52
Antiprotozoal Drugs
CASE STUDY
A 5-year-old American girl presents with a 1-week history of intermittent chills, fever, and sweats. She had returned home 2 weeks earlier after leaving the United States for the first time to spend 3 weeks with her grandparents in Nigeria. She received all standard childhood immunizations, but no additional treatment before travel, since her parents have returned to their native Nigeria frequently without medical consequences. Three days ago, the child was seen in an outpatient clinic and diagnosed with a viral syndrome. Examination reveals a lethargic child, with a temperature of 39.8°C (103.6°F) and splenomegaly. She has no skin rash or lymphadenopathy. Initial laboratory studies are remarkable for hematocrit 29.8%, platelets 45,000/mm3, creatinine 2.5 mg/dL (220 μmol/L), and mildly elevated bilirubin and transaminases. A blood smear shows ring forms of Plasmodium falciparum at 1.5% parasitemia. What treatment should be started?
 MALARIA
MALARIA
Malaria is the most important parasitic disease of humans and causes hundreds of millions of illnesses per year. Four species of plasmodium typically cause human malaria: Plasmodium falciparum, P vivax, P malariae, and P ovale. A fifth species, P knowlesi, is primarily a pathogen of monkeys but has recently been recognized to cause illness, including severe disease, in humans in Asia. Although all of the latter species may cause significant illness, P falciparum is responsible for the majority of serious complications and deaths. Drug resistance is an important therapeutic problem, most notably with P falciparum.
PARASITE LIFE CYCLE
An anopheline mosquito inoculates plasmodium sporozoites to initiate human infection (Figure 52–1). Circulating sporozoites rapidly invade liver cells, and exoerythrocytic stage tissue schizonts mature in the liver. Merozoites are subsequently released from the liver and invade erythrocytes. Only erythrocytic parasites cause clinical illness. Repeated cycles of infection can lead to the infection of many erythrocytes and serious disease. Sexual stage gametocytes also develop in erythrocytes before being taken up by mosquitoes, where they develop into infective sporozoites.
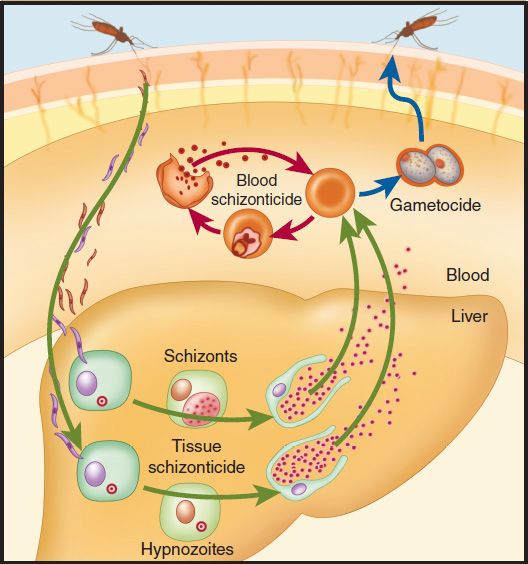
FIGURE 52–1 Life cycle of malaria parasites. Only the asexual erythrocytic stage of infection causes clinical malaria. All effective antimalarial treatments are blood schizonticides that kill this stage. (Reproduced from Baird JK: Effectiveness of antimalarial drugs. N Engl J Med 2005;352:1565.)
In P falciparum and P malariae infection, only one cycle of liver cell invasion and multiplication occurs, and liver infection ceases spontaneously in less than 4 weeks. Thus, treatment that eliminates erythrocytic parasites will cure these infections. In P vivax and P ovale infections, a dormant hepatic stage, the hypnozoite, is not eradicated by most drugs, and relapses can occur after therapy directed against erythrocytic parasites. Eradication of both erythrocytic and hepatic parasites is required to cure these infections.
DRUG CLASSIFICATION
Several classes of antimalarial drugs are available (Table 52–1 and Figure 52–2). Drugs that eliminate developing or dormant liver forms are called tissue schizonticides; those that act on erythrocytic parasites are blood schizonticides; and those that kill sexual stages and prevent transmission to mosquitoes are gametocides. No single available agent can reliably effect a radical cure, ie, eliminate both hepatic and erythrocytic stages. Few available agents are causal prophylactic drugs, ie, capable of preventing erythrocytic infection. However, all effective chemoprophylactic agents kill erythrocytic parasites before they increase sufficiently in number to cause clinical disease.
TABLE 52–1 Major antimalarial drugs.
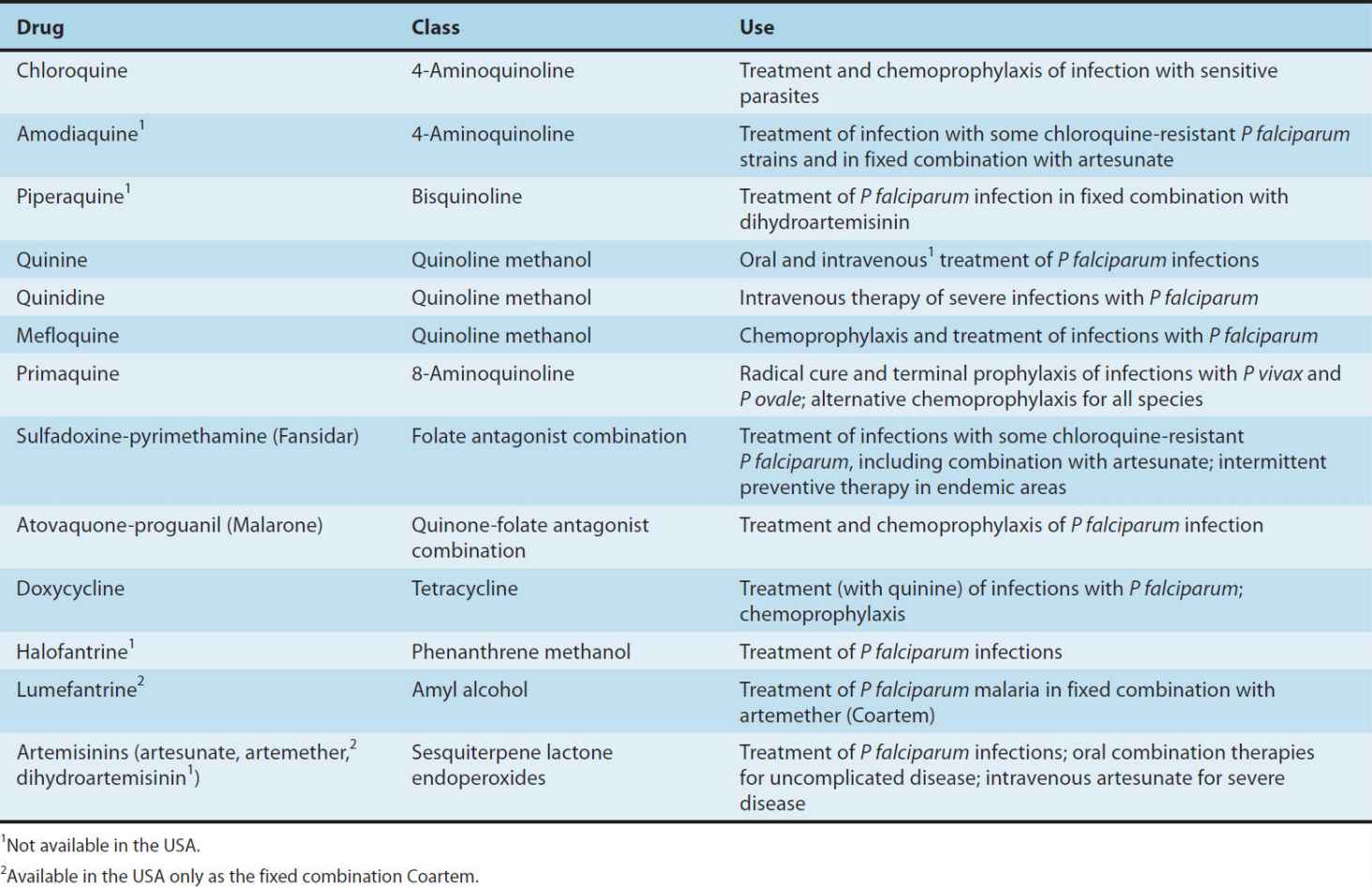
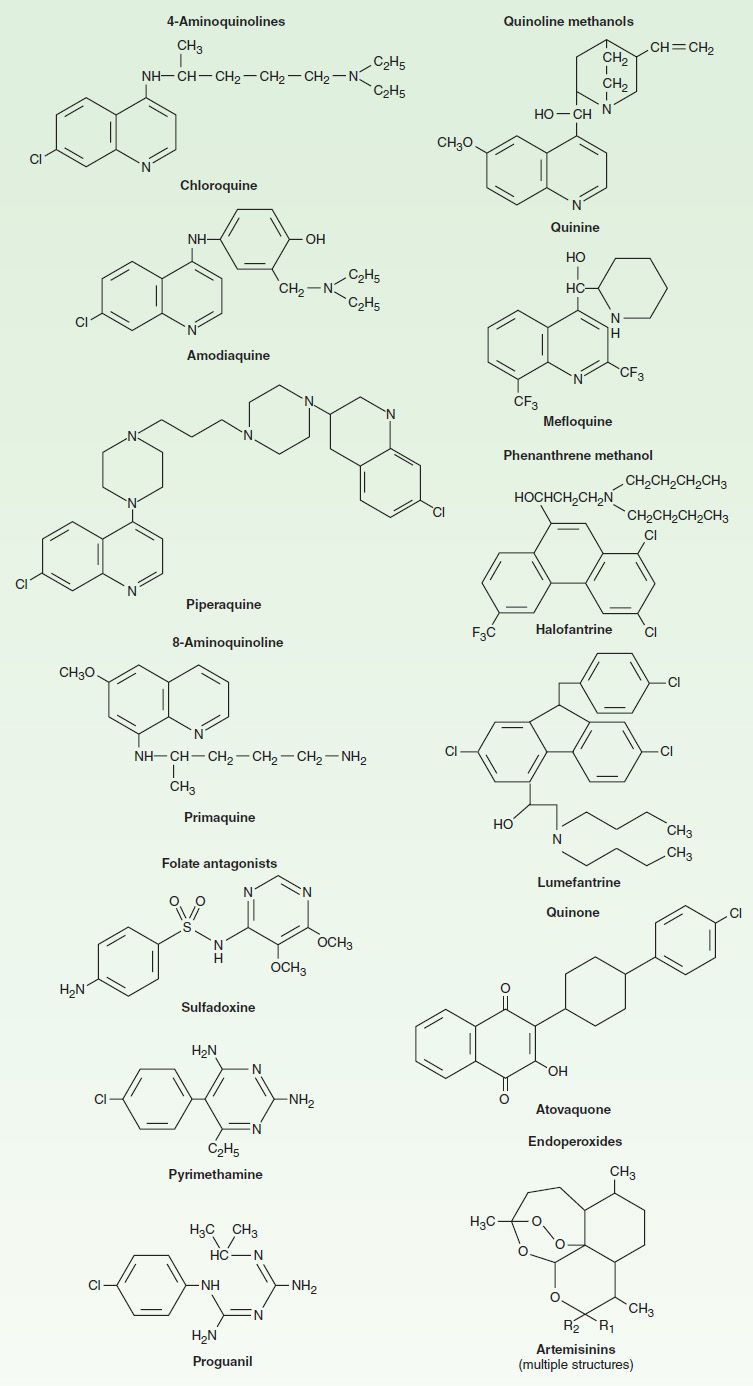
FIGURE 52–2 Structural formulas of some antimalarial drugs.
CHEMOPROPHYLAXIS & TREATMENT
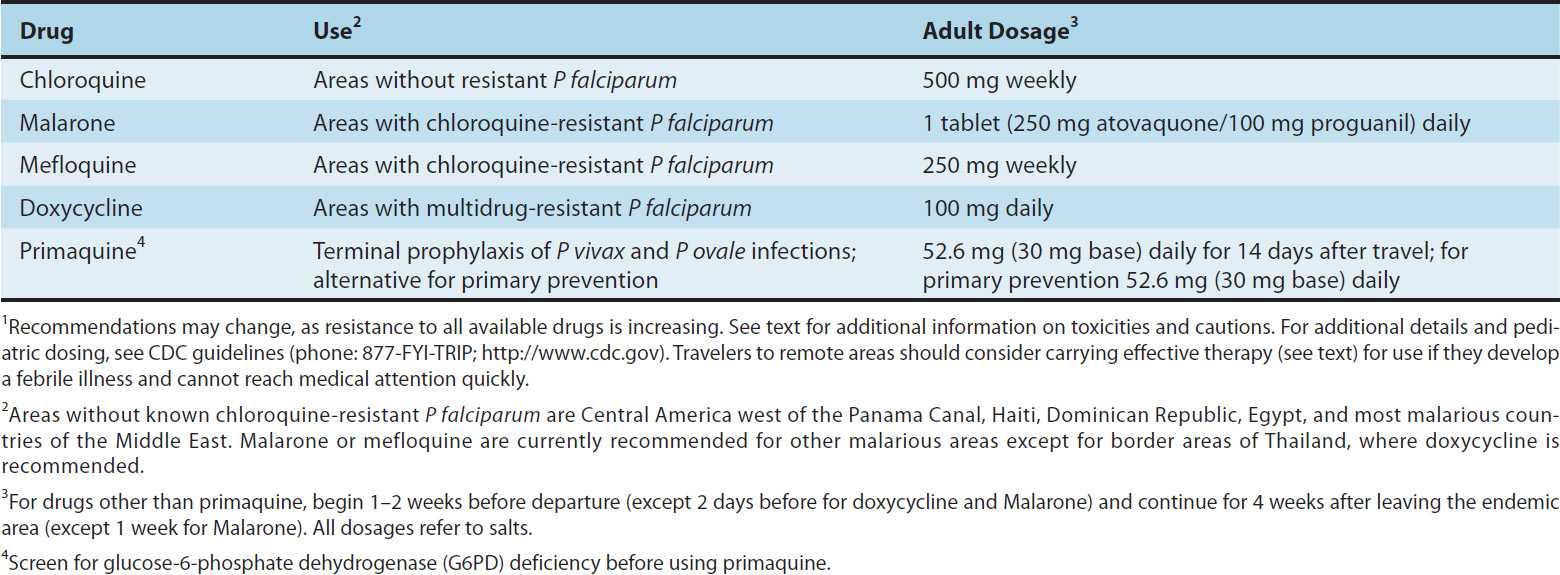
When patients are counseled on the prevention of malaria, it is imperative to emphasize measures to prevent mosquito bites (eg, with insect repellents, insecticides, and bed nets), because parasites are increasingly resistant to multiple drugs and no chemoprophylactic regimen is fully protective. Current recommendations from the Centers for Disease Control and Prevention (CDC) include the use of chloroquine for chemoprophylaxis in the few areas infested by only chloroquine-sensitive malaria parasites (principally Hispaniola and Central America west of the Panama Canal), and mefloquine, Malarone,* or doxycycline for most other malarious areas, with doxycycline preferred for areas with a high prevalence of multidrug-resistant falciparum malaria (principally border areas of Thailand) (Table 52–2). CDC recommendations should be checked regularly (Phone: 770-488-7788; after hours 770-488-7100; Internet: http://www.cdc.gov/malaria), because these may change in response to changing resistance patterns and increasing experience with new drugs. In some circumstances, it may be appropriate for travelers to carry supplies of drugs with them in case they develop a febrile illness when medical attention is unavailable. Regimens for self-treatment include new artemisinin-based combination therapies (see below), which are widely available internationally (and, in the case of Coartem**, in the USA); Malarone; mefloquine; and quinine. Most authorities do not recommend routine terminal chemoprophylaxis with primaquine to eradicate dormant hepatic stages of P vivax and P ovale after travel, but this may be appropriate in some circumstances, especially for travelers with major exposure to these parasites.
TABLE 52–2 Drugs for the prevention of malaria in travelers.1
Multiple drugs are available for the treatment of malaria that presents in the USA (Table 52–3). Most nonfalciparum infections and falciparum malaria from areas without known resistance should be treated with chloroquine. For vivax malaria from areas with suspected chloroquine resistance, including Indonesia and Papua New Guinea, other therapies effective against falciparum malaria may be used. Vivax and ovale malaria should subsequently be treated with primaquine to eradicate liver forms. Uncomplicated falciparum malaria from most areas is most often treated with Malarone, but new artemisinin-based combinations are increasingly the international standard of care, and one combination, Coartem, is now available in the USA. Other agents that are generally effective against resistant falciparum malaria include mefloquine, quinine, and halofantrine, all of which have toxicity concerns at treatment dosages. Severe falciparum malaria is treated with intravenous artesunate, quinidine, or quinine (intravenous quinine is not available in the USA).
TABLE 52–3 Treatment of malaria.
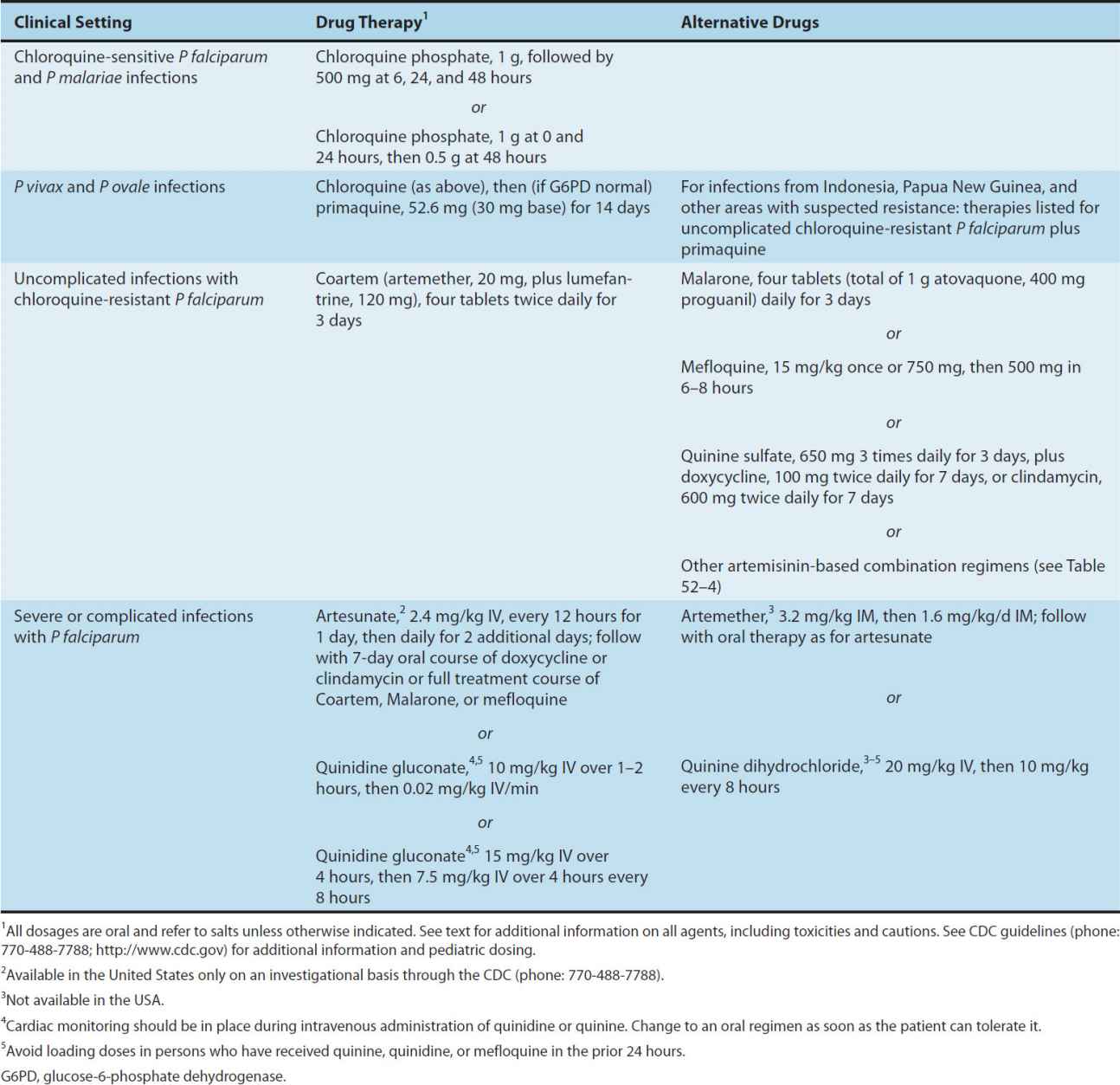
CHLOROQUINE
Chloroquine has been a drug of choice for both treatment and chemoprophylaxis of malaria since the 1940s, but its usefulness against P falciparum has been seriously compromised by drug resistance. It remains the drug of choice in the treatment of sensitive P falciparum and other species of human malaria parasites.
Chemistry & Pharmacokinetics
Chloroquine is a synthetic 4-aminoquinoline (Figure 52–2) formulated as the phosphate salt for oral use. It is rapidly and almost completely absorbed from the gastrointestinal tract, reaches maximum plasma concentrations in about 3 hours, and is rapidly distributed to the tissues. It has a very large apparent volume of distribution of 100–1000 L/kg and is slowly released from tissues and metabolized. Chloroquine is principally excreted in the urine with an initial half-life of 3–5 days but a much longer terminal elimination half-life of 1–2 months.
Antimalarial Action & Resistance
When not limited by resistance, chloroquine is a highly effective blood schizonticide. It is also moderately effective against gametocytes of P vivax, P ovale, and P malariae but not against those of P falciparum. Chloroquine is not active against liver stage parasites. The drug probably acts by concentrating in parasite food vacuoles, preventing the biocrystallization of the hemoglobin breakdown product, heme, into hemozoin, and thus eliciting parasite toxicity due to the buildup of free heme.
Resistance to chloroquine is now very common among strains of P falciparum and uncommon but increasing for P vivax. In P falciparum, mutations in a putative transporter, PfCRT, have been correlated with resistance. Chloroquine resistance can be reversed by certain agents, including verapamil, desipramine, and chlorpheniramine, but the clinical value of resistance-reversing drugs is not established.
Clinical Uses
1. Treatment—Chloroquine is the drug of choice in the treatment of uncomplicated nonfalciparum and sensitive falciparum malaria. It rapidly terminates fever (in 24–48 hours) and clears parasitemia (in 48–72 hours) caused by sensitive parasites. Chloroquine has been replaced by other drugs, principally artemisinin-based combination therapies, as the standard therapy to treat falciparum malaria in most endemic countries. Chloroquine does not eliminate dormant liver forms of P vivax and P ovale, and for that reason primaquine must be added for the radical cure of these species.
2. Chemoprophylaxis—Chloroquine is the preferred chemoprophylactic agent in malarious regions without resistant falciparum malaria. Eradication of P vivax and P ovale requires a course of primaquine to clear hepatic stages.
3. Amebic liver abscess—Chloroquine reaches high liver concentrations and may be used for amebic abscesses that fail initial therapy with metronidazole (see below).
Adverse Effects
Chloroquine is usually very well tolerated, even with prolonged use. Pruritus is common, primarily in Africans. Nausea, vomiting, abdominal pain, headache, anorexia, malaise, blurring of vision, and urticaria are uncommon. Dosing after meals may reduce some adverse effects. Rare reactions include hemolysis in glucose-6-phosphate dehydrogenase (G6PD)-deficient persons, impaired hearing, confusion, psychosis, seizures, agranulocytosis, exfoliative dermatitis, alopecia, bleaching of hair, hypotension, and electrocardiographic changes (QRS widening, T-wave abnormalities). The long-term administration of high doses of chloroquine for rheumatologic diseases (see Chapter 36) can result in irreversible ototoxicity, retinopathy, myopathy, and peripheral neuropathy, but these are rarely seen with standard-dose weekly chemoprophylaxis. Intramuscular injections or intravenous infusions of chloroquine hydrochloride can result in severe hypotension and respiratory and cardiac arrest, and should be avoided.
Contraindications & Cautions
Chloroquine is contraindicated in patients with psoriasis or porphyria. It should generally not be used in those with retinal or visual field abnormalities or myopathy. Chloroquine should be used with caution in patients with liver, neurologic, or hematologic disorders. The antidiarrheal agent kaolin and calcium- and magnesium-containing antacids interfere with the absorption of chloroquine and should not be co-administered. Chloroquine is considered safe in pregnancy and for young children.
OTHER QUINOLINES
Amodiaquine is closely related to chloroquine, and it probably shares mechanisms of action and resistance. Amodiaquine has been widely used to treat malaria because of its low cost, limited toxicity, and, in some areas, effectiveness against chloroquine-resistant strains of P falciparum, but toxicities, including agranulocytosis, aplastic anemia, and hepatotoxicity, have limited its use. However, recent reevaluation has shown that serious toxicity from amodiaquine is uncommon. The most important current use of amodiaquine is in combination therapy. The World Health Organization (WHO) lists artesunate plus amodiaquine as a recommended therapy for falciparum malaria (Table 52–4). This combination is now available as a single tablet (ASAQ, Arsucam, Coarsucam) and is the first-line therapy for the treatment of uncomplicated falciparum malaria in many countries in Africa. Another combination, amodiaquine plus sulfadoxine-pyrimethamine, remains reasonably effective for the treatment of falciparum malaria. Long-term chemoprophylaxis with amodiaquine is best avoided because of its apparent increased toxicity with long-term use, but short-term seasonal malaria chemoprevention with amodiaquine plus sulfadoxine-pyrimethamine (monthly treatment doses for 3–4 months during the transmission season) is now recommended by the WHO for the Sahel sub-region of Africa.
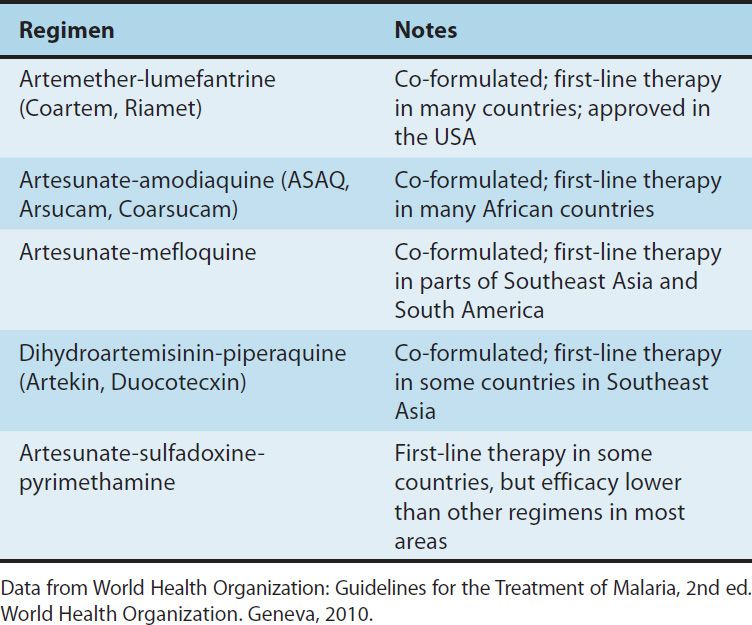
TABLE 52–4 WHO recommendations for the treatment of falciparum malaria.
Piperaquine is a bisquinoline that was used widely to treat chloroquine-resistant falciparum malaria in China in the 1970s–1980s, but its use waned after resistance became widespread. Recently, piperaquine combined with dihydroartemisinin (Artekin, Duocotecxin) has shown excellent efficacy and safety for the treatment of falciparum malaria, without apparent drug resistance. Piperaquine has a longer half-life (~ 28 days) than amodiaquine (~ 14 days), mefloquine (~ 14 days), or lumefantrine (~ 4 days), leading to a longer period of post-treatment prophylaxis with dihydroartemisinin-piperaquine than with the other leading artemisinin-based combinations; this feature should be particularly advantageous in high transmission areas. Dihydroartemisinin-piperaquine is now the first-line therapy for the treatment of uncomplicated falciparum malaria in some countries in Asia.
ARTEMISININ & ITS DERIVATIVES
Artemisinin (qinghaosu) is a sesquiterpene lactone endoperoxide (Figure 52–2), the active component of an herbal medicine that has been used as an antipyretic in China for over 2000 years. Artemisinin is insoluble and can only be used orally. Analogs have been synthesized to increase solubility and improve antimalarial efficacy. The most important of these analogs are artesunate (water-soluble; oral, intravenous, intramuscular, and rectal administration), artemether (lipid-soluble; oral, intramuscular, and rectal administration), and dihydroartemisinin (water-soluble; oral administration).
Chemistry & Pharmacokinetics
Artemisinin and its analogs are rapidly absorbed, with peak plasma levels occurring promptly. Half-lives after oral administration are 30–60 minutes for artesunate and dihydroartemisinin, and 2–3 hours for artemether. Artemisinin, artesunate, and artemether are rapidly metabolized to the active metabolite dihydroartemisinin. Drug levels appear to decrease after a number of days of therapy.
Antimalarial Action & Resistance
The artemisinins are now widely available, but monotherapy for the treatment of uncomplicated malaria is strongly discouraged. Rather, co-formulated artemisinin-based combination therapies are recommended to improve efficacy and prevent the selection of artemisinin-resistant parasites. The oral combination regimen Coartem (artemether-lumefantrine) was approved by the FDA in 2009, and may be considered the new first-line therapy in the USA for uncomplicated falciparum malaria, although it may not be widely available. Intravenous artesunate was made available by the CDC in 2007; use of the drug is initiated by contacting the CDC, which will release it for appropriate indications (falciparum malaria with signs of severe disease or inability to take oral medications) from stocks stored around the USA.
Artemisinin and its analogs are very rapidly acting blood schizonticides against all human malaria parasites. Artemisinins have no effect on hepatic stages. The antimalarial activity of artemisinins appears to result from the production of free radicals that follows the iron-catalyzed cleavage of the artemisinin endoperoxide bridge. Artemisinin resistance is not yet a widespread problem, but delayed clearance of P falciparum infections and decreased treatment efficacy in parts of Southeast Asia demonstrate a worrisome focus of resistance.
Clinical Uses
Artemisinin-based combination therapy is now the standard of care for treatment of uncomplicated falciparum malaria in nearly all areas endemic for falciparum malaria. The leading regimens are highly efficacious, safe, and well tolerated. These regimens were developed because the short plasma half-lives of the artemisinins led to unacceptably high recrudescence rates after short-course therapy, which were reversed by inclusion of longer-acting drugs. Combination therapy also helps to protect against the selection of artemisinin resistance. However, with completion of dosing after 3 days, the artemisinin components are rapidly eliminated, and so selection of resistance to partner drugs is of concern.
The WHO recommends five artemisinin-based combinations for the treatment of uncomplicated falciparum malaria (Table 52–4). One of these, artesunate-sulfadoxine-pyrimethamine is not recommended in many areas owing to unacceptable levels of resistance to sulfadoxine-pyrimethamine, but it is the first-line therapy in some countries. The other recommended regimens are available as combination formulations, although manufacturing standards may vary. Artesunate-mefloquine is highly effective in Southeast Asia, where resistance to many antimalarials is common; it is the first-line therapy in some countries in Southeast Asia and South America. This regimen is less practical for other areas, particularly Africa, because of its relatively high cost and poor tolerability. Either artesunate-amodiaquine or artemether-lumefantrine is the standard treatment for uncomplicated falciparum malaria in most countries in Africa and some additional endemic countries on other continents. Dihydroartemisinin-piperaquine is a newer regimen that has shown excellent efficacy; it is a first-line therapy for falciparum malaria in parts of Southeast Asia.
Artemisinins also have outstanding efficacy in the treatment of complicated falciparum malaria. Large randomized trials and meta-analyses have shown that intramuscular artemether has an efficacy equivalent to that of quinine and that intravenous artesunate is superior to intravenous quinine in terms of parasite clearance time and—most important—patient survival. Intravenous artesunate also has a superior side-effect profile when compared with intravenous quinine or quinidine. Thus, intravenous artesunate has replaced quinine as the standard of care for the treatment of severe falciparum malaria, although it is not yet available in many areas. Artesunate and artemether have also been effective in the treatment of severe malaria when administered rectally, offering a valuable treatment modality when parenteral therapy is not available.
Adverse Effects & Cautions
Artemisinins are generally very well tolerated. The most commonly reported adverse effects are nausea, vomiting, diarrhea, and dizziness, and these may often be due to underlying malaria rather than the medications. Rare serious toxicities include neutropenia, anemia, hemolysis, elevated liver enzymes, and allergic reactions. Irreversible neurotoxicity has been seen in animals, but only after doses much higher than those used to treat malaria. Artemisinins have been embryotoxic in animal studies, but rates of congenital abnormalities, stillbirths, and abortions were not elevated, compared with those of controls, in women who received artemisinins during pregnancy. Based on this information and the significant risk of malaria during pregnancy, the WHO recommends artemisinin-based combination therapies for the treatment of uncomplicated falciparum malaria during the second and third trimesters of pregnancy (quinine plus clindamycin is recommended during the first trimester), intravenous artesunate or quinine for the treatment of severe malaria during the first trimester, and intravenous artesunate for treatment of severe malaria during the second and third trimesters.
QUININE & QUINIDINE
Quinine and quinidine remain important therapies for falciparum malaria—especially severe disease—although toxicity may complicate therapy.
Chemistry & Pharmacokinetics
Quinine is derived from the bark of the cinchona tree, a traditional remedy for intermittent fevers from South America. The alkaloid quinine was purified in 1820, and has been used in the treatment and prevention of malaria since that time. Quinidine, the dextrorotatory stereoisomer of quinine, is at least as effective as parenteral quinine in the treatment of severe falciparum malaria. After oral administration, quinine is rapidly absorbed, reaches peak plasma levels in 1–3 hours, and is widely distributed in body tissues. The use of a loading dose in severe malaria allows the achievement of peak levels within a few hours. The pharmacokinetics of quinine varies among populations. Individuals with malaria develop higher plasma levels of the drug than healthy controls, but toxicity is not increased, apparently because of increased protein binding. The half-life of quinine also is longer in those with severe malaria (18 hours) than in healthy controls (11 hours). Quinidine has a shorter half-life than quinine, mostly as a result of decreased protein binding. Quinine is primarily metabolized in the liver and excreted in the urine.
Antimalarial Action & Resistance
Quinine is a rapid-acting, highly effective blood schizonticide against the four species of human malaria parasites. The drug is gametocidal against P vivax and P ovale but not P falciparum. It is not active against liver stage parasites. The mechanism of action of quinine is unknown. Resistance to quinine is common in some areas of Southeast Asia, especially border areas of Thailand, where the drug may fail if used alone to treat falciparum malaria. However, quinine still provides at least a partial therapeutic effect in most patients.
Clinical Uses
1. Parenteral treatment of severe falciparum malaria—For many years quinine dihydrochloride or quinidine gluconate were the treatments of choice for severe falciparum malaria, although intravenous artesunate is now preferred. Quinine can be administered slowly intravenously or, in a dilute solution, intramuscularly, but parenteral preparations are not available in the USA. Quinidine is available (although not always readily accessible) in the USA for the parenteral treatment of severe falciparum malaria. Quinidine can be administered in divided doses or by continuous intravenous infusion; treatment should begin with a loading dose to achieve effective plasma concentrations promptly. Because of its cardiac toxicity and the relative unpredictability of its pharmacokinetics, intravenous quinidine should be administered slowly with cardiac monitoring. Therapy should be changed to an effective oral agent as soon as the patient has improved and can tolerate oral medications.
2. Oral treatment of falciparum malaria—Quinine sulfate is appropriate therapy for uncomplicated falciparum malaria except when the infection was transmitted in an area without documented chloroquine resistance. Quinine is commonly used with a second drug (most often doxycycline or, in children, clindamycin) to shorten the duration of use (usually to 3 days) and limit toxicity. Quinine is not generally used to treat nonfalciparum malaria, because it is less effective than chloroquine against these parasites and is more toxic.
3. Malarial chemoprophylaxis—Quinine is not generally used in chemoprophylaxis owing to its toxicity, although a daily dose of 325 mg is effective.
4. Babesiosis—Quinine is first-line therapy, in combination with clindamycin, in the treatment of infection with Babesia microti or other human babesial infections.
Adverse Effects
Therapeutic dosages of quinine and quinidine commonly cause tinnitus, headache, nausea, dizziness, flushing, and visual disturbances, a constellation of symptoms termed cinchonism. Mild symptoms of cinchonism do not warrant the discontinuation of therapy. More severe findings, often after prolonged therapy, include more marked visual and auditory abnormalities, vomiting, diarrhea, and abdominal pain. Hypersensitivity reactions include skin rashes, urticaria, angioedema, and bronchospasm. Hematologic abnormalities include hemolysis (especially with G6PD deficiency), leukopenia, agranulocytosis, and thrombocytopenia. Therapeutic doses may cause hypoglycemia through stimulation of insulin release; this is a particular problem in severe infections and in pregnant patients, who have increased sensitivity to insulin. Quinine can stimulate uterine contractions, especially in the third trimester. However, this effect is mild, and quinine and quinidine remain appropriate for treatment of severe falciparum malaria even during pregnancy. Intravenous infusions of the drugs may cause thrombophlebitis.
Severe hypotension can follow too-rapid intravenous infusions of quinine or quinidine. Electrocardiographic abnormalities (QT interval prolongation) are fairly common with intravenous quinidine, but dangerous arrhythmias are uncommon when the drug is administered appropriately in a monitored setting.
Blackwater fever is a rare severe illness that includes marked hemolysis and hemoglobinuria in the setting of quinine therapy for malaria. It appears to be due to a hypersensitivity reaction to the drug, although its pathogenesis is uncertain.
Contraindications & Cautions
Quinine (or quinidine) should be discontinued if signs of severe cinchonism, hemolysis, or hypersensitivity occur. It should be avoided if possible in patients with underlying visual or auditory problems. It must be used with great caution in those with underlying cardiac abnormalities. Quinine should not be given concurrently with mefloquine and should be used with caution in a patient with malaria who has previously received mefloquine chemoprophylaxis. Absorption may be blocked by aluminum-containing antacids. Quinine can raise plasma levels of warfarin and digoxin. Dosage must be reduced in renal insufficiency.
MEFLOQUINE
Mefloquine is effective therapy for many chloroquine-resistant strains of P falciparum and against other species. Although toxicity is a concern, mefloquine is one of the recommended chemoprophylactic drugs for use in most malaria-endemic regions with chloroquine-resistant strains.
Chemistry & Pharmacokinetics
Mefloquine hydrochloride is a synthetic 4-quinoline methanol that is chemically related to quinine. It can only be given orally because severe local irritation occurs with parenteral use. It is well absorbed, and peak plasma concentrations are reached in about 18 hours. Mefloquine is highly protein-bound, extensively distributed in tissues, and eliminated slowly, allowing a single-dose treatment regimen. The terminal elimination half-life is about 20 days, allowing weekly dosing for chemoprophylaxis. With weekly dosing, steady-state drug levels are reached over a number of weeks. Mefloquine and its metabolites are slowly excreted, mainly in the feces.
Antimalarial Action & Resistance
Mefloquine has strong blood schizonticidal activity against P falciparum and P vivax, but it is not active against hepatic stages or gametocytes. The mechanism of action is unknown. Sporadic resistance to mefloquine has been reported from many areas. At present, resistance appears to be uncommon except in regions of Southeast Asia with high rates of multidrug resistance (especially border areas of Thailand). Mefloquine resistance appears to be associated with resistance to quinine and halofantrine, but not with resistance to chloroquine.
Clinical Uses
1. Chemoprophylaxis—Mefloquine is effective in prophylaxis against most strains of P falciparum and probably all other human malarial species. Mefloquine is therefore among the drugs recommended by the CDC for chemoprophylaxis in all malarious areas except those with no chloroquine resistance (where chloroquine is preferred) and some rural areas of Southeast Asia with a high prevalence of mefloquine resistance. As with chloroquine, eradication of P vivax and P ovale requires a course of primaquine.
2. Treatment—Mefloquine is effective in treating uncomplicated falciparum malaria. The drug is not appropriate for treating individuals with severe or complicated malaria, since quinine, quinidine, and artemisinins are more rapidly active, and since drug resistance is less likely with those agents. The combination of artesunate plus mefloquine showed excellent antimalarial efficacy in regions of Southeast Asia with some resistance to mefloquine, and this regimen is now one of the combination therapies recommended by the WHO for the treatment of uncomplicated falciparum malaria (Table 52–4). Artesunate-mefloquine is the first-line therapy for uncomplicated falciparum malaria in a number of countries in Asia and South America.
Adverse Effects
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


