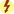http://evolve.elsevier.com/McCuistion/pharmacology/
Reproductive health requires the production of adequate quantities of various hypothalamic, pituitary, and gonadal hormones as well as the appropriate hormone receptors. It requires normal development and patency of the reproductive tract. In addition, reproductive health implies that men and women at developmentally appropriate life stages are fertile (i.e., able to produce gametes [sperm or eggs]). Finally, reproductive health entails the ability to engage in sexual intercourse with ejaculation by the male.
Alterations in male reproductive health reflect a wide range of developmental, endocrine, infectious, inflammatory, hypertrophic, malignant, and psychoemotional processes. Review the introduction to this unit to gain a better understanding of ways in which reproductive health is affected, including anatomy and physiology, sperm production, regulation of male sexual functioning, and sexual intercourse.
The drug family most clearly associated with male reproductive processes is the androgens. Because synthetic anabolic steroids and antiandrogens affect male reproduction, they are also discussed.
Drugs Related to Male Reproductive Disorders
Androgens
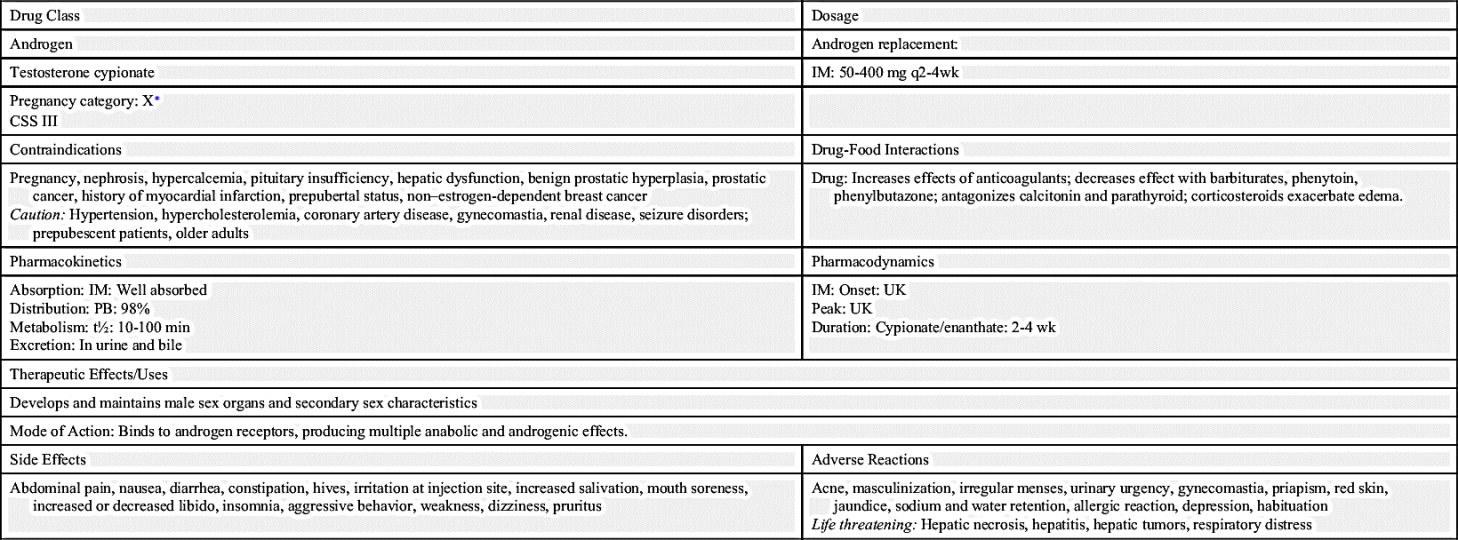
Androgens, or male sex hormones, control the development and maintenance of sexual processes, accessory sexual organs, cellular metabolism, and bone and muscle growth. Testosterone, an anabolic steroid, is the principal male sex hormone. It is the prototype of the androgen hormones, synthesized primarily in the testes and, to a lesser extent, in the adrenal cortex. In women, the ovaries synthesize small amounts of testosterone. In adult males, normal plasma concentrations of testosterone are 270 to 1070 ng/dL, with a slow decline of 1% per year after the age of 30 (Table 53.1). Prototype Drug Chart 53.1 lists the natural and synthetic androgens and their dosages, uses, and considerations.
TABLE 53.1
| Age | T Level (ng/dL) |
| 0-5 months | 75-400 |
| 6 mos.-9 yrs. | <7-20 |
| 10-11 yrs. | <7-130 |
| 12-13 yrs. | <7-800 |
| 14 yrs. | <7-1,200 |
| 15-16 yrs. | 100-1,200 |
| 17-18 yrs. | 300-1,200 |
| 19+ yrs. | 240-950 |
| Avg. Adult Male | 270-1,070 |
| 30+ yrs. | -1% per year |
From: http://www.mayomedicallaboratories.com/test-catalog/Clinical+and+Interpretive/83686
Pharmacokinetics
Testosterone secretion is greater in men than in women in most stages of life. About 98% of circulating testosterone is bound to both sex hormone–binding globulin (SHBG) and albumin protein, leaving about 2% unbound, or circulating free in the plasma; this unbound portion is biologically active. Estrogen elevates the production of SHBG, resulting in more protein-bound testosterone in women than in men. The half-life of endogenous, or naturally occurring, free testosterone in the blood is 10 to 20 minutes.
Because as much as 50% of testosterone is metabolized on its first pass through the hepatic circulation when taken orally, oral testosterone is not available for prescription in the United States. Testosterone can be combined with esters to form esterified testosterone in an oil base for intramuscular (IM) injection.
Ninety percent of testosterone is excreted in the urine as glucuronic and sulfuric acid conjugates and its metabolites. About 6% of the hormone is excreted unconjugated in the feces. Synthetic androgens may be excreted as unaltered hormone or as metabolites. In some tissues the action of testosterone depends on its reduction to 5-alpha-dihydrotestosterone (DHT), whereas in other tissues testosterone itself is the active hormone. In the central nervous system, the metabolite estradiol affects hormonal action.
Pharmacodynamics
Testosterone is responsible for the development of male sex characteristics. The biologic effects of testosterone may be mediated directly by testosterone or by its metabolites. Testosterone and dihydrotestosterone act as androgens by way of a single androgen receptor officially designated NR3A. The hormones bind to sites on certain responsive genes, causing a change to take place in the target cell. The effects of the testosterone depend on which receptor it activates and the tissues in which these effects occur. The manufacture of protein within the target cells results in the buildup of cellular tissue (anabolism), especially in muscles. This leads to development of secondary sex characteristics such as pubic hair growth, beard and body hair growth, baldness, deepening of the male voice, thickening of the skin, sebaceous gland activity, increased musculature, bone development, and red blood cell formation.
Fetal testes begin to produce testosterone during the first 3 months in utero. After birth until just before puberty, production is negligible. During puberty, production increases rapidly and continues until later adulthood. As men age, the number of Leydig cells decreases, sperm production declines, and luteinizing hormone (LH) and follicle-stimulating hormone (FSH) levels rise. Levels of unbound testosterone are reduced in older men to one third to one fifth the peak value. If men experience osteoporosis and anemia, and if their testosterone levels are 300 ng/dL or less, testosterone replacement therapy should be considered.
Indications for Androgen Therapy
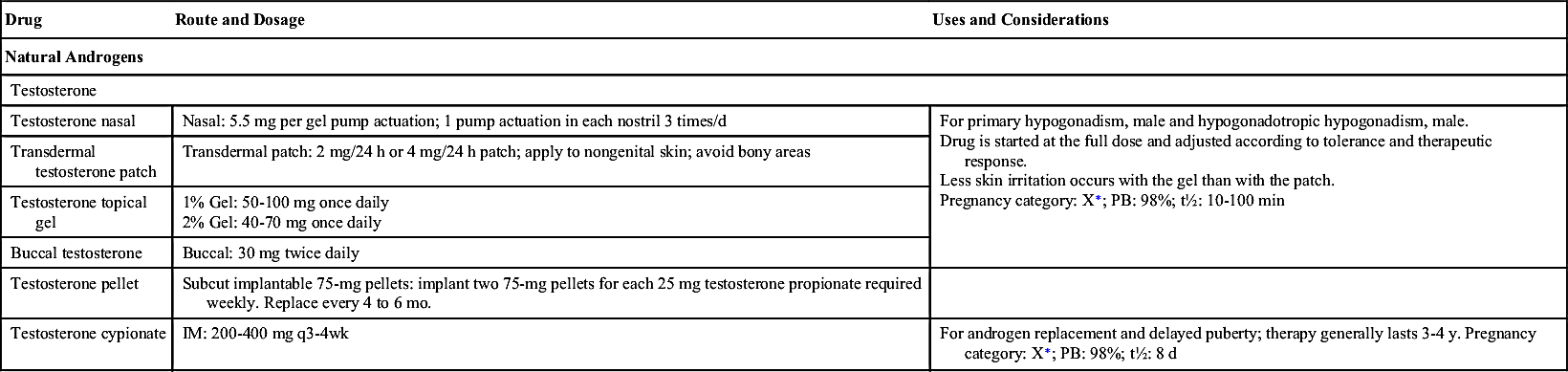
Table 53.2 lists the natural and synthetic androgens with their dosages, uses, and considerations. Androgen therapy is approved for use in androgen deficiency in males, specifically hypogonadism; replacement therapy for testicular failure in adult males; and delayed puberty in adolescents.
Hypogonadism
The clearest indication for exogenous androgen therapy is hypogonadism. Male hypogonadism is a defect of the reproductive system that results in failure of the testes to produce testosterone, sperm, or both. Deficiency of sex hormones can result in defective primary or secondary sexual development, and defective sperm development can result in infertility. Hypogonadism is either primary, reflecting testicular abnormality, or secondary, reflecting hypothalamic or pituitary failure. A combination of disorders can also occur. Inadequate pituitary function will severely affect young boys and results in infertility and a lack of secondary sex characteristics. Adult men may experience testicular atrophy, impotence, decreased libido, decreased bone density, loss of muscle mass, hair loss, gynecomastia, fatigue, difficulty concentrating, or vasomotor flushing.
The timing and extent of treatment depend on the clinical manifestations. Because accelerated bone maturation can lead to premature closure of bone epiphyses and short stature, androgen therapy should be used cautiously in children and only by specialists aware of the adverse effects on bone maturation. Skeletal maturation must be monitored every 6 months by radiography of the hand and wrist. Artificial induction of puberty is undertaken only after boys reach age 15 to 17 years and after hypothalamic and pituitary function has been assessed. A 4- to 6-month trial of androgen therapy is implemented, followed by a similar period of rest for reevaluation. If prolonged therapy is required, testosterone cypionate is used, 50 to 400 mg IM every 2 to 4 weeks. It should be given deep in the IM. Inspect vials visually for particulate matter and discoloration before administration, and warm and shake the vial to dissolve any crystals that may have formed during storage. It takes 3 or 4 years for sexual development to occur, and plasma testosterone levels should be monitored and dosages adjusted as needed to maintain normal levels; if the serum testosterone level is below the normal range, the provider will adjust the dose upward. Therapy may be lifelong.
Testosterone may be administered buccally, nasally, transdermally, or parenterally. Drug selection depends on the balance of growth and sexual maturation desired and on the preferred route of administration. A buccal mucoadhesive system is available that provides a 30-mg dose every 12 hours. Advise the patient to place the rounded surface of the system against the gum above an incisor tooth and hold it firmly in place with finger over the lip and against the product for 30 seconds. To remove, slide gently downward toward the tooth to avoid scratching the gums. Sites must be rotated with each application. If the product falls off within the 12-hour dosing interval, or if it falls out of position within 4 hours before next dose, remove it and apply a new system. The patient should not chew or swallow the system. Advise the patient to regularly inspect gums where the system has been applied. Testosterones are considered Schedule III controlled substances.
Transdermal testosterone (TT) patches achieve adequate serum concentrations when applied to the arm, back, or upper buttocks. TT patches can be applied to any healthy skin site other than the scrotum or bony areas. Daily application of one to two TT 2 mg/24 h or 4 mg/24 h skin patches at 10 PM results in serum testosterone concentrations approaching those of healthy young men. The first day of dosing results in morning serum testosterone concentrations within the normal range. There is no testosterone accumulation with continued use. After removal of TT patches, hypogonadal status returns within 24 hours. Keep testosterone gel out of reach of children.
Testosterone gel is applied to clean dry skin of shoulders or upper arms. It should not be applied to the genitals. Hands should be thoroughly washed with soap and water following application. Testosterone gel carries a boxed warning, as it can be transferred to others through personal contact with skin or clothing. Children should avoid contact with unwashed application sites or application sites not covered by clothing. Children can experience virilization from secondary exposure. Caution is advised. 

Side Effects
Hypogonadal men on androgen therapy may experience frequent erections or priapism (painful, continuous erection), gynecomastia (mammary gland enlargement in men), or urinary urgency. Continued use of androgens by normal men can halt spermatogenesis (formation of spermatozoa). The sperm count may be low (oligospermia) for 3 or more months after therapy is stopped.
Other side effects of androgen therapy include abdominal pain, nausea, insomnia, diarrhea or constipation, hives or redness at the injection site, increased salivation, mouth soreness, and increased or decreased sexual desire. Advise the patient to notify the health care provider if side effects persist, worsen, or are bothersome.
Adverse Reactions
Androgen therapy may cause hypercalcemia by stimulating bone resorption in immobilized patients and those with breast cancer. The drug should be discontinued and appropriate measures instituted if signs of hypercalcemia occur; signs include nausea and vomiting, lethargy, decreased muscle tone, polyuria, and increased urine and serum calcium.
Virilization refers to the development of male secondary sex characteristics in women or hypogonadal males. Such characteristics include growth of facial hair, acne and skin oiliness, and vocal huskiness. Menstrual irregularities or amenorrhea, suppressed ovulation or lactation, baldness or increased hair growth (hirsutism), and hypertrophy of the clitoris may develop in women undergoing androgen therapy. Although most adverse effects slowly reverse themselves after short-term therapy is completed, vocal changes may be permanent. With long-term therapy, as in the treatment of breast cancer, adverse effects may be irreversible.
Children may experience profound virilization as well as impaired bone growth. During pregnancy, androgens can cross the placenta and cause masculinization of the fetus. Virilization can occur in those secondarily exposed to testosterone gel and may cause teratogenic effects in fetuses. Women and children should not handle the gel and should avoid contact with application sites in men using testosterone gel.
Less frequent adverse effects include dizziness, weakness, changes in skin color, frequent headaches, confusion, respiratory distress, depression, pruritus, allergic skin rash, edema of the lower extremities, jaundice, bleeding, paresthesias, chills, polycythemia, muscle cramps, and sodium and water retention. Hepatocellular carcinoma can occur in patients who have received selected androgens for long-term therapy and in cases of abuse of androgenic hormones by athletes.
Serum cholesterol may become elevated during androgen therapy. Other alterations in laboratory tests include increased hematocrit, altered thyroid and liver function tests, and elevated urine 17-ketosteroids (a by-product of the breakdown of androgens). Rare complications of long-term therapy include hepatic necrosis, hepatic peliosis (blood-filled cysts), hepatic tumors, and leukopenia.
Contraindications
Androgen therapy is contraindicated during pregnancy and in individuals with nephrosis or those in the nephrotic phase of nephritis; it is also contraindicated in patients with hypercalcemia, pituitary insufficiency, hepatic dysfunction, benign prostatic hyperplasia (BPH), prostate cancer, or history of myocardial infarction. Men with breast cancer are not treated with androgens, nor are women whose breast cancer is not estrogen dependent.
Caution must be exercised when using androgen therapy in individuals with hypertension, hypercholesterolemia, coronary artery disease, renal disease, or seizure disorder. It is used with caution in infants and prepubertal children because of the potential for growth disturbances and in older men because of their increased risk for BPH and prostate cancer.
Drug Interactions
Androgens potentiate the effects of oral anticoagulants, necessitating a decrease in anticoagulant dosage. Androgens antagonize calcitonin and parathyroid hormones. Because androgens can decrease blood glucose in patients with diabetes, dosages of insulin or other antidiabetic agents may need to be reduced. Concurrent use of corticosteroids exacerbates the edema that can occur with androgen therapy. Barbiturates, phenytoin, and phenylbutazone decrease the effects of androgens.
Anabolic Steroids
Anabolic steroids, or anabolic-androgenic steroids (AASs), are a class of steroid hormones related to the hormone testosterone. They increase protein synthesis within cells, which results in the buildup of cellular tissue (anabolism), especially in muscles. Anabolic steroids also have androgenic and virilizing properties, including the development and maintenance of masculine characteristics such as the growth of the vocal cords and body hair. While the American College of Sports Medicine notes that AASs, combined with sufficient diet and exercise, can contribute to increased lean body mass in some individuals, they also note it is in the best interest of all sports to eradicate the use of AASs by athletes, due to the risk of serious harm or death in those who use AASs.  See Chapter 7, Drugs in Substance Use Disorder, for further information.
See Chapter 7, Drugs in Substance Use Disorder, for further information.
 See Chapter 7, Drugs in Substance Use Disorder, for further information.
See Chapter 7, Drugs in Substance Use Disorder, for further information.Testosterone precursors available as nutritional supplements include androstenediol, androstenedione, and dehydroepiandrosterone (DHEA). Older teens are the heaviest users, but more than one-half million junior high school students also use them. Marketed as “sport supplements” or “teen formulas,” they can be purchased without a prescription in stores and on the Internet. A sudden dramatic increase in weight and body size, increased acne, and changes in mood and behavior can be signs of exogenous anabolic steroid use. Individuals using anabolic steroids may become more aggressive and physical, and health risks can result from long-term use or excessive intake of anabolic steroids; these effects include increased low-density lipoprotein (bad) cholesterol and decreased high-density lipoprotein (good) cholesterol, acne, high blood pressure, liver damage, and dangerous changes in the structure of the left ventricle of the heart. Adverse effects may not be recognized until years later. See Chapter 7, Drugs in Substance Use Disorder, for further information.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree