Musculoskeletal System
The musculoskeletal system is a complex system of bones, joints, muscles, ligaments, tendons, and other tissues that gives the body form and shape. It also protects vital organs, makes movement possible, stores calcium and other minerals in the bony matrix for mobilization if deficiency occurs, and provides sites for hematopoiesis (blood cell production) in the marrow.
Bones
The human skeleton contains 206 bones, which are composed of inorganic salts (primarily calcium and phosphate), embedded in a framework of collagen fibers.
BONE SHAPE AND STRUCTURE
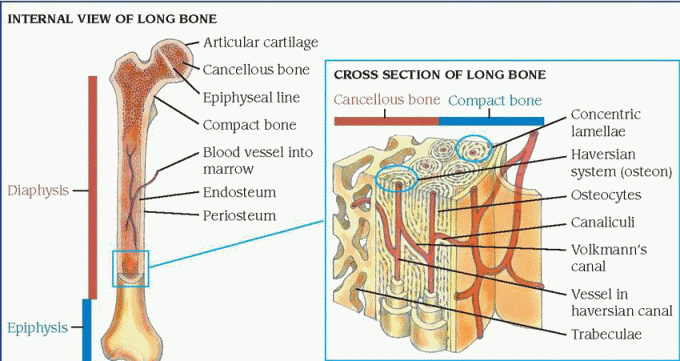
Bones are classified by shape as either long, short, flat, or irregular. Long bones are found in the extremities and include the humerus, radius, and ulna of the arm; the femur, tibia, and fibula of the leg; and the phalanges, metacarpals, and metatarsals of the hands and feet. (See Structure of long bones.) Short bones include the tarsal and carpal bones of the feet and hands, respectively. Flat bones include the frontal and parietal bones of the cranium, ribs, sternum, scapulae, ilium, and pubis. Irregular bones include the bones of the spine (vertebrae, sacrum, coccyx) and certain bones of the skull (the temporal, sphenoid, ethmoid, and mandible).
Classified according to structure, bone is either cortical (compact) or cancellous (spongy or trabecular). Adult cortical bone consists of networks of interconnecting canals, or canaliculi. Each network, or haversian system, runs parallel to the bone’s long axis and consists of a central haversian canal surrounded by layers (lamellae) of bone. Between adjacent lamellae are small openings called lacunae, which contain bone cells or osteocytes. The canaliculi, each containing one capillary or more, provide a route for tissue fluids transport; they connect all the lacunae.
Cancellous bone consists of thin plates (trabeculae) that form the interior meshwork of bone. These trabeculae are arranged in various directions to correspond with the lines of maximum stress or pressure. This gives the bone added structural strength. Chemically, inorganic salts (calcium and phosphate, with small amounts of sodium, potassium carbonate, and magnesium ions) comprise 70% of the mature bone. The salts give bone its elasticity and ability to withstand compression.
BONE GROWTH
Bone formation is ongoing and is determined by hormonal stimulation, dietary factors, and the amount of stress put on the bone. It’s accomplished by the continual actions of bone-forming osteoblasts and bone-reabsorbing cells called osteoclasts. Osteoblasts are present on the outer surface of and within bones. They respond to various stimuli to produce the bony matrix, or osteoid. As calcium salts precipitate on the organic matrix, the bone hardens. As the bone forms, a system of microscopic canals in the bone forms around the osteocytes. Osteoclasts are phagocytic cells that digest old, weakened
bone section by section. As they finish, osteoblasts simultaneously replace the cleared section with new, stronger bone.
bone section by section. As they finish, osteoblasts simultaneously replace the cleared section with new, stronger bone.
Vitamin D supports bone calcification by stimulating osteoblast activity and calcium absorption from the gut to make it available for bone building. When the serum calcium level falls, the parathyroid gland releases parathyroid hormone, which then stimulates osteoclast activity and bone breakdown, freeing calcium into the blood. Parathyroid hormone also increases serum calcium by decreasing renal excretion of calcium and increasing renal excretion of phosphate ion.
Phosphates are essential to bone formation; about 85% of the body’s phosphates are found in bone. The intestine absorbs a considerable amount of phosphates from dietary sources, but adequate levels of vitamin D are necessary for their absorption. Because calcium and phosphates interact in a reciprocal relationship, renal excretion of phosphates increases or decreases inversely proportional to the serum calcium level. Alkaline phosphatase (ALP) influences bone calcification and lipid and metabolite transport. Osteoblasts contain an abundance of ALP. A rise in the serum ALP level can indicate the presence of a skeletal disease, primarily one characterized by marked osteoblastic activity, such as bone metastases or Paget’s disease. It can also point to biliary obstruction, hyperparathyroidism, or excessive ingestion of vitamin D.
In children and young adults, bone growth occurs in the epiphyseal plate, a layer of cartilage between the diaphysis and epiphysis of long bones.
Osteoblasts deposit new bone in the area just beneath the epiphysis, making the bone longer, and osteoclasts model the new bone’s shape by reabsorbing previously deposited bone. These remodeling activities promote longitudinal bone growth, which continues until the epiphyseal growth plates, located at both ends, close during adolescence. Human growth factor and sex hormones influence the rate of ossification. In adults, bone growth is complete, and cartilage is replaced by bone, becoming the epiphyseal line.
Joints
The tendons, ligaments, cartilage, and other tissues that connect two bones constitute a joint. Depending on their structures, joints either predominantly permit motion or provide stability. Joints, like bones, are classified according to structure and function.
JOINT CLASSIFICATION
The three structural types of joints are fibrous, cartilaginous, and synovial.
♦ Fibrous joints, or synarthroses, permit only slight movement and provide stability when tight union is necessary, as in the sutures that join the cranial bones.
♦ Cartilaginous joints, or amphiarthroses, allow limited motion, as between vertebrae.
♦ Synovial joints, or diarthroses, are the most common and permit the greatest degree of movement. These joints include the elbows and knees.
Synovial joints have distinguishing characteristics:
♦ The two articulating surfaces of the bones have a smooth hyaline covering (articular cartilage) that’s resilient to pressure.
♦ Their opposing surfaces are congruous and glide smoothly on each other.
♦ A fibrous (articular) capsule holds them together.
♦ Beneath the capsule, lining the joint cavity, is the synovial membrane, which secretes a clear viscous fluid called synovial fluid. This fluid lubricates the two opposing surfaces during motion and also nourishes the articular cartilage.
♦ Surrounding a synovial joint are ligaments, muscles, and tendons, which strengthen and stabilize the joint but allow free movement.
JOINT MOVEMENT
The two types of synovial joint movement are angular and circular.
Angular movement
Joints of the knees, elbows, and phalanges permit the following angular movements:
♦ flexion (closing of the joint angle)
♦ extension (opening of the joint angle)
♦ hyperextension (extension of the angle beyond the usual arc).
Other joints, including the shoulders and hips, permit:
♦ abduction (movement away from the body’s midline)
♦ adduction (movement toward the midline).
Circular movement
Circular movements include:
♦ rotation (motion around a central axis), as in the ball-and-socket joints of the hips and shoulders
♦ pronation (downward wrist or ankle motion)
♦ supination (upward wrist motion).
Other kinds of movement include inversion (inward turning, as of the foot), eversion (outward turning, as of the foot), protraction (as in the forward motion of the mandible), and retraction (returning protracted part into place).
Muscles
The most specialized feature of muscle tissue— contractility—makes the movement of bones and joints possible. Normal skeletal muscles contract in response to neural impulses. Appropriate contraction of muscle usually applies force to one or more tendons. The force pulls one bone toward, away from, or around a second bone, depending on the type of muscle contraction and the type of joint involved. Abnormal metabolism in the muscle may result in inappropriate contractility. For example, when stored glycogen or lipids can’t be used because the enzyme necessary to convert energy for contraction is lacking, the result may be cramps, fatigue, and exercise intolerance.
Muscles permit and maintain body positions, such as sitting and standing. Muscles also pump blood through the body (cardiac contraction and vessel compression), move food through the intestines (peristalsis), and make breathing possible. Skeletal muscle activity produces heat; it’s an important component in temperature regulation. Deep body temperature regulators are found in the abdominal viscera, spinal cord, and great veins. These receptors detect changes in the body core temperature and stimulate the hypothalamus to institute appropriate temperature changing responses, such as shivering in response to cold. Muscle mass accounts for about 40% of an average man’s weight.
MUSCLE CLASSIFICATION
Muscles are classified according to structure, anatomic location, and function.
♦ Skeletal muscles are attached to bone and have striped (striated) appearances that reflect their cellular structures.
♦ Visceral muscles move contents through internal organs and are smooth (nonstriated).
♦ Cardiac muscles (smooth) constitute the heart wall.
When muscles are classified according to activity, they’re called either voluntary or involuntary. (See chapter 8, Nervous system.) Voluntary muscles can be controlled at will and are under the influence of the somatic nervous system; these are the skeletal muscles. Involuntary muscles, controlled by the autonomic nervous system, include the cardiac and visceral muscles. Some organs contain voluntary and involuntary muscles.
MUSCLE CONTRACTION
Each skeletal muscle consists of many elongated muscle cells, called muscle fibers, through which run slender threads of protein, called myofibrils. Muscle fibers are held together in
bundles by sheaths of fibrous tissues called fascia. Blood vessels and nerves pass into muscles through the fascia to reach the individual muscle fibers. Motoneurons synapse with the motor nerve fibers of voluntary muscles. These fibers reach the membranes of skeletal muscle cells at neuromuscular (myoneural) junctions. When an impulse reaches the myoneural junction, the junction releases the neurotransmitter, acetylcholine, which releases calcium from the sarcoplasmic reticulum, a membranous network in the muscle fiber, which, in turn, triggers muscle contraction. Muscle contraction is isometric or isotonic. Isometric contraction results in an increase in tension without change in length. Isotonic contraction occurs when the muscle shortens as weight is lifted. The energy source for this contraction is adenosine triphosphate (ATP). ATP release is also triggered by the impulse at the myoneural junction. Relaxation of a muscle is believed to take place by reversal of these mechanisms.
bundles by sheaths of fibrous tissues called fascia. Blood vessels and nerves pass into muscles through the fascia to reach the individual muscle fibers. Motoneurons synapse with the motor nerve fibers of voluntary muscles. These fibers reach the membranes of skeletal muscle cells at neuromuscular (myoneural) junctions. When an impulse reaches the myoneural junction, the junction releases the neurotransmitter, acetylcholine, which releases calcium from the sarcoplasmic reticulum, a membranous network in the muscle fiber, which, in turn, triggers muscle contraction. Muscle contraction is isometric or isotonic. Isometric contraction results in an increase in tension without change in length. Isotonic contraction occurs when the muscle shortens as weight is lifted. The energy source for this contraction is adenosine triphosphate (ATP). ATP release is also triggered by the impulse at the myoneural junction. Relaxation of a muscle is believed to take place by reversal of these mechanisms.
Muscle fatigue results when the sources of ATP in a muscle are depleted. If a muscle is deprived of oxygen, fatigue occurs rapidly. As the muscle fatigues, it switches to anaerobic metabolism of glycogen stores, in which the stored glycogen is split into glucose (glycolysis) without the use of oxygen. Lactic acid is a by-product of anaerobic glycolysis and may accumulate in the muscle and blood with intense or prolonged muscle contraction.
Tendons and ligaments
Skeletal muscles are attached to bone directly or indirectly by fibrous cords known as tendons. The least movable end of the muscle attachment (generally proximal) is called the point of origin; the most movable end (generally distal) is called the point of insertion.
Ligaments are fibrous connections that control joint movement between two bones or cartilages. Their purpose is to support and strengthen joints.
Pathophysiologic changes
Alterations of the normal functioning of bones and muscles are described here. Most musculoskeletal disorders are caused by or profoundly affect other body systems.
ALTERATIONS IN BONE
Disease may alter bone density, growth, or strength.
Density
In healthy young adults, the resorption and formation phases are tightly coupled to maintain bone mass in a steady state. Bone loss occurs when the two phases become uncoupled, and resorption exceeds formation. Estrogen not only regulates calcium uptake and release but also regulates osteoblastic activity. A decreased estrogen level may lead to diminished osteoblastic activity and loss of bone mass, called osteoporosis. In children, vitamin D deficiency prevents normal bone growth and leads to rickets.
 Bone density and structural integrity decrease in women after age 30 and in men after age 45. The relatively steady loss of bone matrix can be partially offset by exercise and appropriate dietary calcium intake.
Bone density and structural integrity decrease in women after age 30 and in men after age 45. The relatively steady loss of bone matrix can be partially offset by exercise and appropriate dietary calcium intake.Growth
The osteochondroses are a group of disorders characterized by avascular necrosis of the epiphyseal growth plates in growing children and adolescents. In these disorders, a lack of blood supply to the femoral head leads to septic necrosis, with softening and resorption of bone. Revascularization then initiates new bone formation in the femoral head or tibial tubercle, which leads to a malformed femoral head.
Strength
Both cortical and trabecular bone contribute to skeletal strength. Any loss of the inorganic salts that constitute the chemical structure of bone will weaken bone. Cancellous bone is more sensitive to metabolic influences, so conditions that produce rapid bone loss tend to affect cancellous bone more quickly than cortical bone.
ALTERATIONS OF MUSCLE
Pathologic effects on muscle include atrophy, fatigue, weakness, myotonia, and spasticity.
Atrophy
Atrophy is a decrease in the size of a tissue or cell. In muscles, the myofibrils atrophy after prolonged inactivity from bed rest or trauma (casting), when local nerve damage makes movement impossible, or when illness removes needed nutrients from muscles. The effects of muscular deconditioning associated with lack of physical activity may be apparent in a matter of days. An individual on bed rest loses muscle
strength, as well as muscle mass, from baseline levels at a rate of 3% per day. Conditioning and stretching exercises may help prevent atrophy. If reuse isn’t restored within 1 year, regeneration of muscle fibers is unlikely.
strength, as well as muscle mass, from baseline levels at a rate of 3% per day. Conditioning and stretching exercises may help prevent atrophy. If reuse isn’t restored within 1 year, regeneration of muscle fibers is unlikely.
Fatigue
Pathologic muscle fatigue may be the result of impaired neural stimulation of muscle or energy metabolism or disruption of calcium flux. See chapter 4, Fluids and electrolytes, for a detailed discussion of these events.
Weakness
 Muscle mass and muscle strength may decrease in elderly patients as a result of disuse. This can be reversed with moderate, regular, weight-bearing exercise.
Muscle mass and muscle strength may decrease in elderly patients as a result of disuse. This can be reversed with moderate, regular, weight-bearing exercise.Periodic paralysis is a genetic disorder that can be triggered by exercise or a process or chemical (such as medication) that alters the serum potassium level. This hyperkalemic or hypokalemic periodic paralysis may be caused by a high-carbohydrate diet, emotional stress, prolonged bed rest, or hyperthyroidism. During an attack of periodic paralysis, the muscle membrane is unresponsive to neural stimuli, and the electrical charge needed to initiate the impulse (resting membrane potential) is reduced from -90 to -45 millivolts.
Myotonia and spasticity
Myotonia is delayed relaxation after a voluntary muscle contraction—such as grip, eye closure, or muscle percussion—accompanied by prolonged depolarization of the muscle membrane. Depolarization is the reversal of the resting potential in stimulated cell membranes. It’s the process by which the cell membrane “resets” its positive charge with respect to the negative charge outside the cell. Myotonia occurs in myotonic muscular dystrophy and some forms of periodic paralysis.
Stress-induced muscle tension, or spasticity, is presumably caused by increased activity in the reticular activating system and gamma loop in the muscle fiber. The reticular activating system consists of multiple diffuse pathways in the brain that control wakefulness and response to stimuli. A pathologic contracture is permanent muscle shortening caused by muscle spasticity, seen in central nervous system injury or severe muscle weakness.
Disorders
This section discusses musculoskeletal disorders, some of which have far-reaching effects in other body systems. They include bone fracture, carpal tunnel syndrome, clubfoot, developmental dysplasia of the hip, gout, herniated disk, Legg-Calvé-Perthes disease, muscular dystrophy, osteoarthritis, osteogenesis imperfecta, osteomalacia and rickets, osteomyelitis, osteoporosis, Paget’s disease, rhabdomyolysis, scoliosis, and sprains and strains.
 Many patients with musculoskeletal disorders are elderly, have other concurrent medical conditions, or are victims of trauma. Generally, they face prolonged immobilization. (See Managing musculoskeletal pain.)
Many patients with musculoskeletal disorders are elderly, have other concurrent medical conditions, or are victims of trauma. Generally, they face prolonged immobilization. (See Managing musculoskeletal pain.)BONE FRACTURE
When a force exceeds the compressive or tensile strength (the ability of the bone to hold together) of the bone, a fracture will occur. (For an explanation of the terms used to identify fractures, see Classifying fractures, page 372.)
Each year, an estimated 25% of the population has traumatic musculoskeletal injury, and a significant number of these involve fractures.
The prognosis varies with the extent of disablement or deformity, amount of tissue and vascular damage, adequacy of reduction and immobilization, and patient’s age, health, and nutritional status.
 Children’s bones usually heal rapidly and without deformity. However, epiphyseal plate fractures in children are likely to cause deformity because they interfere with normal bone growth. In elderly people, underlying systemic illness, impaired circulation, or poor nutrition may cause slow or poor healing.
Children’s bones usually heal rapidly and without deformity. However, epiphyseal plate fractures in children are likely to cause deformity because they interfere with normal bone growth. In elderly people, underlying systemic illness, impaired circulation, or poor nutrition may cause slow or poor healing.Causes
♦ Bone tumors
♦ Falls
♦ Medications that cause iatrogenic osteoporosis such as steroids
♦ Metabolic illnesses (such as hypoparathyroidism or hyperparathyroidism)
♦ Motor vehicle crashes
♦ Sports
♦ Use of drugs that impair judgment or mobility
♦ Young age (immaturity of bone)
 The highest incidence of bone fractures occurs in young males between ages 15 and 24 (tibia, clavicle, and lower humerus) and are usually the result of trauma. In elderly people, upper femur, upper humerus, forearm, wrist, vertebrae, and pelvis fractures are commonly associated with osteoporosis and falls.
The highest incidence of bone fractures occurs in young males between ages 15 and 24 (tibia, clavicle, and lower humerus) and are usually the result of trauma. In elderly people, upper femur, upper humerus, forearm, wrist, vertebrae, and pelvis fractures are commonly associated with osteoporosis and falls.Pathophysiology
When a bone is fractured, the periosteum and blood vessels in the cortex, marrow, and surrounding soft tissue are disrupted. A hematoma forms between the broken ends of the bone and beneath the periosteum, and granulation tissue eventually replaces the hematoma.
Damage to bone tissue triggers an intense inflammatory response in which cells from surrounding soft tissue and the marrow cavity invade the fracture area, and blood flow to the entire bone is increased. Osteoblasts in the periosteum, endosteum, and marrow produce osteoid (collagenous, young bone that hasn’t yet calcified, also called callus), which hardens along the outer surface of the shaft and over the broken ends of the bone. Osteoclasts reabsorb dead bone and osteoblasts rebuild bone. Osteoblasts then transform into osteocytes (mature bone cells). Remodeling occurs as excess callus is reabsorbed and trabecular bone is laid down.
Signs and symptoms
♦ Deformity due to unnatural alignment
♦ Swelling due to vasodilation and infiltration by inflammatory leukocytes and mast cells
♦ Muscle spasm and tenderness related to the inflammatory response
♦ Impaired sensation distal to the fracture site due to pinching or severing of neurovascular elements by the trauma or by bone fragments
♦ Limited range of motion due to misalignment, neurovascular compromise, swelling, and pain
♦ Crepitus, or “clicking” sounds on movement, caused by shifting bone fragments
♦ Bruising from blood released at the fracture site.
Complications
♦ Permanent deformity and dysfunction if bones fail to heal (nonunion) or heal improperly (malunion)
♦ Aseptic (not caused by infection) necrosis of bone segments due to impaired circulation
♦ Hypovolemic shock as a result of blood vessel damage (especially with a fractured femur)
♦ Muscle contractures
♦ Compartment syndrome (See Recognizing compartment syndrome, page 373.)
♦ Renal calculi from decalcification due to prolonged immobility
♦ Fat embolism due to disruption of marrow or activation of the sympathetic nervous system after the trauma (may lead to respiratory or central nervous system distress)
Diagnosis
♦ History of traumatic injury and results of the physical examination, including gentle palpation and a cautious attempt by the patient to move parts distal to the injury, reveal bone fracture.
Managing musculoskeletal pain
A patient with a musculoskeletal disorder that causes chronic, nonmalignant pain should be assessed and treated in a stepped approach. Measures include:
♦ nonpharmacologic methods, such as heat, ice, elevation, and rest
♦ acetaminophen (Tylenol)
♦ a nonsteroidal anti-inflammatory drug such as ibuprofen (Motrin)
♦ another nonopioid analgesic, such as tramadol (Ultram), topical capsaicin, (Zostrix) or lidocaine patch (Lidoderm)
♦ a tricyclic antidepressant, such as amitriptyline (Elavil), which may decrease the pain signal at the neurosynaptic junctions
♦ an opioid analgesic alone or with a tricyclic antidepressant.
♦ X-rays of the suspected fracture and the joints above and below confirm the diagnosis. After reduction, X-rays confirm bone alignment.
Treatment
For arm or leg fracture, emergency treatment consists of:
♦ splinting the limb above and below the suspected fracture to immobilize it
♦ applying a cold pack to reduce pain and edema
♦ elevating the limb to reduce pain and edema.
R — Rest
I — Ice
C — Compression
E — Elevation
Treatment of severe bone fracture that causes blood loss includes:
♦ direct pressure to control bleeding
♦ fluid replacement as soon as possible to prevent or treat hypovolemic shock.
After confirming a fracture, treatment begins with a reduction. Closed reduction involves:
♦ manual manipulation
♦ a local anesthetic (such as lidocaine [Xylocaine])
♦ an analgesic (such as morphine)
♦ a muscle relaxant (such as diazepam [Valium] I.V.) or a sedative (such as midazolam [Versed]) to facilitate the muscle stretching necessary to realign the bone.
Classifying fractures
One of the best-known systems for classifying fractures uses a combination of terms that describe general classification, fragment position, and fracture line to describe fractures.
General classification of fractures
♦ Simple (closed) — Bone fragments don’t penetrate the skin.
♦ Compound (open) — Bone fragments penetrate the skin.
♦ Incomplete (partial)—Bone continuity isn’t completely interrupted.
♦ Complete—Bone continuity is completely interrupted.
Classification by fragment position
♦ Comminuted—The bone breaks into small pieces.
♦ Impacted—One bone fragment is forced into another.
♦ Angulated—Fragments lie at an angle to each other.
♦ Displaced—Fracture fragments separate and are deformed.
♦ Nondisplaced—The two sections of bone maintain essentially normal alignment.
♦ Overriding—Fragments overlap, shortening the total bone length.
♦ Segmental—Fractures occur in two adjacent areas with an isolated central segment.
♦ Avulsed—Fragments are pulled from the normal position by muscle contractions or ligament resistance.
Classification by fracture line
♦ Linear—The fracture line runs parallel to the bone’s axis.
♦ Longitudinal—The fracture line extends in a longitudinal (but not parallel) direction along the bone’s axis.
♦ Oblique—The fracture line crosses the bone at about a 45-degree angle to the bone’s axis.
♦ Spiral—The fracture line crosses the bone at an oblique angle, creating a spiral pattern.
♦ Transverse—The fracture line forms a right angle with the bone’s axis.
When closed reduction is impossible, open reduction by surgery involves:
♦ internal fixation, involving immobilization of the fracture by means of rods, plates, or screws and application of a plaster cast
♦ external fixation, a system of surgically placed pins and stabilizing bars to maintain bone alignment
♦ tetanus prophylaxis
♦ a prophylactic antibiotic
♦ surgery to repair soft-tissue damage
♦ thorough wound debridement
♦ physical therapy after cast removal to restore limb mobility.
When a splint or cast fails to maintain the reduction, immobilization requires skin or skeletal traction, using a series of weights and pulleys. This may involve:
♦ elastic bandages and sheepskin coverings to attach traction devices to the patient’s skin (skin traction)
♦ a pin or wire inserted through the bone distal to the fracture and attached to a weight to allow more prolonged traction (skeletal traction).
Special considerations
♦ Watch for signs of shock in the patient with a severe open fracture of a large bone, such as the femur.
♦ Monitor vital signs, and be especially alert for rapid pulse, decreased blood pressure, pallor, and cool, clammy skin—all of which may indicate that the patient is in shock.
♦ Administer I.V. fluids as ordered.
♦ Offer reassurance to the patient, who is likely to be frightened and in pain.
♦ Ease pain with an analgesic as needed.
♦ Help the patient set realistic goals for recovery.
♦ If the bone fracture requires long-term immobilization with traction, reposition the patient often to increase comfort and prevent pressure ulcers. Assist with active range-of-motion exercises to prevent muscle atrophy. Encourage deep breathing and coughing to avoid hypostatic pneumonia.
♦ Urge adequate fluid intake to prevent urinary stasis and constipation. Watch for signs and symptoms of renal calculi (flank pain, nausea, and vomiting).
♦ Provide good cast care, and support the cast with pillows. Observe for skin irritation near cast edges, and check for foul odors or discharge. Tell the patient to report signs or symptoms of impaired circulation (skin coldness, numbness, tingling, or discoloration) immediately. Warn him not to get the cast wet and not to insert foreign objects under the cast.
♦ Encourage the patient to start moving around as soon as he’s able. Help him to walk. (Remember, a patient who has been bedridden for some time may be dizzy at first.) Demonstrate how to use crutches properly.
♦ After cast removal, refer the patient to a physical therapist to restore limb mobility.
CARPAL TUNNEL SYNDROME
Carpal tunnel syndrome, a form of repetitive stress injury, is the most common nerve entrapment syndrome. Carpal tunnel syndrome usually occurs in women between ages 30 and 60 (posing a serious occupational health problem). However, men who are also employed as assembly-line workers and packers and who repeatedly use poorly designed tools are just as likely to develop this disorder. Any strenuous use of the hands—sustained grasping, twisting, or flexing—aggravates this condition.
Causes
Although carpal tunnel syndrome is mostly idiopathic, it may result from:
♦ acromegaly
♦ amyloidosis
♦ benign tumor
♦ conditions that increase fluid pressure in the wrist, including alterations in the endocrine or immune system
♦ diabetes mellitus
♦ flexor tenosynovitis (commonly associated with rheumatic disease)
♦ hypothyroidism
♦ multiple myeloma
♦ nerve compression
♦ obesity
♦ pregnancy
♦ repetitive stress injury
♦ rheumatoid arthritis
♦ sprain or wrist dislocation, including Colles’ fracture followed by edema.
Pathophysiology
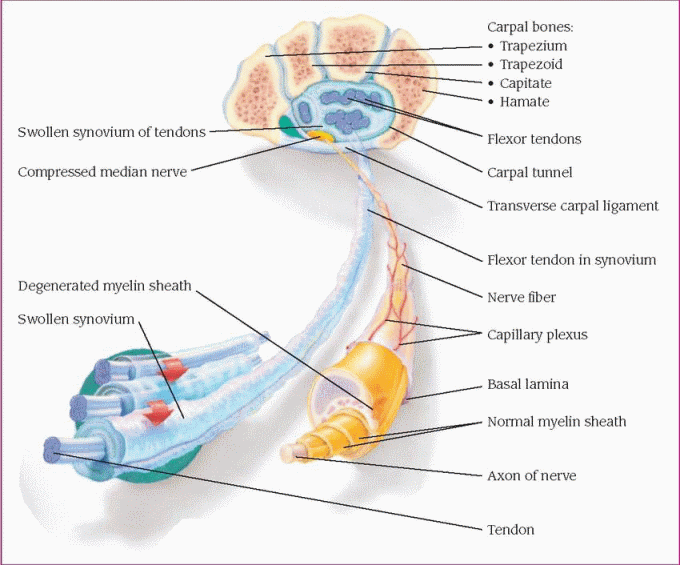
The carpal bones and the transverse carpal ligament form the carpal tunnel. Inflammation or fibrosis of the tendon sheaths that pass through the carpal tunnel usually causes edema and compression of the median nerve. (See Cross section of the wrist with carpal tunnel syndrome, page 374.) This compression neuropathy causes sensory and motor changes in the median distribution of the hands, initially impairing sensory transmission to the thumb, index finger, second finger, and inner aspect of the third finger.
Recognizing compartment syndrome
Compartment syndrome occurs when edema or bleeding increases pressure within a muscle compartment (a smaller section of a muscle), to the point of interfering with circulation. Crush injuries, burns, bites, and fractures requiring casts or dressings may cause this syndrome. Compartment syndrome most commonly occurs in the lower arm, hand, lower leg, or foot.
Signs and symptoms include:
♦ increased pain
♦ decreased touch sensation
♦ increased weakness of the affected part
♦ increased swelling and pallor
♦ decreased pulses and increased capillary refill time.
Treatment of compartment syndrome consists of:
♦ placing the limb at heart level
♦ removing constricting forces
♦ monitoring neurovascular status
♦ subfascial injection of hyaluronidase (Wydase)
♦ emergency fasciotomy.
Signs and symptoms
♦ Weakness, pain, burning, numbness, or tingling in one or both hands due to nerve and blood vessel compression (This paresthesia affects the thumb, forefinger, middle finger, and medial half of the fourth finger.)
♦ Inability to clench the hand into a fist; the nails may be atrophic, the skin dry and shiny
♦ Because of vasodilatation and venous stasis, symptoms that typically worsen at night and in the morning
♦ Pain spreading to the forearm and, in severe cases, as far as the shoulder
♦ Pain relief achieved by shaking or rubbing the hands vigorously or dangling the arms at the side
Complications
♦ Continued use of the affected wrist increasing tendon inflammation, compression, and neural ischemia, causing a decrease in wrist function
♦ Untreated carpal tunnel syndrome producing permanent nerve damage with loss of movement and sensation
Diagnosis
Physical examination reveals decreased sensation to light touch or pinpricks in the affected
fingers. Thenar muscle atrophy occurs in about half of all cases of carpal tunnel syndrome but is usually a late sign.
fingers. Thenar muscle atrophy occurs in about half of all cases of carpal tunnel syndrome but is usually a late sign.
The following tests provide rapid diagnosis of carpal tunnel syndrome:
♦ Tinel’s sign—tingling over the median nerve on light percussion
♦ Phalen’s maneuver—holding the forearms vertically and allowing both hands to drop into complete flexion at the wrists for 1 minute reproduces symptoms of carpal tunnel syndrome
♦ Compression test—blood pressure cuff inflated above systolic pressure on the forearm for 1 to 2 minutes provokes pain and paresthesia along the distribution of the median nerve.
Other tests include electromyography to detect a median nerve motor conduction delay of more than 5 milliseconds and laboratory tests to identify underlying disease.
Treatment
♦ Conservative treatment first, including resting the hands by splinting the wrist in neutral extension for 1 to 2 weeks
♦ A nonsteroidal anti-inflammatory drug for symptomatic relief
♦ Injection of the carpal tunnel with hydrocortisone and lidocaine for significant but temporary relief
♦ Seeking another occupation, if a definite link has been established between the patient’s job and the development of repetitive stress injury
♦ Correction of an underlying disorder
♦ Surgical decompression of the nerve by resecting the entire transverse carpal tunnel ligament or by using endoscopic surgical techniques, when conservative treatment fails
♦ Neurolysis (freeing of the nerve fibers)
To prevent carpal tunnel syndrome, advise your patients to make these lifestyle changes.
Take frequent breaks
Gently stretching and bending the hands and wrists every 15 to 20 minutes gives the hands and wrists a break, especially when using equipment that vibrates or exerts a great amount of force. Tasks should also be alternated to avoid repetitive movements, which can contribute to tendinitis and carpal tunnel syndrome.
Watch hand and wrist positioning
When using a keyboard, bending the wrist all the way up or down should be avoided. A relaxed middle position is best. The keyboard should be kept at elbow height or slightly lower.
Improve posture
Poor posture can cause the shoulders to roll forward, allowing the neck and shoulder muscles to shorten, which can compress the nerves in the neck. This position can affect the wrists, hands, and fingers.
Keep hands warm
Hand stiffness and pain develop more frequently in a cold environment. Using fingerless gloves may help if the temperature can’t be adjusted at work.
Special considerations
♦ Administer a mild analgesic as needed. Encourage the patient to use his hands as much as possible. If his dominant hand has been impaired, you may have to help with eating and bathing.
♦ Teach the patient how to apply a splint. Tell him not to make it too tight. Show him how to remove the splint to perform gentle range-ofmotion exercises, which should be done daily. Make sure the patient knows how to do these exercises before he’s discharged.
♦ After surgery, monitor vital signs and regularly check the color, sensation, and motion of the affected hand.
♦ Advise the patient who is about to be discharged to exercise his hands occasionally in warm water. If the arm is in a sling, tell him to remove the sling several times per day to do exercises for his elbow and shoulder.
♦ Suggest occupational counseling for the patient who has to change jobs because of repetitive stress injury.
♦ Advise all patients on measures to prevent carpal tunnel syndrome, including using correct posture and wrist position, performing stretching exercises, and taking frequent breaks. Ergonomic workstations and tools are designed to allow a worker’s wrist to maintain a natural position, preventing strain and stress to the wrist. (See Preventing carpal tunnel syndrome.)
CLUBFOOT
Clubfoot, also called talipes, is the most common congenital disorder of the lower extremities. It’s marked primarily by a deformed talus and shortened Achilles tendon, which give the foot a characteristic clublike appearance. In talipes equinovarus, the foot points downward (equinus) and turns inward (varus), and the front of the foot curls toward the heel (forefoot adduction).
Clubfoot occurs in about 1 per 1,000 live births, is bilateral in about half of all cases, and is twice as common in boys as in girls. It may be associated with other birth defects, such as myelomeningocele, spina bifida, and arthrogryposis. Clubfoot is correctable with prompt treatment.
Causes
A combination of environmental and genetic factors in utero appears to cause clubfoot, including:
♦ arrested development during the 9th and 10th weeks of embryonic life when the feet are formed (in children without a family history of clubfoot)
♦ heredity (mechanism of transmission is undetermined; the sibling of a child born with clubfoot has 1 chance in 35 of being born with the same anomaly, and a child of a parent with clubfoot has 1 chance in 10)
♦ muscle abnormalities leading to variations in length and tendon insertions
♦ secondary to cerebral palsy (older children), paralysis, or poliomyelitis, in which case treatment includes management of the underlying disease.
Pathophysiology
Abnormal development of the foot during fetal growth leads to abnormal muscles and joints and contracture of soft tissue. Clubfoot can also occur as a result of paralysis, poliomyelitis, or
cerebral palsy. The condition called apparent clubfoot results when a fetus maintains a position in utero that gives his feet a clubfoot appearance at birth; it can usually be corrected manually. Another form of apparent clubfoot is inversion of the feet, resulting from the denervation type of progressive muscular atrophy and progressive muscular dystrophy.
cerebral palsy. The condition called apparent clubfoot results when a fetus maintains a position in utero that gives his feet a clubfoot appearance at birth; it can usually be corrected manually. Another form of apparent clubfoot is inversion of the feet, resulting from the denervation type of progressive muscular atrophy and progressive muscular dystrophy.
Signs and symptoms
Talipes equinovarus varies greatly in severity. Deformity may be so extreme that the toes touch the inside of the ankle, or it may be only vaguely apparent.
Every case includes:
♦ deformed talus because of abnormal development
♦ shortened Achilles tendon from contracture
♦ shortened and flattened calcaneus bone of the heel caused by abnormal development and contracture
♦ shortened, underdeveloped calf muscles and soft-tissue contractures at the site of the deformity (depending on degree of the varus deformity)
♦ foot tight in its deformed position, resisting manual efforts to push it back into normal position, because of shortening of muscles and contractures
♦ no pain, except in elderly patients with arthritis and secondary deformity.
Complications
Possible complications of talipes equinovarus include:
♦ chronic impairment (neglected clubfoot)
♦ incomplete correctable (when severe enough to require surgery).
Diagnosis
Early diagnosis of clubfoot is usually no problem because the deformity is obvious. In subtle deformity, however, true clubfoot must be distinguished from apparent clubfoot (metatarsus varus or pigeon toe), usually by X-rays showing super-imposition of the talus and calcaneus and a ladderlike appearance of the metatarsals (true clubfoot).
Treatment
Treatment of clubfoot is done in three stages: correcting the deformity, maintaining the correction until the foot regains normal muscle balance, and observing the foot closely for several years to prevent the deformity from recurring.
Clubfoot deformities are usually corrected in sequential order: forefoot adduction first, then varus (or inversion), then equinus (or plantar flexion). Trying to correct all three deformities at once only results in a misshapen, rockerbottomed foot.
Other essential parts of management include:
♦ stressing to parents the importance of prompt treatment and orthopedic supervision until growth is completed
♦ teaching parents cast care and how to recognize circulatory impairment before a child in a clubfoot cast is discharged
♦ explaining to an older child and his parents that surgery can improve clubfoot with good function but can’t totally correct it; the affected calf muscle will remain slightly underdeveloped
♦ emphasizing the need for long-term orthopedic care to maintain correction; correcting this defect permanently takes time and patience.
Special considerations
The primary concern in clubfoot is early recognition, preferably in neonates.
♦ Look for any abnormal positioning in an infant’s feet. Make sure you recognize the difference between true clubfoot and apparent clubfoot. Don’t use excessive force in trying to manipulate a clubfoot. The foot with apparent clubfoot moves easily.
♦ Stress to parents the importance of prompt treatment. Clubfoot demands immediate therapy and orthopedic supervision until growth is completed.
♦ After casting, elevate the child’s feet with pillows. Check the toes every 1 to 2 hours for temperature, color, sensation, motion, and capillary refill time; watch for edema. Before a child in a clubfoot cast is discharged, teach parents to recognize circulatory impairment.
♦ Keep in mind that fiberglass casting, the most commonly used kind, doesn’t require petaling. Padding from the cast is turned over and used as a cushioning. (Don’t rub the skin with alcohol, and don’t use oils or powders, which tend to macerate the skin.)
♦ If the child is old enough to walk, caution parents not to let the foot part of the cast get soft and thin from wear. If it does, much of the correction may be lost.
♦ When the wedging method of shaping the cast is being used, check circulatory status frequently; it may be impaired by increased pressure on tissues and blood vessels. The equinus (posterior release) correction especially places considerable strain on ligaments, blood vessels, and tendons.
♦ After surgery, elevate the child’s feet with pillows to decrease swelling and pain. Immediately report any signs of discomfort or pain. Try to locate the source of pain; it may result from cast pressure rather than from the incision. If bleeding
occurs under the cast, document the location and size. If bleeding spreads, report it to the physician.
occurs under the cast, document the location and size. If bleeding spreads, report it to the physician.
♦ Explain to the older child and his parents that surgery can improve clubfoot with good function but can’t totally correct it; the affected calf muscle will remain slightly underdeveloped.
♦ Emphasize the need for long-term orthopedic care to maintain correction. Teach parents the prescribed exercises that the child can do at home. Urge them to make the child wear the corrective shoes ordered and the splints during naps and at night. Make sure they understand that treatment for clubfoot continues during the entire growth period. Correcting this defect permanently takes time and patience.
DEVELOPMENTAL DYSPLASIA OF THE HIP
Developmental dysplasia of the hip (DDH), an abnormality of the hip joint present from birth, is the most common disorder affecting the hip joints in children younger than 3 years. About 85% of affected infants are females.
DDH can be unilateral or bilateral and can affect the femoral head, the acetabulum, or both. This abnormality occurs in three forms of varying severity:
♦ Unstable dysplasia—the hip is positioned normally but can be dislocated by manipulation.
♦ Subluxation or incomplete dislocation—the femoral head rides on the edge of the acetabulum.
♦ Complete dislocation—the femoral head is totally outside the acetabulum.
Causes
Although the causes of DDH aren’t clear, many factors contribute to its development. It’s more likely to occur in the following circumstances:
♦ dislocation after breech delivery (malposition in utero, 10 times more common than after cephalic delivery)
♦ elevated maternal relaxin, a hormone secreted by the corpus luteum during pregnancy that causes relaxation of pubic symphysis and cervical dilation (may promote relaxation of the joint ligaments, predisposing the infant to DDH)
♦ large neonates and twins (more common).
Pathophysiology
The precise cause of congenital dislocation is unknown. Excessive or abnormal movement of the joint during a traumatic birth may cause dislocation. Displacement of bones within the joint may damage joint structures, including articulating surfaces, blood vessels, tendons, ligaments, and nerves. This may lead to ischemic necrosis because of the disruption of blood flow to the joint.
Signs and symptoms
♦ Asymmetrical skin folds due to hip dislocation
♦ Hip riding above the acetabulum, causing the level of the knees to be uneven (Galeazzi sign)
♦ Limited abduction on the dislocated side due to a shortening of the adductor muscle on the medial aspect of the thigh while the femoral head is displaced superiorly
♦ Swaying from side to side (waddling due to uncorrected bilateral dysplasia)
♦ Limpness due to uncorrected unilateral dysplasia
Complications
If corrective treatment isn’t begun until after age 2, DDH may cause:
♦ degenerative hip changes
♦ abnormal acetabular development
♦ lordosis (abnormally increased concave curvature of the lumbar and cervical spine)
♦ joint malformation
♦ sciatic nerve injury (paralysis)
♦ avascular necrosis of femoral head
♦ soft-tissue damage
♦ permanent disability.
Diagnosis
Diagnostic measures may include:
♦ X-rays (rarely used in neonates and infants) to show the location of the femur head and a shallow acetabulum (They also monitor disease or treatment progress.)
♦ sonography and magnetic resonance imaging to help with assessing reduction.
Observations during physical examination of the relaxed child that strongly suggest DDH include:
♦ the number of folds of skin over the thighs on each side when the child is placed on his back (a child in this position usually has an equal number of folds, but a child with subluxation or dislocation may have an extra fold on the affected side, which is also apparent when the child lies in a prone position)
♦ buttock fold on the affected side higher with the child lying in a prone position (also restricted abduction of the affected hip). (See Ortolani’s and Trendelenburg’s signs of DDH, page 378.)
Treatment
The earlier an infant receives treatment, the better the chances are for normal development. Treatment varies with the patient’s age.
In infants younger than 6 months, treatment includes:
♦ gentle manipulation to reduce the dislocation, followed by the use of a splint or harness
to hold the hips in a flexed and abducted position to maintain the reduction
to hold the hips in a flexed and abducted position to maintain the reduction
Ortolani’s and Trendelenburg’s signs of DDH
A positive Ortolani’s or Trendelenburg’s sign confirms developmental dysplasia of the hip (DDH).
Ortolani’s sign
♦ Place the infant on his back, with hip flexed and in abduction. Adduct the hip while pressing the femur downward. This will dislocate the hip.
♦ Then, abduct the hip while moving the femur upward. A click or a jerk (produced by the femoral head moving over the acetabular rim) indicates subluxation in an infant younger than 1 month. The sign indicates subluxation or complete dislocation in an older infant.
Trendelenburg’s sign
♦ When the child rests his weight on the side of the dislocation and lifts his other knee, the pelvis drops on the normal side because abductor muscles in the affected hip are weak.
♦ However, when the child stands with his weight on the normal side and lifts the other knee, the pelvis remains horizontal.
♦ a splint or harness worn continuously for 2 to 3 months, then a night splint for another month to tighten and stabilize the joint capsule in correct alignment.
If treatment doesn’t begin until after age 6 months, it may include:
♦ bilateral skin traction (in infants) or skeletal traction (in children who have started walking) to try to reduce the dislocation by gradually abducting the hips
♦ Bryant’s traction or divarication traction (both extremities placed in traction, even if only one is affected, to help maintain immobilization) for children younger than 3 years and weighing less than 35 lb (16 kg) for 2 to 3 weeks
♦ gentle closed reduction under general anesthesia to further abduct the hips, followed by a spica cast for 3 months (if traction fails)
♦ in children older than 18 months, open reduction and pelvic or femoral osteotomy to correct bony deformity followed by immobilization in a spica cast for 6 to 8 weeks
♦ between 6 and 12 months, immobilization in a spica cast for about 3 months.
In children ages 2 to 5, treatment is difficult and includes skeletal traction and subcutaneous adductor tenotomy (surgical cutting of the tendon).
Treatment begun after age 5 rarely restores satisfactory hip function.
Special considerations
The child who must wear a splint, brace, or body cast needs special personal care that requires parent education.
♦ Teach parents how to correctly splint or brace the hips as ordered. Stress the need for frequent checkups.
♦ Listen sympathetically to the parents’ expressions of anxiety and fear. Explain possible causes of developmental hip dislocation, and give reassurance that early, prompt treatment will probably result in complete correction.
♦ During the child’s first few days in a cast or splint, she may be prone to irritability because of the unaccustomed restricted movement. Encourage her parents to stay with her as much as possible and to calm and reassure her.
♦ Assure the parents that the child will adjust to this restriction and return to normal sleeping, eating, and playing behavior in a few days.
♦ Instruct the parents to remove braces and splints before bathing the infant but to replace them immediately afterward. Stress good hygiene; parents should bathe and change the child frequently and wash her perineum with warm water and soap at each diaper change.
If treatment requires a spica cast:
♦ When transferring the child immediately after casting, use your palms to avoid making dents in the cast. Such dents predispose the patient to pressure sores. Remember that the cast needs 24 to 48 hours to dry naturally. Don’t use heat to make it dry faster because heat also makes it more fragile.
♦ Immediately after the cast is applied, use a plastic sheet to protect it from moisture around the perineum and buttocks. Cut the sheet into strips long enough to cover the outside of the cast, and tuck them about a finger length beneath the cast edges. Using overlapping strips of tape, tack the corner of each petal to the outside of the cast. Remove the plastic under the cast every 4 hours; then wash, dry, and retuck it. Disposable diapers folded lengthwise over the perineum may also be used.
♦ Position the child either on a Bradford frame elevated on blocks, with a bedpan under the frame, or on pillows to support the child’s legs.
Be sure to keep the cast dry, and change the child’s diapers often.
Be sure to keep the cast dry, and change the child’s diapers often.
♦ Wash and dry the skin under the cast edges every 2 to 4 hours. Don’t use oils or powders; they can macerate skin.
♦ Turn the child every 2 hours during the day and every 4 hours at night. Check color, sensation, and motion of the infant’s legs and feet. Be sure to examine all her toes. Notify the physician of dusky, cool, or numb toes.
♦ Shine a flashlight under the cast every 4 hours to check for objects and crumbs. Check the cast daily for odors, which may herald infection.
♦ If the child complains of itching, she may benefit from taking diphenhydramine (Benadryl) or from having a hair dryer set on cool and aimed at the cast edges. Don’t scratch or probe under the cast. Investigate any persistent itching.
♦ Provide adequate nutrition and maintain adequate fluid intake to avoid renal calculi and constipation, both complications of inactivity.
♦ If the child is restless, apply a jacket restraint to keep her from falling out of bed or off the frame.
♦ Provide adequate stimuli to promote growth and development. If the child’s hips are abducted in a froglike position, tell parents that she may be able to fit on a tricycle that the parent can push (if the child is unable to pedal) or in a child’s electric car. Encourage parents to let the child sit at a table by seating her on pillows on a chair, to put her on the floor for short periods of play, and to let her play with other children her age.
♦ Tell parents to watch for signs and symptoms that the child is outgrowing the cast (cyanosis, cool extremities, or pain).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree