28
Structure
CHAPTER CONTENTS
SIZE & SHAPE OF VIRUSES
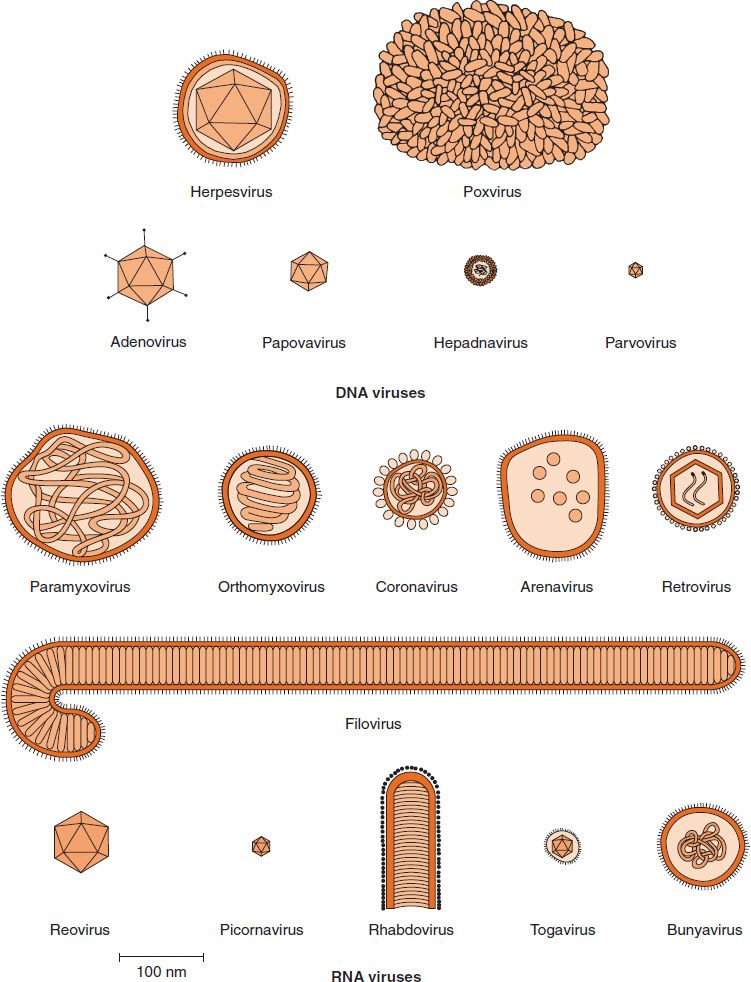
Viruses range from 20 to 300 nm in diameter; this corresponds roughly to a range of sizes from that of the largest protein to that of the smallest cell (see Figure 2–2). Their shapes are frequently referred to in colloquial terms (e.g., spheres, rods, bullets, or bricks), but in reality they are complex structures of precise geometric symmetry (see later). The shape of virus particles is determined by the arrangement of the repeating subunits that form the protein coat (capsid) of the virus. The shapes and sizes of some important viruses are depicted in Figure 28–1.
FIGURE 28–1 Shapes and sizes of medically important viruses. (Modified and reproduced with permission from Fenner F, White DO. Medical Virology. 4th ed. Academic Press. Copyright 1994 Elsevier.)
VIRAL NUCLEIC ACIDS
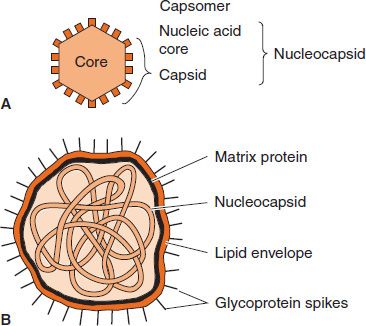
The anatomy of two representative types of virus particles is shown in Figure 28–2. The viral nucleic acid (genome) is located internally and can be either single- or double-stranded DNA or single- or double-stranded RNA.1
FIGURE 28–2 Cross-section of two types of virus particles. A: Nonenveloped virus with an icosahedral nucleocapsid. B: Enveloped virus with a helical nucleocapsid. (Modified and reproduced with permission from Brooks GF et al. Medical Microbiology. 20th ed. Originally published by Appleton & Lange. Copyright 1995 by McGraw-Hill.)
Only viruses have genetic material composed of single-stranded DNA or of single-stranded or double-stranded RNA. The nucleic acid can be either linear or circular. The DNA is always a single molecule; the RNA can exist either as a single molecule or in several pieces. For example, both influenza virus and rotavirus have a segmented RNA genome. Almost all viruses contain only a single copy of their genome (i.e., they are haploid). The exception is the retrovirus family, whose members have two copies of their RNA genome (i.e., they are diploid).
VIRAL CAPSID & SYMMETRY
The nucleic acid is surrounded by a protein coat called a capsid made up of subunits called capsomers. Each capsomer, consisting of one or several proteins, can be seen in the electron microscope as a spherical particle, sometimes with a central hole.
The structure composed of the nucleic acid genome and the capsid proteins is called the nucleocapsid. The arrangement of capsomers gives the virus structure its geometric symmetry. Viral nucleocapsids have two forms of symmetry: (1) icosahedral, in which the capsomers are arranged in 20 triangles that form a symmetric figure (an icosahedron) with the approximate outline of a sphere; and (2) helical, in which the capsomers are arranged in a hollow coil that appears rod-shaped. The helix can be either rigid or flexible. All human viruses that have a helical nucleocapsid are enclosed by an outer membrane called an envelope (i.e., there are no naked helical viruses). Viruses that have an icosahedral nucleocapsid can be either enveloped or naked (see Figure 28–2).
The advantage of building the virus particle from identical protein subunits is twofold: (1) it reduces the need for genetic information, and (2) it promotes self-assembly (i.e., no enzyme or energy is required). In fact, functional virus particles have been assembled in the test tube by combining the purified nucleic acid with the purified proteins in the absence of cells, energy source, and enzymes.
VIRAL PROTEINS
Viral proteins serve several important functions. The capsid proteins protect the genome DNA or RNA from degradation by nucleases. The proteins on the surface of the virus mediate the attachment of the virus to specific receptors on the host cell surface. This interaction of the viral proteins with the cell receptor is the major determinant of species and organ specificity. Outer viral proteins are also important antigens that induce neutralizing antibody and activate cytotoxic T cells to kill virus-infected cells. These outer viral proteins not only induce antibodies, but are also the target of antibodies (i.e., antibodies bind to these viral proteins and prevent [“neutralize”] the virus from entering the cell and replicating). The outer proteins induce these immune responses following both the natural infection and immunization (see later).
The term “serotype” is used to describe a subcategory of a virus based on its surface antigens. For example, measles virus has one serotype, polioviruses have three serotypes, and rhinoviruses have over 100 serotypes. This is because all measles viruses have only one antigenic determinant on their surface protein that induces neutralizing antibody capable of preventing infection. In contrast, polioviruses have three different antigenic determinants on their surface proteins (i.e., poliovirus type 1 has one kind of antigenic determinant, poliovirus type 2 has a different antigenic determinant, and poliovirus type 3 has a different antigenic determinant from types 1 and 2); hence polioviruses have three serotypes. There are two important medical implications of this. First is that a person can be immune (have antibodies) to poliovirus type 1 and still get the disease, poliomyelitis, caused by poliovirus types 2 or 3. The other implication is that the polio vaccine must contain all three serotypes in order to be completely protective.
Some of the internal viral proteins are structural (e.g., the capsid proteins of the enveloped viruses), whereas others are enzymes (e.g., the polymerases that synthesize the viral mRNA). The internal viral proteins vary depending on the virus. Some viruses have a DNA or RNA polymerase attached to the genome; others do not. If a virus has an envelope, then a matrix protein that mediates the interaction between the capsid proteins and the envelope proteins is present.
Some viruses produce proteins that act as “superantigens,” similar in their action to the superantigens produced by bacteria, such as the toxic shock syndrome toxin of Staphylococcus aureus (see Chapters 15 and 58). Viruses known to produce superantigens include two members of the herpesvirus family, namely, Epstein–Barr virus and cytomegalovirus, and the retrovirus mouse mammary tumor virus. The current hypothesis offered to explain why these viruses produce a superantigen is that activation of CD4-positive T cells is required for replication of these viruses to occur.
Some viruses contain regulatory proteins in the virion in a structure called the tegument, which is located between the nucleocapsid and the envelope. These regulatory proteins include transcription and translation factors that control either viral or cellular processes. Members of the herpesvirus family, such as herpes simplex virus and cytomegalovirus, have a prominent, well-characterized tegument.
VIRAL ENVELOPE
In addition to the capsid and internal proteins, there are two other types of proteins, both of which are associated with the envelope. The envelope is a lipoprotein membrane composed of lipid derived from the host cell membrane and protein that is virus-specific. Furthermore, there are frequently glycoproteins in the form of spike-like projections on the surface, which attach to host cell receptors during the entry of the virus into the cell. Another protein, the matrix protein, mediates the interaction between the capsid proteins and the envelope.
The viral envelope is acquired as the virus exits from the cell in a process called “budding” (see Chapter 29). The envelope of most viruses is derived from the cell’s outer membrane, with the notable exception of herpesviruses that derive their envelope from the cell’s nuclear membrane.
In general, the presence of an envelope confers instability on the virus. Enveloped viruses are more sensitive to heat, drying, detergents, and lipid solvents such as alcohol and ether than are nonenveloped (nucleocapsid) viruses, which are composed only of nucleic acid and capsid proteins.
An interesting clinical correlate of this observation is that virtually all viruses that are transmitted by the fecal–oral route (those that have to survive in the environment) do not have an envelope; that is, they are naked nucleocapsid viruses. These include viruses such as hepatitis A virus, poliovirus, Coxsackie virus, echovirus, Norwalk virus, and rotavirus. In contrast, enveloped viruses are most often transmitted by direct contact, such as by blood or by sexual transmission. Examples of these include human immunodeficiency virus, herpes simplex virus type 2, and hepatitis B and C viruses. Other enveloped viruses are transmitted directly by insect bite (e.g., yellow fever virus and West Nile virus) or by animal bite (e.g., rabies virus).
Many other enveloped viruses are transmitted from person to person in respiratory aerosol droplets, such as influenza virus, measles virus, rubella virus, respiratory syncytial virus, and varicella-zoster virus. If the droplets do not infect directly, they can dry out in the environment, and these enveloped viruses are rapidly inactivated. Note that rhinoviruses, which are transmitted by respiratory droplets, are naked nucleocapsid viruses and can survive in the environment for significant periods. Therefore, they can also be transmitted by hands that make contact with the virus on contaminated surfaces.
As described earlier in this chapter, the surface proteins of the virus, whether they are the capsid proteins or the envelope glycoproteins, are the principal antigens against which the host mounts its immune response to viruses. They are also the determinants of type specificity (often called the serotype). There is often little cross-protection between different serotypes. Viruses that have multiple serotypes (i.e., have antigenic variants) have an enhanced ability to evade our host defenses because antibody against one serotype will not protect against another serotype.
ATYPICAL VIRUS-LIKE AGENTS
There are four exceptions to the typical virus as described earlier:
(1) Defective viruses are composed of viral nucleic acid and proteins but cannot replicate without a “helper” virus, which provides the missing function. Defective viruses usually have a mutation or a deletion of part of their genetic material. During the growth of most human viruses, many more defective than infectious virus particles are produced. The ratio of defective to infectious particles can be as high as 100:1. Because these defective particles can interfere with the growth of the infectious particles, it has been hypothesized that the defective viruses may aid in recovery from an infection by limiting the ability of the infectious particles to grow.
(2) Pseudovirions contain host cell DNA instead of viral DNA within the capsid. They are formed during infection with certain viruses when the host cell DNA is fragmented and pieces of it are incorporated within the capsid protein. Pseudovirions can infect cells, but they do not replicate.
(3) Viroids consist solely of a single molecule of circular RNA without a protein coat or envelope. There is extensive homology between bases in the viroid RNA, leading to large double-stranded regions. The RNA is quite small (molecular weight 1 × 105) and apparently does not code for any protein. Nevertheless, viroids replicate, but the mechanism is unclear. They cause several plant diseases but are not implicated in any human disease.
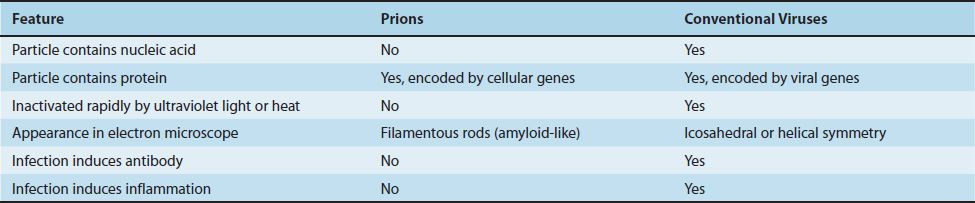
(4) Prions are infectious particles that are composed solely of protein (i.e., they contain no detectable nucleic acid). They are implicated as the cause of certain “slow” diseases called transmissible spongiform encephalopathies, which include such diseases as Creutzfeldt-Jakob disease in humans and scrapie in sheep (see Chapter 44). Because neither DNA nor RNA has been detected in prions, they are clearly different from viruses (Table 28–1). Furthermore, electron microscopy reveals filaments rather than virus particles. Prions are much more resistant to inactivation by ultraviolet light and heat than are viruses. They are remarkably resistant to formaldehyde and nucleases. However, they are inactivated by hypochlorite, NaOH, and autoclaving. Hypochlorite is used to sterilize surgical instruments and other medical supplies that cannot be autoclaved.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree