CHAPTER 14
Phenobarbital and Primidone
DENISE H. RHONEY, PharmD, FCCP, FCCM, FNCS
KAREN J. McALLEN, PharmD
XI LIU-DeRYKE, PharmD
DENNIS PARKER JR., PharmD, BCPS
PHENOBARBITAL
Phenobarbital is a barbituric acid derivative with hypnotic activity and central nervous system (CNS) depressant effects. Phenobarbital is one of the oldest anticonvulsant agents still used in clinical practice. It is FDA-approved for short-term sedation/hypnosis and treatment of generalized or partial onset seizures and provides an alternative to treat refractory status epilepticus. It is less commonly used as the first-line anticonvulsant due to disadvantages such as cognitive impairment, respiratory depression, sedation, and significant drug interactions. The anticonvulsant activity of phenobarbital is thought to be due to its effect on postsynaptic GABA receptors, which increases seizure threshold but the full mechanism is not completely understood.1 Phenobarbital has also been used off-label to treat alcohol withdrawal, neonatal seizures, febrile seizures, neonate hyperbilirubinemia, and adults with congenital nonhemolytic unconjugated hyperbilirubinemia or chronic cholestasis.
DOSING
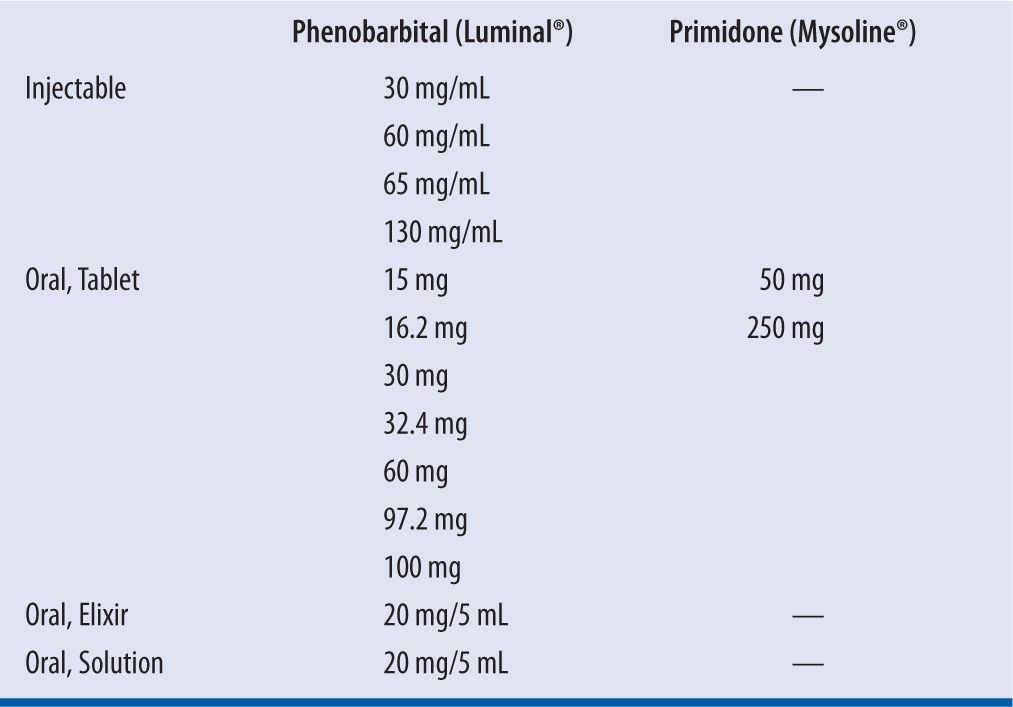
Phenobarbital is available in an injectable formulation, which can be given intravenously or intramuscularly, and oral formulation (tablets and solution) (see Table 14-1). In adult patients, the recommended dose for sedation and hypnosis is 30–100 mg/day and titrated upward slowly to 400 mg/day. For epilepsy management, dosing is usually weight-based where an adult maintenance dose of 2 mg/kg/day would result in a predicted steady-state concentration of 20 mg/L. A case report suggests that dosing should be with total body weight.2 If immediate therapeutic concentrations are required a loading dose of 15–20 mg/kg can be administered intravenously or orally in three divided doses every 2 to 3 hours. When phenobarbital is administered intravenously, a rate of no more than 50 mg/min is recommended to avoid toxicity with the propylene glycol diluent.3 Maintenance dosing in children (1–15 years old) is usually 3–5 mg/kg/day and for neonates (<2 weeks of age) is 2–4 mg/kg/day.4 Because phenobarbital has a long half-life, it can be dosed once daily; however, initially excessive sedation may limit the ability to use once-daily dosing. Excessive sedation can be minimized by gradually increasing the dose using the following schedule: Start with 25 percent of the final daily dose each evening for 5–7 days and if tolerated the dose can then be increased to 50 percent of the final recommended dose for the next 5–7 days and then 75 percent of the final recommended dose for the next 5–7 days. The titration to the final dose should be completed by the fourth week of therapy. Steady-state serum concentrations should be checked about 3–4 weeks after achieving the total maintenance dose. A serum phenobarbital concentration targeting between 10–40 mg/L is considered within optimum range for therapeutic drug monitoring (TDM).5
| TABLE 14-1 | Dosage Formulations and Strengths |
ADVERSE EVENTS
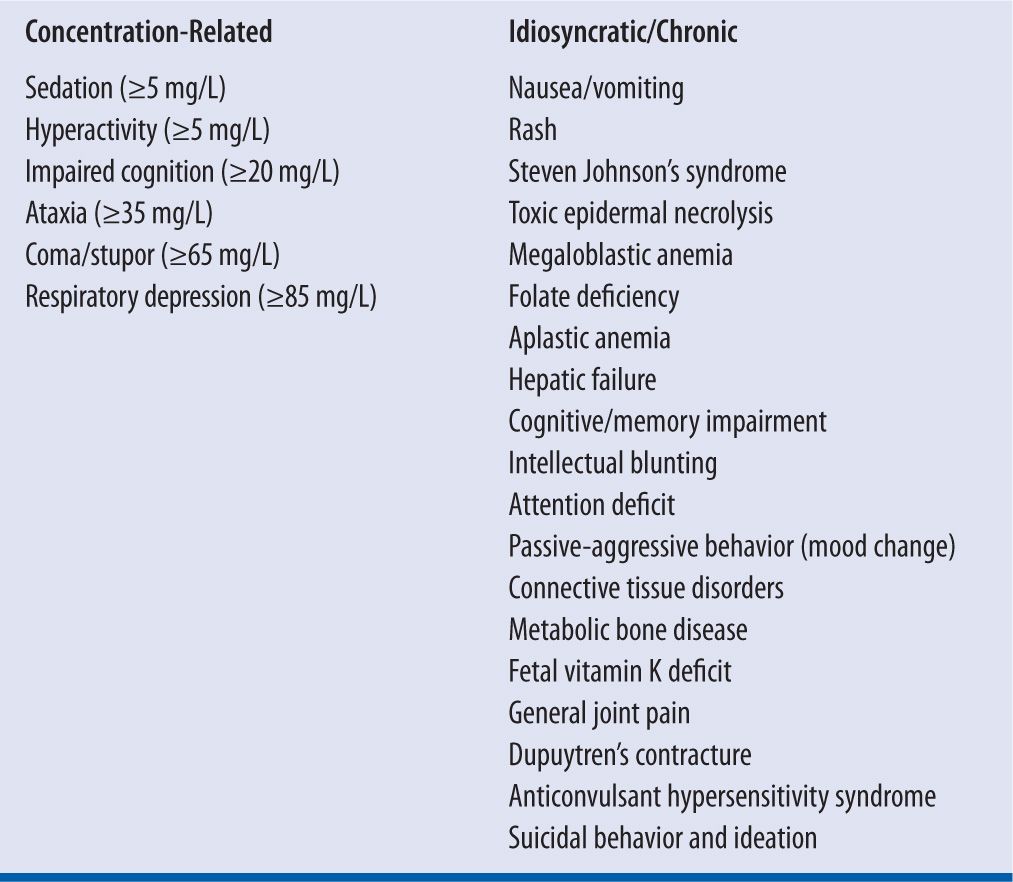
Adverse effects are listed in Table 14-2. The most common adverse effect of phenobarbital is related to CNS depression, which includes sedation, ataxia, fatigue, and confusion. However, in children and elderly a paradoxical effect may be seen producing insomnia and hyperkinetic activity.6 Chronically, phenobarbital is associated with impairment of cognition, which is a major limitation for its use, especially a problem in children where memory impairment and compromised work/school performance may develop independent of the sedation properties. The intelligence quotient of children receiving phenobarbital was 8.4 points lower than other children, and these scores remained 5.2 points lower after the phenobarbital was discontinued.7 Phenobarbital affects calcium and vitamin D metabolism leading to hypocalcemia, osteomalcia, osteopenia, osteoporosis, and fractures.8-10 Although they are rare, dermatologic effects ranging from rash to Stevens-Johnson syndrome and thrombocytopenia/agranulocytosis have been reported with Phenobarbital and primidone. The rash is usually mild maculopapular, morbilliform, or scarlatiniform that disappears upon discontinuation of the drug. Hypersensitivity syndrome is rare (1 per 3,000 exposures, but fatal in 10% of cases) and can start with a severe rash that progresses into a purpuric or exfoliative dermatitis. Other signs of a hypersensitivity syndrome are lymphadenopathy, hepatitis, nephritis, carditis, and eosinophilia. This hypersensitivity is cross-reactive with other aromatic anticonvulsant agents like carbamazepine and phenytoin. Intravenous phenobarbital can also cause bradycardia, hypotension, and syncope. Phenobarbital is classified in Pregnancy Category D due to its propensity to cause serious fetal defects and neonatal hemorrhage and is not recommended during breast-feeding because it is excreted in the breast milk.
| TABLE 14-2 | Adverse Effects Associated with Phenobarbital and Primidone |
PRIMIDONE
Primidone is a 2-deoxy analogue of phenobarbital and was first synthesized in 1949. In the United States, primidone is approved for adjunctive and monotherapy use in generalized tonic-clonic seizures, simple partial seizures, complex partial seizures, and myoclonic seizures. Primidone is also considered to be a first-line therapy for essential tremor along with propranolol. Primidone rapidly converts to phenobarbital and phenylethylmalonamide (PEMA), which both possess anticonvulsant activity. The main anticonvulsant effects are thought to be derived from phenobarbital, but unchanged primidone also has anticonvulsant activity. It is thought that PEMA does not play a large role in the anticonvulsant effects based on its potency of only one-twentieth that of primidone, but PEMA does potentiate the activity of phenobarbital in experimental models.11 The exact mechanism of primidone is unknown but believed to work via interactions with voltage-gated sodium channels that inhibit high-frequency repetitive firing of action potentials.12
DOSING
Primidone is only available in an oral formulation. Patients 8 years of age and older may be started on primidone according to the following regimen:
Days 1 to 3: 100 to 125 mg at bedtime
Days 4 to 6: 100 to 125 mg bid
Days 7 to 9: 100 to 125 mg tid
Day 10 to maintenance: 250 mg tid
For most adults and children 8 years of age and over, the usual maintenance dosage is 250 mg tid or qid. If required, the dose may be increased but daily doses should not exceed 500 mg qid.
For children under 8 years of age, the following regimen of primidone may be used:
Days 1 to 3: 50 mg at bedtime
Days 4 to 6: 50 mg bid
Days 7 to 9: 100 mg bid
Day 10 to maintenance: 125 mg tid to 250 mg tid
For children under 8 years of age, the usual maintenance dosage is 125–250 mg three times daily or, 10–25 mg/kg/day in divided doses.
If serum concentration monitoring is required, then phenobarbital concentrations should be monitored instead of primidone.
ADVERSE EVENTS
The adverse effects of primidone are similar to phenobarbital, so please refer to the previous section. Upon initiation of therapy, primidone may be less well tolerated compared to phenobarbital due to intense dizziness, nausea, and sedation that may be related to high initial concentrations of the parent drug.
BIOAVAILABILITY
PHENOBARBITAL
All routes of administration (oral, intramuscular, or intravenous) of phenobarbital are readily and completely absorbed with an oral bioavailability ranging between 90 percent and 100 percent in adults and children.13,14 In neonates, oral absorption may be variable and appears to be delayed and incomplete.15 The injectable formulation of phenobarbital can be given rectally and is readily absorbed with >90 percent bioavailability.16 The peak concentration following intravenous injection of phenobarbital is reached at 15–30 minutes post-administration, and >4 hours when given orally.13,17 Phenobarbital is administered as the sodium salt, which is 91 percent phenobarbital acid (S = 0.91).
PRIMIDONE
Primidone is absorbed orally with a bioavailability approaching 100 percent. The bioavailability has significant intraindividual variability related to the different formulations/manufacturers. The time to peak concentrations in adults is 2.7 to 3.2 hours and in children 4 to 6 hours.18
VOLUME OF DISTRIBUTION
PHENOBARBITAL
Phenobarbital has a lower lipophilicity compared to other barbiturates, resulting in slower brain penetration, so an immediate onset of action is not expected. A few hours after administration, phenobarbital is found in near-equal concentrations in all tissues of the body. Phenobarbital is well distributed in the brain, and the concentration is in equilibrium with brain, cerebrospinal fluid, and free plasma drug.19 Following intravenous administration, phenobarbital has a two-phase distribution with the early distribution to highly vascular organs including the kidney, liver, muscle, and heart (but not brain) and during the late phase a fairly equal distribution between all tissues except for fat.20,21 Approximately 12–60 minutes are required for maximal entry into the adult brain with the rate of entry related to age; in younger patients, a more rapid entry may be observed.22 The plasma volume of distribution of phenobarbital approximates 0.6–1.0 L/kg, depending on the age of the patient. Children and adults have a smaller volume of distribution (~0.7 L/kg) compared to neonates (~0.9 L/kg). Phenobarbital has minimal protein binding capacity of ~45 percent compared to other AEDs such as phenytoin and valproic acid (>90% protein-bound).1,17,23,24 The drug is primarily bound to plasma albumin.
PRIMIDONE
The volume of distribution of the parent drug (primidone) approximates 0.6 L/kg. Protein binding of primidone the parent compound is approximately 25 percent. The distribution of primidone through tissues is similar to phenobarbital. Maximum brain concentrations occur 2 hours after drug administration. Primidone is also distributed to breast milk at concentrations of 40–96 percent of the maternal serum concentrations. Saliva concentrations seem to correlate with unbound plasma and cerebrospinal fluid concentrations.18
METABOLISM AND CLEARANCE/HALF-LIFE
PHENOBARBITAL
Phenobarbital is primarily metabolized through the liver via cytochrome P-450 and NADPH-cytochrome C reductase with <20 percent eliminated unchanged through the kidney.25 The metabolite, p-hydroxyphenobarbital, is pharmacologically inactive and excreted in the urine in conjugated form. The total body clearance of phenobarbital in adults is 0.004 L/kg/hr (= 0.1 L/kg/day).13,24 Therefore, it would be expected that for every 1 mg/kg/day of phenobarbital sodium administered a steady-state concentration of 10 mg/L would be achieved based upon the following equation:
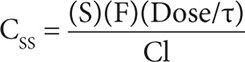
Phenobarbital has a long elimination half-life ranging from 50–160 hours (approximately 5 days) in adults. Therefore, it takes approximately 2–3 weeks for phenobarbital to reach steady state, and slow titration is important to avoid supratherapeutic levels.
PRIMIDONE
Primidone is metabolized by the cytochrome P-450 system (40–60%) and eliminated via renal excretion of the unchanged drug (40–60%), PEMA (50–70%), and phenobarbital (5–10%). Approximately 15–25 percent of an oral dose is metabolized to phenobarbital. The rate of metabolism of primidone into phenobarbital is inversely related to age; the highest rates in oldest patients (the maximum age being 55).26 People aged 70–81, relative to people aged 18–26, have decreased renal clearance of primidone, phenobarbital, and PEMA, with a greater proportion of PEMA in the urine.27 Primidone clearance following an initial dose varies from 0.012 to 0.070 L/kg/hr with a mean of 0.037 L/kg/hr.28 Time-dependent changes in clearance necessitate an increase in dosage over time to maintain stable concentrations. The half-life of primidone ranges from 3 to 22 hours, with a mean half-life of 15.2 hours.29 The half-life of PEMA ranges from 24 to 48 hours. Long-term dosing of primidone can alter its half-life potentially due to accumulation of phenobarbital, which can reduce the half-life of primidone. Thus, it may be necessary to shorten the dosing interval with chronic maintenance therapy.
Several factors can modify phenobarbital and primidone elimination:
• Age: The total body clearance of phenobarbital differs among age groups: the average clearance in neonates is similar to adults and is 0.004 L/kg/hr (0.1 L/kg/day), while clearance in children (1–18 years of age) is 0.008 L/kg/hr (0.2 L/kg/day).24 The difference in the rate of clearance between neonates and children is a result of the maturation of the liver and production of the metabolizing enzymes. The elimination half-life of phenobarbital is therefore faster in children, with a range of 37–133 hours, compared to the adult and neonate ranges of 50–160 hours (approximately 5 days).14,24 Limited information is available on any clearance alterations in elderly but it appears elderly patients require lower doses to achieve therapeutic concentrations.30 The ability of neonates to metabolize primidone to phenobarbital is limited with a primidone half-life of 23 hours in neonates. In elderly patients both total and renal primidone clearance and half-life is similar to adult patients as a result a compensatory increase in nonrenal clearance due to a 30 percent decrease in renal clearance.27 This result suggests a possible increase in the active metabolites of primidone.
• Severe Liver Disease: In severe liver disease, the metabolism of phenobarbital may be decreased where a 50 percent increase in the half-life of phenobarbital has been reported.31 As expected, a lower dose should be considered in patients with liver dysfunction; however, no specific recommendations guide how dosage should be adjusted but should be related to the severity of the liver disease using the Child-Pugh score. A Child-Pugh score greater than 8 may represent a need to initiate therapy at 25–50 percent of the normal recommended dose and then monitor the patient closely for both response and toxicity with serum concentrations and clinical assessment.4 A small study of primidone in patients with acute viral hepatitis did not find any alterations in pharmacokinetic parameters, although it was only a single dose study.32
• Renal Impairment: It is expected that renal dysfunction will increase the half-life of phenobarbital and primidone, and increased toxicity has been reported in patients with renal impairment.33,34 Current data do not suggest empiric dosage adjustments are necessary for phenobarbital in patients with moderate-to-severe renal impairment (CrCl <30 mL/min) but they should be closely monitored.3,35 Approximately 30 percent of phenobarbital and primidone is removed with hemodialysis, 7.5–15 percent is removed with peritoneal dialysis, and a significant amount is removed with hemoperfusion (sieving coefficient 0.86).36-38 Clearance may be higher with high-efficiency hemodialyzers (up to 50%).39 Supplemental dosing following dialysis is often required, but no clear guidelines indicate the amount required.3 In adults on polytherapy and in children, approximately 40 percent of primidone is cleared via the kidney, with another 5–10 percent of an administered dose of primidone excreted via the kidney as phenobarbital.40 Thus, the dosage of primidone should be reduced in patients with renal impairment, with a suggested 25–50 percent reduction in dosage in patients with creatinine clearance less than 30 mL/min.
• Drug Interactions: Drug interactions with phenobarbital have been described and are of varying clinical significance. Phenobarbital is a potent inducer of the cytochrome P-450 isoenzyme systems specifically CYP1A2, CYP2C9, and CYP3A4. Because phenobarbital is a metabolite of primidone, similar drug interactions are expected with primidone administration. In patients receiving concurrent enzyme-inducing anticonvulsant drugs, the half-life of primidone ranges from 3.3 to 11 hours. See Table 14-3 for a description of clinically significant drug interactions.
• pH: The elimination of phenobarbital is sensitive to body pH, where an increase in urine pH increases renal clearance of phenobarbital significantly.41
• Nutritional State: Patients with malnutrition may have increased clearance of phenobarbital and primidone, with a resulting decrease in half-life that can persist for long periods after refeeding.42 These patients may need more aggressive dosing.43
| TABLE 14-3 | Phenobarbital Drug Interactions |

CONCENTRATION MONITORING
PHENOBARBITAL
Several analytical methods can be used to assess phenobarbital concentrations. Gas-liquid chromatography may be performed to detect the unchanged drug or its alkylated metabolites; however, interference with the assay is significant. High-performance liquid chromatography can be used, but it is subject to interference with carbamazepine epoxide. Enzyme-multiplied immunoassay and fluorescence-polarization immunoassay are the most commonly used assay techniques because they have the advantage of rapid and accurate determination of concentrations in serum or plasma, but they also have a potential for cross-reaction with coadministered barbiturates.5,44
Phenobarbital exhibits linear pharmacokinetics where an increase in the dose results in a proportional increase in the serum concentration.5,17 A minimum plasma level of 10 mg/L was found to be effective in controlling seizure activities. The upper limit of phenobarbital is controversial. Some patients exhibit toxic symptoms at a level above 30 mg/L where others tolerate a level greater than 40 mg/L. Common toxic signs and symptoms associated with a level >40–45 mcg/mL include sedation, nystagmus, ataxis, dysarthria, and seizure in severe cases. Concentrations above 100 mg/L may be considered to be potentially lethal.45 Therefore, therapeutic level between 10–40 mg/L should be attained to ensure maximum efficacy and minimal toxicity.
Indications for serum concentration monitoring include loss of seizure control, possible toxicity, dosing changes, significant liver and/or renal impairment, dialysis, and addition or deletion of an interacting drug.46 Ideally phenobarbital concentrations should be drawn immediately before the next dose to ensure a true trough. However, because of the long half-life, a random phenobarbital level can be obtained as long as levels are drawn consistently at similar time points to ensure correct interpretation.5,23 Because steady state is not generally reached until 2–3 weeks, rechecking phenobarbital concentrations immediately following dosage adjustment will not accurately reflect the change. Routine monitoring of phenobarbital should only occur 2–3 weeks after dosing initiation or change; however, serum concentration monitoring may be necessary sooner to assess for the need of an additional loading dose or if toxicity is suspected. If concentrations are assessed following intravenous administration of phenobarbital, the sample should be drawn at least one hour after the end of the infusion to avoid the distribution phase.
PRIMIDONE
A poor correlation is found between serum primidone concentrations and the oral dose of primidone. A range of 5–12 mg/L of primidone has been suggested as therapeutic; however, only limited utility comes from using this guideline in using this clinically. It is recommended to measure phenobarbital concentrations to guide therapy; phenobarbital concentrations generally becomes detectable 24 hours after primidone initiation. A summary of the key pharmacokinetic parameters for phenobarbital and primidone can be found in Table 14-4.
| TABLE 14-4 | Key Pharmacokinetic Parameters of Phenobarbital and Primidone |

CASE STUDIES
CASE 1: LOADING DOSE AND ADMINISTRATION
JP is a 60-year-old female who presents to the emergency department with a diagnosis of intracerebral hemorrhage. She is 5′5″ and weighs 62 kg. Upon arrival in the intensive care unit, she is unresponsive. An EEG is obtained showing nonconvulsive status epilepticus. After administration of 6 mg of lorazepam and 1,000 mg of phenytoin, it is decided to start her on phenobarbital.
QUESTION 1
What loading dose of phenobarbital should be administered?
Answer:
Two methods can be used to calculate an initial loading dose of phenobarbital, the literature-based recommended dosing and the pharmacokinetic dosing method. Each method has advantages and disadvantages. The literature-based method is a popular method due to the ease of calculation, but it does not offer the flexibility of targeting a specific serum concentration. This method will generally achieve concentrations in the middle to lower portion of the therapeutic range. In addition, this method cannot be used if the patient presents with a subtherapeutic phenobarbital level and needs a loading dose to increase the serum level into the therapeutic range or if the patient has recently received a suboptimal loading dose. The pharmacokinetic dosing method is commonly based on population pharmacokinetic parameters and allows the flexibility of targeting a specific level.
If the literature-based recommended dosing method is chosen, it can be calculated in the following manner:
Loading dose = 15–20 mg/kg
Loading dose = 62 kg × 20 mg
Loading dose = 1,240 mg
The targeted serum concentration dosing/pharmacokinetic dosing method can be calculated by using the following equation:
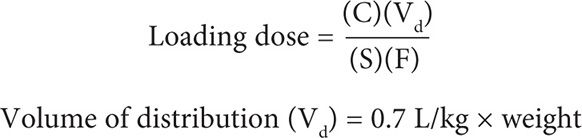
Because this dosage is given intravenously, the bioavailability F = 1.0 and the salt fraction S = 0.9 for phenobarbital sodium. In adults, the volume of distribution can be estimated at 0.7 L/kg. In most patients, a targeted serum concentration of 30 mg/L would be appropriate to achieve a serum level of 10–40 mg/L.
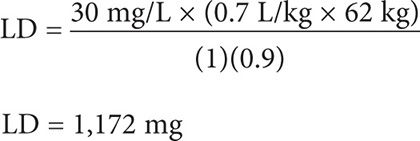
JP should receive a loading dose rounded up to 1,200 mg to achieve a serum concentration of 30 mg/L.
QUESTION 2
How should phenobarbital be administered?
Answer:
Phenobarbital can be administered either by intravenous push or diluted in an appropriate volume of fluid and given intravenously over a specific amount of time. The maximum infusion rate recommended to avoid toxicity (specifically hypotension) is 50 mg/min even though higher maximum rates may be found in the official product information. It is commonly divided into two or three portions and administered over several hours. This strategy can be used in patients to avoid cardiac toxicity from the propylene glycol diluents in the injectable dosage form. Phenobarbital loading doses can be rounded depending on the dosage forms available at each specific institution. It is important to monitor level of consciousness because higher doses of phenobarbital can produce somnolence and may require intubation of the patient. As mentioned earlier, phenobarbital is a cardiac depressant and likely will cause hypotension, particularly during infusion of the loading dose. Phenobarbital is stable in normal saline, dextrose 5 percent, and lactated ringers.47
In order to calculate the administration rate, you must first calculate the total administration time by using the following equation:

Next, the infusion rate in mL per hour should be calculated. The total volume to be infused is divided by the administration time. Because this calculation will give you the infusion rate in mL per minute, if the infusion pump is to be programmed in mL/hour, the rate should be multiplied by 60 minutes.

Loading Dose Targeted to Increase to a Specific Serum Concentration
JP was given a loading dose of 500 mg over 10 minutes. A serum phenobarbital level was obtained and it was 10 mg/L.
QUESTION 3
What dose of phenobarbital should be given to achieve a therapeutic concentration of 30 mg/L?
Answer:
Using the pharmacokinetic dosing strategy, an additional loading dose can be calculated if a patient has a subtherapeutic phenobarbital level. The same principles are used as with initial loading doses, altering the targeted serum concentration to reflect the degree of increase desired. The additional loading dose to increase by a specific concentration can be calculated by the following equation:
LD = ΔC (Cdesired – Cmeasured) × Vd
LD = (30 mg/L – 10 mg/L) × (0.7 L/kg × 62 kg)
LD = 868 mg, rounded up to 900 mg
Measurement of Serum Concentration
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


