HISTOLOGY
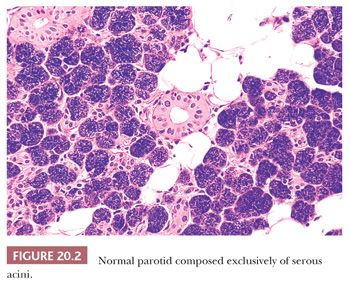
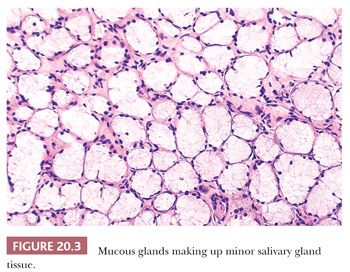
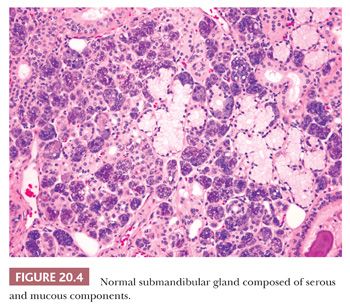
Both the major and minor salivary glands possess acinar and ductal systems. These glands may be of the serous, mucous, or mixed seromucous type. The parotid gland and Ebner glands of the tongue are exclusively of serous type (Fig. 20.2). The palatal salivary glands and those situated at the base and the lateral border of the tongue are predominantly of mucinous type (Fig. 20.3). The submandibular and sublingual salivary glands have both serous and mucinous components (Fig. 20.4), with mucous cells being more prominent in the latter. Mixed salivary glands are also found among the minor salivary glands present in the lip, cheek, and the apex of the tongue, although most minor salivary glands are exclusively mucinous in type.
Saliva is formed by the acinar cells; it is high in amylase content when secreted by the serous glands, and it contains acidic and neutral sialomucin if it is formed by the mucinous acini. The serous acinar cells display intracytoplasmic periodic acid-Schiff (PAS)–positive secretory granules. Large basally located intercellular capillaries characterize these acinar cells. The mucinous acinar cells are arranged around an empty lumen, and they have a well-rounded, basally located nucleus. Myoepithelial cells surround the individual acini and contract during secretion.
The intricate duct system is composed of intercalated, striated, and interlobular ducts. The intercalated duct is quite short, and it is lined by a single layer of cuboidal epithelial cells that are backed by myoepithelial cells on the outside. The striated ducts are lined by a columnar epithelium featuring a luminal brush border. The most important function of these ducts is active saliva secretion. The interlobular ducts form the terminal portion of the duct system. Depending on the circumference of these ducts, multiple stratified layers of epithelial cells are present. Elastic and collagen fibers surround the periphery, facilitating the active transport of the saliva through the system.
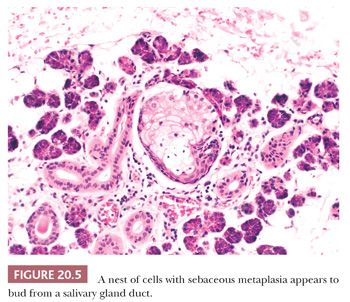
Scattered sebaceous cells arising from intercalated and striated ducts are a normal occurrence in the parotid gland (Fig. 20.5). Oncocytic metaplasia in salivary glands is a common finding with advancing age.
Heterotopic salivary gland tissue has been identified in a wide variety of anatomic sites, including the following: the external and middle ear, the mastoid region, the thyroglossal duct, the thyroid capsule, and even the parathyroid glands. By far, cervical (periparotid) lymph nodes are the most common sites for benign salivary gland inclusions. The embryologic basis for this is discussed earlier. Intramandibular salivary gland tissue may appear on the lingual surface of the bone within surface indentations, most often situated in the angle of the mandible. These heterotopic salivary rests may explain the rare occurrence of salivary tumors arising within the mandibular bone.
NONNEOPLASTIC PROCESSES
SIALOLITHIASIS
Sialoliths are calcified concretions arising in and partially or completely obstructing distal salivary gland ducts. They are primarily composed of calcium salts, along with cellular debris from ductal epithelium and bacterial colonies. About three-fourths of all sialoliths involve the major salivary glands, particularly the submandibular gland, which accounts for almost half of all cases. The tortuous nature of the submandibular gland duct as well as the thicker mucin-rich nature of its secretions has been suggested as reasons for this predilection (1).
The clinical diagnosis of sialolithiasis is usually straightforward. The stones may be expressed clinically and are infrequently submitted to pathology. The importance of sialolithiasis for surgical pathologists lies in the secondary histologic changes that occur proximal to the obstruction when these lesions are surgically resected. Depending on the duration and completeness of the obstruction, there will be chronic inflammatory changes and varying degrees of acinar atrophy. The duct proximal to the obstruction may be dilated and undergo squamous, mucinous, or oncocytic metaplasia, leading to confusion with a cystic mucoepidermoid carcinoma. The history of sialolithiasis, associated inflammatory changes, and lack of a true proliferative process lining the cyst wall should aid in this distinction.
MUCOCELE
Mucoceles (mucous retention and extravasation cysts) arising from the minor salivary glands are common. Favored sites in the submucosa of the oral cavity are the lower lip, the cheeks, the dorsal surface of the tip of the tongue, and the floor of the mouth. In superficial locations, the cyst is well circumscribed, blue-white, and feels like a tiny nodule. Large nodules are distinctly unusual. Viscous fluid erupts if the cyst spontaneously ruptures. Without proper evacuation, repeated rupture and reformation are often the case.
Microscopically, the cyst wall is made up of compressed connective tissue with a denuded epithelial lining and granulation tissue. Chronic inflammatory cells usually infiltrate the surrounding connective tissue. The minor salivary glands in close proximity to the cystic space are also affected by this inflammatory reaction, and the salivary ductules in this location are often ectatic. An incomplete, stratified squamous epithelial lining is rarely observed within the cyst.
RANULA
A ranula is a collection of extravasated mucin from the sublingual glands that presents as a cyst of the floor of the mouth (2). The term is derived from the Latin word for little frog (rana) and is based on its likeness to the throat pouch of frogs. A ranula may be extraoral (also called a cervical or plunging ranula), and it may even extend to the supraclavicular area, upper mediastinum, or skull base (3).
The exact etiology of these mucous cysts is not clearly established. Inflammation, trauma, and mechanical obstruction have all been invoked in the pathogenesis of mucous cysts.
LYMPHOEPITHELIAL CYST
Most lymphoepithelial cysts occur in the region of the parotid gland where they appear to arise from cystic dilation and squamous metaplasia of intranodal salivary gland inclusions. The cysts are usually lined by flattened to stratified squamous epithelium, surrounded by lymphoid stroma (Fig. 20.6). Occasional cysts may be lined by glandular epithelium. The resultant microscopic image may be identical to that of a second branchial cleft cyst, and lymphoepithelial cysts are often misdiagnosed as such. True cysts of the second branchial cleft must be located in the appropriate area of the lateral neck and are often associated with a well-defined sinus tract. In our experience, lymphoepithelial cysts are far more common than branchial cleft cysts.
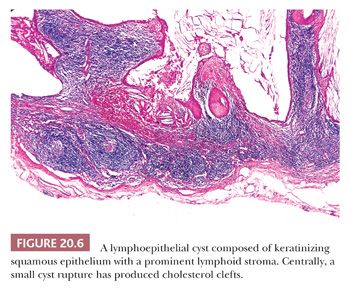
Lymphoepithelial cysts are often encountered in human immunodeficiency virus (HIV)–positive patients in association with the striking lymphoid hyperplasia seen early in the course of that disease. The term cystic lymphoid hyperplasia has been applied to this condition. Typically, these patients have a single cyst or multiple cysts lined by a flattened epithelium and surrounded by a prominent lymphoid infiltrate with germinal centers. The pathogenesis of cystic lymphoid hyperplasia remains unresolved (4). Cyst formation may be caused by obstruction of the ducts by the lymphoid infiltrate (5).
SCLEROSING POLYCYSTIC ADENOSIS
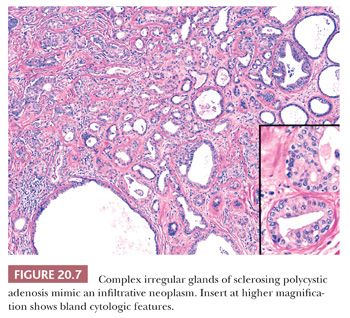
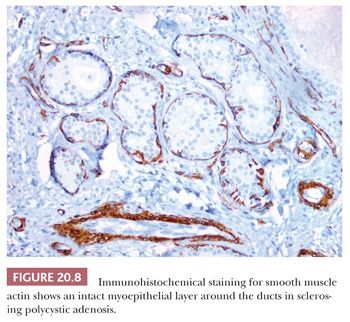
Sclerosing polycystic adenosis is a recently described, mass-forming lesion with a striking predilection for the parotid gland (6,7). Grossly, there is a mass several centimeters in diameter with a sclerotic, variably cystic appearance. Microscopically, there is a mixture of cystic, sclerotic stroma with entrapped glands and inflammatory changes. Apocrine metaplasia is a common finding, as are areas of complex ductal hyperplasia reminiscent of mammary ductal hyperplasia (Fig. 20.7). Intraductal necrosis may be present, and areas of epithelial atypia are commonly seen. The exact nature of this process remains unclear, but one recent study showed clonality in six cases, suggesting that this may represent a neoplastic process (8). Sclerosing polycystic adenosis can easily be confused with adenocarcinoma because of the infiltrative nature of this process. In our experience, the presence of myoepithelial cells around the proliferating ducts and acini aids in this distinction (Fig. 20.8).
MISCELLANEOUS CYSTS
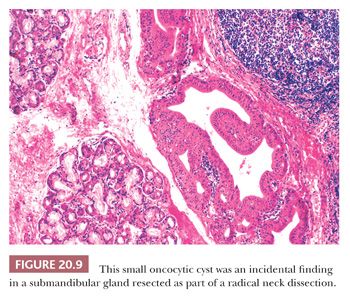
The rarely observed submandibular salivary gland cyst is often lined by flattened epithelium. Parotid duct cysts occur mostly in elderly men. They vary from 1 to 3 cm in size, and they are unilocular. Histologically, they closely resemble the mucous cysts of minor salivary glands (9). The lining of salivary duct cysts can exhibit epithelial proliferation and various types of metaplasia (goblet cell, clear cell, squamous cell, and oncocytic) (Fig. 20.9), or rarely, they can even give rise to a malignancy. The lumen may contain calcospherites or crystalline deposits in addition to retained mucus.
ONCOCYTOSIS/DIFFUSE ONCOCYTIC HYPERPLASIA
Rarely, diffuse oncocytosis may affect the parotid gland with complete oncocytic transformation of the ductal and acinar epithelium. It is always unilateral and occurs almost exclusively in the elderly. Nodular adenomatous (oncocytic) hyperplasia diffusely involves the salivary ducts. The multifocal nodular oncocytic foci may measure up to 1 cm in diameter, and they have a solid trabecular growth pattern. These nodular hyperplastic areas may, on occasion, progress into an oncocytoma. Multifocal oncocytic metaplasia should not be confused with an infiltrative neoplastic process.
NECROTIZING SIALOMETAPLASIA
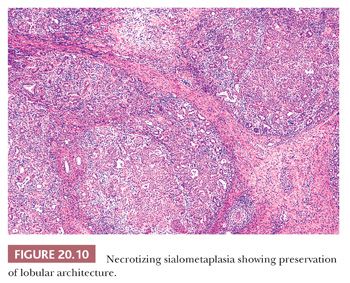
Necrotizing sialometaplasia is an infarctive, inflammatory, metaplastic process that can occur wherever there are seromucinous glands. Typically, there is vascular compromise leading to necrosis of ducts and glandular acini with secondary inflammatory changes and eventual repopulation of the acini by metaplastic squamous epithelium. Often there is a history of prior surgery or trauma. As a spontaneous process, necrotizing sialometaplasia most commonly involves the palatal minor salivary glands. The blood vessels supplying this region are not large and are subject to trauma as a result of compression against the hard palate during mastication. In this location, it is generally encountered in white men with a mean age of 46 years (10).
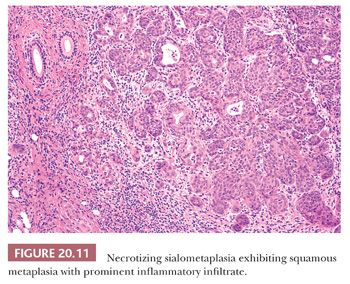
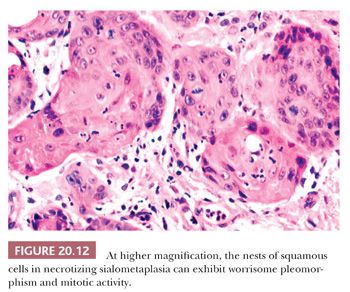
Regardless of location or causation, the microscopic features are stereotypical and related to the age of the lesion. Initially, there is infarction of seromucinous glands with extravasation of luminal contents and a surrounding acute and chronic inflammatory reaction (Fig. 20.10). Within a few days, the necrotic acini become filled with metaplastic squamous cells (Fig. 20.11) that may show prominent mitotic activity and mild cytologic atypia (Fig. 20.12). The squamous cells often coexist with residual noninfarcted mucous cells, creating a pattern that may be confused with mucoepidermoid carcinoma. Because necrotizing sialometaplasia is a process superimposed on preexistent normal salivary acini, it retains a lobular pattern at low magnification. Mucoepidermoid carcinoma, in contrast, does not have a lobular pattern; tends to be infiltrative; is usually not inflamed, especially not with neutrophils; and is composed of varying combinations of basal, intermediate, clear, squamous, mucous, and oncocytic cells.
RADIATION-RELATED CHANGE
Salivary glands, particularly their serous acini, are relatively sensitive to radiation therapy, accounting for the dry mouth frequently encountered after radiation to the head and neck. Submandibular glands included in radical neck dissections will often show marked atrophy with variable degrees of chronic inflammation if there has been a protracted interval (several months) between radiation therapy and resection. More acutely, radiation-related changes are ischemic in nature and identical to the changes of necrotizing sialometaplasia described earlier, with the additional imposition of radiation-related atypia. The latter consists of cells with enlarged, often densely hyperchromatic, “smudged” nuclei and similarly increased amounts of cytoplasm such that the nuclear-to-cytoplasmic ratio remains nearly normal. As with other causes of necrotizing sialometaplasia, confusion with mucoepidermoid carcinoma or even squamous cell carcinoma is possible, particularly on frozen sections, and the superimposed radiation atypia may add to this confusion. Attention to the low magnification pattern with maintenance of the lobular architecture, inflammation, and lack of other cell types (e.g., basal, intermediate, oncocytic) will allow distinction.
ACUTE SIALADENITIS
Salivary gland tissue is relatively resistant to bacterial infection, although acute suppurative sialadenitis may occasionally be encountered, often secondary to a variety of predisposing conditions including trauma, immunosuppression, and duct obstruction caused by sialolithiasis. Responsible agents are usually staphylococcal or streptococcal species, although Gram-negative organisms may also be involved.
Historically, viral sialadenitis has most commonly been caused by mumps virus, although coxsackie species, enteric cytopathic human orphan (ECHO) viruses, Epstein-Barr virus (EBV), and cytomegalovirus, among others, have also been encountered. With the advent of vaccination, mumps parotiditis has become rare in much of the world. Although seldom biopsied, even when more common, the microscopic changes of mumps parotiditis have been described as interstitial edema with dense lymphoplasmacytic infiltrate, swelling and vacuolization of acinar cells, and dilation of ductal lumina as a result of accumulation of secretions and desquamated lining cells (11).
CHRONIC SIALADENITIS
Lymphocytic infiltration of a salivary gland as an isolated finding unassociated with Sjögren syndrome or other autoimmune disease is most likely a result of chronic duct obstruction secondary to sialolithiasis (see earlier discussion). Over time, there is progressive glandular atrophy with varying degrees of fibrosis and chronic inflammation. The latter is much less intense than in autoimmune-related conditions.
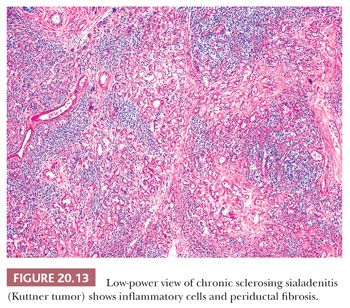
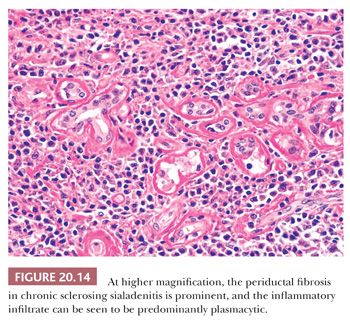
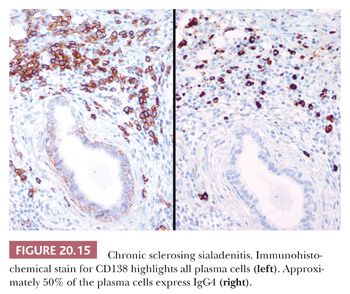
A specific variant of chronic sialadenitis, termed chronic sclerosing sialadenitis or Kuttner tumor, deserves mention (12). This process almost exclusively involves the submandibular gland and usually produces a palpable mass clinically mimicking a neoplasm. Microscopically, the lobular architecture is usually preserved, but a lymphoplasmacytic infiltrate surrounds the ducts and venules with accompanying periductal fibrosis (Figs. 20.13 and 20.14) (13). As the process progresses, the salivary acini proximal to the duct obstruction become atrophic. Reactive lymphoid follicles may be present. The overall appearance is similar to that of sclerosing autoimmune pancreatitis, and this process appears to be a member of the growing family of immunoglobulin (Ig) G4–related sclerosing diseases (13). Immunohistochemistry documents a predominance of T cells (except in germinal centers) with prominent IgG4-positive plasma cells (Fig. 20.15). A marginal zone (mucosa-associated lymphoma tissue [MALT]–type) lymphoma has been reported in the setting of chronic sclerosing sialadenitis (14).
GRANULOMATOUS SIALADENITIS
Virtually any granuloma-producing process can affect the salivary glands. In addition, involvement of intraparotid lymph nodes by a granulomatous process will be clinically indistinguishable from disease of the parotid parenchyma. Sarcoidosis is commonly encountered in these locations, and infections with fungi or mycobacteria are occasionally seen. Cat scratch disease frequently involves the neck region and may produce necrotizing granulomatous inflammation in intraparotid lymph nodes. Approximately 2% to 3% of patients with cat scratch disease will present with parotid gland swelling and pain (15).
SJÖGREN SYNDROME
The San Diego diagnostic criteria for primary Sjögren syndrome are as follows: keratoconjunctivitis sicca, xerostomia, extensive lymphocytic infiltrate on minor salivary gland biopsy, and laboratory evidence of a systemic autoimmune disorder (16). Specific exclusions are preexisting lymphoma, graft-versus-host disease, acquired immunodeficiency syndrome (AIDS), and sarcoidosis. The patients may develop primary biliary cirrhosis, sclerosing cholangitis, pancreatitis, interstitial nephritis, interstitial lymphocytic pneumonitis, and peripheral vasculitis.
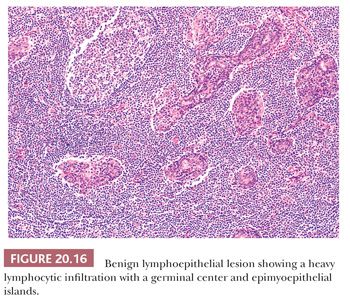
Microscopic examination of the enlarged salivary glands, both major and minor, may reveal a lymphoepithelial sialadenitis, also known as benign lymphoepithelial lesion (BLL). This is characterized by heavy infiltration of the ductal epithelium by lymphocytes of marginal zone or monocytoid B-cell type. The result is the formation of cellular aggregates commonly known as epimyoepithelial islands (Fig. 20.16), although the myoepithelial component of these predominantly epithelial structures may be sparse or absent by immunohistochemistry. BLL must be distinguished from lymphoepithelioma-like undifferentiated carcinoma of the salivary gland, also referred to somewhat ambiguously as a malignant lymphoepithelial lesion. This entity and its distinction from BLL are discussed in more detail later under the section on “Malignant Salivary Gland Neoplasms.”
B-cell clones are detected in more than 50% of BLL, but they do not correlate with morphologic or clinical evidence of lymphoma (17). Patients with BLL have a 44-fold increased risk of developing salivary gland or extrasalivary lymphoma, of which 80% are the marginal zone type (4). Salivary gland lymphomas are discussed later.
For a minor salivary gland biopsy specimen to be diagnostic of Sjögren syndrome, it should contain at least four lobules with at least two foci of lymphocytes per 4 mm2; a focus is defined as a cluster of 50 or more lymphocytes (18). The labial salivary gland biopsy has a sensitivity of 70% to 83%, and it is most useful in patients with partial San Diego criteria, not in patients in whom the index of suspicion is low (19).
The terms Mikulicz disease and Mikulicz syndrome have been used in the past for salivary or lacrimal gland swellings resulting from Sjögren syndrome and various other conditions. These terms have become so ambiguous with regard to the underlying pathologic process that they should no longer be used (20).
GENERAL COMMENTS ON SALIVARY NEOPLASIA
Salivary gland neoplasms occur frequently enough that even pathologists in small practices can expect to encounter them from time to time. Yet, they are infrequent enough that pathologists in large, high-volume medical centers often feel uncomfortable diagnosing them. This discomfort is compounded by the fact that a broad spectrum of tumors arises in this location, there is considerable morphologic overlap between some subtypes, and a substantial minority of tumors do not fit easily into existing diagnostic categories.
Fine-needle aspiration is a useful adjunct for presumptively or, in many instances, definitively diagnosing salivary gland neoplasms. Definitively diagnosing a salivary gland tumor on frozen section may vary from extremely easy to impossible. Experienced ear, nose, and throat (ENT) surgeons are aware of this difficulty, and most use frozen sections only to assess margin status and not to provide a definitive diagnosis. Even when a definitive diagnosis cannot be made on frozen section, some assessment can be provided about the nature of the lesion based on standard histologic features such as the cellularity, pleomorphism, and nature of the margin (encapsulated, pushing, or diffusely invasive). Thus, it should be perfectly acceptable, if frozen section interpretation is requested, to render diagnoses such as “low-grade neoplasm, adenoma versus low-grade carcinoma” or “high-grade carcinoma, await permanents for definitive classification.”
BENIGN EPITHELIAL NEOPLASMS
MIXED TUMOR (“PLEOMORPHIC ADENOMA”)
Although the terms benign mixed tumor or simply mixed tumor are the author’s preferred designations for this entity, the unfortunate designation of pleomorphic adenoma is entrenched in the pathology literature as a classic example of nosologic imprecision. “Pleomorphic” is used by pathologists to indicate nuclear variability, a feature not commonly encountered in this neoplasm. The more appropriate designation would have been polymorphic adenoma to correctly identify the variable cell types seen in these lesions.
Mixed tumors typically present as a painless, persistent swelling, and they can occur at any age. They are most common in adults during the third through fifth decades of life, but they may be found during childhood. Rare bilateral synchronous or metachronous and even familial mixed tumors have been reported (21). Approximately 75% of mixed tumors arise in the parotid gland. The rest occur in the submandibular gland (approximately 5% to 10%) and the minor salivary glands (approximately 10%). The most common minor salivary gland sites are the palate (approximately 60% to 65%), cheek (15%), tongue, and floor of mouth (approximately 10%). Occasionally, mixed tumors arise in intraparotid or periparotid lymph nodes or in the heterotopic salivary gland tissues (22). Approximately 70% of all tumors in the parotid gland are mixed tumors. This compares with 50% of the submandibular salivary gland tumors and 45% of the minor salivary gland tumors. The vast majority (>90%) of mixed tumors arise in the superficial portion of the parotid gland, whereas the rest present in the deep lobe occupying the parapharyngeal space. These tumors of the deep lobe result in oropharyngeal swelling that is often misdiagnosed as unrelated to the parotid gland, and the patients are subjected to intraoral biopsy. It should be noted that tumors histologically similar or identical to salivary gland mixed tumors may occasionally arise from closely related glands of the breast, skin (chondroid syringoma), or ceruminal glands of the external ear.
On gross examination, the lesional tissue is made up of nodules connected by a delicate network of fibrous connective tissue (Figs. 20.17 and 20.18). Multicentric tumor at the time of initial surgery is virtually nonexistent, but conversely, improperly resected tumor will often recur as multicentric nodules, a pattern referred to as satellitosis. This multifocal recurrence is a result of improper initial resection of the tumor by a “shelling out” procedure that leaves tiny pseudopod-like transcapsular processes of the tumor in the adjacent salivary gland. Such recurrences may be quite difficult to eradicate and can be avoided by performing a superficial parotidectomy or, if necessary for large or deep lesions, a more complete resection. After proper resection, the recurrence rate is low (≤2%) (23,24).
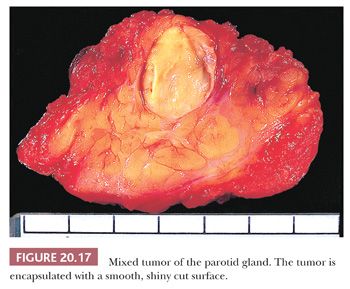
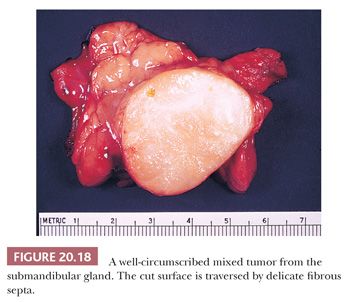
Grossly, mixed tumors appear sharply demarcated or encapsulated. The appearance of their cut surface varies tremendously depending on the epithelial and stromal elements present. Areas of chondroid differentiation are often easily visible and allow virtual diagnosis on gross examination. Small cysts may be present grossly, and some may appear hemorrhagic.
Microscopic examination confirms the capsule seen grossly, but as noted earlier, fingerlike projections of tumor can often be found extending through the capsule for a short distance into the surrounding stroma. The presence and extent of these pseudopods of extracapsular growth do not correlate with recurrence rate, provided that proper resection technique is used and they should not be interpreted as evidence of malignancy. In addition, intravascular tumor deposits may rarely be encountered and appear to be of no clinical significance, provided the tumor is an otherwise conventional mixed tumor (25). Such foci may well represent artifactual transfer secondary to fine-needle aspiration or surgical manipulation.
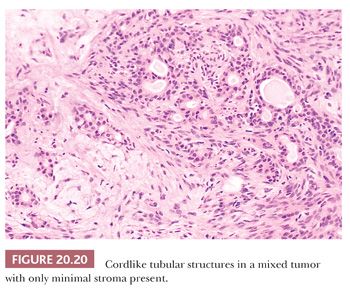
The complex and highly variable histologic patterns seen in mixed tumors are a result of the interplay of the epithelial and stromal elements (Fig. 20.19) (26). The epithelial components may form trabeculae, tubules, ductules, nonkeratinizing squamous cell aggregates, keratinizing squamous cysts, mucous cysts, aggregates of plasmacytoid or spindled myoepithelial cells, scattered sebaceous cells, or sheets of nondescript epithelium. Some ductal structures may display hyperplastic epithelial linings with cribriforming. The ductules typically include both epithelial and myoepithelial components (Fig. 20.20) (27,28).
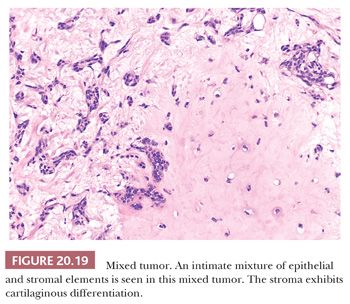
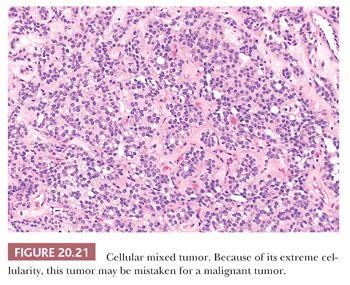
The stromal elements are nearly as variable as the epithelial and may include mucoid, myxoid, fibroblastic, cartilaginous, osseous, and lipogenic elements. The osseous component may be replete with hematopoietic bone marrow. It has been postulated that mixed tumor is a fundamentally epithelial (or myoepithelial) neoplasm with a propensity for multifocal stromal metaplasia. Often, the epithelial and stromal components are so delicately intertwined that their separation from each other is not feasible. Ultrastructural studies of this blurred epithelial–stromal junction have shown “transitional cells” with both epithelial and mesenchymal properties in this region, with modulation to more definite epithelial or stromal elements at respective edges of the junction. The amount of epithelium versus stroma varies tremendously from lesion to lesion. Some mixed tumors are predominantly stromal and often highly cartilaginous. Others are so lacking in stromal elements as to border on the appearance of a monomorphic adenoma. Often, when epithelial elements are predominant, particularly when they assume a nearly sheetlike pattern, the lesion may display high cellularity that leads to concern for malignancy (cellular mixed tumor) (Fig. 20.21). Areas of squamous metaplasia combined with mucinous differentiation may cause the unwary to classify the lesion as a mucoepidermoid carcinoma.
Cytogenetic and molecular studies in mixed tumors have repeatedly shown chromosomal abnormalities involving the long arm of chromosomes of 8 and 12 (29). Translocations in these regions probably promote tumor development or progression through the activation of proto-oncogenes. Loss of heterozygosity has been implicated as an early event in the development of mixed tumor (30).
WARTHIN TUMOR
Warthin tumor, or papillary cystadenoma lymphomatosum, represents 15% of epithelial salivary gland tumors. It is almost exclusively a tumor of the parotid gland and periparotid lymph nodes. Rare reports in the submandibular gland may represent origin in the closely adjacent tail of the parotid gland. Cases reported in unusual locations such as the larynx invariably represent oncocytic cysts with a smattering of chronic inflammation. The reason for the sharp localization to the parotid and periparotid lymph nodes relates to the unique admixture of salivary and lymphoid elements in this region during embryologic development (see earlier “Embryology” section).
Studies suggest a strong association of Warthin tumor with smoking (31,32). This tumor more commonly affects men in their sixth and seventh decades of life, but the incidence in females is increasing, probably because of the increase in smoking in women (33). Bilateral or multifocal tumor involvement is observed in approximately 10% of patients (34). However, because bilateral involvement is so uncommon in salivary neoplasia, Warthin tumor is by far the most common bilateral tumor in this location.
The typical Warthin tumor is well delineated and, on average, is 2 cm in greatest dimension. The cut surface is frequently cystic, with cyst contents that may be mucinous, proteinaceous, or even resemble used (dark) motor oil. Because of the cystic aspects and the occasional presence of friable amorphous material in the central or cystic regions, a branchial cleft cyst or even caseating tuberculous lymphadenitis may be part of the differential diagnosis. Some of the lesions are tiny and are incidental histologic findings at parotid resections for unrelated reasons.
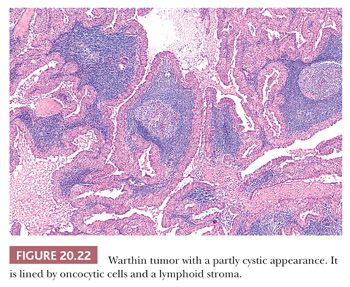
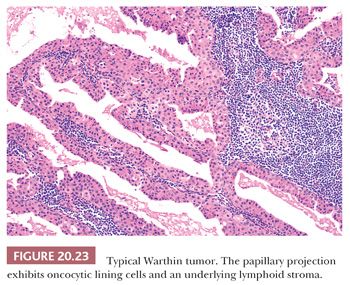
The histologic appearance of Warthin tumor is one of the most distinctive images in all of surgical pathology. An oncocytic epithelial component and a prominent lymphoid stroma with well-developed follicles (Fig. 20.22) blend together in this tumor. The oncocytic epithelial elements have (by definition) strikingly eosinophilic granular cytoplasm as a result of large numbers of mitochondria. This component consists of two or occasionally multiple layers of cells, which often form papillary projections into the cystic lumina (Fig. 20.23). The cystic spaces may contain cast-off epithelial cells, inflammatory cells, crystalline structures, or corpora amylacea. Apocrine or squamous metaplasia of the luminal epithelium may also be present focally. The latter is often associated with areas of infarction and, if prominent, may lead to confusion with squamous or mucoepidermoid carcinoma.
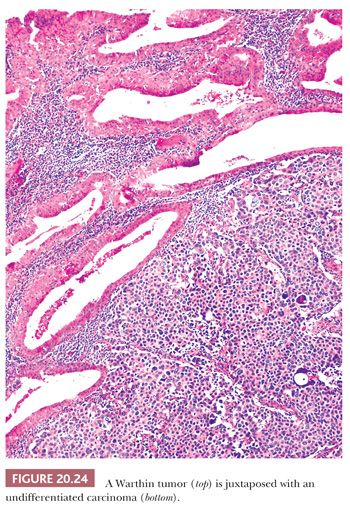
The lymphoid stroma closely resembles a normal lymph node, and it is composed of T and B cells with occasional germinal centers. Immunohistochemical characterization of Ig reveals that approximately 50% of the B lymphocytes contain IgG, with roughly 33% containing IgA. The lymphoid stroma can serve as a metastatic site for carcinoma or melanoma, or it may give rise to malignant lymphomas. Carcinomas can, on rare occasion, develop in the epithelium of a Warthin tumor. Among these, adenocarcinomas, squamous carcinomas, and undifferentiated carcinomas have been reported (Fig. 20.24) (35).
Cytogenetic studies on a small series of cases revealed 6p rearrangements and t(11;19) as being specific for Warthin tumor (36).
BASAL CELL ADENOMA
Approximately 70% of basal cell adenomas occur in the parotid gland, with the remainder arising in the submandibular gland and a variety of sites in the oral cavity, including the upper lip. Although the age range is broad, most patients are in their sixth decade of life or older. Grossly, these are sharply circumscribed, encapsulated masses that resemble hyperplastic lymph nodes. The exception is the uncommon membranous variant, which closely resembles and may be associated with a nearby dermal eccrine cylindroma. These membranous tumors are often multinodular in the parotid and lack well-formed capsules.
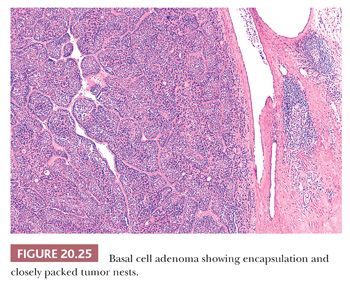
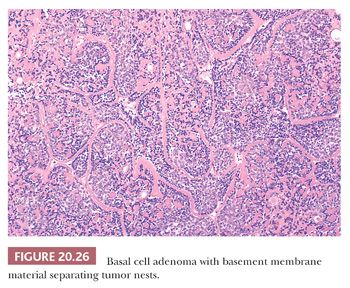
Microscopically, uniform basal cells form, in order of decreasing frequency, solid, trabecular, tubular, or membranous structures (Fig. 20.25). With the exception of approximately half of the membranous subtype lesions, a distinct capsule should be present and is important for distinguishing this entity from basal cell adenocarcinoma and adenoid cystic carcinoma (see “Basal Cell Adenocarcinoma” and “Adenoid Cystic Carcinoma” sections later in this chapter). The tumor cells are clearly separated from the nonmucoid stroma by a well-defined basement membrane (Fig. 20.26). Peripheral cell palisading is characteristic and is best seen in the larger nests of the solid pattern. The cells within the more central portions of the solid nests often have more cytoplasm and show varying degrees of squamoid differentiation ranging from only slightly more prominent eosinophilic cytoplasm to overt keratin pearl formation. The trabecular pattern is often mixed with the solid pattern. It can be thought of as elongated solid nests that vary in size down to thin structures only two cells in thickness. The tubular pattern consists of the trabecular pattern with superimposed ductlike lumina. In many instances, the cells are haphazardly arranged around the openings such that they appear to represent extracellular spaces rather than true glandular lumina. In other foci, however, a distinctly glandlike cell orientation is present. The membranous pattern consists of nests similar to those of the solid pattern but with interposed hyaline stroma within, around, and between the nests of basaloid cells. The multinodular pattern seen grossly in approximately half of these cases will be reflected microscopically as multiple, nonencapsulated nodules (37–39).
Immunohistochemically, the tumor cells are reactive for carcinoembryonic antigen (CEA), epithelial membrane antigen (EMA), cytokeratin, and, focally, S-100 protein (40). Two recent markers noted to be positive in adenoid cystic carcinomas, CD117 and CD43, may also be seen in basal cell adenomas (and basal cell adenocarcinomas), rendering them of little value in this distinction (13).
CANALICULAR ADENOMA
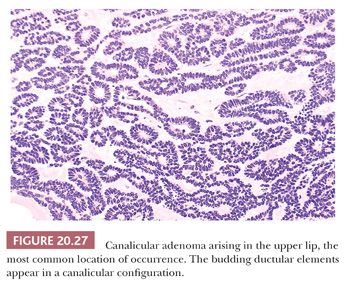
Canalicular adenoma is closely related to basal cell adenoma, and cases with hybrid microscopic features are occasionally encountered. Indeed, the distinction has often been confused in the literature. Although lumping the two together under such terms as basal cell adenoma or even monomorphic adenoma will cause no harm to the patient, there are sufficient clinicopathologic differences to warrant pathologic distinction. Canalicular adenoma is a tumor of adults that approximately three-fourths of the time occurs in the seromucinous glands of the upper lip (37). Grossly, there may be a single encapsulated nodule, a well-demarcated but unencapsulated nodule, or a distinctly multinodular growth pattern. Microscopically, the tumor displays elongated tubules or ducts lined by small cuboidal to columnar cells with scant to moderate amounts of eosinophilic cytoplasm, a morphologic spectrum that clearly encompasses basaloid cells. Scattered mucous or oncocytic cells may be present (37). Papillary structures and psammoma bodies have also been described. The cords of cells form intermittent expansions with “canal-like” lumina that create a beaded pattern (Fig. 20.27). The stroma typically has a loose myxoid quality with prominent vascularity. Canalicular adenomas are positive for cytokeratin and S-100 protein and, focally, for glial fibrillary acidic protein (GFAP) (41).
ONCOCYTOMA
Oncocytic cells are recognized components of a wide variety of salivary gland neoplasms including Warthin tumor, mucoepidermoid carcinoma, acinic cell carcinoma, mixed tumor, and multiple other benign and malignant processes. It is doubtful that any salivary gland neoplasm is completely free of their presence as a scattered component. They are also frequent in nonneoplastic conditions including incidental foci of oncocytic metaplasia and oncocytic cysts of the seromucinous glands, particularly in the false vocal cords.
Monomorphic adenomas composed entirely of oncocytes (oncocytomas) account for approximately 1% of all salivary gland neoplasms (42). Most involve the parotid, followed by the submandibular gland and minor salivary glands from the lower lip, palate, pharynx, and oral mucosa. In addition, approximately 5% arise in salivary rests in periparotid lymph nodes (42). Oncocytomas occur in middle-aged and older adults, with a mean age of 58 years. Approximately 20% of all patients have a history of radiation therapy to the face or upper torso or long-term occupational radiation exposure that occurred 5 or more years before tumor discovery (42). Patients with previous radiation exposure are, on average, 20 years younger at tumor discovery than are those without a documented history. The exact nature of oncocytomas with regard to the neoplastic role of nuclear versus mitochondrial DNA is a fascinating but unresolved topic.
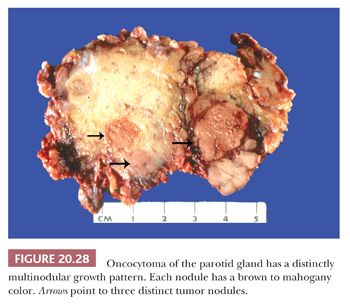
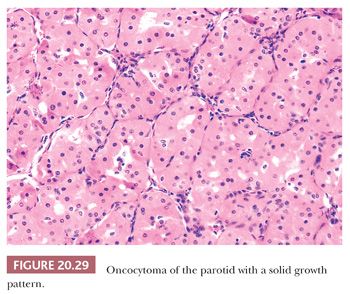
On gross examination, oncocytomas are usually 3 to 4 cm in size, possess a well-defined capsule, and have a light brown to mahogany color. Larger lesions may show a lobular or multinodular pattern (Fig. 20.28). Microscopically, the oncocytic cells are arranged in a solid (Fig. 20.29) or trabecular pattern. Microcyst formation is rarely observed. By definition, oncocytes have ample, distinctly granular acidophilic cytoplasm that ultrastructurally corresponds to large numbers of cytoplasmic mitochondria to the virtual exclusion of other organelles. The nuclei are small and pyknotic. Rarely, oncocytomas present with large polyhedral clear cells in an organoid distribution, separated by a thin fibrovascular stroma. Occasional clear cells may be encountered in oncocytomas, and tumors with a predominantly clear cell component are referred to by the apparent oxymoron of clear cell oncocytoma. The optically clear cell appearance is a result of fixation artifact or intracytoplasmic glycogen, in addition to the numerous mitochondria (43,44).
Histochemical studies demonstrate phosphotungstic acid hematoxylin (PTAH)–positive cytoplasmic staining. PAS staining before and after diastase digestion demonstrates granular cytoplasmic positivity that represents the numerous, tightly packed mitochondria. Antimitochondrial antibodies will show strong positivity in oncocytomas, but this stain is seldom needed for diagnosis. By electron microscopy, the mitochondria have elongated cristae and a partial lamellar internal structure (45). The nuclei of the oncocytes are irregular, and they contain inclusions and glycogen granules.
The clear-cut separation of oncocytic adenomatous (nodular) hyperplasia of the parotid gland from a multinodular oncocytoma (a true neoplasm) is not always possible because the two entities overlap histologically (46–48). The multinodular feature may impart an undeserved sinister, malignant appearance to the lesion. In our experience, these nodules are separated by normal salivary gland, whereas the rare oncocytic carcinoma has multinodularity with intervening fibrotic or desmoplastic stroma (see “Oncocytic Carcinoma” section later in this chapter).
Oncocytomas only rarely recur, and when they do, recurrences are often multiple and bilateral, suggesting that they, in reality, represent new lesions arising in multifocal nodular oncocytic hyperplasia. The differential diagnosis of oncocytoma includes the extremely rare oncocytic carcinoma discussed later and a wide variety of salivary gland lesions that can have at least a minor oncocytic component, as well as metastatic neoplasms such as renal cell carcinoma. In our experience, acinic cell carcinomas and mucoepidermoid carcinomas with prominent oncocytes, also discussed later, present the greatest diagnostic challenges. In both cases, careful search for other diagnostic cell types will allow distinction. With the exception of focal clear cell change, oncocytomas should have a monomorphous appearance. The presence of adjacent oncocytic foci (oncocytosis, nodular oncocytic metaplasia) adds support for a primary oncocytoma as opposed to a metastatic lesion.
MYOEPITHELIOMA
Cells with myoepithelial differentiation figure prominently in a wide variety of salivary gland neoplasms, and immunohistochemical markers for this differentiation (e.g., p63, smooth muscle actin, calponin, CD10, etc.) can be valuable in differentiating tumors with myoepithelial cells from those without. Although recognition of myoepithelial differentiation has significant diagnostic practicality, this feature tells us nothing about the origin of the neoplastic cells, and complex lineage charts for salivary neoplasms based on the flawed assumption that differentiation equates with origin are of no practical value.
Benign salivary gland tumors composed entirely of myoepithelial cells (i.e., myoepitheliomas) are rare encapsulated or sharply demarcated neoplasms. Approximately 50% of such lesions involve the parotid gland, 40% arise in minor salivary glands, and a few affect the submandibular salivary gland (49–52). The vast majority of the intraoral minor salivary gland lesions are of palatal origin. The incidence in males and females is equal. The lesions present as asymptomatic, slowly growing masses in patients who have an average age of 40 years (age range, 6 to 81 years).
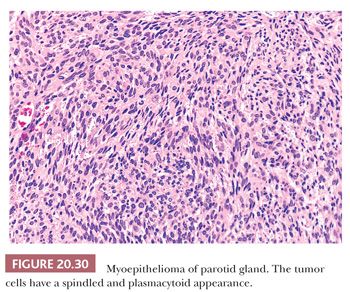
Several distinct cell types are encountered in these tumors, including spindle cells, plasmacytoid (hyaline) cells, and, possibly, clear cells. Each is associated with a different group of differential diagnoses. Mixtures of cell types in a single tumor are common. The spindle cell tumors usually exhibit cellular growth with little or no ground substance (Fig. 20.30). The neoplastic cells form storiform, swirling, herringbone, or fascicular patterns. The differential diagnosis accordingly includes benign fibrous histiocytoma, leiomyoma, benign fibroblastic lesions, and benign peripheral nerve sheath tumor.
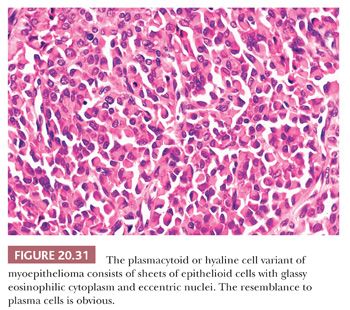
In the plasmacytoid or hyaline cell variant, the tumor cells are distributed in nests and groups that are separated by an abundant myxoid stroma containing hyaluronic acid and lacking mucin (Fig. 20.31). The individual cells are polygonal to round and typically have prominent glassy (nongranular) eosinophilic cytoplasm, often with eccentrically placed nuclei. The resultant appearance may mimic a plasma cell proliferation or any of a variety of neoplasms noted to have a “rhabdoid” phenotype, including malignant melanoma. The glassy cytoplasm corresponds ultrastructurally to aggregates of intermediate filaments. In our experience, these are predominantly vimentin rather than actin, and immunohistochemical evidence of myoepithelial differentiation may be difficult or impossible in some cases (53).
Myoepithelial cells can have completely clear cytoplasm and such cells are most commonly encountered in epithelial-myoepithelial carcinoma. However, the existence of a pure clear cell myoepithelioma is questionable, and we have not encountered, or at least recognized, an example. Diagnosis would require an encapsulated or sharply demarcated, benign-appearing (and acting) neoplasm with immunohistochemical or ultrastructural demonstration of myoepithelial differentiation. Entities to be excluded in the differential diagnosis would include the extremely rare clear cell adenoma, a lesion of glycogen-filled epithelial cells lacking myoepithelial differentiation; the hyalinizing clear cell carcinoma, also lacking myoepithelial differentiation; and an epithelial-myoepithelial carcinoma with prominent clear cells.
Myoepitheliomas are closely related to salivary gland mixed tumors. The latter tumors may contain all of the cell types seen in myoepitheliomas, and we have encountered multiple cases in which the distinction of myoepithelioma from mixed tumor rested solely on the presence of a small focus of stromal (often cartilaginous) differentiation. This distinction has no clinical importance.
SEBACEOUS ADENOMA AND LYMPHADENOMA
These tumors are rare, and they occur almost exclusively in the parotid gland and periparotid lymph nodes (54–56). The localization of sebaceous adenoma to this location may relate to the predilection of the parotid to show sebaceous metaplasia, a rare finding in other salivary tissues (57). In addition to this factor, the sharp localization of lymphadenoma to this region, as with Warthin tumor and lymphoepithelial cyst, is a result of the unique intermingling of salivary and lymphoid tissue in the parotid and periparotid lymph nodes. The average age of patients with sebaceous neoplasms is approximately 60 years, but they can occur from the second through the ninth decades of life. The tumors vary in size from 1 to 3 cm in diameter, and they are well encapsulated or at least sharply circumscribed. The clinical behavior is entirely benign, and local recurrence is virtually unheard of. There have been isolated reports of carcinomas arising in sebaceous lymphadenomas, and these have been referred to as sebaceous lymphadenocarcinomas. Examples that we have encountered resembled basal cell adenocarcinoma, described later.
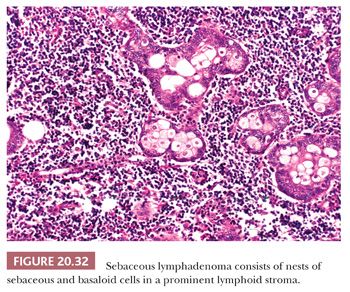
Sebaceous adenomas and lymphadenomas are usually solid, but on occasion, they may have a cystic quality. The well-differentiated sebaceous cells show minimal, if any, cellular pleomorphism, unlike sebaceous carcinomas discussed later. They have practically no tendency for local invasion. In sebaceous lymphadenoma, in addition to the sebaceous cell nests, a background of lymphoid follicles and lymphocytes is seen (Fig. 20.32). In our experience, the large majority of cells in sebaceous adenomas have distinctly sebaceous differentiation with prominent foamy cytoplasm. In contrast, sebaceous lymphadenomas are typically composed primarily of more nonspecific ductal or basaloid epithelial cells with only scattered small nests of sebaceous cells. In some cases, the sebaceous nests may be quite rare.
Although some sebaceous lymphadenomas are clearly within the parotid gland and probably arise from intraparotid lymph nodes, others are distinct from the parotid, although immediately adjacent to it, and appear to arise from salivary inclusions within periparotid nodes. The latter scenario leads to the potential to misdiagnose the lesion as a metastatic carcinoma. We have encountered several sebaceous lymphadenomas in periparotid lymph nodes submitted for consultation with a diagnosis of metastatic mucoepidermoid carcinoma, with the sebaceous cells being misinterpreted as mucinous.
Lesions similar to sebaceous lymphadenoma except completely lacking sebaceous nests have been referred to as simply lymphadenomas. These may represent basal cell adenomas involving lymphoid tissue. Fortunately, these lesions are rare because they can be difficult to distinguish from BLL when present within the parotid gland and from a metastatic carcinoma when present in a periparotid lymph node. In the former situation, the sharp circumscription or encapsulation of the lesion with surrounding normal salivary gland should allow distinction from the more diffuse involvement of BLL. In the latter situation, the small size of the cell nests, the paucity of mitotic figures, and the lack of pleomorphism should suggest the diagnosis.
DUCTAL PAPILLOMAS
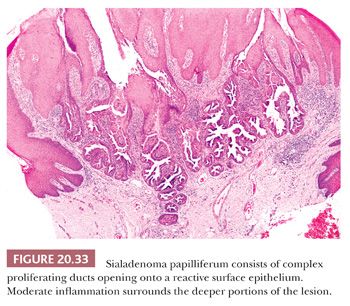
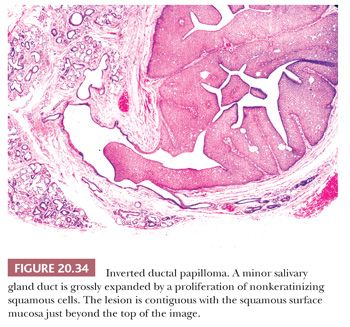
The term ductal papilloma currently encompasses three benign and probably related tumors predominantly arising in the minor salivary glands: sialadenoma papilliferum, inverted ductal papilloma, and intraductal papilloma (58). These occur in the sixth to eighth decades of life and are slightly more common in men. Sialadenoma papilliferum has an exophytic surface and is usually mistaken clinically for a squamous papilloma. It shows a strong predilection for the minor salivary glands of the palate. Microscopically, it has a cauliflower-like surface that merges at its base with underlying often cystically dilated minor salivary gland ducts (Fig. 20.33). The lining of the complex papillary structures varies from columnar glandular epithelium to keratinizing squamous cells. A mild to intense mixed inflammatory infiltrate is located in the fibrovascular cores of the papilla. The appearance greatly resembles syringocystadenoma papilliferum of the skin adnexae. Inverted ductal papilloma has an endophytic growth that is contiguous with the surface epithelium but does not extend above it (Fig. 20.34). It consists of a mixture of squamous and basaloid cells, often in a ribbonlike growth pattern, that project into a cystic space, representing a dilated salivary gland duct. Intraductal papilloma is characterized by complex branching papillary fronds within a cystic expansion of a salivary gland duct. The epithelium is predominantly columnar or cuboidal with scattered mucous cells (59).
INTERCALATED DUCT LESION/ADENOMA
Microscopic proliferations of intercalated ducts are occasionally encountered in salivary gland, particularly parotid gland tissue, removed for other reasons (60). Terms applied to these proliferations have included hyperplasia of intercalated ducts and adenomatous ductal proliferation. Some have noted an association with epithelial-myoepithelial carcinoma and basal cell adenoma and proposed this process as a precursor lesion, but the proliferations may also be encountered as isolated findings in salivary tissue removed for any reason.
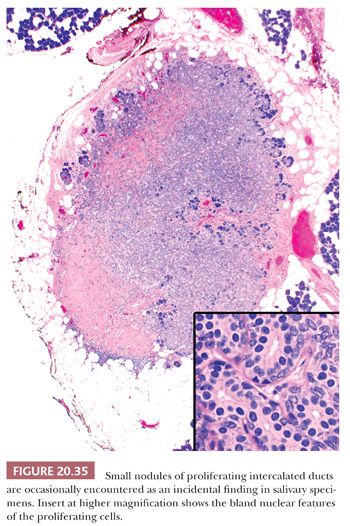
Weinreb and colleagues (60) proposed dividing these lesions, somewhat arbitrarily, into two groups. Intercalated duct hyperplasia consists of small solitary or multifocal nonencapsulated proliferations that blended with the adjacent salivary acinar tissue (60). Intercalated duct adenomas were discrete, rounded, partially or completely encapsulated nodules of proliferating intercalated ducts with little or no intervening stroma. Hybrid lesions with features of both were also encountered.
The epithelial cells in both lesions are typical of those of normal intercalated ducts and consist of small cuboidal cells with uniform nuclei and modest amounts of eosinophilic cytoplasm (Fig. 20.35). The proliferating cells, like their normal counterparts, are positive for CK7 and lysozyme, with focal positivity for estrogen receptor. Myoepithelial markers including calponin and CK14 demonstrate a thin layer of myoepithelial cells around all ducts (60).
MALIGNANT SALIVARY GLAND NEOPLASMS
ADENOID CYSTIC CARCINOMA
Adenoid cystic carcinoma accounts for approximately 10% of all salivary gland tumors. It is the most common malignant tumor of the submandibular and minor salivary glands (61). The ages of patients range from 20 to 84 years, with a median age of 52 years (62). Among the 264 adenoid cystic carcinomas studied at Memorial Sloan Kettering Cancer Center, 45 (17%) arose in the parotid, and 41 (15.5%) involved the submandibular gland (62). The rest were in minor salivary gland sites. Adenoid cystic carcinomas within the mandibular bone are rare occurrences (63,64).
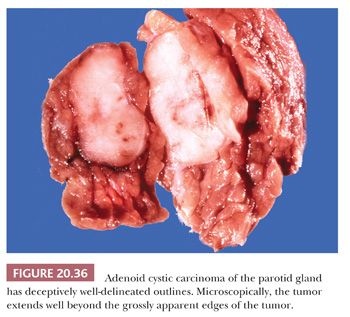
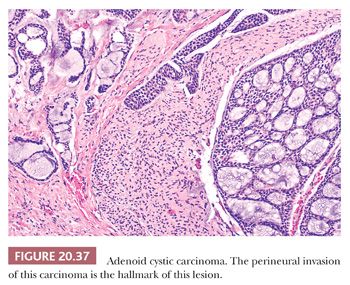
The tumor grossly appears solid and circumscribed, but microscopically, it extends well beyond the grossly visible and palpable limits of the lesion (Fig. 20.36). This infiltrative capacity is a hallmark of this salivary gland carcinoma. Spreading along nerve sheaths with associated severe pain is often noted (Fig. 20.37). Facial nerve paralysis may be the first presenting symptom, appearing before the lesion becomes otherwise obvious. The usual clinical course is lengthy, often spanning decades, with multiple late local recurrences after operative intervention (62). Occasional patients have a rapid demise. Although adenoid cystic carcinomas may involve lymph nodes by direct extension, particularly when they arise in the submandibular gland, embolic lymph node metastases are rare. A review of a large registry of these tumors at our institution revealed only two examples of small, solitary metastases to regional nodes. Clearly, radical neck dissection should not be a component of normal treatment. In contrast, hematogenous tumor spread, often to the lungs, is quite characteristic. Lung metastases, although clearly identifiable on routine chest radiographs, may remain stable for years.
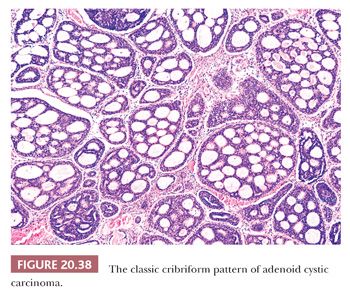
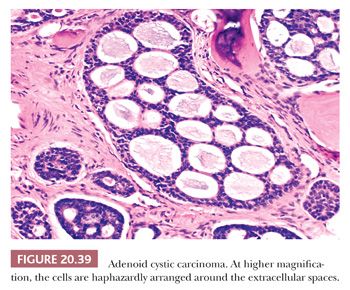
Three histologic patterns are typically encountered in these tumors: cribriform, tubular, and solid (65–67). Multiple patterns are often present in a single tumor. The cribriform, or classic, pattern consists of basaloid epithelial cells forming sharply demarcated nests containing multiple extracellular spaces (Fig. 20.38). The spaces contain PAS-positive connective tissue mucin or eosinophilic hyaline-like material. The cells surrounding the spaces are haphazardly oriented or stretched out around them, indicating that these are extracellular spaces and not true glandular lumina with radially oriented, polarized surrounding cells (Fig. 20.39). This can be confirmed ultrastructurally. This pattern is encountered, at least focally, in the majority of adenoid cystic carcinomas.
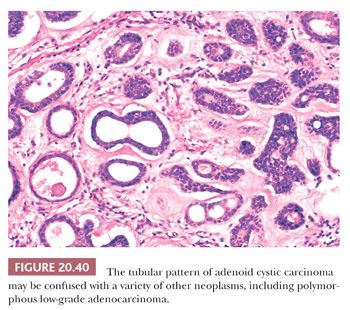
The tubular pattern is characterized by smaller ductlike arrays of basaloid epithelial cells surrounding a single central lumenlike space (Fig. 20.40). The surrounding stroma is typically fibrous or hyalinized. The lumenlike space may contain PAS-positive mucin or hyaline material or appear nearly empty. Close examination shows that the cells surrounding the spaces are haphazardly oriented, as in the cribriform pattern, and these again represent extracellular spaces rather than true glandular lumina. Approximately 20% to 30% of adenoid cystic carcinomas contain this pattern.
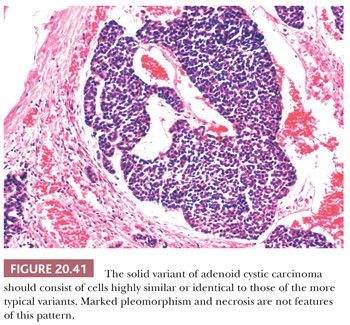
The solid pattern is the least frequently encountered (Fig. 20.41) and is most often mixed with one of the other variants. It consists of solid nests of basaloid cells, often in a hyalinized stroma. The cells should be cytologically similar or identical to those seen in the cribriform and tubular patterns. Nuclei should be relatively uniform with dark, coarse nuclear chromatin. Mitotic rate may be slightly increased in this variant, but marked nuclear pleomorphism, enlarged nuclei with prominent nucleoli, or substantial areas of necrosis should lead to alternate diagnostic considerations.
Some studies have emphasized that histologic pattern correlates with prognosis (tubular = best, cribriform = intermediate, and solid = worst) (66,67). However, two large studies and our own experience with a substantial number of these lesions failed to duplicate these prognostic assertions (61,62). This discrepancy may relate to diagnostic misadventures at each end of the morphologic spectrum. Polymorphous low-grade adenocarcinoma (PLGA) is an indolent neoplasm that often has a distinctly tubular growth pattern. Misinterpretation as a tubular adenoid cystic carcinoma would improve the apparent prognosis of that variant. Conversely, the solid pattern of adenoid cystic carcinoma is relatively nonspecific, and similar or identical patterns can be seen in basaloid squamous cell carcinomas, sinonasal undifferentiated carcinomas, and high-grade neuroendocrine carcinomas. These lesions are highly aggressive, and their misinterpretation as the solid variant would greatly decrease the apparent prognosis of the latter subtype.
In reality, predicting the prognosis of these tumors is difficult or impossible on an individual basis. Statistically important prognostic factors include the size and site of the primary tumor and the presence of metastases. Of these, the clinical stage is not surprisingly the most reliable guide (68). Proliferative activity measured by MIB-1 (Ki-67 antigen) has been found to be higher in those cases in which treatment failed, and it may provide additional information on the short-term prognosis (69). The p53 oncoprotein has been reported to be an adverse prognostic marker in adenoid cystic carcinoma (70). In a limited study, it was detected more frequently in recurrent tumors than in primary ones, which may be a reflection of its involvement in the later stages of tumor progression (71). As an important caveat, p53 mutations are also seen in “dedifferentiated” adenoid cystic carcinomas and these highly aggressive tumors are distinct from pure adenoid cystic carcinoma and should not be lumped with them. CD117 expression has been demonstrated in the majority of adenoid cystic carcinomas (72), but the mutation associated with sensitivity to imatinib mesylate does not appear to be present in these tumors. Approximately 70% of adenoid cystic carcinomas exhibit a t(6;9) translocation resulting in a MYB-NFIB gene fusion (73).
The prototypical cribriform pattern is easy to diagnose. As mentioned earlier, the tubular pattern may be confused with PLGA. Attention to cytologic detail usually will allow distinction. PLGA is a tumor of larger cells with more prominent eosinophilic cytoplasm than the distinctly basaloid cells of adenoid cystic carcinoma. Many of the tubules in PLGA have cells oriented radially around them, indicating true ductal or luminal differentiation. As its name implies, PLGA is noted for its histologic variability (and cytologic uniformity). Many of the patterns present are well beyond the spectrum of adenoid cystic carcinoma. Immunohistochemically, adenoid cystic carcinomas typically express CD117 and stain weakly for S-100 protein. PLGA shows a reverse pattern, with weak staining for CD117 and strong positivity for S-100 protein. Recently, CD43 has been touted as a marker for adenoid cystic carcinoma, but this antigen requires more complete study in salivary neoplasia (13).
Fortunately, the solid variant of adenoid cystic carcinoma is uncommon and rarely occurs in pure form. If the cells in the solid nests vary more than minimally from the cells of the more typical cribriform pattern, if there is obvious necrosis, if the mitotic rate is extremely high, or if abrupt squamous differentiation is noted, strong consideration should be given to other basaloid neoplasms. In our experience, squamous differentiation in particular is extremely rare in salivary adenoid cystic carcinomas and, conversely, is a classic feature of basaloid squamous cell carcinoma. This distinction is important because the latter tumor frequently metastasizes to regional lymph nodes, and a formal lymph node dissection is often warranted. High-grade “solid” carcinoma may arise through “dedifferentiation” in recurrent adenoid cystic carcinoma, and these high-grade solid nests should be clearly distinguished from the solid variant of conventional adenoid cystic carcinoma.
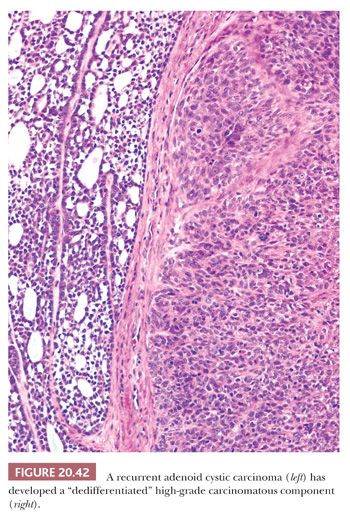
As alluded to earlier, adenoid cystic carcinomas may rarely undergo dedifferentiation to a high-grade glandular or undifferentiated neoplasm (Fig. 20.42) (74). This change usually arises after multiple recurrences of a typical adenoid cystic carcinoma. It should be recognized and distinguished from the usual patterns of adenoid cystic carcinoma, as it heralds a different much more aggressive clinical course. Strong, diffuse immunohistochemical staining for p53, presumably representing p53 mutation, typically accompanies this transformation (74).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


