnosis of LRT infections is largely dependent upon culturing sputum. Unfortunately this may be contaminated with the normal bacterial flora of the upper respiratory tract during expectoration. In hospitalized patients, the empirical use of antibiotics before admission substantially diminishes the value of sputum culture and may result in overgrowth by nonpathogenic microbes, thus causing difficulty with the interpretation of sputum culture results. Alternative diagnostic samples include needle aspiration of sputum directly from the trachea or of fluid within the pleural cavity. Blood may also be cultured and serum examined for antibody responses or microbial antigens. In the community, few patients will have their LRT infection diagnosed microbiologically and the choice of antibiotic is based on clinical diagnosis.
3.1.2.1 Pneumonia
The range of pathogens causing acute pneumonia includes viruses, bacteria and, in the immunocompromised host, parasites and fungi. Table 14.2 summarizes these pathogens and indicates drugs appropriate for their treatment. Clinical assessment includes details of the evolution of the infection, any evidence of a recent viral infection, the age of the patient and risk factors such as corticosteroid therapy or preexisting lung disease. The extent of the pneumonia, as assessed clinically or by X ray, is also important.
Table 14.2 Microorganisms responsible for pneumonia and the therapeutic agent of choice
| Pathogen | Drug(s) of choice |
| Streptococcus pneumoniae | Penicillin |
| Staphylococcus aureus (MSSA) | Flucloxacillin ± fusidic acid |
| Staphylococcus aureus (MRSA) | Vancomycin or linezolid |
| Haemophilus influenzae | Cefotaxime or ciprofloxacin |
| Klebsiella pneumoniae | Cefotaxime ± gentamicin |
| Pseudomonas aeruginosa | Ceftazidime ± g entamicin or piperacillin-tazobactam ± gentamicin |
| Mycoplasma pneumoniae | Erythromycin or tetracycline |
| Legionella pneumophila | Erythromycin ± rifampicin |
| Chlamydia psittaci | Tetracycline |
| Mycobacterium tuberculosis | R ifampicin + isoniazid + ethambutol + pyrazinamidea |
| Herpes simplex, varicella/zoster | Aciclovir |
| Candida spp. | Fluconazole or echinocandins (caspofungin, anidulafungin) |
| Aspergillus spp. | Amphotericin B or broad-spectrum triazoles (e.g. voriconazole) |
| Anaerobic bacteria | Penicillin or metronidazole |
aReduce to two drugs after 6–8 weeks.
Streptococcus pneumoniae remains the commonest cause of pneumonia and still responds well to penicillin despite a global increase in isolates showing reduced susceptibility to this agent. So called ‘respiratory quinolones ’ such as levofloxacin and moxifloxacin, which exhibit increased activity against Gram positive organisms compared to ciprofloxacin, are an alternative. A number of atypical infections may cause pneumonia and include Mycoplasma pneumoniae, Legionella pneumophila, psittacosis and occasionally Q fever. With psittacosis there may be a history of contact with parrots or budgerigars; while legionnaires ’ disease has often been acquired during hotel holidays in the Mediterranean area. The atypical pneumonias, unlike pneumococcal pneumonia, do not respond to penicillin. Legionnaires’ disease is treated with erythromycin and, in the presence of severe pneumonia, rifampicin is added to the regimen. Mycoplasma infections are best treated with either erythromycin or tetracycline, while the latter drug is indicated for both psittacosis and Q fever.
3.1.2.2 Lung abscess
Destruction of lung tissue may lead to abscess formation and is a feature of aerobic Gramnegative bacillary and Staph. aureus infections. In addition, aspiration of oropharyngeal secretion can lead to chronic lowgrade sepsis with abscess formation and the expectoration of foul smelling sputum that characterizes anaerobic sepsis. The latter condition responds to highdose penicillin, which is active against most of the normal oropharyngeal flora, while metronidazole may be appropriate for strictly anaerobic infections. In the case of aerobic Gram-negative bacillary sepsis, aminoglycosides, with orwithout a broad-spectrum cephalosporin, are the agents of choice. Acute staphylococcal pneumonia is an extremely serious infection and requires treatment with high dose flucloxacillin alone or in combination with fusidic acid.
3.1.2.3 Cystic fibrosis
Cystic fibrosis is a multisystem congenital abnormality that often affects the lungs and results in recurrent infections, initially with Staph. aureus, subsequently with H. influenzae and eventually leads on to recurrent Pseudomonas aeruginosa infection. The last organism is associated with copious quantities of purulent sputum that are extremely difficult to expectorate. Ps. aeruginosa is a co factor in the progressive lung damage that is eventually fatal in these patients. Repeated courses of antibiotics are prescribed and although they have improved the quality and longevity of life, infections caused by Ps. aeruginosa are difficult to treat and require repeated hospitalization and administration of parenteral antibiotics such as an aminoglycoside, either alone or in combination with an antipseudomonal penicillin or cephalosporin. The dose of aminoglycosides tolerated by these patients is often higher than in normal individuals and is associated with larger volumes of distribution for these and other agents. Some benefit may also be obtained from inhaled aerosolized antibiotics. Unfortunately drug resistance may emerge and makes drug selection more dependent upon laboratory guidance.
3.2 Urinary tract infections
Urinary tract infection is a common problem in both community and hospital practice. Although occurring throughout life, infections are more common in preschool girls and women during their childbearing years, although in the elderly the sex distribution is similar. Infection is predisposed by factors that impair urine flow. These include congenital abnormalities, reflux of urine from the bladder into the ureters, kidney stones and tumours and, in males, enlargement of the prostate gland. Bladder catheterization is an important cause of urinary tract infection in hospitalized patients.
3.2.1 Pathogenesis
In those with structural or drainage problems the risk exists of ascending infection to involve the kidney and occasionally the bloodstream. Although structural abnormalities may be absent in women of childbearing years, infection can become recurrent, symptomatic and extremely distressing. Of greater concern is the occurrence of infection in the pre-school child, as normal maturation of the kidney may be impaired and may result in progressive damage which presents as renal failure in later life.
From a therapeutic point of view, it is essential to confirm the presence of bacteriuria (a condition in which there are bacteria in the urine), as symptoms alone are not a reliable method of documenting infection. This applies particularly to bladder infection, where the symptoms of burning micturition (dysuria) and frequency can be associated with a variety of nonbacteriuric conditions. Patients with symptomatic bacteriuria should always be treated. However, the necessity to treat asymptomatic bacteriuric patients varies with age and the presence or absence of underlying urinary tract abnormalities. In the preschool child it is essential to treat all urinary tract infections and maintain the urine in a sterile state so that normal kidney maturation can proceed. Likewise in pregnancy there is a risk of infection ascending from the bladder to involve the kidney. This is a serious complication and may result in premature labour. Other indications for treating asymptomatic bacteriuria include the presence of underlying renal abnormalities such as stones, which may be associated with repeated infections caused by Proteus spp.
3.2.2 Drug therapy
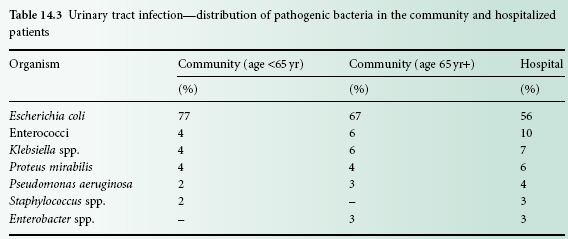
The antimicrobial treatment of urinary tract infection presents a number of interesting challenges. Drugs must be selected for their ability to achieve high urinary concentrations and, if the kidney is involved, adequate tissue concentrations. Safety in childhood or pregnancy is important as repeated or prolonged medication may be necessary. The choice of agent will be dictated by the microbial aetiology and susceptibility findings, because the latter can vary widely among Gramnegative enteric bacilli, especially in patients who are hospitalized. Table 14.3 shows the distribution of bacteria causing urinary tract infection in the community and in hospitalized patients. The greater tendency towards infections caused by Klebsiella spp. and Ps. aeruginosa should be noted as antibiotic sensitivity is more variable for these pathogens. Drug resistance has increased substantially in recent years and has reduced the value of formerly widely prescribed agents such as the sulphonamides and ampicillin.
Table 14.3 Urinary tract infection—distribution of pathogenic bacteria in the community and hospitalized patients
Reprinted from Journal of Infection, 46(2):94-100, Farrell, D.J etal, Urinary tract infection—distribution of pathogenic bacteria in the community and hospitalized patients, Copyright (2003), with permission from Elsevier