46
Sulfonamides, Trimethoprim, & Quinolones
CASE STUDY
A 59-year-old woman presents to an urgent care clinic with a 4-day history of frequent and painful urination. She has had fevers, chills, and flank pain for the past 2 days. Her physician advised her to come immediately to the clinic for evaluation. In the clinic she is febrile (38.5°C [101.3°F]) but otherwise stable and states she is not experiencing any nausea or vomiting. Her urine dipstick test is positive for leukocyte esterase. Urinalysis and urine culture are ordered. Her past medical history is significant for three urinary tract infections in the past year. Each episode was uncomplicated, treated with trimethoprim-sulfamethoxazole, and promptly resolved. She also has osteoporosis for which she takes a daily calcium supplement. The decision is made to treat her with oral antibiotics for a complicated urinary tract infection with close follow-up. Given her history, what would be a reasonable empiric antibiotic choice? Depending on the antibiotic choice are there potential drug interactions?
 ANTIFOLATE DRUGS
ANTIFOLATE DRUGS
SULFONAMIDES
Chemistry
The basic formulas of the sulfonamides and their structural similarity to p-aminobenzoic acid (PABA) are shown in Figure 46–1. Sulfonamides with varying physical, chemical, pharmacologic, and antibacterial properties are produced by attaching substituents to the amido group (–SO2–NH–R) or the amino group (–NH2) of the sulfanilamide nucleus. Sulfonamides tend to be much more soluble at alkaline than at acid pH. Most can be prepared as sodium salts, which are used for intravenous administration.
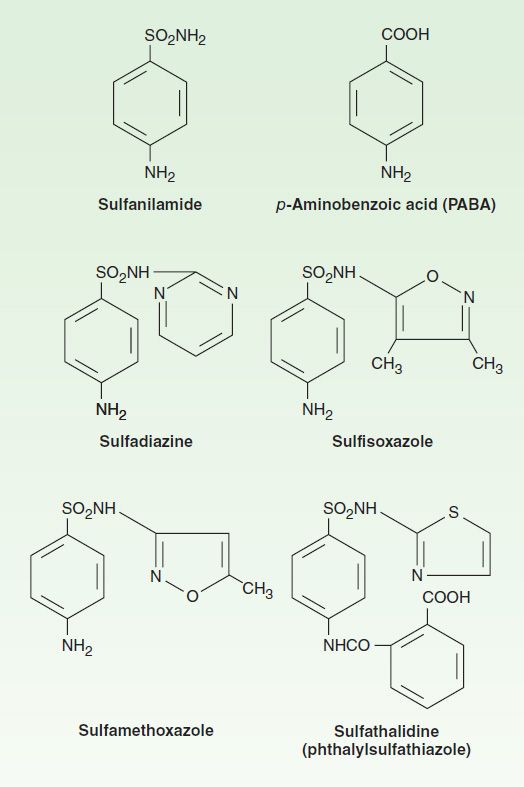
FIGURE 46–1 Structures of some sulfonamides and p-aminobenzoic acid.
Mechanism of Action & Antimicrobial Activity
Sulfonamide-susceptible organisms, unlike mammals, cannot use exogenous folate but must synthesize it from PABA. This pathway (Figure 46–2) is thus essential for production of purines and nucleic acid synthesis. As structural analogs of PABA, sulfonamides inhibit dihydropteroate synthase and folate production. Sulfonamides inhibit both gram-positive and gram-negative bacteria, Nocardia sp, Chlamydia trachomatis, and some protozoa. Some enteric bacteria, such as Escherichia coli, Klebsiella pneumoniae, Salmonella, Shigella, and Enterobacter sp are also inhibited. It is interesting that rickettsiae are not inhibited by sulfonamides but are instead stimulated in their growth. Activity is poor against anaerobes. Pseudomonas aeruginosa is intrinsically resistant to sulfonamide antibiotics.
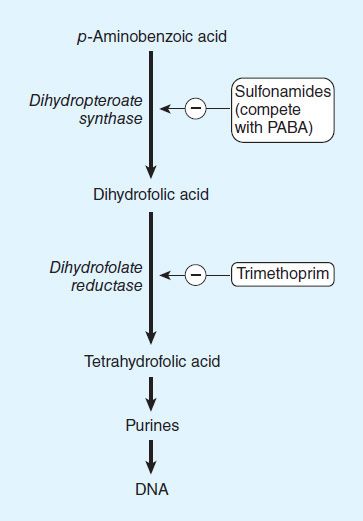
FIGURE 46–2 Actions of sulfonamides and trimethoprim.
Combination of a sulfonamide with an inhibitor of dihydrofolate reductase (trimethoprim or pyrimethamine) provides synergistic activity because of sequential inhibition of folate synthesis (Figure 46–2).
Resistance
Some bacteria lack the enzymes required for folate synthesis from PABA and, like mammals, depend on exogenous sources of folate; therefore, they are not susceptible to sulfonamides. Sulfonamide resistance may also occur as a result of mutations that (1) cause overproduction of PABA, (2) cause production of a folic acid-synthesizing enzyme that has low affinity for sulfonamides, or (3) impair permeability to the sulfonamide. Dihydropteroate synthase with low sulfonamide affinity is often encoded on a plasmid that is transmissible and can disseminate rapidly and widely. Sulfonamide-resistant dihydropteroate synthase mutants also can emerge under selective pressure.
Pharmacokinetics
Sulfonamides can be divided into three major groups: (1) oral, absorbable; (2) oral, nonabsorbable; and (3) topical. The oral, absorbable sulfonamides can be classified as short-, intermediate-, or long-acting on the basis of their half-lives (Table 46–1). They are absorbed from the stomach and small intestine and distributed widely to tissues and body fluids (including the central nervous system and cerebrospinal fluid), placenta, and fetus. Protein binding varies from 20% to over 90%. Therapeutic concentrations are in the range of 40–100 mcg/mL of blood. Blood levels generally peak 2–6 hours after oral administration.
TABLE 46–1 Pharmacokinetic properties of some sulfonamides and pyrimidines.
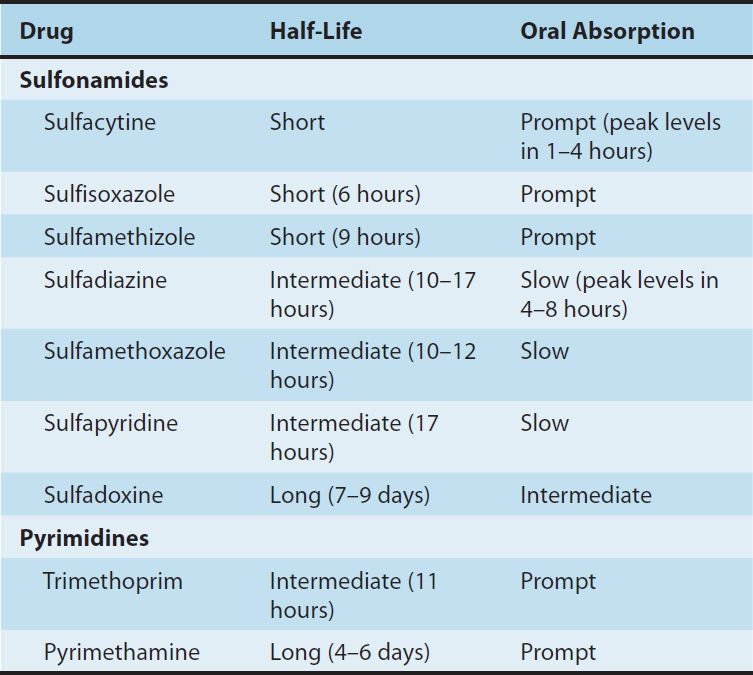
A portion of absorbed drug is acetylated or glucuronidated in the liver. Sulfonamides and inactive metabolites are then excreted into the urine, mainly by glomerular filtration. In significant renal failure, the dosage of sulfonamides must be reduced.
Clinical Uses
Sulfonamides are infrequently used as single agents. Many strains of formerly susceptible species, including meningococci, pneumococci, streptococci, staphylococci, and gonococci, are now resistant. The fixed-drug combination of trimethoprim-sulfamethoxazole is the drug of choice for infections such as Pneumocystis jiroveci (formerly P carinii) pneumonia, toxoplasmosis, nocardiosis, and occasionally other bacterial infections.
A. Oral Absorbable Agents
Sulfisoxazole and sulfamethoxazole are short- to medium-acting agents used almost exclusively to treat urinary tract infections. The usual adult dosage is 1 g of sulfisoxazole four times daily or 1 g of sulfamethoxazole two or three times daily.
Sulfadiazine in combination with pyrimethamine is first-line therapy for treatment of acute toxoplasmosis. The combination of sulfadiazine with pyrimethamine, a potent inhibitor of dihydrofolate reductase, is synergistic because these drugs block sequential steps in the folate synthesis pathway (Figure 46–2). The dosage of sulfadiazine is 1 g four times daily, with pyrimethamine given as a 75 mg loading dose followed by a 25 mg once-daily dose. Folinic acid, 10 mg orally each day, should also be administered to minimize bone marrow suppression.
Sulfadoxine is a long-acting sulfonamide that is coformulated with pyrimethamine (Fansidar). This combination is no longer commercially available in the USA but may be found in other parts of the world where it is used as a second-line treatment of malaria (see Chapter 52).
B. Oral Nonabsorbable Agents
Sulfasalazine (salicylazosulfapyridine) is widely used in ulcerative colitis, enteritis, and other inflammatory bowel disease (see Chapter 62).
C. Topical Agents
Sodium sulfacetamide ophthalmic solution or ointment is effective in the treatment of bacterial conjunctivitis and as adjunctive therapy for trachoma. Another sulfonamide, mafenide acetate, is used topically but can be absorbed from burn sites. The drug and its primary metabolite inhibit carbonic anhydrase and can cause metabolic acidosis, a side effect that limits its usefulness. Silver sulfadiazine is a less toxic topical sulfonamide and is preferred to mafenide for prevention of infection of burn wounds.
Adverse Reactions
Historically, drugs containing a sulfonamide moiety, including antimicrobial sulfas, diuretics, diazoxide, and the sulfonylurea hypoglycemic agents, were considered to be cross-allergenic. However, recent evidence suggests cross-reactivity is uncommon and patients who are allergic to nonantibiotic sulfonamides may safely receive sulfonamide antibiotics. The most common adverse effects are fever, skin rashes, exfoliative dermatitis, photosensitivity, urticaria, nausea, vomiting, diarrhea, and difficulties referable to the urinary tract (see below). Stevens-Johnson syndrome, although relatively uncommon (< 1% of treatment courses), is a particularly serious and potentially fatal type of skin and mucous membrane eruption associated with sulfonamide use. Other unwanted effects include stomatitis, conjunctivitis, arthritis, hematopoietic disturbances (see below), hepatitis, and, rarely, polyarteritis nodosa and psychosis.
A. Urinary Tract Disturbances
Sulfonamides may precipitate in urine, especially at neutral or acid pH, producing crystalluria, hematuria, or even obstruction. This is rarely a problem with the more soluble sulfonamides (eg, sulfisoxazole). Sulfadiazine when given in large doses, particularly if fluid intake is poor, can cause crystalluria. Crystalluria is treated by administration of sodium bicarbonate to alkalinize the urine and fluids to increase urine flow. Sulfonamides have also been implicated in various types of nephrosis and in allergic nephritis.
B. Hematopoietic Disturbances
Sulfonamides can cause hemolytic or aplastic anemia, granulocytopenia, thrombocytopenia, or leukemoid reactions. Sulfonamides may provoke hemolytic reactions in patients with glucose-6-phosphate dehydrogenase deficiency. Sulfonamides taken near the end of pregnancy increase the risk of kernicterus in newborns.
TRIMETHOPRIM & TRIMETHOPRIM-SULFAMETHOXAZOLE MIXTURES
Mechanism of Action
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


