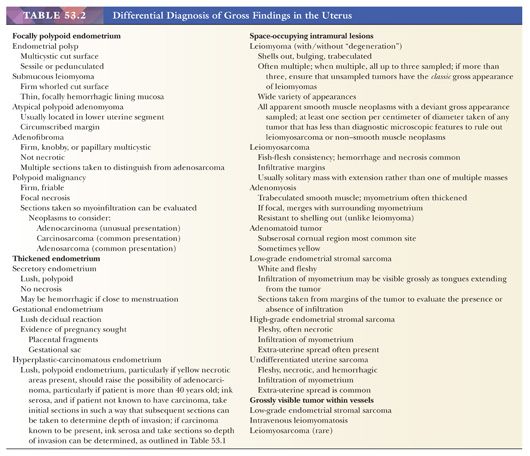
MYOMECTOMY SPECIMENS
Myomectomies are performed in women who wish to retain their uterus, usually to relieve symptoms produced by the neoplasm. Although there is no evidence to support this, myomectomies have also been performed to enhance women’s fertility. Myomectomy can be conducted in various ways: in the course of laparotomy, laparoscopically, or hysteroscopically (6,7). The gross appearance of these specimens is particularly important with regard to the question of possible leiomyosarcoma. Almost all leiomyosarcomas deviate from the characteristic gross features of leiomyomas by being soft, hemorrhagic, and/or necrotic, and almost all infiltrate such that they are difficult to remove. Conversely, however, most grossly peculiar myomectomy specimens ultimately prove to be benign when their histologic appearances have been evaluated. The preoperative administration of gonadotropin-releasing hormone has been shown to reduce the size of myomas, and these agents may modify the appearance of benign smooth muscle tumors (6–9); however, the gross and histologic correlates of this clinical shrinkage have been difficult to establish with certainty (10). Leiomyosarcoma in the reproductive years is very unusual. The tissue samples taken should be well fixed and, when possible, should include the interface between normal myometrium and the gross lesion. Histologic sections prepared from these samples must be suitable for obtaining an accurate mitotic index; that is, they must be thinly cut and well stained (Table 53.3).

APPROACH TO UTERINE CORPUS DIAGNOSIS: PATTERN DIAGNOSIS
Even in the absence of any previous information, the range of differential diagnostic possibilities is narrowed initially by the overall low-power microscopic appearance of the endometrium and myometrium. Most specimens fall into one of six scanning power groups. In our view, it is very helpful in formulating differential diagnoses to examine the endometrium and myometrium at low-power magnification and to attempt to place it into one of the following six categories. The location in the text of the differential diagnosis of each of these patterns is presented in Table 53.4.

PATTERN 1: PROLIFERATIONS COMPOSED OF GLANDS AND SUPPORTIVE NONNEOPLASTIC ENDOMETRIAL STROMA
This is the most common pattern encountered in daily practice. Diagnosis is facilitated by a systematic examination of the glands-to-stroma ratio, glandular features, stromal features, the appearance of the vessels, and pattern uniformity.
Glands-to-Stroma Ratio
The low-power evaluation of the ratio of glands to stroma leads to a further sorting into three categories. The first group shows a roughly 1:1 ratio of glands to stroma, a pattern exhibited by most normally cycling endometria and also encountered in the majority of endometria associated with dysfunctional uterine bleeding and infertility. Second, there are endometria marked by a shift in the glands-to-stroma ratio in favor of glands. This group includes some fully developed late secretory endometria, menstrual endometria, endometrial hyperplasias, and carcinomas. The final group comprises endometria featuring a predominance of stroma, a group that includes normal decidua, some examples of atrophy, and all the pattern 3 monophasic stromal proliferations listed later.
Glandular Features
The next step in narrowing the differential diagnosis of proliferations composed of glands and supportive stroma involves an examination of the cytologic features and the architecture of the endometrial glands.
Cytologic Features. Atrophic and weakly proliferative glands are lined by low cuboidal to columnar cells with sparse or absent mitotic figures. Proliferating glands are lined by columnar epithelium exhibiting nuclear pseudostratification, mitotic figures, high nuclear-to-cytoplasmic ratios, and elongated, ovoid nuclei with dense basophilic chromatin. Cytologic atypia in such glands is manifest by an increase in nuclear (and cell) size, increasing prominence of nucleoli, rounding of nuclei, nuclear pleomorphism, and a tendency of the cells to become stratified. These nuclear features are most pronounced in carcinoma. Parenthetically, in many carcinomas, the epithelium tends to grow in a complex stratified, sheetlike pattern punctuated by sharply rounded spaces (cribriform pattern).
Secretory epithelia can be broadly divided into those manifesting early secretory, midsecretory, and late secretory changes. Early secretory epithelia feature large cytoplasmic vacuoles either in a subnuclear or supranuclear position; the nuclear features are those of late proliferative phase cells. Midsecretory glands are coiled, the epithelium is low columnar to cuboidal, and the cells possess an oval to round, vesicular, usually basilar nucleus with a small but discernible nucleolus. The cytoplasm is eosinophilic to clear. A dominant secretory vacuole is typically absent. Late secretory glands are fully coiled and dilated, and they frequently show luminal secretion and luminal border fraying. Marked cytoplasmic vacuolization associated with nuclear hyperchromasia and pleomorphism in the absence of mitotic figures is characteristic of the Arias-Stella reaction. The epithelial cells of disintegrating glands often contain karyorrhectic fragments (apoptosis).
Architecture. The normal endometrial gland is a nonbranched, coiled structure. Branching and budding of varying degrees of complexity are the hallmarks of endometrial hyperplasia and carcinoma. Occasionally, epithelial secretory changes are superimposed on the budding pattern of hyperplasia or even of carcinoma.
Stromal Features
Characteristically, endometrial stromal cells have either oblong or spindled nuclei with scant cytoplasm that is difficult to discern (proliferative phase); alternatively, they show varying degrees of deciduation (secretory phase and pregnancy). The term deciduation refers to the transformation of a proliferative phase stromal cell, with its dense nucleus and sparse, ill-defined cytoplasm to one with a large, ovoid to round vesicular nucleus and abundant well-defined eosinophilic to clear cytoplasm. Lesser degrees of this transformation are known as predecidua and are characteristic of the normal late secretory phase of the menstrual cycle. Fibrotic, spindled stroma is characteristic of endometrial polyps.
Appearance of the Vessels
The distinctive feature of the normal developing endometrium is a synchronous proliferation of blood vessels, glands, and stroma. The endometrial vasculature begins as a delicate arborizing network of vessels ramifying through the developing proliferative endometrial stroma. It is this early vascular pattern that is so characteristic of endometrial stromal neoplasms. As the vasculature continues its development during the normal secretory phase of the menstrual cycle, the vessels acquire thicker walls and become coiled; this appearance led to the designation spiral arteries. This coiling is particularly evident in the middle and superficial regions of the functionalis. Poorly developed, thin-walled vessels are typically seen in the endometria in anovulatory cycles and in those reflecting the effect of progestational agents. Larger, thick-walled vessels embedded in fibrotic stroma are features of endometrial polyps.
Endometrial Pattern Uniformity
The hallmark of the normal endometrium is a uniformity of development from one portion of the stratum functionalis to another. In curettage or biopsy specimens, this implies uniformity from one fragment of endometrium to another. There are two important exceptions: fragments that derive from the region of the lower uterine segment or isthmus and fragments of the stratum basalis comprising the deepest layer of the endometrium. The lower uterine segment is typically less responsive to hormonal stimulation than the fundal functionalis, and the appearance of this region does not reflect the full impact of prevailing steroid hormone levels.
Fragments from the lower uterine segment are very often present in endometrial samples and can be identified by their spindled stromal cells separated by collagen fibers and by the presence of hybrid endometrial–endocervical glands. The stratum basalis maintains an essentially constant appearance throughout the menstrual cycle and does not exhibit the striking glandular and stromal changes of the overlying cycling stratum functionalis. The basalis is composed of small-caliber, minimally tortuous, weakly proliferative glands embedded in a cellular stroma. This stroma frequently intermingles with wisps of superficial myometrium. As a result, evaluation of the functional state of the endometrium should be carried out only on fragments of functionalis, which are lined by surface epithelium.
PATTERN 2: BIPHASIC PROLIFERATIONS COMPOSED OF GLANDS AND ABUNDANT (POSSIBLY NEOPLASTIC) STROMA
Entities that figure in the differential diagnosis of this pattern include endometrial polyps, atypical polypoid adenomyomas, mixed müllerian proliferations, and some of the patterns with prominent stroma discussed earlier in “Pattern 1: Proliferations Composed of Glands and Supportive Nonneoplastic Endometrial Stroma.”
PATTERN 3: PREDOMINANTLY MONOPHASIC SPINDLE CELL PROLIFERATIONS
This pattern figures most prominently in the evaluation of hysterectomy or myomectomy specimens, although, from time to time, an endometrial sample will contain fragments of spindled stroma devoid or largely devoid of glands. The major differential diagnostic considerations raised by this low-power pattern are smooth muscle neoplasms, endometrial stromal neoplasms, spindled epithelial neoplasms, pure heterologous uterine sarcomas, and undifferentiated sarcoma.
Epithelial versus Mesenchymal Differentiation
Strategies useful in separating spindled epithelial neoplasms from spindled mesenchymal neoplasms include immunohistochemical staining and preparation of additional hematoxylin and eosin (H&E)–stained sections to search for areas of less equivocal epithelial differentiation. Normal endometrial epithelial cells express keratins, epithelial membrane antigen (EMA), and vimentin; this same pattern is found in most epithelial neoplasms including carcinoma. Smooth muscle cells can express keratins, usually AE1, and less often CAM 5.2. Smooth muscle cells usually show positive results for muscle actin and very often for desmin and caldesmon. Although neoplastic endometrial stromal cells may have staining patterns similar to those of smooth muscle (smooth muscle cells may express CD10, and endometrial stromal cells may express desmin and/or caldesmon), strong diffuse desmin staining in conjunction with caldesmon staining supports smooth muscle differentiation, and strong diffuse CD10 staining supports endometrial stromal differentiation. Distinguishing uterine epithelial proliferations from mesenchymal processes should not be based on immunohistochemical staining patterns alone, although antigen expression may provide support for interpretation reached on the basis of H&E sections (11,12). Nonneoplastic stromal cells do not express cytokeratin or EMA, and desmin staining, if positive, is usually focal; however, almost all express CD10.
Smooth Muscle versus Stromal versus Heterologous Differentiation
Under the assumption that a proliferation is mesenchymal, certain strategies may be useful in distinguishing the possibilities. These are set out in Table 53.5. This topic is further discussed in “Smooth Muscle Neoplasms.”
PATTERN 4: SHEETLIKE PROLIFERATIONS COMPOSED OF LARGE, ROUND, UNDIFFERENTIATED CELLS
The major consideration that falls under this heading is the uterine version of the problem of the undifferentiated malignant lesion. In whatever organ system this diagnostic problem arises, it is important to cast one’s net widely in terms of differential diagnosis; the undifferentiated uterine neoplasm is no exception. In addition to poorly differentiated uterine corpus primary tumors—grade 3 adenocarcinoma, carcinosarcoma, and high-grade undifferentiated uterine sarcomas—one must consider extension of a primary uterine cervix malignancy to the uterine corpus, metastatic carcinoma (particularly from an extragenital primary), metastatic melanoma, leukemia (including granulocytic sarcoma), and lymphoma (the usual problem being the large cell diffuse type). The most useful maneuvers in sorting out this differential diagnosis in our experience are submitting more tissue, if it is available, and performing an immunohistochemical panel that usually includes keratin, desmin, caldesmon, and CD10. If the results of these tests are not informative, S-100 protein; lymphoid markers, particularly CD30; and EMA may be helpful (see “Epithelial versus Mesenchymal Differentiation”). When tissue is limited, repeat biopsy may be necessary. When a proliferation’s histologic appearance raises the possibility of a metastatic neoplasm, clinical history and additional clinical studies are often indicated. We have encountered several cases of cytologically bland, lobular carcinoma of the breast infiltrating the endometrial stroma.
Usually, these maneuvers serve to narrow the possibilities to one or two processes, and examination of the hysterectomy specimen most often establishes the diagnosis. It is obviously important not to misidentify leukemia, lymphoma, melanoma, and metastatic carcinomas as primary uterine carcinomas or sarcomas, and it is rare for the procedures listed previously not to prevent such an error. However, there are occasions when even complete sampling of a malignant uterine primary neoplasm fails to reveal the direction of differentiation of the tumor. It is of some comfort that failure to distinguish primary high-grade uterine sarcoma from carcinosarcoma or poorly differentiated high-grade carcinoma in a uterine sampling is usually of little clinical consequence; treatment is based on the posthysterectomy diagnosis.
PATTERN 5: SAMPLES THAT FEATURE EXTENSIVE NECROSIS, INFLAMMATION, OR DISINTEGRATION
Extensive necrosis in endometrial samples should always raise the possibility of a malignancy, especially in a menopausal woman. A careful search should be made for isolated malignant cells in such a sample. Inflammatory cells are most often a normal finding in the postpartum endometrium, but sheets of polymorphonuclear leukocytes (pus) should suggest the possibility of a postpartum bacterial infection. Necrotic endometrial samples are obtained when cervical stenosis and pyometra are present; xanthomatous endometritis occurs in this context. Various infectious diseases may cause inflammation and necrosis when they affect the endometrium, and these are outlined at the end of this chapter in Table 53.17a.
Disintegrating endometria may be encountered in numerous situations, most commonly at the time of menstruation. The features of menstrual endometrium are familiar. Glands exhibiting secretory exhaustion are surrounded by a halo of disintegrating, swollen predecidual cells; often, both are suspended in fibrin and blood and typically are infiltrated by varying numbers of neutrophils. Karyorrhectic fragments are often present in epithelial cells (apoptosis). Nonsecretory endometria, including hyperplastic endometria, may also disintegrate, and this is often marked by fibrin thrombi in vessels and stroma. On occasion, the degenerative changes in the epithelial and stromal cells of these endometria cause the stroma to collapse, and the result is glandular approximation mimicking hyperplasia or carcinoma. For the same reason that a diagnosis of malignancy should be avoided in cytologic specimens composed of degenerated cells, such a diagnosis should be made only when cytologic features are well preserved in tissue sections. If the specimen exhibits degeneration and disintegration and if carcinoma is a possibility, a second tissue sampling is warranted.
PATTERN 6: SCANTY SAMPLES THAT RAISE THE QUESTION OF SAMPLING ADEQUACY
Scanty endometrial samples are commonly encountered but do not necessarily indicate an inadequate sample. Specimen adequacy depends on the clinical presentation and the question being posed. For example, a scant (and inadequate) specimen may arise as a result of an obstructing lesion that shields the endometrium from the sampling instrument. However, more often, the specimen contains little or no endometrium either because the endometrium was atrophic (and what is present is all there is) or because of prior endometrial shedding or removal; in either scenario, the specimen is adequate. When neoplasm is suspected, inquiries about the clinical and operative particulars—the presence of a thickened endometrium on imaging, the thoroughness of the procedure and its extent, and so on—should be made, and consideration should be given to resampling the endometrium if there is any discrepancy. Scant benign endometrial surface epithelium and stroma with minimal or no glands may be entirely consistent with atrophy in a postmenopausal woman, provided the sampling is thorough and there is no evidence of an enlarged uterus or abnormal endometrial lining; however, similar findings in a premenopausal or perimenopausal woman should be interpreted with caution.
NORMAL MENSTRUAL CYCLE ENDOMETRIUM
The particulars of the normal menstrual cycle are chiefly important for setting a morphologic baseline for discussion of endometrial abnormalities and for the interpretation of EMB performed for infertility (13,14). Today, biopsies are infrequently undertaken for the latter purpose in many institutions for reasons discussed later (see “Endometrial Dating”). In women being evaluated for infertility, there are three clinical questions of major interest.
CLINICAL QUESTIONS
Clinical Question 1
Is there an intrinsic abnormality of the endometrium that could explain the couple’s infertility (e.g., endometrial polyps, submucous leiomyomas, endometritis, hyperplasia, carcinoma)? Much of this information is provided by hysterosalpingogram, hysteroscopy, ultrasound, and laparoscopy that are increasingly routine procedures for the workup of the infertile patient (15–19).
Clinical Question 2
If the endometrium is normal, do its morphologic features provide evidence that the patient ovulated in the biopsy cycle? This amounts to deciding whether or not the endometrium is secretory. The answer to this question is also relevant for the patient undergoing EMB for abnormal bleeding in the reproductive years; the approach to the therapy of dysfunctional uterine bleeding depends on whether such bleeding is ovulatory (secretory pattern of some sort) or anovulatory (nonsecretory pattern of some sort).
Clinical Question 3
If the endometrium is secretory, is it appropriately developed for the patient’s chronologic dates (see “Endometrial Dating”)? The only importance of this observation is that, under certain circumstances, the establishment of endometrial maturation delay warrants the diagnosis of luteal phase defect, a relatively uncommon condition that is thought by some researchers to be responsible for infertility and is correctable by various therapies. As discussed later, luteal phase defect is defined in terms of a more than 3-day disparity between the woman’s date of ovulation and her endometrial morphologic date, when this disparity occurs more than sporadically (see “Other Secretory Patterns”). Thus, after determining that the endometrium is normal, evaluation of the EMB from the infertile patient requires assigning the endometrium a morphologic date based on an examination of an H&E slide (13,14,20). Because of the observer agreement issues discussed later, the value of the EMB in the evaluation of the infertile couple is subject to controversy (21,22).
The following discussion provides a brief review of normal endometrial histologic characteristics.
ENDOMETRIAL DATING
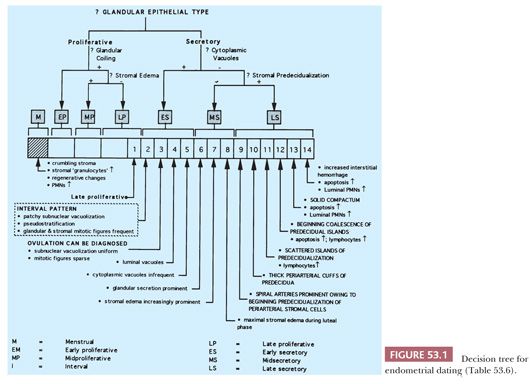
The first day of the menstrual cycle has conventionally been identified as the first day of menstrual flow. Menses usually last for fewer than 5 days and are followed by the endometrial proliferative phase, the length of which exhibits great variation (9 to 20 days); on average, it lasts 10 days. After ovulation, the coordinated and highly predictable series of stromal and glandular changes characteristic of the secretory (luteal) phase take place. The traditional view is that the length of this phase is constant (14 days), and this alleged constancy provides the basis for endometrial dating.
In evaluating the endometrium, it is important to distinguish carefully between the morphologic postovulatory date assigned to a morphologically normal endometrium and the chronologic postovulatory date. The morphologic date is a summary characterization of the histologic development of the endometrium based on an assessment of glandular and stromal features. Endometrial development is characterized either in terms of conventional light microscopic features or, in recent years (and in research settings), in terms of more sophisticated and reproducible morphometric semiquantitated features. The morphologic findings may be summarized in terms of postovulatory days (PODs), cycle days (CDs), or phases. For example, the morphologic pattern associated with a particular “standard” POD is assigned the number of that day; POD 12 refers to the pattern seen on the 12th POD of the standard cycle. Equivalently, this morphologic information can be conveyed using CD (e.g., CD 26) or phase (e.g., late secretory). The precise details of the morphologic patterns corresponding to standard cycle dates are presented in Table 53.6 and Figure 53.1. In practice, normal endometria may exhibit a range of patterns, which may span a 3-day interval; in these cases, the assigned date should reflect the most advanced pattern(s) and is expressed as an interval (e.g., POD 10–11).

NORMAL ENDOMETRIAL PATTERNS
Normal Proliferative Endometrium
Description. Refer to Figure 53.1 and Table 53.6 in conjunction with the text in this section. The stratum functionalis of the normally cycling proliferative endometrium is characterized by nonbranching, nonbudding, similarly shaped glands evenly distributed throughout a stroma composed of monomorphous, undifferentiated stromal cells with scant cytoplasm and indistinct cell margins supplied by a uniformly developed, arborizing vasculature with thin walls (Fig. 53.2). The glands-to-stroma ratio is typically 1:1. Early in proliferation, the glands are tubular and of narrow caliber; as proliferation continues, they become increasingly coiled, and their caliber increases. Normal proliferative endometria are further marked by pseudostratified, mitotically active, elongated epithelial cells with dense chromatin and mitotically active stromal cells. Most of the vessels are inconspicuous and resemble capillaries, particularly near the endometrial surface.
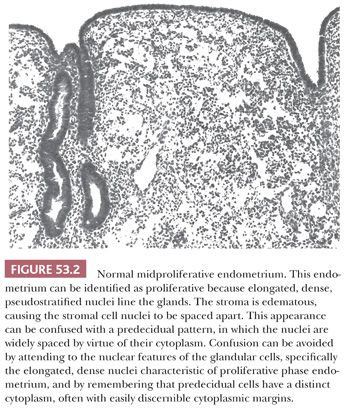
Clinicopathologic Correlation. A normal proliferative pattern may also be seen in association with anovulatory cycles and with exogenous estrogen therapy.
Interval Endometrium
Description. In essence, an interval endometrium is a late proliferative endometrium in which the glands are coiled and in which fewer than half of the epithelial cells feature spotty, nonuniform subnuclear vacuolation. Two points about the ultrastructural appearance of the endometrium during this period should be made. First, there is ultrastructural evidence of secretion throughout the menstrual cycle; the relative increase in secretory activity (visible on H&E preparations) after ovulation motivates the designation secretory phase. Second, the first unambiguous evidence of ovulation seen in the endometrium on ultrastructural examination is the appearance of the distinctive nucleolar channel system.
Clinicopathologic Correlation. The presence of this pattern is no guarantee that ovulation has occurred, even though the normal endometrium from POD 1 to POD 2 has this appearance.
Normal Early Secretory Endometrium: Postovulatory Days 2 to 5
Description. The endometrium during this time features coiled glands composed of cells resembling those found in the proliferative phase, but more than half of the glandular cells contain relatively large cytoplasmic vacuoles. These vacuoles serve as the marker of early secretory endometria. The glands are set within a nonpredeciduated stroma. The precise date assigned to such patterns depends on the location of the cytoplasmic vacuole (subnuclear or supranuclear) and the number of mitotic figures present (Table 53.6; Figs. 53.1 and 53.3).
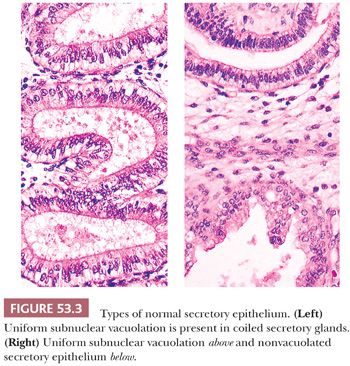
Clinicopathologic Correlation. This pattern may be accompanied by a clinical history of midcycle spotting and mittelschmerz.
Normal Midsecretory Endometrium: Postovulatory Days 6 to 8
Description. The midsecretory endometrium is characterized by fully coiled secretory glands lined by cells with round, often vesicular nuclei. The cytoplasm of such cells does not contain large cytoplasmic vacuoles, but luminal secretions may well be present. The stroma has not begun to undergo predeciduation. The fine tuning of the postovulatory date within 48 hours in this segment of the secretory phase depends on an evaluation of the extent of stromal edema and the prominence of the glandular luminal secretion, as summarized in Figure 53.1 and Table 53.6. The absence of extensive vacuolation and of predecidua is the most useful marker of midsecretory endometria (Fig. 53.1).
Clinicopathologic Correlation. Implantation occurs during this part of the cycle. This morphologically normal pattern may be clinicopathologically (functionally) abnormal, depending on the time of ovulation.
Normal Late Secretory Endometrium: Postovulatory Days 9 to 14
Description. At the beginning of this phase in the cycle, low-power examination reveals that the spiral arteries are prominent, not only in part as a result of the thickness of their walls but also largely because of the cuffs of predeciduated stromal cells around them. Predeciduation begins initially around spiral arteries (POD 10) and then extends to form islands in the superficial reaches of the endometrial stroma. It marks the beginning of the last third of the secretory phase (Fig. 53.4). At the end of the late secretory phase, these decidual islands become confluent and then, as menstruation becomes imminent, are dissected by interstitial hemorrhage. Associated with the progressive predeciduation of the endometrial stroma is the increase in the number of stromal granulocytes (Fig. 53.5). These cells have bean-shaped, dense nuclei; inconspicuous cytoplasm; and cytoplasmic granules visible with special stains and occasionally on H&E preparations. These enigmatic cells, once thought to derive from the endometrial stroma, have been shown by immunoperoxidase techniques to be endometrial granular lymphocytes (natural killer [NK] cells) (23).
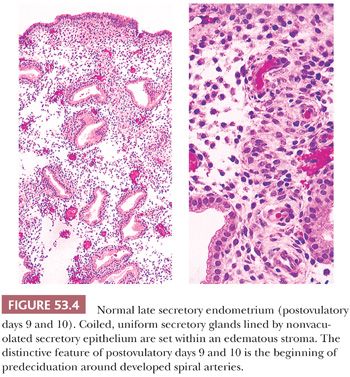
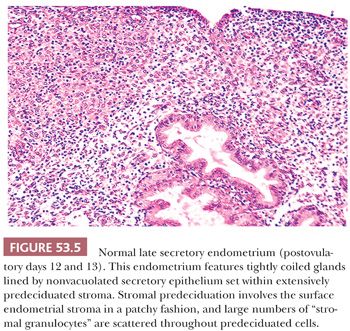
Menstrual Endometrium
Description. The menstrual pattern features disintegrating fragments of fully developed secretory endometrium. The glands are dilated and lined by flattened cells, often with frayed borders (secretory exhaustion), and the stroma is fully predeciduated. Karyorrhectic fragments are present in the subnuclear area of some glands (apoptosis), the cell margins are frayed, the epithelial nuclei are pyknotic, and fibrin thrombi are present in vessels and sometimes within the stroma. Fibrin thrombi are a very useful feature in the identification of endometrial disintegration. As menstruation proceeds, the glands break up into strips, the stroma crumbles, and the epithelial cells lose cohesion.
Clinicopathologic Correlation. Menstrual shedding should be distinguished from shedding of abnormal endometrial tissue, for example, premature shedding of proliferative endometria, shedding of hyperplastic endometria, and sloughing of disintegrating fragments of carcinoma.
Differential Diagnosis
Compact Late Proliferative Stroma versus Predeciduated Stroma. Predeciduation begins around spiral arteries and only later becomes confluent (Fig. 53.4). It is invariably accompanied by prominent spiral arteries with thicker walls than the thin-walled, capillary-like vessels of the late proliferative endometrium. Proliferative phase stromal cells have indistinct cell margins and scant cytoplasm, whereas decidual cells have abundant cytoplasm and more distinct cell margins. When edema is prominent, proliferative stromal cell nuclei may be spread apart and may appear to have abundant cytoplasm, and this can cause them to resemble decidual cells. Attention to stromal cell margins, the vascular pattern, and the nuclear features of the glandular cells will help one to avoid misclassifying such proliferative stromal changes as secretory.
Interval versus Early Secretory Pattern. The importance of this distinction is that an early secretory pattern (PODs 2 and 3) is required before ovulation can be confidently diagnosed. We require that at least 50% of the glandular cells show uniform subnuclear vacuolation before we assert that ovulation has occurred. The reason for this prerequisite is that scattered subnuclear vacuoles can be seen in many different nonsecretory conditions, including endometrial hyperplasia and well-differentiated “secretory” carcinoma. If the biopsy is performed only during the presumed last third of the secretory phase of the cycle, this 50% rule will need to only rarely be invoked; if the biopsy is, in fact, taken in the presumed late luteal phase, the endometrium’s maturation is substantially delayed, and it would be abnormal on that account.
Secretions in Nonsecretory Glands. The finding of secretory material in glands is entirely nonspecific and may be seen in various contexts other than the midsecretory phase endometrium. In particular, luminal glandular secretions may be found in disordered proliferative endometria, hyperplasia, and carcinoma.
Nonsecretory Epithelial Cell Vacuolation. The presence of a large secretory vacuole in the cytoplasm of endometrial epithelial cells is only one cause of epithelial cell clearing. In some stages of their evolution, ciliated cells (normally present in proliferative endometria) are cleared, and lymphocytes migrating through epithelium may produce cytoplasmic clearing. Cleared cells can be seen in some patterns of squamous or morular differentiation and also in mucinous metaplasia, mucinous carcinoma, clear cell carcinoma, and secretory carcinoma.
Nonmenstrual Hemorrhage in Endometrial Biopsies. Hemorrhage in an endometrial sampling may be produced by numerous processes other than menstruation. Hemorrhage associated with interstitial and intravascular fibrin is a feature of any disintegrating endometrium, including those produced by estrogen-withdrawal bleeding in an anovulatory patient. It is, of course, common to see blood without fibrin thrombi in any endometrial sampling, simply as a result of the operative procedure.
Isolated, Out-of-Step, Cystically Dilated Glands. Isolated, cystically dilated, or budded glands are commonly encountered in otherwise normal endometria, and as isolated findings, they do not provide an adequate explanation for a patient’s infertility.
Midsecretory versus Late Secretory Endometria. In late secretory endometria, the predecidua first forms cuffs around vessels and then coalesces to form islands in the superficial endometrium. Later, these islands become confluent. Predecidua is absent in midsecretory endometria.
OTHER SECRETORY PATTERNS
Deviations from the patterns expected in endometria during the normal secretory phase may sometimes be encountered, particularly in infertile women (14). The pathogenesis and significance of these changes are the subject of debate. Some researchers believe these endometria are the result of inadequate corpus luteum function, whereas others implicate a defect in the endometrium itself. Indeed, some of these patterns, particularly underdeveloped (out-of-phase) endometria, may be normal variations of no particular significance in explaining a couple’s infertility. Patients with unusual secretory patterns may experience oligomenorrhea or hypomenorrhea as well as infertility.
LUTEAL PHASE DEFECT
There is considerable controversy concerning the accuracy and reproducibility of endometrial dating, the prevalence of luteal phase defect, and its clinical relevance as a cause of infertility (21,22,24–26). Although some physicians may continue to use EMB in the evaluation of a select group of infertile women, this procedure has been greatly de-emphasized in most referral fertility centers, especially in view of the finding that virtually all infertile patients are treated with a progestational agent regardless of the underlying cause of infertility.
MIXED (ABNORMAL) SECRETORY PATTERNS
Description
This term is used when there is disparity of more than 3 days in the development of the secretory endometrium from region to region of the stratum functionalis or other abnormal secretory pattern. The specific abnormality should be described.
Clinicopathologic Correlation
The significance of abnormal secretory patterns is unknown and should be determined by the clinician in combination with other clinical findings.
SECRETORY CHANGE SUPERIMPOSED ON ABNORMAL NONSECRETORY PATTERNS
Description
Secretory change may be superimposed on a disordered proliferative endometrium, endometrial hyperplasia, or carcinoma. This condition can usually be recognized because the glands in these processes are frequently branched, whereas the glands of the normal secretory endometrium are coiled. The diagnostic importance of this phenomenon is that the features used to distinguish atypical hyperplasia from grade 1 (well-differentiated) carcinoma may be lost or substantially altered in these circumstances. Extensive secretory change in grade 1 (well-differentiated) endometrial adenocarcinoma is referred to as secretory carcinoma. We require that the architectural and cytologic features of carcinoma be present before this diagnosis is made (see “Usual Endometrial Adenocarcinoma”).
Clinicopathologic Correlation
Secretory changes of this sort may be produced by spontaneous ovulation or prebiopsy administration of progestational agents.
DIFFERENTIAL DIAGNOSIS
Atypical Hyperplasia with Secretory Change versus Endometrioid Carcinoma with Secretory Change
Although endometrioid carcinomas with secretory change are very low grade, the architectural and cytologic features of well-differentiated carcinoma must be present before that diagnosis is made. These characteristics include a rather extensive cribriform pattern, some degree of nucleomegaly, nuclear rounding, chromatin abnormalities, and mitotic figures. If most of these features are not present but the architecture is complex, we designate the process atypical hyperplasia with a superimposed secretory effect and specify the degree of cytologic atypia (Fig. 53.6; see also Fig. 53.64).
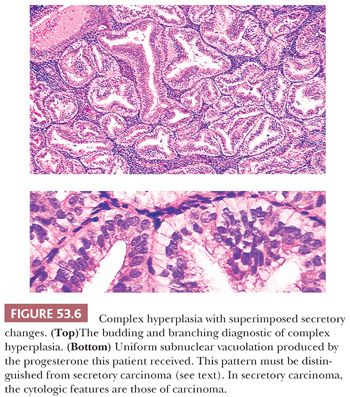
GESTATIONAL ENDOMETRIUM
After implantation, the secretory changes in the endometrium become more pronounced, and the fully developed gestational endometrium can usually be recognized on the basis of the features discussed here. Such changes in the endometrium, however, also can be seen in patients harboring an ectopic pregnancy as well as in patients receiving progestogen therapy (14).
EARLY GESTATIONAL ENDOMETRIUM
Description
Both Hertig (27) and Mazur et al. (28) described changes that they considered to be indicative, but not diagnostic, of early gestation; these include the coincidence of prominent glandular luminal secretion, prominent predeciduation, and prominent stromal edema. In the cycling endometrium, these changes assume their maxima in a sequential fashion. In early gestational endometria, their maximal development is simultaneous. In biopsy material, these secretory changes may not be particularly conspicuous (28).
Clinicopathologic Correlation
When these changes are encountered in an EMB, they are highly suggestive of early gestation, but because there is considerable overlap with late secretory changes in the cycling endometrium, they are not diagnostic. Obviously, extrauterine and intrauterine pregnancy can cause such endometrial changes.
FULLY DEVELOPED GESTATIONAL ENDOMETRIUM
Description
The fully developed gestational endometrium is characterized by sheets of decidua surrounding glands lined by relatively low cuboidal or flattened cells. Some of the glands may be tubular or gaping rather than coiled. Glandular cells with inclusion-like cleared chromatin can be seen in the presence of trophoblasts. Although they resemble nuclei infected with herpesvirus, eosinophilic inclusions and nuclear molding are absent, and there is no evidence that virus is present in such cells. This change may be related to intranuclear accumulation of biotin (29,30). Associated findings in curettage specimens containing fully developed gestational endometria depend, of course, on the age of the gestation, and they range from primary villi and anchoring cytotrophoblastic tissue to fetal parts and placental fragments in more advanced gestations. Intermediate trophoblasts infiltrate the endometrium and the underlying myometrium in normal gestation, and these often large, bizarre-appearing cells must not be misconstrued as evidence of a gestational trophoblastic neoplasm or sarcoma.
Clinicopathologic Correlation
The unequivocal diagnosis of intrauterine pregnancy requires, in our opinion, the presence of chorionic villi, fetal parts, or unambiguous trophoblastic cells within the uterus. Immunohistochemical techniques may help to identify trophoblastic cells, but this is rarely necessary (31,32).
ARIAS-STELLA REACTION
Description
This change is marked (Fig. 53.7) by hypersecretory glands lined by large cells with abundant clear to eosinophilic cytoplasm and irregularly shaped, hyperchromatic, smudged, enlarged nuclei exhibiting striking pleomorphism (33–35). Mitotic figures are very rare. The stroma is often deciduated. The Arias-Stella reaction may be focal, and the remainder of the endometrium may or may not exhibit a secretory reaction.
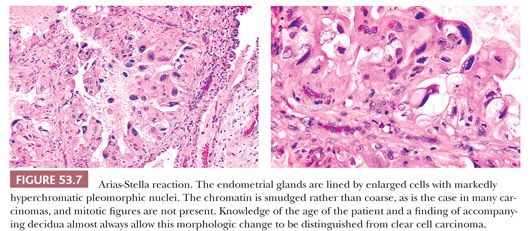
Clinicopathologic Correlation
The Arias-Stella phenomenon may be found in the endometrium in various contexts, including normal pregnancy, gestational trophoblastic disease, ectopic gestation, and in association with the administration of exogenous hormones (36,37). This glandular reaction may also develop in extraendometrial sites, such as the cervix or fallopian tubes, and in foci of endometriosis (38–40).
GESTATIONAL PATTERN WITHOUT PLACENTAL TISSUE OR FETAL PARTS
Description
Endometrial samples composed entirely of decidua and secretory glands with or without an Arias-Stella reaction but unassociated with fetal or placental tissues are not diagnostic of intrauterine pregnancy. On occasion, such endometria may be shed intact (decidual cast). Typically, the secretory development is florid and is characterized by dilated glands and inspissated secretions.
Clinicopathologic Correlation
These changes are not infrequently found in the endometria of patients who have spontaneously aborted an intrauterine gestation. They may also be seen in endometria associated with ectopic gestations (41) or with a corpus luteum cyst or persistent corpus luteum or in endometria that have responded to the administration of progestational agents (37). Endometrial vascular changes similar to those of atherosclerosis are commonly present in the decidual vessels of intrauterine gestations but not in those associated with ectopic pregnancy (42). It is imperative to alert the clinician to the possibility of an ectopic gestation when the patient is suspected of being pregnant and the tissue recovered from the uterus does not show unequivocal features of intrauterine gestation.
IMPLANTATION SITE REACTION, PLACENTAL SITE NODULES, AND SUBINVOLUTION OF THE PLACENTAL SITE
The endometrium and myometrium beneath an implantation site are invaded by trophoblasts shortly after implantation occurs. This phenomenon is termed (exaggerated) implantation site reaction. Trophoblasts have enlarged, often atypical nuclei, which may be arranged in a syncytium; they contain mitotic figures, including abnormal forms; and they may invade blood vessels. Intermediate trophoblasts, cytotrophoblasts, and syncytial trophoblasts are all present in varying numbers. The resemblance to a malignant neoplasm can be striking, particularly if the infiltration is composed predominantly of intermediate trophoblasts. The latter often resemble smooth muscle cells, and their nuclear atypia often raises the question of leiomyosarcoma. When the implantation site reaction does not promptly regress after abortion or delivery, it is designated subinvolution (43). Nodular aggregates of intermediate trophoblasts embedded in a hyaline matrix have been designated placental site nodules or plaques (44–46). The constituent cells are amphophilic, vacuolated, and mitotically inactive. There typically is no histologic or clinical evidence of a recent pregnancy (44–46). On occasion, multiple nodules may be present. Intermediate trophoblast cells can be identified by their keratin, epithelial membrane, and human placental lactogen immunoreactivity. A cautionary note: Smooth muscle cells may cross-react with keratin markers (47).
Distinguishing choriocarcinoma from an implantation site is based on the bilaminar pattern of large numbers of cytotrophoblasts and syncytial trophoblasts in choriocarcinoma and the extensive hemorrhagic necrosis that is a feature of choriocarcinoma but not usually of an implantation site. Moreover, chorionic villi may be associated with an implantation site but are almost always absent in choriocarcinoma. Placental site trophoblastic tumor is composed of intermediate trophoblasts and shares many features with an implantation site. It is distinguished by the presence of confluent masses of intermediate trophoblasts and the absence of villi.
Syncytial trophoblasts and cytotrophoblasts predominantly produce human chorionic gonadotropin (hCG), whereas intermediate trophoblasts express large amounts of placental lactogen. hCG is absent in the latter cells or expressed only in small quantities. In addition to trophoblasts, subinvolution of the placental site is marked by patchy chronic inflammation and distended spiral arteries whose walls are composed, in part, of hyalinized decidua. These vessels (rather than retained placental fragments) are usually the culprits in postpartum hemorrhage. The chronic inflammation routinely encountered in this situation does not indicate clinically significant infection. Gestational trophoblastic disease is discussed in detail in Chapter 49.
DIFFERENTIAL DIAGNOSIS
Arias-Stella Reaction versus Clear Cell Carcinoma
Patients with the Arias-Stella reaction are in the childbearing years; they will have experienced a recent pregnancy or be pregnant, will have an hCG-producing tumor, or will have undergone hormonal therapy. The patient may be unaware of a pregnancy, however, especially if it is ectopic or has aborted. Tumors producing hCG and resulting in the Arias-Stella reaction may also be silent. Patients with clear cell carcinoma of the endometrium are almost always postmenopausal, and their tumors are mitotically active. The cells in the Arias-Stella reaction are either not mitotically active or feature only rare mitotic figures, and the chromatin is smudged rather than coarsely granular. Immunohistochemical staining with Ki-67 may help distinguish endometrial Arias-Stella reaction from endometrial clear cell carcinoma in especially problematic cases (48).
A stromal decidual reaction is often associated with the glands demonstrating the Arias-Stella reaction but not with the tumor glands of clear cell carcinoma, unless the patient has received progestogen therapy. A distinctive localized endometrial proliferation associated with pregnancy has been described. It consists of a focal, microscopic epithelial proliferation featuring smoothly contoured cribriform endometrial glands lined by stratified, cytologically bland cells. The appearance is quite distinct from that of the Arias-Stella reaction (Fig. 53.7; see also Fig. 53.65). Despite its alarming appearance, this proliferation is unassociated with any clinical consequences (49).
Infiltrating Normal Trophoblastic Cells versus Mimics
Normal trophoblasts invading beneath an implantation site can mimic malignant neoplasms, particularly leiomyosarcoma and choriocarcinoma. The clinical history and a high index of suspicion will usually allow accurate interpretation; occasionally, immunohistochemical stains for hCG, placental lactogen, and muscle filaments will be indicated (14). An implantation site must be distinguished from choriocarcinoma (characterized by a lamellar pattern of syncytial trophoblasts and cytotrophoblasts as well as by extensive hemorrhage and necrosis), invasive hydatidiform mole (presence of villi), and placental site trophoblastic tumor (a mass composed mainly of intermediate trophoblasts). These neoplasms are further discussed in Chapter 49.
INFLAMMATION, NECROSIS, AND INFECTIONS
ENDOMETRITIS
The endometrium is normally populated by various inflammatory cells, including lymphocytes (occasionally organized into follicles and germinal centers), macrophages, and mast cells (see Table 53.17) (13). Neutrophils and eosinophils are barely detectable in proliferative endometria but increase dramatically immediately before menstruation. Neutrophils may be seen in the vicinity of physiologic necrosis of the decidua during pregnancy. Granular lymphocytes infiltrate the stroma of late secretory, menstrual, and gestational endometria. These observations imply that specific morphologic criteria must be present before diagnosing clinically significant endometrial inflammation. These criteria are discussed in the following sections.
Acute Endometritis
This diagnosis requires the presence of confluent aggregates of polymorphonuclear cells (microabscesses), as well as infiltration and destruction of glandular epithelium. The diffuse infiltration of the endometrium by granular lymphocytes and neutrophils during menstruation should not be construed as evidence of acute endometritis. Chlamydial endometritis may be an acute or mixed inflammatory process (50,51).
Nonspecific Chronic Endometritis
Of the usual inflammatory cells, plasma cells are probably the only cell type normally absent from the endometrium. For this reason, most researchers in this field have regarded the presence of plasma cells as the sine qua non for establishing the diagnosis of chronic endometritis. Scattered lymphocytes and lymphoid follicles in the basalis are a normal finding. Low numbers of T lymphocytes and B lymphocytes can also be detected in nonbasalis regions of the endometrium of physiologically normal women. Undoubtedly, large numbers of lymphocytes, lymphoid follicles, and germinal centers should be considered abnormal, but when they are present in significant numbers, there is almost always an associated plasma cell infiltrate, which should be documented before a diagnosis of chronic endometritis is made.
Description. A diagnosis of chronic endometritis requires the presence of more than rare plasma cells (52). In convincing examples, lymphocytes, lymphoid follicles, neutrophils, and histiocytes are also usually evident; the inflammatory infiltrate is often mixed. In addition, the stroma is spindled or fibroblastic, and there is often glandular destruction with stromal breakdown. Xanthomatous endometritis is a form of chronic endometritis characterized by sheets of xanthoma cells.
Clinicopathologic Correlation. Chronic endometritis (as defined earlier) is usually encountered in the context of pelvic inflammatory disease, in association with the use of an intrauterine device or in connection with retained products of conception. Dense stromal lymphocytic inflammatory cell infiltrates in association with neutrophils in gland lumens and surface epithelium may be a predictor of upper genital tract infection. Kiviat et al. (52) found that the combination of acute inflammation (>5 polymorphonuclear leukocytes/×400 field in endometrial surface epithelium) and stromal plasma cells (>1 plasma cell/×120 field in endometrial stroma) was a highly sensitive and specific rule for predicting the presence of culture-positive and laparoscopically visible salpingitis. Mild, nonspecific chronic endometritis has been associated with symptomatic bacterial vaginosis, a condition connected to high concentrations of potentially pathogenic aerobic and anaerobic organisms that replace the normal flora of the lower genital tract (53).
Chronic endometritis is also present in the endometrium immediately postpartum, but in this circumstance, it is considered a normal finding. Most patients with chronic endometritis have menstrual abnormalities, and one-half of them experience pelvic pain. Although florid endometritis may be a cause of infertility, the relevance of the occasional plasma cell to a couple’s infertility is much less clear. Xanthomatous endometritis is seen most often in elderly women and is almost exclusively associated with cervical stenosis and pyometra (54).
Granulomatous Endometritis and Specific Infections
Most uterine granulomas are focal and are related to prior instrumentation and, even when diffuse, are only infrequently related to systemic disease (55). Uterine ablation using the neodymium: yttrium-aluminum-garnet laser often produces a striking foreign body giant cell response (56). Tuberculous endometritis, which is characterized by a granulomatous inflammatory response, is rare in the United States, but it is a relatively common cause of infertility in other countries (57). Other causes of granulomatous endometritis as well as specific infections are listed in Table 53.17.
DIFFERENTIAL DIAGNOSIS
Endometritis versus Menstrual Endometrium
Normal menstrual and gestational endometria often exhibit focal acute neutrophilic infiltrates in association with focal necrosis. Physiologic inflammation and necrosis are distinguished from endometritis on the basis of the clinical setting, appearance of the background endometrium, and often, the focality of the inflammation.
Endometritis versus Glandular and Stromal Breakdown
Endometrial and glandular stromal breakdown typically occurs in nonsecretory endometria and can be focal or diffusely involve the entire endometrium. When diffuse, it has a nonuniform pattern, unlike menstrual endometrium. It is commonly encountered in women undergoing evaluation for dysfunctional uterine bleeding and can be seen in a wide variety of settings but does not feature a significant plasma cell infiltrate, unless associated with endometritis. Classic features of stromal breakdown are stromal collapse with formation of “stromal blue balls,” fibrin thrombi, and stromal fibrosis with hyalinization. Stromal foam cells and hemosiderin may be present. Nuclear (apoptotic) debris forms aggregates along the basal cytoplasm of the glandular epithelium, which coalesces into eosinophilic syncytia at the surface, often surrounding the detached clusters of condensed stroma. Neutrophils are commonly present in the syncytial aggregates, but large, coalescent collections of neutrophils (pus) are absent.
Actinomyces versus Pseudosulfur Granules
Actinomyces israelii may cause an intense endometritis in association with an intrauterine device (IUD). The organisms stain with tissue Gram stain or Gomori methenamine silver, whereas pseudoactinomycotic radiate granules, which may also be encountered in association with an IUD, do not. Pseudoactinomycotic (pseudosulfur) radiate granules represent an unusual response to foreign material and/or bacteria and may be seen in pelvic inflammatory disease but are not a cause of endometritis (58–60).
IATROGENIC PATTERNS
A full discussion of iatrogenic patterns is beyond the scope of this chapter. The reader is referred to Mazur and Kurman (14) and the contained references for a thorough discussion. What follows are brief notes on the main effects that are commonly encountered in practice.
ESTROGENS
In general, estrogens, whether endogenous or exogenous, promote the growth of a nonsecretory endometrium, which may range from normal proliferative endometrium to patterns diagnostic of well-differentiated carcinoma.
Tamoxifen, a selective estrogen receptor modulator has been linked to a wide range of glandular and combined glandular and stromal uterine proliferations, extending from hyperplasia to carcinoma and from adenofibroma to adenosarcoma and carcinosarcoma (61–65). The estimated relative risk for the development of carcinoma and carcinosarcoma ranges from 2- to 15-fold and increases with the dose and duration of use (66). Although endometrial carcinoma may develop during tamoxifen treatment, the development of carcinosarcoma generally occurs much later, following the completion of the 5-year treatment regimen (66).
Various other tamoxifen associations have been claimed. Endometrial polyps are reported to be increased in users compared with nonusers. The morphology in some tamoxifen-related polyps is distinctive; they tend to be large and to have myxoid edematous stroma and distinctive staghorn vessels. Metaplasias, particularly mucinous metaplasia, and carcinomas have been reported in these polyps (67,68).
PROGESTINS AND ESTROGEN–PROGESTIN COMBINATIONS
These agents produce various patterns, depending on the combination of estrogen and progestin (14,69). Perhaps the most common progestin effect is the co-occurrence of small and atrophic, noncoiled glands embedded in a spindled cell predecidualized or decidualized stroma. Another pattern, particularly seen after high-dose progestin therapy, is profound decidualization of the endometrial stroma; in this setting, the glands show marked secretory changes or secretory exhaustion. Progestins administered to patients with hyperplastic endometria or well-differentiated carcinomas sometimes show a striking subnuclear vacuolization producing a parody of the normal early secretory endometrium. Arias-Stella change may also be seen (37). Combined estrogen and progestin replacement therapy in menopausal women may simulate weakly proliferative or secretory pattern endometria (14).
SYNTHETIC PROGESTERONE RECEPTOR MODULATORS
Synthetic progesterone receptor modulators (PRMs) interact with the progesterone receptor to alter hormonal response. They have potential use as contraceptives and for treatment of endometriosis and leiomyomas. Several of these agents are being studied in trials, and one, mifepristone, has been approved in Europe and the United States as a short-term contraceptive. Preliminary studies have revealed a variety of endometrial patterns in women receiving PRMs, including some that do not fit recognized endometrial diagnostic categories. Recognized alterations that have been identified in association with PRM therapy include atrophic or inactive epithelium, variable glandular secretory activity, and dyssynchrony between glands and stroma. Unique changes include thick-walled corded vessels close to or beneath the surface epithelium; shift in the glands-to-stroma ratio in favor of the stroma so that there is stromal prominence; and cystically dilated glands, usually lined by atrophic or inactive epithelium (70). Metaplasia, particularly ciliary metaplasia, is common. Further details on the morphologic alterations and long-term effects of PRMs await the conclusion of the ongoing trials.
INTRAUTERINE DEVICES
In their immediate surroundings, these devices produce localized acute and chronic inflammation. Progestin-releasing devices also induce a localized progestin effect in the neighboring endometrium.
UTERINE ABLATION
Electrosurgical rollerball ablation characteristically produces striking changes including extensive necrosis and inflammation shortly after treatment and, subsequently, scarring and residual inflammation that often features a giant cell granulomatous reaction (56). Uterine artery embolization for leiomyomas produces characteristic changes (71).
NONSECRETORY ENDOMETRIAL PATTERNS OTHER THAN NORMAL PROLIFERATIVE INCLUDING ENDOMETRIOID ADENOCARCINOMA
In addition to the normal proliferative endometrium of the menstrual cycle (discussed earlier), a spectrum of other nonsecretory patterns in which stroma plays a supportive role is often encountered in day-to-day practice (Fig. 53.8). We use the terms atrophic, weakly proliferative, disordered proliferative, hyperplastic, and carcinomatous to label the various morphologic patterns along this spectrum. With the exception of atypical hyperplasia and carcinoma, these patterns are normal in certain clinical conditions. For example, an atrophic endometrium is normal for prepubertal girls and for elderly postmenopausal women; however, it is distinctly abnormal during the reproductive years, unless the patient has been treated with hormones or suffers from ovarian failure. As a result, the clinical significance of many of these histopathologic patterns emerges only after clinicopathologic correlation. Patients with nonsecretory-type endometria may be asymptomatic; when they have symptoms, they most often have experienced abnormal uterine bleeding.

If abnormal uterine bleeding occurs during the reproductive years and is not associated with a detectable uterine abnormality other than these nonsecretory patterns—again excluding atypical hyperplasia and carcinoma, it is termed dysfunctional uterine bleeding (72). Uterine bleeding is designated abnormal if it is excessive or scant or if it occurs at the wrong time. The clinical diagnosis of dysfunctional uterine bleeding should be made only after excluding other causes of uterine bleeding, including focal abnormalities (e.g., submucous leiomyomas and polyps), atypical hyperplasia, carcinoma and other malignant neoplasms, gestational side effects, endometritis, and systemic coagulation abnormalities. The treatment of patients with abnormal bleeding is beyond the scope of this chapter and is discussed in several reviews (72–74).
Several generalizations can be derived from these sources:
1. Tissue sampling is mandatory in any patient who, on clinical grounds, is at significant risk of harboring a malignant or premalignant proliferation of the endometrium. The clinical criteria defining significant risk vary from authority to authority.
2. Tissue sampling is rarely indicated in the adolescent because abnormal bleeding is almost invariably the result of anovulatory cycles, and the risk of malignancy in this age group is extremely low (75,76).
3. The decision to obtain a tissue sample in patients taking hormone replacement therapy who bleed during their artificial cycle is complicated, and there are circumstances when it may not be necessary.
4. Monitoring of patients taking tamoxifen is also a complicated topic beyond the scope of this chapter (see “Iatrogenic Patterns”), but it involves endometrial sampling in patients judged to be at high risk. Most patients who develop bleeding during treatment with tamoxifen undergo tissue sampling.
5. Assessing response of hyperplastic endometria (or low-grade carcinoma) should be done in patients treated with progestin therapy. Adequacy of response to therapy is determined not only on the histologic evaluation for persistent or progressive hyperplasia and/or atypia but also on the adequacy of dosage (and duration) of progestin treatment.
The endometrial patterns discussed later, with the exception of endometrial carcinoma and probably some forms of atypical hyperplasia, are the morphologic reflections of alterations in prevailing serum steroid levels, whether derived from endogenous or exogenous sources. Atrophic and weakly proliferative endometria represent the appropriate response of a physiologically competent endometrium to depressed estrogen levels. Disordered proliferative endometria and hyperplastic endometria represent the physiologic normal endometrial response to elevated estrogen levels or prolonged estrogen stimulation. The reversibility of some grade 1 carcinomas using progestational agents or ovulation induction suggests that at least a subset of these proliferations may represent extreme responses of a physiologically normal endometrium to prolonged estrogen stimulation. In short, the endometrium in these circumstances can be thought of as functioning as a bioassay, reflecting in its changing morphologic features a response to shifting serum estrogen levels.
ATROPHIC AND WEAKLY PROLIFERATIVE (INACTIVE) ENDOMETRIA
Description
Both of these terms refer to the appearance (and inferred activity) of the epithelium lining the glands of the endometria. The epithelium tends to be mitotically inactive and bland in terms of cytologic appearance. The glands are embedded in a similarly “inactive” spindled stroma, which exhibits varying degrees of collagenation and practically no mitotic activity (Fig. 53.9). The ratio of glands to stroma is usually near unity, although pattern uniformity is variable. The glandular architecture may be cystic or budded, but the cells are not proliferative; typically, however, the glands are tubular. Occasionally, the epithelium may be metaplastic (Fig. 53.10). Weakly proliferative endometria differ from those that are atrophic (Figs. 53.11 to 53.14) by virtue of cells with pseudostratified, elongated, densely basophilic nuclei, rather than the cuboidal or flattened and mitotically inactive nuclei characteristic of the cells that populate an atrophic endometrium. Cystic atrophy is the term applied to endometria composed predominantly of cystically dilated glands lined by cuboidal to flattened (and consequently atrophic) epithelial cells. In many cases of postmenopausal bleeding, the recovered endometrial tissue is scanty, consisting of detached, fragmented strips of atrophic endometrial epithelium with minimal supporting stroma; despite the clinical history, evidence of breakdown or bleeding may be absent.
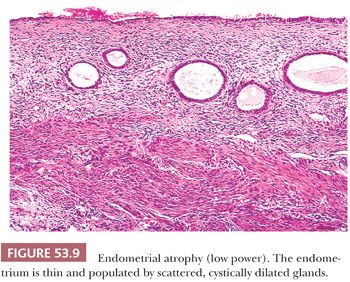
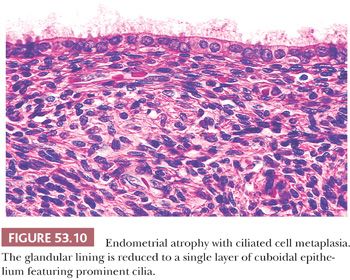
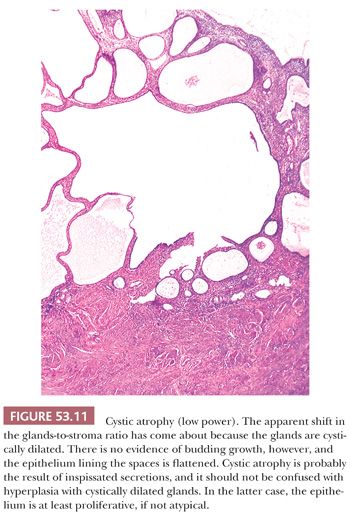
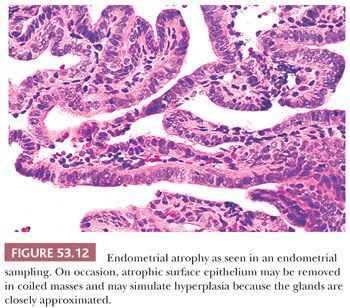
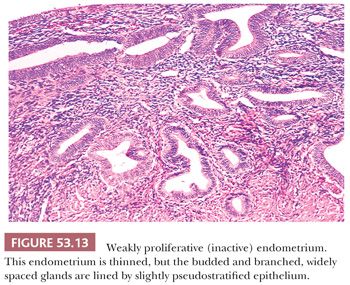
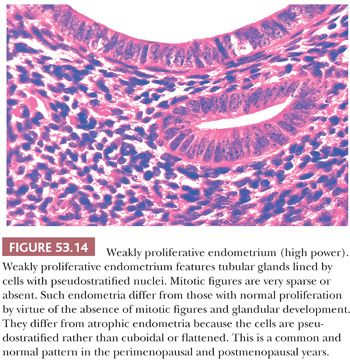
Clinicopathologic Correlation
Atrophy and weakly proliferative patterns are normal in menopausal and perimenopausal women, as well as in prepubertal girls. Atrophic endometria are most commonly recovered in the EMB sample from patients with postmenopausal bleeding and, provided that the uterine lining has been well sampled, should not be regarded as “insufficient for diagnosis.” However, such patterns are distinctly abnormal during the reproductive years, unless there is a history of hormonal medication or premature ovarian failure. When seemingly atrophic endometrium is recovered, it is important to consider these alternatives: (a) scant endometrial samples that consist only of basalis (make sure there is surface endometrium to evaluate); (b) samples that consist of only the lower uterine segment (look for hybrid endocervical–endometrial glands characteristic of the lower uterine segment); (c) the endometrial covering of a submucous myoma (look for rounded aggregates of smooth muscle underneath the endometrium); (d) fragments of endometrial polyp (look for other fragments in the sample showing normal features and thick-walled vessels in the atrophic fragment); and (e) a progestin effect (look for predecidual changes associated with underdeveloped, noncoiled glands).
DISORDERED PROLIFERATIVE ENDOMETRIUM
Description
The disordered proliferative endometrium resembles normal proliferative tissue in consisting of glands lined by cytologically bland, pseudostratified, proliferative, mitotically active epithelium and in having a roughly normal (unitary) ratio of glands to stroma. It differs from the normal proliferative endometrium in the absence of uniform glandular development (Fig. 53.15). The uniform appearance of the normal proliferative endometrium results from synchronous and coordinated growth of the fundal functionalis under the influence of estradiol. In contrast, the absence of pattern uniformity—a principal defining feature of disordered proliferative endometrium—is a result of dyssynchronous growth of the functionalis (Fig. 53.16).
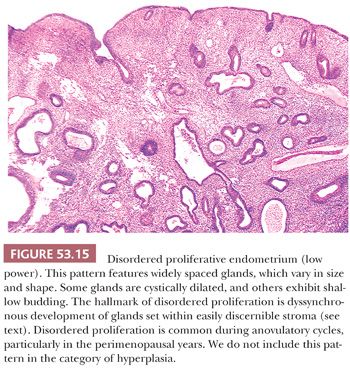
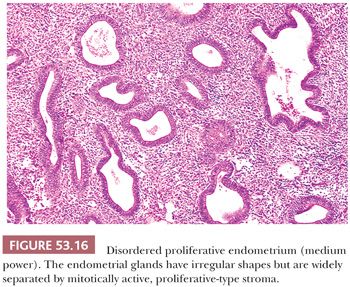
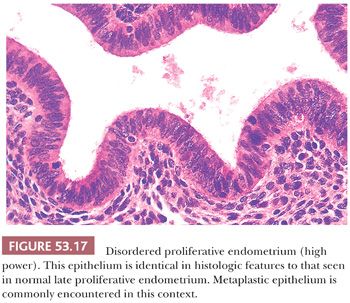
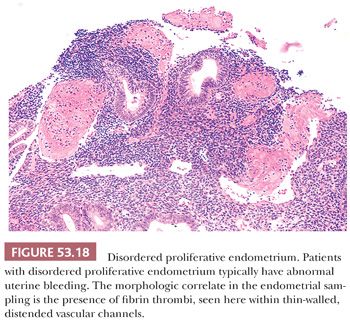
In some areas, the glands may be cystically dilated or may demonstrate varying degrees of shallow budding, whereas in other regions, the glands are tubular, of narrow caliber, and set within abundant stroma. Metaplastic epithelium, particularly ciliated epithelium, is commonly encountered (Fig. 53.17). No significant cytologic atypia is present. Evidence of endometrial breakdown and hemorrhage (the morphologic correlate of abnormal bleeding) may be present and is characterized by thrombosed, thin-walled vessels and interstitial fibrin and hemorrhage (Fig. 53.18). Disordered proliferation differs from hyperplasia without cytologic atypia by virtue of its relatively normal ratio of glands to stroma (1:1); that is, the significant shift in the glands-to-stroma ratio (3:1) in favor of glands that we require for a diagnosis of hyperplasia is absent (see following section “Hyperplasia”). Thus, disordered proliferation serves as a morphologic “bridge” between normal proliferation and hyperplasia.
Clinicopathologic Correlation
This is a common and normal pattern in the perimenarchal and postmenopausal years. The functional correlates are anovulatory cycles and exogenous estrogen therapy (77). This pattern is considered by some researchers to represent hyperplasia, but hyperplasia also connotes to many clinicians a proliferation that puts the patient at increased risk of endometrial carcinoma. Because there is no evidence that patients with disordered proliferative endometria are at any greater risk of endometrial carcinoma than those without this pattern, we do not think that the label of hyperplasia is appropriate.
HYPERPLASIA
Description
The term endometrial hyperplasia denotes a proliferating endometrium featuring glandular architectural abnormalities that result in glandular crowding and take the form of either cystic dilation of glands or glandular budding (78–80). Current taxonomy stratifies hyperplastic endometria on the basis of their cytologic features into atypical endometrial hyperplasia and nonatypical endometrial hyperplasia, with the latter term implying that significant cytologic atypia is absent (Table 53.7).

The characteristic architectural features of hyperplasia are glandular enlargement and budding. When it is excessive, budding leads to complex epithelial structures with numerous branching channels and papillary infoldings. Sectioning of these channels produces a pattern of closely approximated, narrow-caliber glands that, in extreme cases, can mimic the cribriform pattern characteristic of carcinoma. When growth is predominantly exophytic, “villoglandular” architecture reminiscent of villous adenoma of the gastrointestinal tract may be produced, with the formation of elongated, thin-stalked (foliate) structures.
As emphasized earlier, nonsecretory endometria constitute a morphologic spectrum, and the lines separating neighboring entities along this spectrum are ultimately conventional; that is, they attempt to approximately demarcate morphologic categories associated with different levels of risk of subsequent invasive endometrial carcinoma. The groupings are probably more or less right; the precise lines are underspecified by currently available data. Before invoking the term endometrial hyperplasia (rather than disordered proliferative endometrium), we require that the glandular overgrowth be sufficiently pronounced to shift the glands-to-stroma ratio to 2:1 to 3:1 (i.e., such that the stroma comprises less than one-third of the volume—or cross-sectional area—of the proliferation). We incorporate in the glandular fraction both glands (including their lumina) and villoglandular structures (Figs. 53.19 and 53.20).
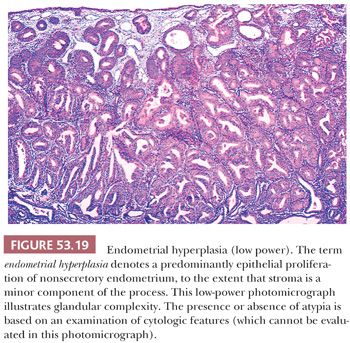
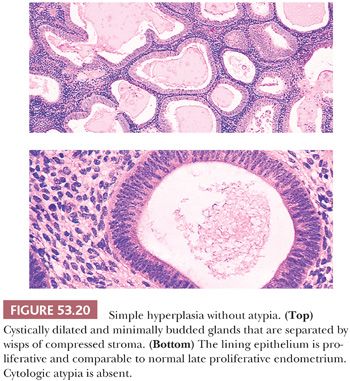
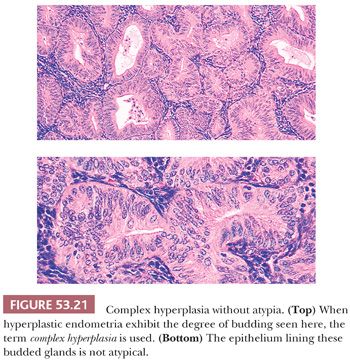
To qualify as hyperplastic, the epithelium must also be proliferative in its morphologic features. As a result, it should exhibit, at the very least, prominent pseudostratification; the cells may be focally stratified. The constituent cells in a hyperplastic endometrium vary from those that resemble the cells of the normal proliferative endometrium (Figs. 53.20 and 53.21) to those that show varying degrees of nuclear atypia, characterized by nuclear enlargement and irregularity, chromatin abnormalities, and prominent nucleoli. Again, these last nuclear features grouped as atypia, not architectural complexity, are the defining features of atypical hyperplasia. These epithelial requirements immediately eliminate from the category of hyperplasia architecturally complex endometria with atrophic or weakly proliferative epithelium.
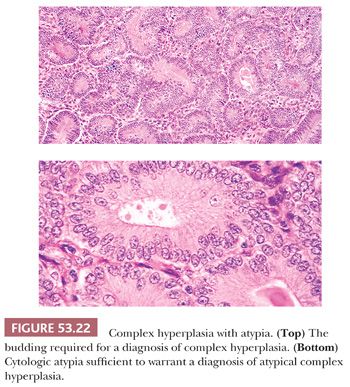
For the distinction between atypical and nonatypical hyperplasia, we use outcome-based criteria extrapolated from the study by Kurman et al. (78) in 1985. In that study, the risk of progression to carcinoma was 23% for atypical endometrial hyperplasia, whereas it was only 2% for nonatypical hyperplasia (78). The criteria for atypical hyperplasia include nuclear enlargement and very often nuclear rounding, some degree of pleomorphism, loss of nuclear polarity, and a shift in the nuclear-to-cytoplasmic ratio in favor of the nuclei. The relative size of the nuclei can be estimated by comparing them to the surrounding stromal cell nuclei or those of residual normal epithelial elements. Other features frequently present in cytologic atypia include prominent nucleoli, irregularity of nuclear size and shape, and dispersed (vesicular) and clumped chromatin (Fig. 53.22). There may be disarray and jumbling of the epithelium. Intraluminal tufting and focal stratification may be found, but when stratification is extensive, it is a criterion of malignancy (see “Grade 1 [Well-Differentiated] Endometrioid Adenocarcinoma”). Mitotic figures are almost always present in atypical hyperplasia and may be numerous, but abnormal division figures are sparse or absent.
The glandular architectural changes encountered in hyperplasia range from endometria which contain glands with shapes similar to those found in disordered proliferation to endometria which approach the architectural complexity and cytologic atypia of well-differentiated endometrioid adenocarcinoma.
For the purposes of clinical decision making, we think that any taxonomic scheme which is used to classify hyperplasia should reflect what is known about cancer risk and hyperplasia. To do this, hyperplastic endometria are separated into two categories: hyperplasia without cytologic atypia (little or no known risk) and hyperplasia with cytologic atypia (implies significantly increased risk) (Figs. 53.23 and 53.24; Table 53.7). Apart from our unwillingness to trade the label of disordered proliferation for simple hyperplasia, we endorse this classification for those endometria with a significant shift in the glands-to-stroma ratio and use it in our diagnostic practice.
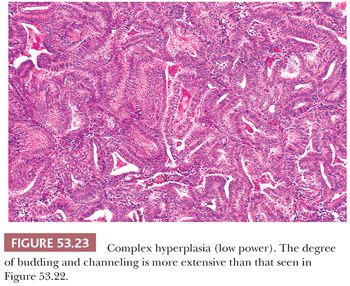
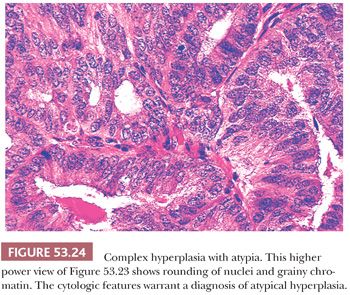
In most instances, hyperplastic endometria have an increased volume. On occasion, however, the architectural and cytologic criteria for hyperplasia are fulfilled, but the proliferation represents a small focus in an otherwise nonhyperplastic endometrium. The term focal hyperplasia seems reasonable for this finding. In other instances, the features of hyperplasia are present throughout a thin endometrium (or scant curetting). The prognostic and therapeutic implications of these variations of hyperplasia are probably those that would attach to a more voluminous proliferation with these features.
The term epithelial metaplasia denotes the presence of epithelial cells expressing differentiated features not usually encountered to any noticeable degree in endometrial cells. This phenomenon is most often encountered in hyperplastic endometria; the details of specific metaplastic cell types and their relation to hyperplasia are discussed in “Endometria with Alternative Differentiated Epithelium: Metaplasias and Special Variant Carcinomas.”
Clinicopathologic Correlation
Most patients with either nonatypical endometrial hyperplasia or atypical endometrial hyperplasia experience abnormal uterine bleeding. Endometrial hyperplasia usually occurs in the perimenopausal or postmenopausal years; in rare instances, adolescents show signs of atypical hyperplasia (81). Most hyperplastic endometria represent the normal endometrium’s response to sustained estrogen levels unrelieved by progesterone. The estrogen responsible for this process may be either endogenous (e.g., chronic anovulation) or exogenous (e.g., estrogen hormone replacement therapy). Adjuvant tamoxifen (a nonsteroidal antiestrogen with some estrogenic effects) has been reported to be associated with endometrial hyperplasia and to increase the relative risk of endometrial carcinoma (see “Iatrogenic Patterns”). Hyperplastic endometria without atypia and those with minimal atypia can usually be successfully treated by curettage and hormonal manipulation.
Although there is little doubt that atypical hyperplasia is a marker for an increased risk of adenocarcinoma, the magnitude of that risk is the subject of some controversy because our knowledge of the relative risk of endometrial carcinoma that attaches to various patterns found in hyperplastic endometria is meager, and acquisition of new knowledge is beset by formidable methodologic problems. The morphologic definition of the target event (invasive carcinoma, usually grade 1 adenocarcinoma) is ill defined and a matter of ongoing debate. Moreover, the willingness of the surgeon and patient to retain the organ involved by the putative precancerous lesion to wait out its natural history is extremely low in the usual age group in which these hyperplasias develop. In addition, the precursor lesions are anatomically unstable; they can be shed spontaneously, or they can be reversed by a change in the patient’s hormonal milieu, whether through alterations in physiology or alterations produced iatrogenically. It is as though one were attempting to develop a classification of intraepithelial breast lesions in a world where researchers had no universally shared morphologic definition of invasive breast cancer (e.g., the separation of ductal carcinoma in situ and invasive ductal cancer was typically challenging), where the intraductal lesion often changed its appearance, and where any atypical lesion was treated by bilateral mastectomies.
Despite these logistical problems, most reports provide evidence that approximately 20% to 30% of women with endometrial hyperplasia characterized by glands with marked architectural complexity and crowding, in addition to cytologic atypia, progress to a pattern, which the investigators deem morphologic adenocarcinoma (82). That is to say, not all cases progress to malignancy, either in the form of myoinvasion or clinical relapse. In the study published by Kurman et al. (78), once a patient had atypical hyperplasia, no further insight into risk was provided by grading the degree of atypia; that is, varying degrees of cytologic atypia were not reflected in a greater or lesser risk of adenocarcinoma once it was determined that the endometrium was lined by cytologically atypical cells.
Reaching beyond Conventional Light Microscopy for a Definition of Endometrial Carcinoma Precursors
Energetic attempts have been made to identify the endometrial carcinoma precursor using various techniques. Morphometric analyses by Baak and his colleagues have attempted, over the years, to provide computerized morphometric criteria for assigning levels of risk to hyperplastic endometria for the subsequent development of carcinoma. It would appear that both cytologic and architectural features are required to produce a discriminant function (D-score) that performs well (83–86). Other workers have recruited molecular techniques to the task of identifying precursor lesions, and these have been largely successful in demonstrating monoclonal proliferations. Mutter and colleagues (87–92) have translated these molecular observations into conventional light microscopy correlates, some of which correspond to the morphologic features traditionally used to identify complex atypical hyperplasia. However, endometrial intraepithelial neoplasia, as defined by Mutter and coworkers, does not escape the old problems of interobserver disagreement, the absence of a uniformly accepted and clinically credible morphologic definition of grade 1 adenocarcinoma, and most importantly, a real-world and independently verified clinical risk assessment on which valid and reproducible treatment recommendations can be based at this time.
To ascertain whether a patient with endometrial hyperplasia should be treated by hysterectomy, two classes of hysterectomies need to be distinguished: one performed on the climacteric or menopausal patient who is a low-risk surgical candidate (“low-penalty” hysterectomy) and one performed on the patient interested in preserving her fertility or on the climacteric or menopausal patient who is a high-risk surgical candidate or simply wishes to retain her uterus (“high-penalty” hysterectomy). Patients in the first group often undergo hysterectomy for benign disease (e.g., uterine prolapse, leiomyomas, dysfunctional bleeding) simply to deal with troublesome symptoms. Because fertility is no longer a relevant consideration, the risk of a life-threatening disease typically offsets whatever advantage (e.g., body image, perceived change in sexual function) there could be to retaining the uterus. In this situation, a diagnosis of atypical hyperplasia is sufficient to warrant hysterectomy.
A diagnosis of nonatypical hyperplasia does not, in itself, warrant hysterectomy, but the severity of the patient’s symptoms (e.g., dysfunctional bleeding) and the need for continued endometrial samplings may tip the balance enough to make hysterectomy the most attractive option. Patients in the high-penalty group are in a very different situation because substantial risks are attached to hysterectomy—infertility in the first subset and an increased risk of dying at the time of surgery or in the postoperative period in the second subset. In this high-penalty group, there is literature to support treating even well-differentiated carcinoma in a nonsurgical fashion.
Is there use in grading the degree of cytologic atypia in this clinicopathologic setting? Certainly, a diagnosis of nonatypical hyperplasia, by itself, is not sufficient grounds to remove the uterus; however, the same may be true of a diagnosis of atypical hyperplasia for patients in the high-penalty group. For these patients, it would seem that there are (or should be) no substantial changes in clinical treatment (i.e., withholding hysterectomy and managing the proliferation medically) anywhere along the spectrum from disordered proliferation to atypical hyperplasia. The critical end of the classification for high-penalty patients is at the boundary between atypical hyperplasia and grade 1 carcinoma or even at the interface of grade 1 and grade 2 carcinoma and not across any boundary within the hyperplasia group (93–96). When the cytologic atypia is that of grade 1 adenocarcinoma but the degree of architectural complexity is insufficient for a diagnosis of carcinoma, terminology such as “atypical hyperplasia, well-differentiated adenocarcinoma cannot be excluded” is warranted.
In summary, climacteric and menopausal women with atypical hyperplasia are usually treated by hysterectomy if they are suitable surgical candidates. Thus, the pathologist need not label borderline, worrisome lesions as adenocarcinoma to remove the uterus. Occasionally, curettage samples contain only atypical hyperplasia, whereas grade 1 adenocarcinoma is found in the hysterectomy specimen. No harm has been done in this circumstance by the misinterpretation because a diagnosis of carcinoma would have resulted in the identical treatment. Premenopausal women with atypical hyperplasia who desire uterine preservation may be treated by hormonal eradication of the lesion, recurettage, and induction of ovulation (80).
ENDOMETRIAL POLYP
Description
The usual endometrial polyp is a grossly pedunculated lesion whose bulk is composed predominantly of collagenated fibrous stroma populated by cystically dilated and occasionally crowded glands lined by inactive, atrophic to weakly proliferative endometrium (Fig. 53.25). Sometimes endometrial stromal cells are admixed with the fibrous tissue, and on rare occasions, the stromal cells may exhibit striking nuclear atypia or increased mitotic activity (97,98). All types of endometria, however, including those showing signs of hyperplasia, can be found in polyps; now and then, adenocarcinoma, including serous carcinoma or even carcinosarcoma, will be found in focal areas or diffusely throughout an otherwise characteristic polyp (99). The central portion of a polyp contains large, thick-walled, coiled blood vessels, and metaplastic epithelium may also be present (Fig. 53.26). Thus, the definition of an endometrial polyp requires a polypoid structure with a fibrous core containing large, thick-walled vessels. Rarely, polyps may be sessile. Polyps containing cycling endometrium are called functional polyps. If the stroma is made up predominantly of smooth muscle but the glands are not atypical, the term adenomyoma is used. If atypical endometrial glands are set in a predominantly smooth muscle stroma, the polyp is an atypical polypoid adenomyoma (see “Adenomyoma, Atypical Polypoid Adenomyoma, and Atypical Polypoid Adenomyoma of Low Malignant Potential”).
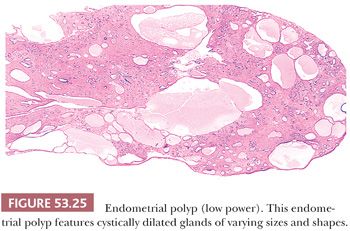
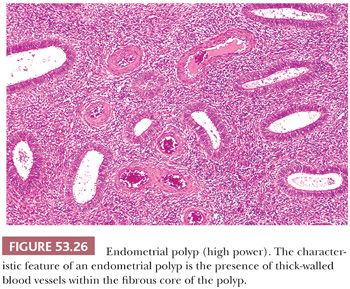
Clinicopathologic Correlation
Endometrial polyps are characteristically asymptomatic, incidental findings in perimenopausal women, but they may cause clinically significant bleeding or spotting. They are thought to be foci of retained endometrium that have become pedunculated and fibrotic over several cycles; many of these lesions undergo spontaneous regression (100). The relation of endometrial polyps to the subsequent development of endometrial carcinoma is uncertain. One case control study showed a somewhat increased risk of carcinoma in this context (101). Endometrial polyps with severe focal epithelial atypia have been associated with the administration of tamoxifen (102,103). Carcinomas developing in patients using tamoxifen are more often associated with endometrial polyps than are carcinomas developing in patients not treated with tamoxifen (104). Polypoid adenomyomas produce similar symptoms but are distinctly less common than polyps with predominantly fibrous stroma (105).
USUAL ENDOMETRIAL ADENOCARCINOMA
Description
Endometrial adenocarcinomas, by far the most common malignant tumors of the uterine corpus, are divided into endometrioid (usual) and special variant types. In terms of their architectural and cytologic features, better differentiated examples of endometrioid adenocarcinoma are composed of glands or villoglandular structures that resemble those found in endometrial hyperplasia; by convention, the endometrioid group includes neoplasms, which have a squamous component (Fig. 53.27). The remaining few endometrial adenocarcinomas are termed special variant carcinomas and are composed of cells demonstrating a type of differentiation usually encountered elsewhere in the female genital tract (Table 53.8). More than 80% of endometrial adenocarcinomas in most large series are of the endometrioid type. Because endometrioid adenocarcinomas represent the terminus along the morphologic spectrum of nonsecretory endometrium that we have been describing, it is natural to discuss them in this section, accompanied by an overview of endometrial carcinoma; the specifics concerning the special variant carcinomas are discussed in “Endometria with Alternative Differentiated Epithelium: Metaplasias and Special Variant Carcinomas.”
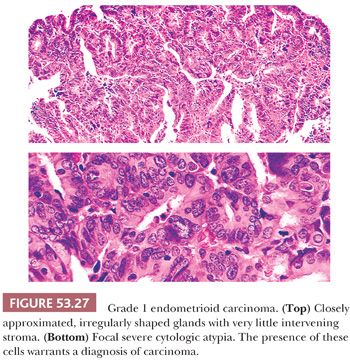

Endometrioid adenocarcinomas exhibit a continuous spectrum of appearances. This continuum is bracketed at one end by proliferations difficult to distinguish from atypical endometrial hyperplasia and at the other end by sheetlike proliferations of malignant cells difficult to distinguish from uterine sarcomas. The typical carcinoma in the endometrioid group is easy to recognize as an adenocarcinoma because gland formation is prominent; these neoplasms are sometimes associated with squamous elements that may have malignant cytologic features but more often are bland.
The low-power appearance of the nonsquamous elements typically conforms to one or more of three broad architectural patterns (Figs. 53.28 through 53.32). The first pattern consists of large, back-to-back, architecturally complex, compressive “macroglands” (Figs. 53.30 and 53.31); these macroglands may, in turn, be composed of closely approximated back-to-back buds set within a delicate stromal supportive framework whose presence may be suggested only by the radial polarization of the epithelial cells. The second pattern is represented by extensively budding and branching glands with little intervening stroma, forming a complex labyrinth (Fig. 53.29). Third, there may be a branching, exophytic thin-stalked papillary (villoglandular) pattern that is visible at low power and is reminiscent of villous adenoma of the gastrointestinal tract. Less often, the malignant glands are composed of extensively stratified epithelium forming a gland-within-gland (cribriform) pattern uninterrupted by stromal scaffolding (Fig. 53.32). Although most endometrioid carcinomas form easily found glands or complex, branching villoglandular structures, in high-grade carcinomas, these features may be focal and, in some cases, require extensive sampling to pinpoint. A detailed morphologic description of endometrial adenocarcinoma is provided later.
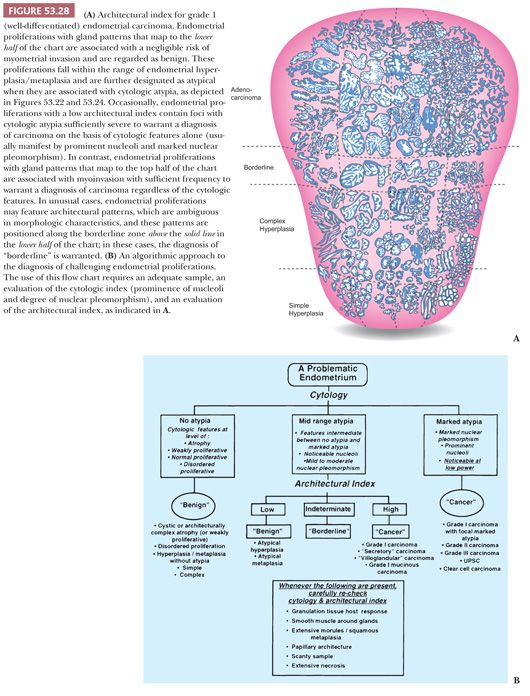
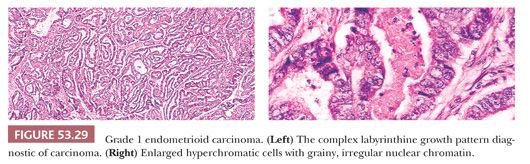
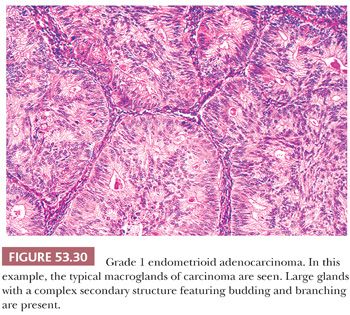
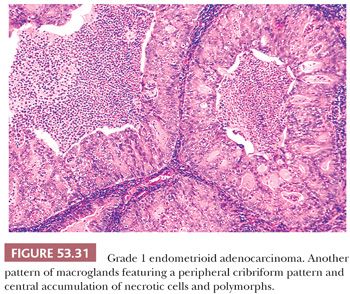
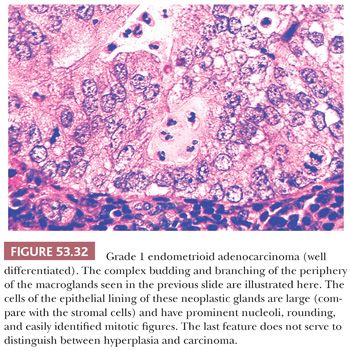
Many clinicopathologic studies of patients with endometrioid adenocarcinoma have shown that increasing loss of glandular and villoglandular architecture (and their displacement by more and more areas of sheetlike growth) is positively correlated with progressive cytologic atypia; both of these features, in turn, are correlated with higher stages (as determined at surgery) and diminished survival rates. These clinicopathologic correlations can be charted by a numeric grading system. Similarly, some types of special variant carcinoma are associated with high-grade clinical behavior. Thus, the histologic appearance of endometrial carcinomas (whether endometrioid or a special variant subtype) provides important insight into prognosis and plays a role in determining therapy (see “Clinicopathologic Correlation”). The histologic assessment of an endometrial carcinoma should proceed in a stepwise fashion.
First, histologic subtyping of endometrial carcinomas selects out two clinically aggressive special variant carcinomas—uterine serous carcinoma and clear cell carcinoma—which do not need to be graded because the subtype dictates outcome, and one clinically low-grade carcinoma—secretory carcinoma—which does not need to be graded because it is definitionally grade 1. The remaining subtypes of endometrial carcinoma are graded using the current modified International Federation of Gynecology and Obstetrics (FIGO) system, which is a three-tiered classification based on an evaluation of both the low-power architectural appearance and the cytologic features of a neoplasm (Fig. 53.33). This scheme is set out in Table 53.9 (Figs. 53.29, 53.31, 53.32, 53.34, and 53.35) (106,107).
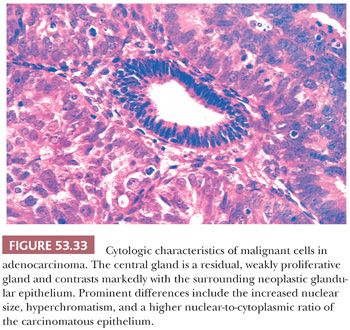
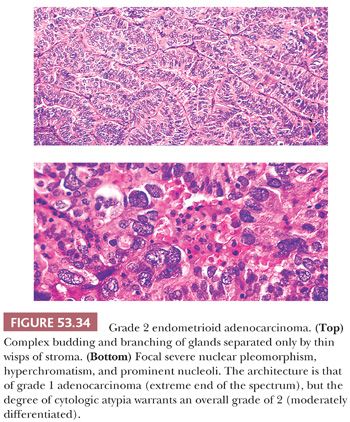
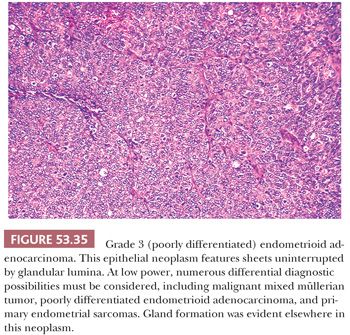

The cytologic features we principally rely on are nuclear pleomorphism and the prominence of nucleoli (Figs. 53.33 and 53.36). Additional features evaluated in assigning a cytologic grade to endometrioid carcinomas include nuclear size, nuclear irregularity, and the degree of chromatin clumping. When there is discordance between the cytologic and architectural grade, we supply both in the body of the pathology report but use the cytologic grade on the diagnosis line. The reproducibility of the FIGO modified system has been assessed, and it was found that interobserver agreement was acceptable for architectural grading but, not surprisingly, rather poor for cytologic grading (108). Observer agreement is improved by changing from a three-tiered system to a two-tiered system (109–112). These systems, although plausible, have not been generally adopted, and we do not currently recommend it.
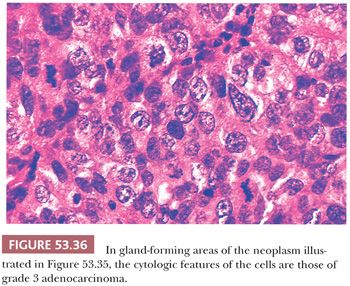
There are a variety of unusual morphologic patterns that can be seen in low-grade endometrioid adenocarcinoma, including sertoliform and other sex cord–like patterns (113–115), prominent spindle cell elements, corded and/or hyalinized stroma (115), and prominent intracytoplasmic clearing (116). Tumors with sertoliform differentiation typically exhibit slender cords and uniform, small hollow tubules lined by columnar cells with clear or pale cytoplasm (113). Tumors with spindle cell elements and prominent corded and hyalinized stroma usually feature areas of typical squamous differentiation (115), whereas the typical endometrioid carcinoma with prominent clear cell change features subnuclear or, occasionally, supranuclear vacuoles, resembling secretory endometrium. Whenever these unusual patterns are encountered, the diagnosis of low-grade endometrioid carcinoma is established on the basis of the underlying cytoarchitectural features or on the identification of typical endometrioid adenocarcinoma elsewhere in the tumor.
Clinicopathologic Correlation
Most patients with endometrial carcinoma show initial signs of uterine bleeding. Given uterine bleeding, the probability of carcinoma is a strong function of the patient’s age. Although rates may vary depending on the population and other risk factors, one group estimated the rate to be 9% for women in their 50s, 16% for those in their 60s, 28% for those in their 70s, and 60% for those in their 80s (117). Sampling issues are discussed earlier.
It is now apparent that many patients with endometrial adenocarcinoma fall into two loose clinicopathologic clusters or syndromes (118,119). Patients in the first group (type I) tend to be between 40 and 60 years of age, although carcinoma can develop in younger women, including, in rare instances, those in their 20s. These patients may have a history of chronic anovulation or estrogen hormone replacement therapy, and the carcinomas are usually well-differentiated, stage I nonmyoinvasive tumors with endometrioid histology, often associated with endometrial hyperplasia (found either concurrently or in previous endometrial samplings) (81). Most of these tumors are estrogen receptor (ER) and progesterone receptor (PR) positive and p53 negative and express low levels of the proliferation antigen Ki-67 (120). Patients with type I endometrial adenocarcinoma tend to have a very favorable prognosis after hysterectomy.
In contrast, patients in the second group (type II) tend to be elderly and typically have no history of hyperestrogenism (either endogenous or exogenous). In these cases, the surrounding nonneoplastic endometrium is almost always atrophic, and there is often an in situ component with high-grade cytologic features. The carcinomas that develop in this group of patients are usually of the special variant type with a poor prognosis or high-grade endometrioid neoplasms that are found at surgery to be high stage with deep myoinvasion. The type II tumors tend to be ER/PR negative, strongly express p53, and show high Ki-67 labeling (121,122). Needless to say, these patients are often not cured by hysterectomy. Lax and Kurman (122) summarized these clinical differences as well as a body of molecular genetic differences; they proposed that type I carcinomas conform closely to the adenoma–carcinoma sequence of colonic cancer, whereas type II carcinomas arise rapidly from an in situ lesion in the context of endometrial atrophy. A higher incidence of type II carcinomas has been observed in African American women (123). Several recent studies have also found an increased frequency of breast carcinoma in women with uterine serous carcinoma (124–126).
As noted earlier, almost all patients with adenocarcinomas of the endometrium are older than age 40 years at the time of diagnosis; of those few patients younger than 45 years of age, most fall into the first clinicopathologic group. In other words, they are hyperestrogenic and often obese, and their neoplasms are most often well differentiated and confined to the endometrium (127–130). They frequently have a history of anovulatory cycles or exogenous estrogen therapy; rarely, they may have a functioning ovarian tumor. Given the clinical indolence of the usual well-differentiated carcinoma in women younger than 45 years of age, we think a nonsurgical approach is acceptable for selected young women with grade 1 endometrial adenocarcinoma who desire uterine preservation. Those women in the reproductive years with grade 2 or 3 carcinomas should be treated like older patients with carcinomas of comparable grade (131).
Historically, estrogen therapy has also been linked to an increased risk of adenocarcinoma of the endometrium among perimenopausal and postmenopausal women. Almost all of these carcinomas are well differentiated (132,133).
Tamoxifen is a synthetic antiestrogen used in the treatment of breast cancer, but it has a paradoxically agonist effect on the endometrium and has been associated with the development of certain estrogen-driven proliferations, including endometrial carcinoma (see “Iatrogenic Patterns”).
In general, the outlook for patients with endometrial adenocarcinoma depends primarily on three factors (in order of importance): the stage of the neoplasm, the depth of myometrial invasion, and the histologic subtype of the tumor and its grade (Table 53.9) (134,135). Accurate assignment of histologic subtype and grade (when appropriate) requires adequate sampling. Other features reported to worsen the prognosis in endometrial carcinoma include the presence of lymphovascular invasion and uterine serosal involvement; these features should be noted in the body of the pathology report (136,137).
The presence of malignant cells in peritoneal fluids in women with endometrial adenocarcinoma is an adverse prognostic finding; however, the independent prognostic significance of carcinoma cells in peritoneal washings is unclear (138–141).
Lynch Syndrome
Endometrial carcinoma is the most common extracolonic cancer in women with Lynch syndrome (hereditary nonpolyposis colorectal cancer syndrome [HNPCC]), with an estimated lifetime risk of 42% or more (142). Lynch syndrome is attributed to germline mutations in mismatch repair genes, MLH1, MSH2, MSH6, and PMS2, as well as germline deletions of epithelial cell adhesion molecule, which produce transcriptional read-through, leading to hypermethylation of the MSH2 promoter (143). Up to 50% of women with Lynch syndrome may develop endometrial carcinoma as the first or sentinel cancer, often a full decade or more earlier than women with sporadic endometrial carcinoma (144). The frequency of Lynch syndrome–associated germline mutations in endometrial carcinomas has been estimated at 1.8% to 2.1%, which is similar to that in colon cancer (145–147). Mutations in the DNA mismatch repair genes involved in Lynch syndrome most frequently lead to loss of function and to microsatellite instability (MSI). Microsatellites are repetitive widely dispersed DNA sequences and prone to replication errors that are normally corrected by the DNA mismatch repair system (DNA-MMR). Deficiencies in the DNA-MMR therefore results in MSI (148). Because MSI can be a consequence of both genetic (related to mismatch repair gene mutations or Lynch syndrome) and epigenetic changes (sporadic due to MLH1 promoter methylation) (149), Lynch syndrome and MSI should not be used synonymously. Although 20% to 25% of all endometrial carcinomas show MSI, the majority (75%) result from sporadic MLH1 promoter methylation; only a minority are Lynch syndrome-associated (143).
Lynch syndrome endometrial cancers show a predilection for the lower uterine segment (this refers to tumors that arise in the lower uterine segment and does not include fundic tumors that may involve the lower uterine segment). Although initially as many as one-third of tumors arising in the lower uterine segment were reported to be Lynch syndrome-associated (150), the proportion is probably closer to 10% to 15% (151). The endometrial carcinomas can show a wide spectrum of histologic subtypes. Endometrioid carcinomas are the most common type, but nonendometrioid carcinomas also occur, including serous carcinoma, clear cell carcinoma, and carcinosarcomas, often at relatively younger ages (151–153).
An association between Lynch syndrome and presence of synchronous endometrioid carcinomas of the ovary and endometrium has been reported (142), but this association has not been uniformly substantiated (143,154,155). Synchronous endometrioid carcinomas of the ovary and endometrium are relatively common, particularly in young patients with estrogen excess. Synchronous uterine endometrioid carcinoma and ovarian clear cell carcinoma has been seen in association with Lynch syndrome, but the numbers of reported cases are few (155,156). There is no well established association between Lynch syndrome and endocervical adenocarcinoma (157), endometrial stromal sarcoma, or adenosarcoma.
MOLECULAR CLASSIFICATION OF ENDOMETRIAL CARCINOMA
The Cancer Genome Atlas (TCGA) (158) Research Network has recently classified endometrial cancers into four broad molecular groups:
1. Ultramutated cancers with DNA polymerase epsilon (POLE) mutations
2. Hypermutated cancers with defective mismatch repair and MSI
3. Low mutation rate cancers with low-frequency DNA copy number abnormalities
4. Low mutation rate cancers with high-frequency DNA copy number abnormalities
These molecularly defined groups reduplicate morphologic classification to an extent, although some of the molecular groups cross morphologic boundaries and appear to encompass apparent disparate morphologic groups. The classically defined morphologic and more recently identified molecular endometrial tumor groups are presented in Table 53.8. The ultramutated cancers with POLE mutations are microsatellite-stable tumors that account for 6% of low-grade endometrioid endometrial carcinomas and 17% of high-grade endometrioid endometrial carcinomas. The progression-free survival of patients in the ultramutated subgroup appears to be more favorable than for the other three molecular subgroups, although the number of ultramutated endometrial carcinomas that have been described thus far is small. The hypermutated cancers with defective mismatch repair and MSI exhibit frequent MLH1 promoter methylation and reduced MLH1gene expression. Hypermutated and microsatellite-unstable tumors are also associated with a heavily methylated subgroup suggestive of a CpG methylator phenotype. The receptor tyrosine kinase (RTK)/RAS/catenin pathway is altered in 69% of hypermutated and microsatellite-unstable tumors, and the PIK3CA/PIK3R1/PTEN axis is genomically altered in 95% of cases. KRAS alterations also occur in 35% of hypermutated and microsatellite-unstable endometrial tumors. In the TCGA tumor cohort, 29% of low-grade endometrioid endometrial carcinomas and 54% of high-grade endometrioid endometrial carcinomas were within the hypermutated and microsatellite-unstable subgroup. The low copy number/microsatellite-stable subgroup described by TCGA included 60% of low-grade endometrioid carcinomas, 9% of high-grade endometrioid carcinomas, 2% of serous carcinomas, and 25% of mixed histology carcinomas. Almost all (92%) of the tumors in this subgroup have a somatically altered PI3K pathway. KRAS is altered in only 16% of cases. The RTK/RAS/β-catenin pathway is also altered at high frequency (83%), and within this pathway, somatic mutations in CTNNB1 are particularly prevalent. More than 95% of serous carcinomas and 75% of mixed histology carcinomas fall into the high copy number tumor subgroup.
SURGICAL STAGING OF ENDOMETRIAL CARCINOMA
Before the 1980s, endometrial carcinoma was most often staged clinically by means of preoperative clinical studies combined with a careful scrutiny of the differential curettage samples. The preoperative clinical stage was used to formulate therapy, which, in the past, could well have included preoperative radiation therapy. Since that time, preoperative radiation has largely been abandoned (except for bulky extrauterine or cervical disease), and now the initial procedure is almost always abdominal hysterectomy. Staging and subsequent therapy are based on the histopathologic analysis of tissue removed at the time of hysterectomy. In addition to the uterus and adnexa, peritoneal biopsies, cytologic preparations, and lymph node samplings are frequently obtained.
In many institutions, the pathologist bears some responsibility for determining which patients will undergo surgical staging. Patients with grade 3 and high-risk special variant carcinomas, as determined by preoperative biopsies, are often considered candidates for a staging procedure regardless of the presence or absence of myometrial invasion or its depth. For patients with grade 1 or 2 endometrial carcinomas, gross examination of the myometrium at the time of operation and selected frozen section examination of apparent areas of invasion are often used to determine which patients will undergo staging. The final surgical pathologic stage assigned to a patient with endometrial carcinoma is based on complete examination of the hysterectomy specimen using permanent sections as well as intraoperative biopsies. Thus, the pathologist bears the chief responsibility for assigning the stage. The current FIGO staging and grading systems are set out in Table 53.9.
Assessing Myoinvasion
To obtain accurate staging information, it is essential to examine and section the hysterectomy specimen carefully and intelligently in a way that unambiguously reveals whether myoinvasion is present (Table 53.1). If it is, sections must be cut so the maximum depth of invasion can be determined. Invasive adenocarcinomas typically infiltrate the myometrium in the form of jagged, irregular branching glands, and inflamed granulation tissue often forms around these invasive foci (Figs. 53.37 to 53.39). In some instances, the invasive glands exhibit a distinctive microcystic, elongated, and fragmented pattern within the myometrium (159). In a large number of cases, however, establishing the presence of myoinvasion is not straightforward because the normal endometrial–myometrial junction is irregular, and the normal endometrium often interdigitates deeply with the myometrium. As a result, the basalis can lie deep in the myometrium.
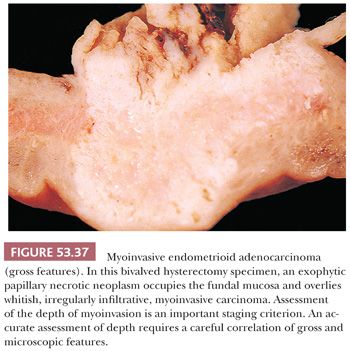
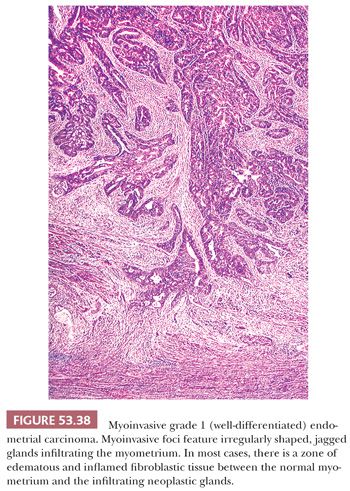
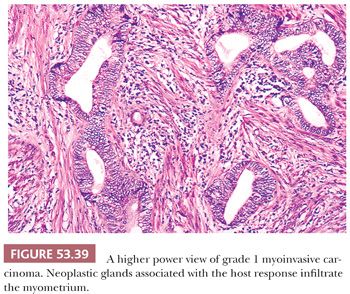
If deeply lying basalis is involved by carcinoma, it may be difficult (sometimes impossible) to distinguish this carcinomatous involvement from superficial myoinvasion. Unless a granulation tissue host inflammatory reaction is identified, we withhold a diagnosis of myoinvasion in challenging cases involving the superficial myometrium. One is unlikely to be proved wrong by this maneuver because the behavior of endometrial carcinomas with superficial myoinvasion is essentially that of nonmyoinvasive carcinomas of identical grade and type. Likewise, involvement of adenomyosis by carcinoma should be distinguished from myoinvasion because the former case does not imply a worse prognosis than carcinoma strictly limited to the endometrium (160).
Thorough sectioning to identify residual benign endometrial glands and/or endometrial stroma in suspected foci of myoinvasion is a useful way of establishing that carcinoma is confined to the endometrium or to an area of adenomyosis (Figs. 53.40 and 53.41). Moreover, neoplastic glands within deep invaginations of the endometrium or within adenomyosis usually have a blunted advancing front, a growth pattern different from the ragged infiltration found in many carcinomas, particularly of grades 2 and 3. Foci of adenomyosis are often surrounded by hyperplastic struts of myometrium, a feature not present around myoinvasive foci of carcinoma. The use of CD10 to highlight endometrial stromal cells has been proposed to be useful in distinguishing carcinoma involving adenomyosis from myoinvasion (161), but we and others have not found this to be particularly useful because some foci of adenomyosis may contain minimal or absent CD10-positive cells, whereas the mesenchymal cells surrounding foci of unequivocal myoinvasion may express CD10 (162,163).
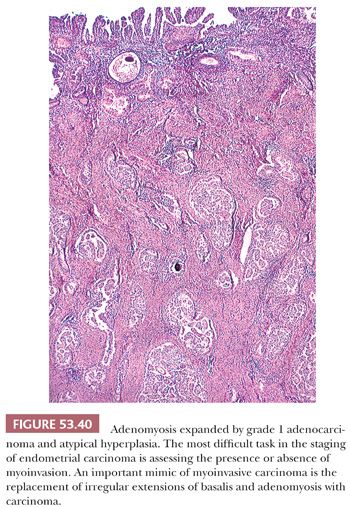
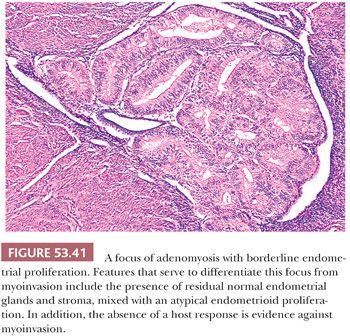
Despite these efforts, in some cases, determining whether carcinoma in the myometrium represents invasion or involvement of basalis or adenomyosis may be particularly problematic. If the tumor in an ambiguous case is grade 1, we tend to consider that it represents adenomyosis or basalis involvement; if it is grade 2 or 3, we are more inclined toward the myoinvasion interpretation. In either event, when uncertainty exists after culling many sections, we state the problem, provide our best determination of whether invasion is present, and, if appropriate, append the equivocation “invasion cannot be excluded.” Myometrial invasion by grade 1 endometrial adenocarcinoma has been reported to be more common in uteri containing carcinomatous involvement of adenomyosis than in those without such involvement (164).
Invasion from foci of adenomyosis involved by adenocarcinoma may rarely occur in absence of a detectable surface component (165–167). Whether minimal or early invasion from deep adenomyosis conveys the same prognosis as the equivalent level of invasion from a surface carcinoma is unknown.
Nonmyoinvasive carcinoma involving the intramural fallopian tube may mimic deep myoinvasion, and sections should be taken to avoid this pitfall whenever possible.
Occasionally, the glands forming well-differentiated carcinomas “melt” through the myometrium without inducing a host response; this condition almost always manifests in the form of massive tumor involvement of much of the myometrium, thus facilitating the diagnosis of myoinvasion (Fig. 53.42) (168).
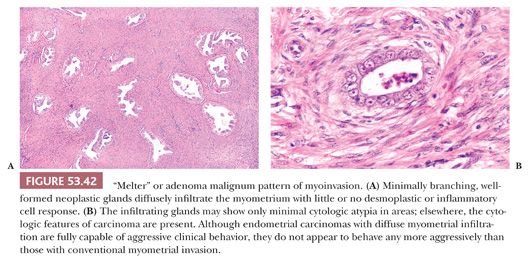
FIGO staging determines whether carcinoma has invaded the inner half or the outer half of the myometrium, with measurements taken from the junction of the endometrium and myometrium (Table 53.9) (106,107). The pathologist should report the raw measurements (uterine wall thickness and maximum depth of myoinvasion) and the manner in which they were obtained (e.g., measuring from the endomyometrial junction to both the deepest myoinvasive point and the serosa versus measuring from the surface of the carcinoma to those two points), so a ratio can be derived. More important, the pathologist’s reporting practices should conform to those of the treating physician. We report myoinvasion in accordance with the FIGO scheme and take our measurements from the endometrial/myometrial junction.
Lymphovascular involvement has been reported in several series to be an independent risk factor for recurrence and for death and should be noted in the pathology report. Lymphovascular involvement may be subtle, closely mimicking intravascular histiocytes (169). An association with perivascular lymphocytic infiltrates has been reported (136). The overall incidence of lymphovascular space involvement is 15% in stage I carcinoma; the percentages increase with increasing grade and increasing depth of myoinvasion (170).
Assessing the Status of the Cervix
Sampling of the cervix and lower uterine segment should be sufficient to determine whether the cervix is involved by carcinoma. Although the staging of endometrial carcinoma is now done on surgical rather than clinical grounds and the involvement of the cervix is definitively determined on the hysterectomy specimen, endocervical curettage is sometimes still performed on patients with endometrial carcinoma before hysterectomy in an attempt to determine the status of the cervix. Contamination of such specimens by carcinoma actually originating from the endometrium is a common phenomenon. Consequently, it is widely held that cervical stromal involvement should not be diagnosed unless the carcinoma actually anatomically involves the cervical stromal tissue. If fragments of endometrial adenocarcinoma and fragments of normal cervix are present but separate from each other in the endocervical curettage specimen and the adenocarcinoma is primary in the endometrium, the prognostic implications seem to be the same as when the endocervical curettage material does not contain adenocarcinoma. When cervical involvement is present, it is important to distinguish cervical glandular involvement without invasion of the stroma from cervical stromal invasion (FIGO II) because the latter is associated with a significantly poorer prognosis (171).
The finding of carcinoma involving the cervix should always raise the possibility of a cervical primary tumor. To some extent, the differentiated type of carcinoma is helpful in assigning primary site; pure squamous carcinomas only very rarely arise primarily in the endometrium. Unfortunately, all adenocarcinoma subtypes, including endometrioid, can potentially arise from either uterine corpus or cervix, and correlation with immunohistochemical studies, imaging, and colposcopic findings is required (see “Mucinous Carcinoma”) (172). A deceptively bland pattern of cervical stromal invasion by endometrial carcinoma featuring variably shaped, often tubular glands with little or no stromal response may mimic a primary endocervical process; this pattern is often associated with the “melter” or diffusely infiltrative pattern of invasion in the myometrium (168,173).
Evaluating Extrauterine Specimens Produced by the Staging Procedure
Occasionally, endometrioid adenocarcinoma is present in both the uterus and the ovary but only at these two sites. Whether these tumors are considered to represent a primary carcinoma and a metastasis to one site from the other (and hence high-stage neoplasms arising from one of the sites) or are interpreted as simultaneous primary tumors (hence both stage I) has significant implications for therapy and prognosis. When both neoplasms are grade 1 and the endometrial neoplasm is not myoinvasive, we think the available published evidence supports treatment as simultaneous primary tumors (174–178). Women with synchronous primary cancers of the endometrium and ovary exclusive of serous histologic type tend to be young, obese, nulliparous, and premenopausal (176). Most (~70%) have concordant endometrioid histology. When both neoplasms are high grade, assigning the stage is less definitive, but such carcinomas probably warrant adjuvant therapy whether they are simultaneous primary tumors or one is a metastasis, particularly if the endometrial tumor is serous or deeply myoinvasive. Foci of atypical hyperplasia can sometimes be seen in the uterus in association with endometrioid tumors of the ovary.
Simultaneous endometrioid carcinomas of the uterus and fallopian tube are far less common (179). The tumors that have been reported are predominantly well or moderately differentiated, often with discordant grades. The fallopian tube carcinoma is usually unilateral and located at the distal end of the tube (179). These tumors tend to exhibit a more aggressive clinical course than the simultaneous low-grade endometrial and ovarian endometrioid carcinomas.
Lymph nodes are often sampled in staging procedures, and foci of endosalpingiosis may be present; these foci should not be misinterpreted as adenocarcinoma (see Chapter 58). Peritoneal metastasis from an endometrial carcinoma may be mimicked by a peritoneal foreign body reaction to keratin, produced by endometrial carcinomas with prominent squamous elements that are regurgitated through the fallopian tubes (180,181). An erroneous diagnosis of carcinoma in this circumstance can be avoided by not interpreting bland squamous cells on the peritoneum as carcinoma.
Nearly three-fourths of patients with adenocarcinoma of the endometrium are found to have tumors limited to the corpus at the time of hysterectomy (stage I). Another 10% have cervical involvement (stage II), whereas the remainder are fairly evenly divided between stages III and IV (Table 53.9) (182). Cervical and/or occult lymph node metastasis is detected at the time of hysterectomy in approximately 10% of patients judged to have clinical stage I disease. These are almost exclusively patients with grade 2 or 3 carcinomas. Although patients with high-stage (stages III and IV) neoplasms have a poor prognosis, those with stage I or II endometrial adenocarcinoma of the endometrioid type have excellent outcomes (stage I survival is >90%, whereas stage II is roughly 80%) (183). These survival figures are higher if the carcinoma is grade 1 and myoinvasion is minimal or absent. Stage, grade, and depth of myometrial invasion are not simply the parameters used to gauge prognosis; they also determine the need for adjuvant therapy (183).
There has been interest concerning the correlation between the level of ER/PR in endometrial adenocarcinoma cells and the clinical outcome using current therapies. The results of numerous studies indicate that, in general, the increasing levels of receptor protein are positively correlated with low histologic grade (183). The presence of ER-α in metastatic endometrial carcinoma tissue prior to hormonal therapy is significantly related to clinical response to daily tamoxifen and intermittent medroxyprogesterone acetate therapy (184).
GRADE 1 (WELL-DIFFERENTIATED) ENDOMETRIOID ADENOCARCINOMA
Description
The morphologic boundaries (diagnostic criteria) of grade 1 adenocarcinoma have been variously drawn over the years (185). Controversy has surrounded both the atypical hyperplasia/grade 1 adenocarcinoma morphologic threshold and the grade 1/grades 2 and 3 carcinoma morphologic threshold. Different choices of boundaries, not surprisingly, result in different statistical (or average) properties of grade 1 adenocarcinomas, including disparate percentages of myoinvasive cases, varying relapse-free survival rates, and diverse descriptions of the typical case of grade 1 adenocarcinoma. What follows is a description of the typical grade 1 endometrioid adenocarcinoma using our diagnostic criteria; other commonly employed criteria, although differing on the assignment of cases near a boundary, would yield roughly the same description (185–187). In terms of its definition, grade 1 endometrioid adenocarcinoma must meet specific architectural and cytologic requirements (Figs. 53.27 to 53.33).
Architectural Abnormalities
Grade 1 endometrioid adenocarcinoma must consist almost entirely of epithelium with very little intervening stroma; in fact, an extensive, complex epithelial growth pattern is essential to the concept of well-differentiated endometrial adenocarcinoma. Usually, the tumor has a complex glandular pattern featuring budding and branching of enlarged neoplastic macroglands, which result in the formation of side channels and papillary infoldings (Figs. 53.27 and 53.29 to 53.32). In many cases, the constituent glands form long, complex, and intertwining channels which cannot be easily traced and are best detected by the microscopist’s ability to view large extents of luminal spaces and epithelial bridges without crossing intervening stroma (complex budding or labyrinthine glands). It is not uncommon, however, for the tumor to have a low-power villoglandular pattern reminiscent of villous adenoma of the gastrointestinal tract, featuring either pseudostratified epithelial cells on thin, delicate stromal stalks (really sheets or folia) or a uniform layer of budded glands growing out over stalks. The stromal stalks may be elongated and rigid or short and filiform, forming so-called nonvillous papillae (188). In both cases, there is a complex secondary branching structure, often with extensive nuclear pseudostratification; it is this feature that distinguishes grade 1 adenocarcinoma from atypical hyperplasia. Both glandular and villoglandular patterns may be found in the same tumor.
Less often seen is a pattern featuring true papillae formed by collagenated stromal stalks lined by relatively bland epithelial cells; this architectural pattern is much more characteristic of clear cell and serous carcinomas that are composed of cells with marked cytologic atypia (see “Uterine Serous Carcinoma”). For reasons outlined later, we require that at least 30% of the proliferation in an endometrial sampling that is considered to be well-differentiated (grade 1) adenocarcinoma exhibit these architectural abnormalities, which we label a high architectural index, as defined in Figure 53.28. Epithelial stratification uninterrupted by stroma so pronounced as to produce a cribriform pattern is relatively unusual in grade 1 adenocarcinoma, but it is more common in higher grade carcinomas. Conversely, back-to-back buds with thin, delicate intervening stroma (confusingly also referred to as cribriform) is characteristic of grade 1 adenocarcinoma.
Cytologic Abnormalities
A spectrum of cytologic atypia may be encountered in grade 1 endometrial adenocarcinoma. In brief, atypia falls short of that required for grade 2 carcinoma but is at least as severe as that of atypical hyperplasia (Figs. 53.32 and 53.33). This means that the degree of cytologic atypia for atypical hyperplasia and grade 1 adenocarcinoma may be identical; in this circumstance, grade 1 adenocarcinoma is recognized on the basis of the architectural changes described in the last paragraph. The typical cytologic features of grade 1 adenocarcinoma include cytomegaly, nucleomegaly, identifiable nucleoli, high nuclear-to-cytoplasmic ratios, mild hyperchromasia, and easily found mitotic figures. Atypical mitotic figures may be present, but they are usually sparse. Although they are relatively unusual features, significant nuclear pleomorphism and large eosinophilic nucleoli may be seen in endometrial proliferations that otherwise do not meet the complex architectural criteria delineated in Figure 53.28. In these exceptional cases, cytologic abnormalities are often only focal and are of a degree that, if they were present to a greater extent, would confer a grade 2 diagnosis under the revised FIGO grading system. Our data indicate that some of these lesions invade the myometrium; hence, endometrial proliferations with this degree of cytologic abnormality should be considered carcinomas, even though the architectural complexity found in the usual carcinoma is absent. In other words, this degree of cytologic atypia overrides the architectural requirements for grade 1 adenocarcinoma. Conversely, a few endometrial proliferations associated with myoinvasion have the glandular complexity of carcinoma but have cytologic abnormalities less severe than those usually seen in atypical hyperplasia. The aggressive potential of these latter cases cannot be recognized in biopsy and curettage specimens. The extreme architectural complexity, however, should lead the pathologist to interpret these as borderline lesions, which will result in appropriate treatment.
Extensive, marked nuclear pleomorphism with prominent, variably sized and shaped nucleoli are features of grades 2 and 3 adenocarcinoma, not of grade 1 endometrioid carcinomas. Squamous differentiation is often present; in grade 1 adenocarcinoma, the squamous elements characteristically are cytologically bland (adenoacanthoma) (Table 53.10). An approach to the evaluation of a challenging endometrial curettage sample is presented in Figure 53.28.

Although the pattern of irregular, atypical glands surrounded by a granulation tissue–like host fibrous stromal response in an endometrial curettage specimen is highly associated with the presence of myoinvasion in the subsequent hysterectomy specimen, care must be taken to exclude mimics of this infrequently encountered pattern. These mimics include foreign body reaction to keratin; nonspecific-associated endometritis; and the eosinophilic, cellular fibrous stroma, which is relatively nonspecifically associated with various endometrial lesions but is most notably seen in benign endometrial polyps and some cases of grade 1 adenocarcinoma. The latter type of fibrous tissue has very low predictive value for myoinvasion.
We find distinctions among types of stroma to be most challenging, and consequently, we place less reliance on the finding of fibroblastic stroma than other researchers. Foam cells may be present in either hyperplastic endometria or those harboring adenocarcinoma; thus, their presence provides no additional information regarding the aggressive potential of a proliferation. Extensive areas of squamous epithelium are more common in myoinvasive endometrial proliferations than in nonmyoinvasive proliferations, but the differences are insufficient for this to be a reliable discriminating feature. The problem of distinguishing atypical hyperplasia from grade 1 adenocarcinoma is discussed later in the “Differential Diagnosis” section.
Not infrequently, grade 1 adenocarcinoma cells demonstrate variation in cytoplasmic differentiation. In addition to the expected endometrioid cells, scattered ciliated cells, cells with eosinophilic cytoplasm without cilia, squamous cells and morules, cells with intracytoplasmic mucin, and cells with secretory-like features may be found. Most often, these different types of cells are mixed with the endometrioid cells, but grade 1 adenocarcinomas may feature populations of mixed cells alternating with areas of monotypic differentiation. When the alternatively differentiated cells come to predominate, a diagnosis of special variant carcinoma should be considered (see “Special Variant Carcinomas”).
Clinicopathologic Correlation
Grade 1 adenocarcinomas of the endometrium are almost always stage I, and most are not myoinvasive. In the Stanford series, the overall relapse-free survival rate for patients with grade 1, stage I tumors was nearly 95% (189). The figure is essentially 100% for patients with grade 1, stage I tumors confined to the endometrium. Hysterectomy is the treatment of choice for patients with grade 1 adenocarcinoma for whom fertility is not a consideration. Young women with grade 1 adenocarcinoma interested in preserving their childbearing capacity may be treated nonsurgically under some circumstances (see earlier discussion). Postoperative radiation therapy is often used for those patients in whom grade 1 adenocarcinoma has invaded more than a 50% thickness of the uterine wall.
GRADE 2 ENDOMETRIOID ADENOCARCINOMA
Description
The usual grade 2 adenocarcinoma (Fig. 53.34) has all the architectural features of a grade 1 adenocarcinoma, except 6% to 50% of the tumor is composed of a sheetlike proliferation of malignant cells in which glandular differentiation is either absent or barely discernible (Table 53.9). The constituent cells almost always have cytologic features characterized by pleomorphism and large, prominent nucleoli, which are easily recognized as malignant. Care must be taken to distinguish between a sheetlike proliferation of cytologically malignant cells and sheetlike formations produced by cytologically bland squamous cells or morules in what is otherwise grade 1 carcinoma; the latter should be ignored in assigning a grade to the carcinoma (see “Squamous Differentiation”; Table 53.10).
Clinicopathologic Correlation
The initial treatment for patients with grade 2 endometrial adenocarcinoma is hysterectomy and, in some patients, surgical staging. Postoperative radiation therapy is administered to patients whose carcinomas are myoinvasive. Because the clinical behavior of grades 2 and 3 adenocarcinomas of the endometrium is similar and both are distinctly more aggressive than grade 1 carcinomas, it has been suggested that grades 2 and 3 be collapsed into a single high-grade group; this would result in a simplified (and perhaps more clinically relevant) stratification of endometrial carcinoma into low-grade and high-grade groups. These considerations notwithstanding, we prefer to retain the three-tiered grading scheme because it allows more flexibility in the grading process.
GRADE 3 ENDOMETRIOID ADENOCARCINOMA
Description
The definition of grade 3 adenocarcinoma (Figs. 53.35 and 53.36) requires that more than 50% of the tumor be solid or sheetlike, with cytologically high-grade undifferentiated cells (Table 53.9). Even if better differentiated areas are present, the glands tend to be poorly formed. The constituent cells are easily recognized as malignant and feature marked nuclear pleomorphism and prominent nucleoli of varying sizes. Grade 3 endometrioid carcinomas sometimes contain areas of cytologically malignant squamous elements; not infrequently, grade 3 endometrioid adenocarcinomas are mixed with poor-prognosis special variant carcinomas (see “Squamous Metaplasia Associated with Benign Glandular Elements”; Table 53.10). Vascular invasion is not unusual; it should be searched for and reported if present.
Clinicopathologic Correlation
Most grade 3 carcinomas have invaded the myometrium by the time of hysterectomy. We think that any degree of myometrial invasion by grade 3 carcinoma warrants adjuvant therapy.
UNDIFFERENTIATED CARCINOMA
Undifferentiated carcinoma is distinguished from FIGO grade 3 endometrioid adenocarcinoma by the complete absence of glandular formation. These tumors are characterized by the presence of solid, dyshesive sheets of round or polygonal cells with vesicular nuclei and prominent nucleoli (190). Some may exhibit foci with myxoid matrix and rhabdoid cells; occasional cases may show lymphoepithelioma-like areas, defined as sheets of undifferentiated cells with a prominent lymphocytic infiltrate. Such tumors typically express cytokeratin and/or EMA focally or not at all. When accompanied by a component of well- to moderately differentiated (FIGO grade 1 or 2) endometrioid carcinoma, they have been referred to as dedifferentiated endometrial carcinomas (191). A significant proportion of undifferentiated carcinomas have sporadic methylation of MLH1 and are microsatellite unstable (192).
DIFFERENTIAL DIAGNOSIS: NONSECRETORY PATTERNS
Atrophic versus “Insufficient for Diagnosis”
At times, the endometrial sample may contain only fragments of the lower uterine segment. The glands in such a specimen are similar to those found in endometrial atrophy. Misinterpretation of such fragments as atrophy would mislead the clinician into believing that the endometrium had been adequately sampled when in fact it had not. This error can be avoided by paying attention to the prominent fibrotic stroma characteristic of the lower uterine segment. Even if the specimen consists entirely of atrophic endometrium, a scant specimen may not always be representative of the entire endometrium, and correlation with clinical and radiologic findings is required. In the interest of maintaining clinicopathologic harmony, it should be remembered that “insufficient for diagnosis” is not to be equated with “inadequate gynecologist”; there may be nothing more in the uterus for the sampling to obtain.
Atrophic versus Weakly Proliferative
In the usual case, weakly proliferative glands are tubular and are lined by small (relative to cells in normal proliferative glands), pseudostratified, elongated, bland cells with densely basophilic nuclei (Fig. 53.14). Mitoses are distinctly rare or absent. Atrophic glands are lined by flattened to cuboidal cells and may be tubular or cystically dilated (Figs. 53.10 and 53.11). Both of these patterns are normal in the perimenopausal and postmenopausal years and may be seen in endometria from patients experiencing abnormal uterine bleeding. It is of no importance to distinguish between atrophic and weakly proliferative endometria. It is important, however, to ensure that the sample is representative and that nothing else is present in curettage samples with this pattern. In this regard, many cases of high-grade carcinoma in postmenopausal women are unaccompanied by hyperplastic endometria. In some cases of uterine serous carcinoma, for example, the only evidence of the malignant neoplasm in curettage samples will be individual malignant cells scattered among otherwise atrophic endometrial fragments.
Atrophic Endometrium versus Endometrial Hyperplasia
Occasionally, the atrophic surface epithelium and underlying thin endometrium may be rolled up like a carpet by the sampling curette. Sectioning of this low-volume sample may yield a low-power pattern, which resembles complex hyperplasia in its predominance of epithelium over stroma (Fig. 53.12). The addition of a few atypical-looking ciliated cells may further prompt a diagnosis of atypical hyperplasia (Fig. 53.10). An awareness of this possibility and attention to the overall inactive appearance of the epithelium should lead to the correct diagnosis. Atrophic endometria may be abundant and may contain cystically dilated glands (Fig. 53.11), which may be crowded. Distinguishing this condition from hyperplasia is based on the flattened to cuboidal cells lining the atrophic glands rather than the mitotically active, elongated to round, and often enlarged cells characteristic of hyperplasia (Figs. 53.9 to 53.11, 53.19, and 53.20).
Disordered Proliferative versus Normal Proliferative
Both of these patterns are encountered in patients who are experiencing sporadic anovulation at the beginning and the end of the reproductive years—the perimenarche and the climacteric. The normal proliferative pattern is, of course, the usual follicular phase pattern found during the reproductive years. In addition, both these patterns are typically seen in patients receiving exogenous estrogens. Distinguishing disordered proliferative endometria (Figs. 53.15 to 53.17) from proliferative endometria is of no particular importance. The essential information conveyed by a diagnosis of disordered proliferation is that the patient has not ovulated, neither atypical hyperplasia nor carcinoma is present, and the specimen is judged to be representative of the uterine lining. Curettage is usually sufficient to control acute bleeding and, at least for the time being, to eradicate the responsible proliferation. Attention can then turn to tracking down the source of what is, in essence, estrogen-withdrawal bleeding and instituting appropriate hormonal manipulations.
Morphologic features that distinguish normal and disordered proliferation are presented earlier. Weakly proliferative endometria lack the mitotic activity found in both disordered proliferative and normal proliferative endometria, and the constituent nuclei are smaller and thinner.
Disordered Proliferative versus Hyperplasia
For a diagnosis of hyperplasia, rather than disordered proliferative endometrium (Figs. 53.15 to 53.21), we require that glandular overgrowth be sufficiently pronounced to shift the glands-to-stroma ratio to 2:1 to 3:1, that is, the stroma comprises less than one-third the volume of the proliferation. The glandular fraction includes both glands (and their lumina) and villoglandular structures.
Hyperplasia: Nonatypical versus Atypical
When evaluating hyperplastic endometria, the important observation is cytologic atypia in an architecturally simple or complex endometrium; only endometria with significant cytologic atypia should be labeled atypical hyperplasia. Cytologic atypia implies that most of the following features are present: enlargement and rounding of nuclei, prominence of nucleoli, chromatin clearing, hyperchromatism, and a tendency for the cells to appear piled up, with a loss of polarity. Cells with minimal atypia deviate only very slightly from those in proliferative endometria; they are larger but still elongated, and they have increased chromatin. Cells demonstrating severe atypia have the features found in grade 1 carcinoma cells, but their architectural arrangement is not as complex as is required for carcinoma (see earlier discussion). This said, we must add that there are substantial observer agreement problems in assessing atypia in complex hyperplasia (185–188).
Atypical Hyperplasia versus Grade 1 Adenocarcinoma
The diagnostic decision-making dilemmas in the range of atypical hyperplasia/grade 1 adenocarcinoma are notoriously challenging and are fully discussed earlier (Figs. 53.22 to 53.24 and 53.27 to 53.33). Our approach is also presented in Table 53.11 and Figure 53.28 (185). There are no known abrupt biochemical, cellular, kinetic, chromosomal, ultrastructural, or light microscopic discontinuities as one travels along the morphologic continuum from atypical hyperplasia to proliferations whose light microscopic features would compel a diagnosis of malignancy by all responsible observers. In short, special technology provides us with no gold standard for distinguishing the biologically benign from the biologically (and clinically relevant) malignant in the atypical hyperplasia/grade 1 adenocarcinoma range.
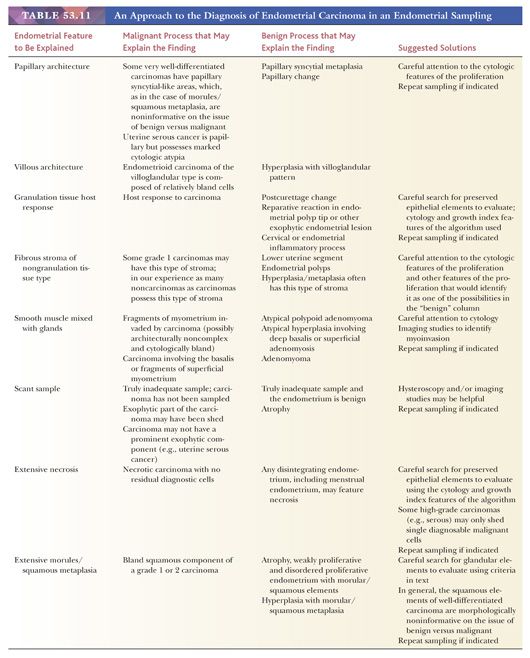
This leaves us with drawing subjective lines using standard H&E cytologic and architectural features. This line drawing is as much dependent on patients’ values, risks of current therapies, medical-legal considerations, and professional self-interest as it is on the “objectively evaluated” morphologic features of these proliferations. It follows that even given agreement on the clinicopathologic facts of the matter (e.g., given that a pattern is correlated with some probability of finding occult distant disease at the time of diagnosis or myoinvasion in the hysterectomy specimen), many plausible morphologic definitions of grade 1 adenocarcinoma are still possible—one for every combination of choices about the nonhistopathologic issues listed earlier. All of these problems also arise in the separation of atypical metaplasia and the corresponding well-differentiated special variant carcinomas, which feature cellular differentiation other than endometrioid.
Kurman and Norris (186) suggested one set of morphologic criteria for the diagnosis of grade 1 adenocarcinoma. These workers required that one of four patterns be present before making a diagnosis of carcinoma: irregularly shaped glands embedded in fibrous tumor stroma, confluent glandular growth, an extensive papillary growth, or extensive squamous epithelium. The latter three patterns must be present in a contiguous area of at least 2.1 mm. Using these criteria, Kurman and Norris (186) achieved a 90% sensitivity for detecting myoinvasion in the hysterectomy specimen (false-negative rate of 10%) and a 70% specificity for identifying myoinvasion (false-positive rate of 30%). In their series, the false-negative cases (diagnosis of atypical hyperplasia on the sampling when myoinvasive carcinoma was present in the hysterectomy specimen) were all ones in which the tumors were only superficially myoinvasive. In our experience, these criteria have a sensitivity of 80% and a specificity of 70%.
We have explored alternative sets of criteria and, using myoinvasion in the hysterectomy specimen as the outcome variable, we find that essentially all the predictive power of H&E criteria for distinguishing myoinvasion in these well-differentiated proliferations resides in the complex glandular and villoglandular patterns enumerated in the section devoted to well-differentiated adenocarcinoma, as long as the required minimal cytologic features are also present. For these well-differentiated borderline lesions, more elaborate rules incorporating the degree of cytologic abnormalities (nuclear pleomorphism, prominence of nucleoli), stromal features (presence of granulation tissue–like stroma, fibrous stroma, adenofibromatous or smooth muscle), and the differentiated features of the epithelium (squamous, mucinous, ciliated) could be constructed that marginally improved on the sensitivity and specificity figures cited, but none of them stood up under cross-validation.
This impression was confirmed when various rules were tested on an entirely different set of endometria from those used to construct the classification rule. Thus, of the four Kurman and Norris (186) criteria, confluent glandular growth and extensive papillary growth appeared to be the best at differentiating potentially myoinvasive proliferations and are predictive, whereas the extensive squamous epithelium and glands embedded in fibrous stroma did not add to the predictive power of these architectural features.
Most proliferations in this atypical hyperplasia/grade 1 adenocarcinoma spectrum are readily placed, with practice, on one side of the line or the other (Fig. 53.28 and Table 53.11). Problems are created by those cases that straddle the line or represent variations on the atypical hyperplasia/grade 1 adenocarcinoma theme that were unanticipated by the criteria. We find the term borderline lesion useful in these circumstances. Again, taking a broader analytic view is often more helpful than prolonged staring at the curettage specimen to divine its true nature. In the vocabulary used in the atypical hyperplasia section, it is clear that for low-risk hysterectomy patients, there is no question of not performing a hysterectomy when the endometrial lesion is borderline. The only thing at stake is rendering a possibly wrong diagnosis of malignancy, with the attendant concern that this engenders in the patient. The diagnosis of borderline lesion has the advantage of warranting a hysterectomy and tabling the issue of cancer until all the evidence is available.
Matters are more complicated for the high-penalty hysterectomy group of patients. The issue for them is whether to remove the uterus at all or to attempt reversal of the process pharmacologically. Again, the diagnosis of “borderline” leaves the clinician’s options open: hysterectomy or another sampling, attempted pharmacologic reversal, and resampling. In our opinion, the responsible way of handling this situation is not to make a dogmatic pronouncement in the presence of what realistically is fairly complete ignorance about the potential of a borderline lesion but (unpopular as it often is) to pass the uncertainty onto the clinician, who, in possession of the relevant clinical facts, is in a better position to deal with them in a more constructive and informed fashion than the pathologist.
As in the identification of atypia in hyperplasia, problems of observer agreement plague the distinction of atypical hyperplasia versus grade 1 adenocarcinoma (192–195). We addressed this problem in our study and found that with suitable pretest training, respectable κ values were attained (185).
Disintegrating Endometria versus Endometrial Hyperplasia or Adenocarcinoma
This may seem like a trivial differential diagnostic issue, but in reality, it is a fairly common and occasionally difficult problem; in some instances, it is unresolvable and requires rebiopsy. When the stroma of benign endometria (with any of the patterns discussed earlier) disintegrates and collapses, the glands may remain intact and may become closely approximated. This pattern can simulate a significant shift in the glands-to-stroma ratio in favor of glands and may thus imitate the low-power appearance of hyperplasia or carcinoma. Careful attention to nuclear features usually allows for distinction of this pattern from carcinoma. Care must be taken in evaluating nuclear features, however, because degenerated nuclei may become swollen and enlarged. Level sections to search for glands and intact stroma may be helpful. In menstrual endometria, a search for evidence of secretory changes in the glands, glandular apoptosis, and predecidual reaction in the stroma may also be helpful (see earlier discussion).
Polypoid Fragments of Endometrium versus Endometrial Polyp
Normal endometria, particularly secretory endometria, may have grossly polypoid areas, but they are histologically indistinguishable from surrounding nonpolypoid fragments. At the microscopic level, we require a fibrous stroma containing large, thick-walled vessels for a diagnosis of endometrial polyp.
Endometrial Polyp versus Adenofibroma versus Adenosarcoma versus Adenomyoma versus Atypical Polypoid Adenomyoma
Most adenofibromas and some adenosarcomas (see “Mixed Müllerian Neoplasia”) have a gross papillary architecture manifest microscopically either as long, epithelial-lined clefts extending into the stroma or as rounded papillae, some of which may be inserted into cystic spaces. Endometrial polyps do not have this architecture (Figs. 53.25 and 53.26; compare with Figs. 53.70 to 53.74). Adenomyomas have a myofibromatous stroma. Atypical polypoid adenomyomas also have smooth muscle or hybrid smooth muscle and fibrous stroma, and the glandular elements are architecturally complex and cytologically atypical (see Figs. 53.66 to 53.69). Morules and squamous differentiation are encountered in more than 90% of atypical polypoid adenomyomas.
Grading of Adenocarcinoma: Grade 1 versus Grade 2
The delimiting of endometrial carcinoma grades is as arbitrary as distinguishing atypical hyperplasia from grade 1 adenocarcinoma. The statistics relating grade to prognosis are summary statistics that reflect the behavior of the majority of cases that concentrate toward the centers of each of the grades; it is doubtful that those statistics would change significantly with the shifting of ambiguous cases from one side to the other of the grades 1 and 2 or 2 and 3 boundaries. We are nevertheless called on to assign an unambiguous grade to a malignant lesion, and the conventions for doing so are indicated under “Clinicopathologic Correlation” in the “Usual Endometrial Adenocarcinoma” section and in Table 53.9.
Grade 3 Adenocarcinoma versus Other Neoplasms Featuring Sheetlike Proliferation of Malignant Cells
Grade 3 adenocarcinomas are, by definition, largely undifferentiated. This feature raises numerous differential diagnostic difficulties, which are discussed in “Pattern 4: Sheetlike Proliferations Composed of Large, Round, Undifferentiated Cells” (see pattern 4 in Table 53.4).
PROGESTIN THERAPY FOR ENDOMETRIAL HYPERPLASIA AND LOW-GRADE ENDOMETRIAL ADENOCARCINOMA
Patients with atypical hyperplasia and well-differentiated endometrioid carcinoma are candidates for progestin treatment in lieu of hysterectomy. Although this therapy is typically reserved for younger women for whom reproductive conservation is an issue, older patients who wish to avoid surgery or who are poor candidates for surgery may also be treated with progestins. In younger women, a successful pregnancy rate of approximately 25% has been reported in patients so treated (196). Success with this therapy is predicated on (a) accurate diagnosis of the endometrial lesion to be treated, (b) sufficiently high-dose progestin delivered for a sufficiently prolonged period prior to reevaluation, and (c) the eradication of the lesion on follow-up EMB and/or curettage. Failures occur most often as a result of inadequate dosage, insufficient duration of treatment, and early or premature follow-up evaluation of the treated endometrium. An additional source of failure is misdiagnosis of the initial lesion; for example, atypical polypoid adenomyoma may persist following adequate progestin treatment, but this is not necessarily a warrant for hysterectomy because it is a focal lesion, poses minimal risk if carefully followed, and is not incompatible with successful pregnancy (see later section “Adenomyoma, Atypical Polypoid Adenomyoma, and Atypical Polypoid Adenomyoma of Low Malignant Potential”). When the pathologist receives a follow-up uterine sampling in a patient who has undergone progestin therapy for hyperplasia or carcinoma, the prior lesion(s) should be compared to determine degree of response. The glandular complexity, gland-to-stroma ratio, and degree of atypia present, if any, should be commented on. Complete response is manifested by diffuse stromal decidual reaction with inactive or weakly proliferative-appearing glands. Often, there may be a more pronounced mucinous, squamous, or eosinophilic metaplastic change than was evident in the initial biopsy because many of these metaplasias appear to be hormonally driven (197). In some instances, partial response followed by progression occurs, and so all biopsies should ideally be reviewed.
ENDOMETRIA WITH ALTERNATIVE DIFFERENTIATED EPITHELIUM: METAPLASIAS AND SPECIAL VARIANT CARCINOMAS
At times, benign cells with differentiation typically encountered elsewhere in the female genital tract may be discovered in endometrial proliferations (Fig. 53.8) (198). Similarly, carcinomas arising in the endometrium may, on occasion, demonstrate differentiation found more often in neoplasms arising in other parts of the female genitalia. The concept of the extended müllerian system is a useful construct for understanding these observations (198). The ovarian surface epithelium, fallopian tubes, uterus, and upper third of the vagina share a common embryologic history, and all these structures act, in many ways, as a single extended organ system. Many of these components show similar changes during pregnancy, develop comparable epithelial metaplasias, and share a common set of differentiated neoplasms. Furthermore, several neoplasms (often of identical histologic types) may arise in different components of the müllerian system metachronously or synchronously.
Although the full range of müllerian epithelial neoplasms may develop at any site within the female genital tract, the incidence of a particular differentiated type varies from one anatomic site to another. For example, most of the surface epithelial carcinomas that arise in the ovary may also be primary in the endometrium. In the endometrium, however, endometrioid carcinomas are by far the most common type; in the ovary, endometrioid carcinomas identical to those arising in the uterus are less common than serous neoplasms. In general, there is nothing in the intrinsic histologic characteristics of a müllerian neoplasm that pinpoints its anatomic site of origin. Papillary serous carcinoma looks much the same whether it arises in the endometrium (an infrequent occurrence) or in the ovary (a common occurrence).
These considerations have certain important implications for the histopathologist. First, clinicopathologic correlation is often required to establish the primary site of a gynecologic malignancy. In the endometrium, this is particularly true for neoplasms exhibiting mucinous (endocervical primary?) or serous (ovarian primary?) differentiation. Second, the diagnosis of benign or malignant, given a particular type of differentiation, may depend on the primary site. For example, considerably more cytologic atypia, mitotic activity, and epithelial stratification are allowed in benign endometrial proliferations with mucinous areas (mucinous metaplasia) than in benign endocervical proliferations with mucinous areas. Finally, the occurrence of synchronous, primary, müllerian neoplasms at several sites in the female genital tract has important implications for staging, prognosis, and therapy.
ENDOMETRIAL EPITHELIAL METAPLASIAS
In various circumstances, benign endometrial cells may exhibit epithelial differentiation other than the well-known differentiated patterns seen in proliferative and secretory endometria (198). The commonly encountered alternative epithelial types (metaplasias) are discussed here. These benign alternative epithelial types may be present in association with any of the nonsecretory endometria (i.e., atrophic, weakly proliferative, disordered proliferative, hyperplastic including atypical hyperplasia, and endometrioid carcinoma) (Fig. 53.8). The clinical significance that attaches to these patterns is, as far as we know, that of the underlying associated nonsecretory pattern.
For example, patients with atypical hyperplasia containing squamous metaplastic areas presumably have the same risk of endometrial carcinoma as those patients with hyperplasia with the same degree of cytologic atypia but uncomplicated by this differentiated feature. Benign metaplastic cells often cause diagnostic difficulties because of their unusual appearance and because they may grow in architectural arrangements also found in complex hyperplasia and in carcinoma. In particular, they may stratify (e.g., morules), or they may have cribriform patterns (e.g., ciliary metaplasia). When benign metaplastic epithelium coexists with carcinoma, the metaplastic cells do not warrant a change in the classification of the carcinoma; the classification of the carcinoma is based on the morphologic features of the malignant epithelium.
Cells with aberrant differentiation may themselves be malignant, however. In this circumstance, the term metaplasia is no longer appropriate; instead, the designation special variant carcinoma is employed. A final important differential diagnostic point is as follows: The subclassification of metaplasias into various types is for descriptive and differential diagnostic purposes. Once a proliferation is determined to be a metaplasia and its degree of atypia is determined, an unequivocal assignment to a particular metaplastic type is of little clinical (and probably less scientific) interest. Indeed, most metaplastic endometria are of mixed type, and it is likely that many of the patterns overlap (e.g., morules and papillary syncytial metaplasia).
SPECIAL VARIANT CARCINOMAS
The endometrium gives rise to widely varied differentiated carcinomas, but more than 80% are glandular neoplasms that resemble the epithelium found in endometrial hyperplasia (Table 53.8). Squamous or squamoid (morular) differentiation is commonly encountered in this endometrioid or usual adenocarcinoma. Other müllerian differentiated types make up the remainder of endometrial carcinomas, the so-called special variants. Serous carcinoma and clear cell carcinoma are important because of their notorious aggressiveness. Mucinous carcinoma raises questions concerning localization because both the cervix and the endometrium give rise to mucinous neoplasms and the treatment of carcinomas at these two sites differs. Carcinomas composed predominantly or exclusively of ciliated cells define the category of ciliated cell carcinoma and are a curiosity; however, scattered ciliated cells are frequently found in ordinary endometrioid carcinomas.
SPECIFICS ABOUT VARIOUS TYPES OF DIFFERENTIATION
Squamous Differentiation
Squamous differentiation in the endometrium takes many forms, ranging from highly keratinized epithelia that exfoliate anucleate squames to sheetlike proliferations of cells with indistinct cytoplasmic margins, no obvious keratinization, and often a rounded contour (morules) (Figs. 53.43 to 53.46; Table 53.10) (199). Occasionally, the sheetlike morules may feature either abrupt central hyaline keratinization or a central accumulation of granular necrotic material. Sometimes, morular cells are spindled. None of these cytoplasmic features in and of itself is diagnostic of malignancy.
The nuclear features of squamous cells encountered in the endometrium range from completely bland to obviously malignant. Cytologically bland squamous elements may be associated with normal endometrial glands, hyperplasias, and glandular proliferations with the features of carcinoma. Cytologically malignant squamous elements may, on extremely rare occasions, be present in isolation (pure squamous carcinoma), but they are more often seen in association with high-grade carcinomatous glandular elements. When the squamous or morular elements lack malignant cytologic features, the prognostic and therapeutic implications of proliferations containing squamous elements are essentially those that attach to the associated glandular part of the process. In other words, proliferations containing benign squamous or morular elements mixed with carcinomatous glands behave like the glandular component, whereas proliferations with identical squamous elements in a hyperplastic endometria behave like hyperplasia.
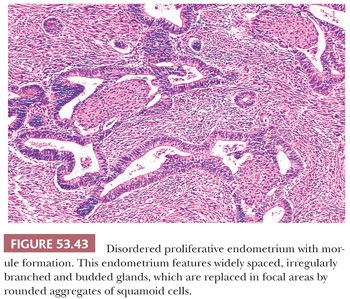
Squamous Metaplasia Associated with Benign Glandular Elements. This pattern of metaplasia may be seen in association with foreign body reactions, chemical irritants, and endometritis, but it is found most often and most extensively in hyperestrogenic states, particularly endometrial hyperplasia (Figs. 53.44 to 53.46). Ichthyosis uteri refers to the complete replacement of the endometrium by benign squamous epithelium (200). Progestin treatment of hyperplasia may produce striking squamous metaplasia (201).
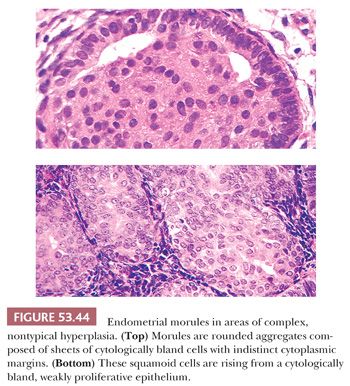
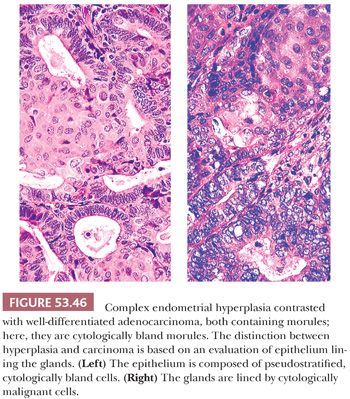
Carcinoma with Squamous Elements. The current view is that the squamous elements encountered in endometrioid carcinoma exhibit a spectrum of cytologic atypia, ranging from cytologically benign to cytologically malignant squamous proliferations and including the high-grade, large cell nonkeratinizing type; the cytologic atypia of the squamous elements more or less correlates with that of the glandular elements (202,203). Accordingly, we do not distinguish grade 3 endometrioid carcinomas from adenosquamous carcinomas of the large cell nonkeratinizing type. Our terminology is adenocarcinoma (specify grade) with squamous or morular metaplasia. When grade 3 adenocarcinoma with squamous or nonkeratinizing large cell elements is present, however, it may be appropriate to point out that such neoplasms are classified by some as adenosquamous carcinoma because clinicians are attuned to this term. We consider glassy cell carcinoma to be a variant of carcinoma with high-grade squamous elements. Carcinomas of the endometrium with squamous differentiation can be associated with a foreign body reaction on the peritoneum in the absence of metastases, apparently stemming from discharge of keratin through the fallopian tubes (180).
Differential Diagnosis. Not all endometrial proliferations with squamous elements are malignant. The differential diagnosis of bland squamous elements includes squamous metaplasia and morular metaplasia. The distinction between atypical hyperplasia with squamous metaplasia and grade 1 adenocarcinoma with bland squamous elements rests on an evaluation of the glandular component; the squamous component in this situation is not informative in terms of its morphologic features regarding whether the proliferation is benign or malignant. Most cytologically atypical squamous proliferations of the endometrium are intimately admixed with endometrial glandular elements easily recognized as malignant. When this is not the case, the possibility of a primary squamous cell carcinoma should be considered. The recovery of pure squamous cell carcinoma in a curettage specimen always raises the possibility of a cervical primary tumor, and suitable clinical localizing studies should be undertaken. Although a few cases of primary pure squamous cell carcinoma in the endometrium have been reported, this is indeed rare (204,205). Even more of a curiosity is verrucous carcinoma primary of the endometrium (206,207). On rare occasions, there may be extension of cervical carcinoma in situ into the uterine fundus (208–210).
Sheetlike Growth of Poorly Differentiated Carcinoma versus Sheetlike Growth of Morules. This is an important distinction, both in deciding between benign and malignant proliferations and (particularly if one uses the FIGO grading scheme) in establishing the grade of a carcinoma. The distinction is cytologic. High-grade carcinomas with a sheetlike architecture of stratified cells have obvious malignant nuclear features and easily found mitotic figures. Abnormal division figures are common. In contrast, although the constituent cells of morules are stratified into nodules or sheets, they lack malignant features, and mitotic figures are very difficult to find (Fig. 53.44). Central necrosis of morules is common, whether the glandular proliferation is benign or malignant (Fig. 53.45). When there is uncertainty about whether the squamous elements are malignant (high grade), the glandular elements can be used for grading. Most lesions containing squamous cells that are ambiguous are low grade.
Recovery of Abundant Bland Squamous Elements Unassociated with Glands in an Endometrial Sample. This problem occasionally arises; although carcinoma is discovered in the hysterectomy specimens of such patients more often than not, this finding by no means establishes that diagnosis. Endometria ranging from atrophic to acutely hyperplastic may be associated with large zones of squamous metaplasia or large areas of morular metaplasia.
Mucinous Differentiation
Mucinous differentiation is relatively common in the endometrium and may be encountered in association with the entire spectrum of nonsecretory endometrial patterns (185,211,212). It takes many different forms. On occasion, the epithelium may be highly reminiscent of normal endocervix, and when such fragments are discovered in an endometrial curettage specimen, they may masquerade as an endocervical fragment. On other occasions, endometrial cells containing cytoplasmic mucin are arranged on delicate fibrovascular stalks. Goblet cells may be present. On still other occasions, mucin-containing cells have delicately grainy, eosinophilic to slightly basophilic cytoplasm. When mucinous differentiation is found in glands with a tubuloalveolar pattern, acute inflammatory cells are usually embedded in the pools of mucin, a useful low-power marker of mucinous differentiation. Predominantly mucinous carcinomas develop in the endometrium and sometimes resemble those found more often in the endocervix or ovary. This histologic similarity occasionally raises difficult questions of localization.
Mucinous Metaplasia. Mucinous metaplasia is most often encountered in the context of hyperestrogenic states and, in rare instances, when associated with an element of cervical stenosis, may produce dramatic mucometra (Figs. 53.47 to 53.49). Diffuse or focal mucinous metaplasia is typically present in endometrial polyps and is not uncommon in postmenopausal endometria, usually in association with other metaplasias. Most forms of mucinous metaplasia simulate endocervical-type mucinous epithelium; intestinal metaplasia is rare but may also occur (213,214). Multifocal mucinous metaplasia may occur throughout the female genital tract (213,215).
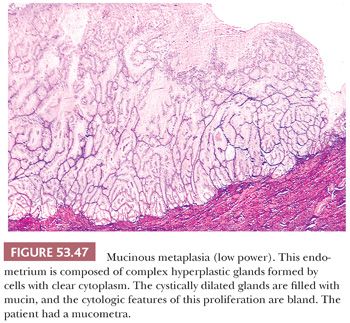
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


