Measles Virus
Diane E. Griffin
Measles is a highly contagious disease characterized by a prodromal illness of fever, coryza, cough, and conjunctivitis followed by the appearance of a generalized maculopapular rash. Introduction of measles into virgin populations and endemic transmission in populations with inadequate medical care are associated with high mortality. Despite the development of a successful live attenuated vaccine, measles remains a major cause of mortality in children, particularly in developing countries, and a cause of continuing outbreaks in industrialized nations.
History
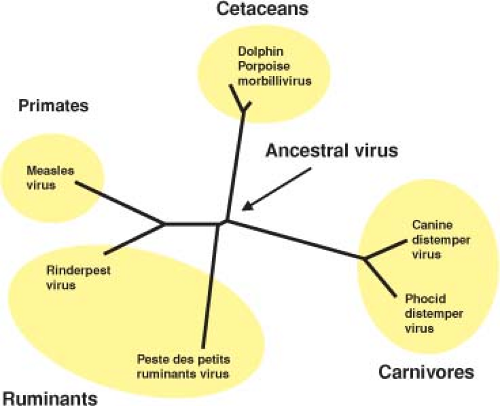
Measles is a relatively new disease of humans and probably evolved from an animal morbillivirus. Phylogenetically, measles virus (MeV) is most closely related to rinderpest virus (RPV), a pathogen of cattle (Fig. 36.1), and it is postulated that MeV evolved in an environment where cattle and humans lived in close proximity.444 Because large numbers of people are required to generate sufficient susceptible individuals to maintain measles in a population, measles may have evolved in the early centers of civilization in the Middle East where populations attained sufficient densities to sustain continued transmission.444
Abu Becr, an Arab physician known as Rhazes of Baghdad, is generally credited with distinguishing smallpox from measles in the 9th century. He dated its first description to the 6th century. Rhazes referred to measles as hasbah (eruption) and regarded it as a modification of smallpox with the distinction that “anxiety of mind, sick qualms and heaviness of heart oppress more in the measles than in the smallpox”.585 Repeated epidemics of illnesses characterized by a rash are recorded in European and Far Eastern populations between 1 and 1200 AD.444 It appears that measles spread across the Pyrenees into France with the Saracen invasion of the 8th century.600 Repeated epidemics identified as measles were recorded in the 11th and 12th centuries, and it is first mentioned as a childhood disease in 1224.444
In the European literature the name applied was “morbilli,” derived from the Italian “little diseases” to distinguish it from plague, “il morbo,” but morbilli included several exanthemata. Sanvages in 1763 defined morbilli as measles but called it rubeola (derived from the Spanish600), leading to a common confusion with rubella that persists to the present. Introduction of measles into previously unexposed populations has been associated with high morbidity and mortality.68,600,655,667 Epidemics of rash illnesses were associated with episodes of depopulation in China, India, and the Mediterranean region. Introduction of measles into the Fiji Islands in 1875 resulted in 26% mortality.600 Approximately 56 million people died as a result of European exploration of the New World, largely due to the introduction into native Amerindian populations of Old World diseases, notably smallpox and measles. Decreases in population are likely to have facilitated the transfer of Spanish culture to South America.444
Many of the basic principles of measles epidemiology were elucidated by Peter Panum, a Danish physician who worked in the Faroe Islands during a large measles epidemic in 1846.530 Panum deduced the highly contagious nature of the disease, the 14-day incubation period, and the lifelong immunity present in older residents and postulated a respiratory route of transmission.
Complications of measles were first described in the 18th century. In 1790, James Lucas, an English surgeon, described the first case of postmeasles encephalomyelitis in a young woman who developed paraparesis as the rash was fading.412 Nineteenth-century medical textbooks associated measles with the exacerbation of tuberculosis, and in 1908, while working at a tuberculosis hospital in Vienna, von Pirquet recorded the disappearance of
delayed-type hypersensitivity skin test responses to tuberculin,750 the first experimental evidence of measles-induced immune suppression.
delayed-type hypersensitivity skin test responses to tuberculin,750 the first experimental evidence of measles-induced immune suppression.
In 1933, Dawson described subacute sclerosing panencephalitis (SSPE) in a 16-year-old boy with progressive neurologic deterioration. Histologic examination of the brain showed eosinophilic intranuclear and intracytoplasmic inclusions in neurons and glial cells.142 After reports of paramyxovirus-like particles in the inclusions,717 observations of elevated levels of MeV antibody in serum and cerebrospinal fluid (CSF) and the reactivity of the inclusions with antibody to MeV identified the paramyxovirus as MeV.130
Infectious Agent
In 1757, an infectious agent was formally shown to be the cause of measles when the Scottish physician Francis Home, attempting immunization, transmitted the disease to naive individuals using blood taken from measles patients during the early stages of the rash.297 In 1905, Hektoen transmitted disease to volunteers with blood “free of bacteria” taken from measles cases in the acute stage and observed an incubation period of 13 days.280 In 1911, Goldberger and Anderson transmitted measles to rhesus macaques with filtered respiratory tract secretions from measles patients and successfully passaged disease from one monkey to another.238,531
Propagation and Assay in Cell Culture
Primary Isolation
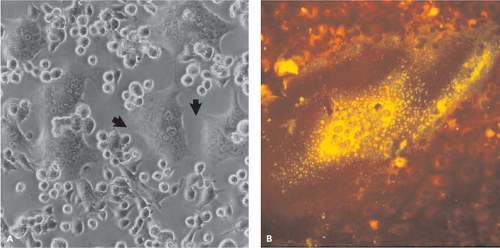
In 1954, Enders and Peebles first isolated MeV in tissue culture by inoculating primary human kidney cells with the blood of David Edmonston, a child with measles.184 Isolates were also made using primary monkey kidney cells184 and later continuous monkey kidney cell lines (e.g., Vero, CV-1).677 However, isolation of wild-type strains of MeV is most often successful using an Epstein-Barr virus–transformed marmoset B-lymphocyte line, B95-8365; a human T-cell line from cord blood, COBL-a364; or Vero cells engineered to express the MeV receptor signaling lymphocyte activation molecule (SLAM).521 Generally, the first observable sign of virus growth is cell–cell fusion and syncytia formation (Fig. 36.2).
Laboratory Propagation and Assay
Growth of MeV led to the development of live-attenuated vaccine strains by adaptation of MeV to growth in cells from foreign hosts, such as the chick embryo and canine and bovine kidney cells.349 Most experimental work is done with tissue culture–adapted strains so virus stocks are generally grown in Vero cells using a low multiplicity of infection to avoid accumulation of defective interfering (DI) particles. Vero and Vero/SLAM cells are useful for virus titration by plaque formation.10 The virus replicates slowly and 3 to 5 days of culture are often needed for plaques to become visible. Wild-type strains can also be assayed by syncytia formation in B95-8 or human cord blood mononuclear cells.
Biological Characteristics
The morbilliviruses form two genetically distinct groups of viruses related either to canine distemper virus (CDV) or to RPV (Fig. 36.1)50 and differ from other paramyxoviruses in formation of intranuclear inclusion bodies. Virions are pleomorphic and range in size from 100 to 300 nm. The envelope carries surface projections that are composed of the viral transmembrane hemagglutinin (H) and fusion (F) glycoproteins (Fig. 36.3). The matrix (M) protein lines the interior of the virion envelope. The helical ribonucleocapsid (total length of 1.2 μm) formed from the 16 kb genomic RNA wrapped with the nucleocapsid (N) protein is packed within the envelope in the form of a symmetrical coil with the phosphoprotein (P) and large polymerase (L) proteins attached.
Proteins
Nucleocapsid Protein
The N protein messenger RNA (mRNA) is the first transcribed from the genome and the N protein (525 amino acids [aa]) is the most abundant of the viral proteins. N appears as a 60-kD band on polyacrylamide gels and can self-assemble but usually surrounds viral genomic or antigenomic RNAs to form helical ribonucleocapsid structures.100 This conformationally flexible structure62,643 is the required template for both replication and transcription. Each N monomer binds six nucleotides, and viral genomes must be multiples of six for replication.367 Monomeric N (N0) can be transported into the nucleus but is usually retained in the cytoplasm by binding to P.309,679 Phosphorylation regulates oligomer formation and activation of transcription.243,267
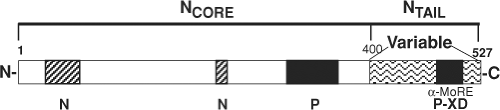
N is organized into two functionally distinct regions. The N-terminal portion of the protein (aa 1 to 400, NCORE) is conserved and is required for self-assembly into nucleocapsids and for RNA binding44,242,342,403,679 (Fig. 36.4). NCORE forms a globular domain located toward the helical axis of the nucleocapsid,643 includes a nuclear localization signal,628 and
can also react with the cell surface through FcγRII.579 The C-terminal portion of the protein (aa 401 to 525, NTAIL) is more variable, intrinsically disordered, acidic, and phosphorylated.40,110,267,341,408,567 N interacts with P through residues in NCORE and NTAIL.44,77,356,792 NTAIL is required for nucleocapsid flexibility,62 but its location in the nucleocapsid structure is not clear.151 NTAIL contains an α-helical molecular recognition element (α-MoRE, aa 488 to 499) reversibly involved in induced α-helical folding upon interaction with the X domain (XD) in the C-terminus of P (PCT) that likely positions the polymerase
complex near the RNA in the nucleocapsid.44,76,77,151,333,408,468 The intrinsic disorder of NTAIL provides a structural plasticity that allows strain- and cell type–specific interactions with several host proteins including heat shock protein (Hsp) 72, interferon regulatory factor (IRF)-3, the cellular protein responsible for nuclear export of N, the p40 subunit of eukaryotic initiation factor 3, cyclophilins A and B, and an unidentified cell surface nucleocapsid receptor.128,379,627,718,760,803
can also react with the cell surface through FcγRII.579 The C-terminal portion of the protein (aa 401 to 525, NTAIL) is more variable, intrinsically disordered, acidic, and phosphorylated.40,110,267,341,408,567 N interacts with P through residues in NCORE and NTAIL.44,77,356,792 NTAIL is required for nucleocapsid flexibility,62 but its location in the nucleocapsid structure is not clear.151 NTAIL contains an α-helical molecular recognition element (α-MoRE, aa 488 to 499) reversibly involved in induced α-helical folding upon interaction with the X domain (XD) in the C-terminus of P (PCT) that likely positions the polymerase
complex near the RNA in the nucleocapsid.44,76,77,151,333,408,468 The intrinsic disorder of NTAIL provides a structural plasticity that allows strain- and cell type–specific interactions with several host proteins including heat shock protein (Hsp) 72, interferon regulatory factor (IRF)-3, the cellular protein responsible for nuclear export of N, the p40 subunit of eukaryotic initiation factor 3, cyclophilins A and B, and an unidentified cell surface nucleocapsid receptor.128,379,627,718,760,803
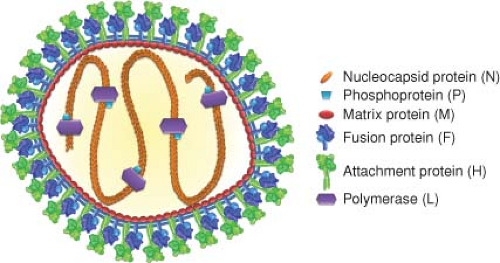 Figure 36.3. Schematic diagram of measles virus. The lipid bilayer of the pleomorphic particle is represented by blue lines, under which the matrix protein layer (red) resides. The viral membrane is densely packed with envelope glycoprotein complexes consisting of fusion protein trimers (shades of blue) and attachment protein tetramers (shades of green). The negative-strand RNA genome and the nucleocapsid protein (orange) form the nucleocapsid, which interacts with the phosphoprotein (light blue) and the polymerase (purple). In addition to contacts between the nucleocapsid and matrix proteins,325 the luminal tails of the glycoproteins are thought to contact the matrix layer. Individual viral components are not drawn to scale. Structural renderings of the glycoprotein complexes are based on original crystal structures (H head domains277); homology models of measles virus (MeV) F386,552 derived from coordinates reported for pre- and postfusion PIV5 and PIV3 F, respectively794,795; or hypothetical structural models (F prehairpin intermediate). High-resolution structural models were aligned at the level of the transmembrane domain (viral envelope) as described527 and then morphed into low-resolution images using the Sculptor (resolution 12, voxel size 3) package.64 (Adapted from Plemper RK, Brindley MA, Iorio RM. Structural and mechanistic studies of measles virus illuminate paramyxovirus entry. PLoS Pathog 2011;7:e1002058; courtesy of M. A. Brindley and R. K. Plemper.) |
P, C and V Proteins
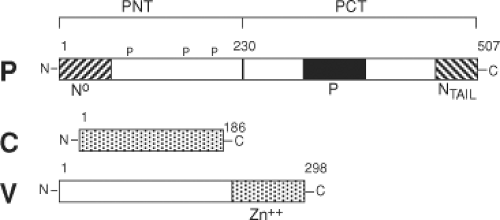
The P (phospho) protein (507 aa) is a polymerase co-factor that is activated by phosphorylation, forms tetramers, and links L to N to form the replicase complex.135 While the 72-kD P protein is abundant in the infected cell, only small amounts are present in the packaged virus. P is a multifunctional protein with a modular organization (Fig. 36.5). The N-terminus (PNT) is poorly conserved, intrinsically unstructured, acidic, phosphorylated, and required for replication. PNT binds to NCORE and this interaction induces folding of PNT.343 PNT is a chaperone for N0 that prevents binding to cellular RNAs, illegitimate N self-assembly, and nuclear translocation. PNT initiates encapsidation of genomic viral RNA by sequence-specific binding of the leader RNA.135,309 Elongation of the nucleocapsid structure is sequence independent. PNT also interacts with cellular proteins to regulate the response to interferon (IFN) (see discussion of V later).
PCT is conserved45 and contains all domains required for transcription. The region between amino acids 204 and 321 contains the α-helical domain that binds to NTAIL as part of the nucleocapsid structure and is responsible for tethering the polymerase L to its template.309,356 A coiled-coil domain between aa 344 to 411 is sufficient for P oligomerization.135 The unique XD portion of PCT (aa 459 to 507) has three α-helices arranged in an antiparallel triple helix bundle that binds the NTAIL α-MoRE with 1:1 stoichiometry and induces its folding.65,229,336,356,408 Binding affinity is weak, consistent with a model in which XD is responsible for tethering L to the ribonucleocapsid in a way that allows it to progress during RNA synthesis.356,792 XD also interacts with and stabilizes the ubiquitin ligase p53-induced-RING-H2.118
The P gene of MeV, like other members of the Paramyxoviridae, encodes proteins in addition to P that, together with P, regulate the innate response to infection (Fig. 36.5). The C protein is a basic protein of 186 amino acids encoded by the same mRNA but translated using an initiator methionine codon 19 nucleotides downstream from that for P and an overlapping reading frame.60 The V protein shares the initiator methionine and the amino terminal 231 amino acids of the P protein, but RNA editing adds an extra non-template-directed guanosine (G) residue at position 751. This shifts the reading frame and results in a different 68–amino acid cysteine-rich C-terminus with zinc-binding properties.105,404 Neither C nor V is necessary for MeV replication in Vero cells,572,636 but both C and V interact with cellular proteins and regulate the response to infection.155,405,646,722
C interferes with innate immune responses by inhibition of IFN signaling, modulates viral polymerase activity, and has been implicated in prevention of cell death.47,584,654,711 Deletion of C decreases MeV replication in monkeys, peripheral blood mononuclear cells (PBMCs), and thymic epithelial cells and decreases neurovirulence for CD46 transgenic mice, suggesting that C has an important in vivo role.155,194,537,711,731 In some cells decreased replication is associated with inhibition of translation and induction of IFN.491 C suppresses IFN induction by regulating viral RNA synthesis490 and preventing the activation of the cellular protein kinase PKR.435,723
Matrix (M) Protein
The envelope of the virion (Fig. 36.3) consists of the M protein (335 aa) and the two transmembrane glycoproteins F and H. M is a basic protein with several conserved hydrophobic domains.59 The mRNAs for morbillivirus M proteins contain approximately 400 nucleotides of noncoding sequence at the 3′
end that increases M protein production.59,705 In infected cells M is associated with nucleocapsids and with detergent-resistant regions of the inner layer of the plasma membrane where it regulates MeV RNA synthesis and assembly.291,325,556 M also interacts with the intracytoplasmic regions of one or both transmembrane glycoproteins, modulates the targeting and fusogenic capacity of the envelope glycoproteins, and directs release of virus from the apical surface of polarized epithelial cells.71,487,556,618 Deletion of M increases cell-to-cell fusion and decreases production of infectious virus.102 These properties are often defective in the mutated M proteins of viruses causing persistent infection.290,693
end that increases M protein production.59,705 In infected cells M is associated with nucleocapsids and with detergent-resistant regions of the inner layer of the plasma membrane where it regulates MeV RNA synthesis and assembly.291,325,556 M also interacts with the intracytoplasmic regions of one or both transmembrane glycoproteins, modulates the targeting and fusogenic capacity of the envelope glycoproteins, and directs release of virus from the apical surface of polarized epithelial cells.71,487,556,618 Deletion of M increases cell-to-cell fusion and decreases production of infectious virus.102 These properties are often defective in the mutated M proteins of viruses causing persistent infection.290,693
Fusion (F) Protein
F is a highly conserved type I transmembrane glycoprotein synthesized as an inactive precursor (Fo) of about 60 kD. The mRNAs for morbillivirus F0 proteins contain unusually long (460 to 585 nucleotides) G-C rich 5′ nontranslated regions (NTRs) that are predicted to have extensive secondary structure and are followed by clusters of three to four AUGs.588 The 5′ NTR influences the choice of AUG and decreases translation of F, virus production, and cytopathogenicity.101,705 There is a 28-residue signal sequence and after translation, F is glycosylated and trimerized in the endoplasmic reticulum (ER).550 F0 is cleaved at a multibasic site (108 to 112: Arg-Arg-His-Lys-Arg) by furin in the trans-Golgi to yield the 41-kD (F1) and 18-kD (F2) disulfide-linked fusion-competent mature protein.73,761 Mutation of Arg 112 results in a reduced rate of F transport to the cell surface, aberrant cleavage, and abolition of the fusogenic activity necessary for infection.13,761 Restricted processing of F is associated with persistent infection.447 The 33-residue cytoplasmic tail of F1 possesses basolateral sorting and endocytosis signals.461 F2 has all of the predicted N-linked glycosylation sites (aa 29, 61, and 67). Mutation of any of these asparagines decreases transport to the cell surface and impairs proteolytic cleavage, stability, and the fusion capacity of F, perhaps because F2 is an integral part of the prefusion F head.14,746
F1 contains a highly conserved stretch of hydrophobic amino acid residues at the new N-terminus (aa 113 to 145) that constitutes the fusion peptide. Oligopeptides that mimic this segment of F1 inhibit fusion.587 Mutants resistant to the fusion inhibitory effect of these oligopeptides have amino acid alterations in a cysteine-rich region (aa 337 to 381) of F1 important for interaction with H311 (Fig. 36.6). There are two predicted heptad repeat amphipathic α-helices: one adjacent to the fusion peptide and another N-terminal to the transmembrane region. Partial membrane-proximal cleavage between the heptad repeat and the transmembrane region enhances fusion.748 Synthetic peptides representing the heptad repeat regions inhibit fusion,381 as does mutagenesis of the leucines in the zipper region.83 F2 possesses a third heptad repeat region that modulates fusogenicity through a microdomain around residue 94.548
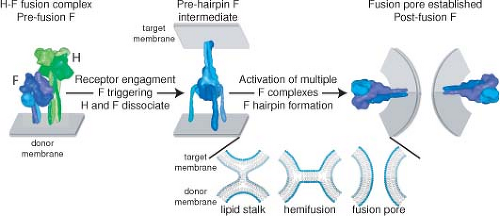 Figure 36.6. Schematic of measles virus (MeV) membrane fusion. MeV H and F envelope glycoproteins exist as hetero-oligomeric complexes on the surface of infectious viral particles (left panel). Receptor binding by the H protein triggers major conformational changes in prefusion F, resulting in insertion of the F fusion peptide domain into the target membrane in a hypothetical prehairpin intermediate conformation (center). Most likely, the concerted refolding of several prehairpin F complexes into the thermodynamically stable postfusion conformation is required to open a fusion pore and enable infection (right panel). For clarity, MeV H is represented as a single tetramer and F as a single trimer in the hetero-oligomeric fusion complex. More than one F trimer may interact with each individual H tetramer. The insert shows enlarged representations of hypothetical lipid mixing intermediates. Formation of a local fusion nipple is thought to be followed by merger of the outer lipid layers (lipid stalk; hemifusion stage) and ultimately merger of the inner lipid layers and the opening of a fusion pore. F complexes have been eliminated from the lipid mixing representation. Structural renderings were prepared as described in Figure 36.3. (Adapted from Plemper RK, Brindley MA, Iorio RM. Structural and mechanistic studies of measles virus illuminate paramyxovirus entry. PLoS Pathog 2011;7:e1002058; courtesy of M. A. Brindley and R. K. Plemper.) |
Fusion requires the expression of both H and F, with a predicted interaction of the F head with the H stalk (Fig. 36.6), and binding of H to a cell surface receptor (Fig. 36.7).385,527,802 Modeling of pre- and postfusion conformations of the F trimer indicates large conformational changes that result in formation of a six-helical bundle.568 Basolateral expression of H and F is important for syncytia formation and increases cell-to-cell spread in vitro and in vivo.461 In polarized epithelial cells transport is directed by interaction of the F luminal tail with M to the apical surface where virus is released.71,487 F tail mutations in viruses causing persistent infection include premature stop codons, missense mutations, altered reading frames, and
nonconservative amino acid substitutions.104,635 These changes interfere with virus envelope assembly and budding and increase cell-to-cell fusion and neurovirulence in hamsters.36,103
nonconservative amino acid substitutions.104,635 These changes interfere with virus envelope assembly and budding and increase cell-to-cell fusion and neurovirulence in hamsters.36,103
Hemagglutinin (H) Protein
H (617 aa) is the receptor-binding and hemagglutinating (HA) protein and an important determinant of morbillivirus cellular tropism. H is a type II transmembrane glycoprotein that resides on the surfaces of infected cells and virions as disulfide-linked homodimers that associate in the ER to form tetramers (Figs. 36.3 and 36.7).79,277,550 The mature H protein has a cytoplasmic tail of 34 amino acids preceding a single hydrophobic transmembrane region and a large C-terminal ectodomain with 13 strongly conserved cysteines. The cytoplasmic tail is essential for efficient transport to the cell surface and includes signals for basolateral sorting and endocytosis. However, H can be redirected to the apical surface for efficient particle formation and virus release.71,462,463
The H protein of the Edmonston strain of MeV has five predicted N-linked glycosylation sites clustered between positions 168 and 238 (Fig. 36.8). The first four of these sites are used.305 More recent MeV isolates often have an additional glycosylation site at residue 416, and this correlates with a loss of HA activity.615 Glycosylation is necessary for proper folding, antigenicity, dimerization, and export of H from the Golgi.305 H processing and intracellular transport is relatively slow, taking approximately 30 minutes for oligomerization and an hour to reach the medial Golgi.306 During persistent infections H proteins often accumulate mutations that affect glycosylation, oligomerization, and intracellular transport.104
Structural studies of H indicate that the N-terminus forms an α-helical stalk supporting a cubic-shaped six-blade β-propeller head structure (Fig. 36.7).127,276,385 Each of the blade modules contains four antiparallel β-strands connected sequentially through extended loops. In the dimer, N-linked carbohydrates cover the top pocket of the head domain and
cause the two molecules to tilt away from each other, optimizing exposure of neutralizing epitopes and the receptor-binding sites away from the dimer interface on the lateral surface (Fig. 36.8)276,308,749,751 (see section on Cellular Receptors). Cysteine residues at 139 and 154 are responsible for intermolecular disulfide bonding of monomeric H glycoproteins.549
cause the two molecules to tilt away from each other, optimizing exposure of neutralizing epitopes and the receptor-binding sites away from the dimer interface on the lateral surface (Fig. 36.8)276,308,749,751 (see section on Cellular Receptors). Cysteine residues at 139 and 154 are responsible for intermolecular disulfide bonding of monomeric H glycoproteins.549
H acts in conjunction with F for budding and for cell-to-cell fusion and entry (Fig. 36.6). Fusion occurs through conformational changes in both proteins triggered by the binding of H to a cellular receptor (Fig. 36.7).277,321,499,500,624 Hetero-oligomerization occurs in the ER.550 Mutagenesis has identified separate regions in the H stalk required for interacting with F and for triggering F fusion.385,527,547 MeV fusogenicity correlates inversely with the strength of the interaction between F and H133,134,189,321,551 (see section on Entry).
L Protein
The L (large) protein (2,183 aa) is a multidomain protein with several highly conserved regions. One contains the Gly-Asp-Asn-Gln motif common to the RNA polymerases of negative-strand viruses.174,669 L is present in small quantities in the infected cell, interacts with and functions in association with P, and is part of the viral nucleocapsid both in the cell and in the virion. A domain in the N-terminal 408 amino acids binds to a trihelical binding domain in PCT that links it to the nucleocapsid for transcription and replication.300,357
Cellular Receptors
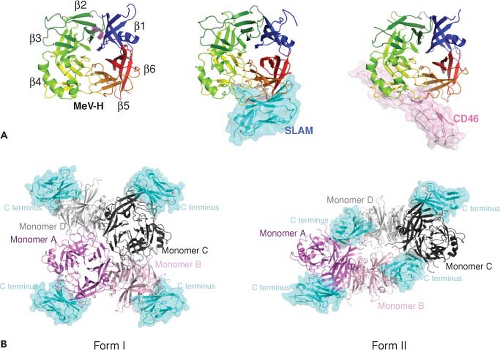
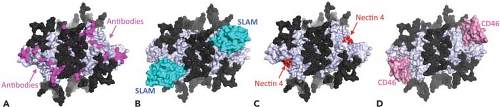
MeV can infect several types of cells and uses multiple receptors in a virus strain and cell type–specific manner. Three of these receptors have been identified: membrane co-factor protein or CD46,168,497 SLAM or CD150,715 and polio virus receptor–related 4 (PVRL4) or nectin 4.479,508 The binding sites for these cellular receptors are all found on the lateral surface of the head structure of H277,622 (Figs. 36.7 and 36.8).
CD46
CD46 is a widely distributed human complement regulatory protein expressed on all nucleated cells and preferentially on the apical surface of polarized epithelial cells.71,445 It normally acts as a co-factor in the proteolytic inactivation of C3b/C4b by factor I.591 Monkeys have a CD46 homolog that is expressed on erythrocytes, but such a protein has not been identified in mice. Multiple mRNAs are produced by alternative splicing of CD46 transcripts. All code for proteins that contain an N-terminal signal peptide, four short consensus repeats (SCRs), a transmembrane region, and an anchor. Isoforms differ in the length and composition of an extracellular serine/threonine/proline domain near the transmembrane segment and in having one of two alternative cytoplasmic tails.591 The cytoplasmic tail of CD46 is associated with intracellular kinases and adaptor proteins, and cross-linking of CD46 can induce autophagy and regulate inflammatory responses.338,591 The four isoforms common on human cells can all serve as receptors for MeV.231,423 SCR1 and SCR2 interact with the MeV H protein, while SCRs 2, 3, and 4 bind C3b/C4b.591 MeV infection of cells or expression of the H protein alone can lead to rapid internalization of CD46 from the cell surface.373 In persistently infected cells CD46 down-regulation is accomplished through a membrane-proximal Tyr-X-X-Leu motif in the cytoplasmic domain.791
The H binding site involves one planar face of SCR1 and SCR2 with an important role for the N-linked carbohydrates on SCR2.99,304,417 Most vaccine strains use CD46 efficiently, while wild-type strains often do not.191,790 A tyrosine at position 481 of H and glycine at 546 are key determinants of the affinity of H for CD46,53,660 but several additional residues are also important.432,603,637,695 The crystal structure of Edmonston H with SCR1 and SCR2 shows that CD46 binds to the side of the β-propeller through three contact regions on blades 4 and 5 (Figs. 36.7 and 36.8).622
SLAM
SLAM/CD150 is a 70-kD glycoprotein expressed on cells of the immune system including immature thymocytes, activated T and B lymphocytes, activated monocytes, and mature dendritic cells.124 SLAM is a member of a family of immunomodulatory type I transmembrane proteins93,153 and is the most important receptor for MeV infection of lymphoid tissue.143
A recombinant MeV that interacts inefficiently with SLAM is attenuated in macaques.389 CDV, RPV, and peste des petits ruminants virus also use SLAM as a receptor, suggesting that this is a common feature of morbilliviruses.5,49,716,749
A recombinant MeV that interacts inefficiently with SLAM is attenuated in macaques.389 CDV, RPV, and peste des petits ruminants virus also use SLAM as a receptor, suggesting that this is a common feature of morbilliviruses.5,49,716,749
SLAM has two highly glycosylated immunoglobulin-like domains (V and C2) and structural features of the CD2 family of membrane proteins.670,790 The cytoplasmic domain has immunoreceptor tyrosine-based switch motifs that bind small SH2 (src homology 2) domain adaptor proteins, such as SLAM-associated protein (SAP) and Ewing sarcoma–associated transcript-2 (EAT-2), important for cell signaling.93,153,515,670,790 MeV H binds to the V domain of human, but not mouse, SLAM,515 and this results in down-regulation of SLAM expression on the surface of infected cells.767 Mutagenesis studies have identified MeV H residues Ile194, Asp505, Asp507, Asp530, Arg533, Phe552, and Pro554 as important for binding SLAM.431,500,751 The crystal structure of H with the V domain of SLAM shows that these residues contribute to four components of the binding interface located primarily on the side of H blade 5 contiguous to the binding site for CD46277 (Fig. 36.7).
Studies of different strains of MeV have shown that both vaccine and wild-type strains can use SLAM as a receptor and that most H proteins can bind both CD46 and SLAM, but receptor affinity and efficiency of entry differ.191,422,432,521,621,637,751,790 In general, binding to SLAM is of higher affinity than binding to CD46.432 Viruses with asparagine at H481 use SLAM and enter PBMCs more efficiently than viruses with tyrosine at this position.191,637 Differences in efficient receptor usage likely involve interactions with MeV proteins in addition to H.370,710
Nectin 4
The distributions of SLAM and CD46 in tissues do not account for MeV replication in epithelial cells in vivo or in vitro.18,197,278,460,519,661,696,708,709,710 Recently, poliovirus receptor–like 4/nectin 4, an adherens junction protein of the immunoglobulin superfamily, has been identified as a receptor on epithelial cells.479,508 This is consistent with previous studies that indicated that an epithelial receptor is expressed on the basolateral surface of polarized cells and involved in formation of tight junctions.665,674 Nectin 4 is a transmembrane protein with two C2-type immunoglobulin domains and a V domain that interacts with H.479
Other Receptors
Several pieces of information suggest that MeV uses additional receptors. The currently known receptors do not account for the ability of MeV to infect endothelial cells in acute infections197 or cells of the central nervous system in chronic infections.445,661 Receptors used by attenuated vaccine strains adapted to growth in cells from nonsusceptible hosts (e.g., chickens) probably represent an additional category of MeV receptors that have yet to be identified.193
Other cell surface molecules interact with MeV but do not serve as entry receptors. For instance, MeV H can bind Toll-like receptor 2 (TLR2) and induce signaling.63 Dendritic cell–specific intercellular adhesion molecule 3 (ICAM-3) grabbing nonintegrin (DC-SIGN) is an attachment receptor that enhances infection and modulates function of DCs.31,147,263 Incorporation of cyclophilin B into MeV virions by binding to N leads to interaction with the cyclophilin ligand CD147/EMMPRIN, a multifunctional transmembrane protein expressed on epithelial and neural cells.760
Hemagglutination, Hemadsorption, and Hemolysis
Some strains of MeV bind to and agglutinate the erythrocytes of Old World monkeys, particularly African green, patas, and rhesus macaques. HA occurs optimally at physiologic pH and 37°C. Infected cells can also adsorb monkey erythrocytes (hemadsorption). Both HA and hemadsorption are properties of the H glycoprotein. Many wild-type MeV isolates require high salt or have little HA activity.594 HA of monkey erythrocytes is indicative of binding to CD46, is improved by adaptation to growth in Vero cells, and is dependent on the C-terminal 18 amino acids of H, amino acids 451 and 481, and absence of glycosylation at 416.616,630,660 This is consistent with the distribution of the CD46 molecule, which is not present on human red blood cells, and with the molecular characteristics of primate CD46.303 In baboons, lack of HA is due to an amino acid substitution in SCR2 and in New World monkeys to an absence of SCR1.303 HA is followed within a few hours by lysis of the agglutinated erythrocytes. Hemolysis is a consequence of fusion and dependent on F, as well as H.14,108,779
Entry
H attachment to a cellular receptor is followed by fusion of the virus envelope with the plasma membrane and delivery of the viral ribonucleocapsid into the cytoplasm for initiation of infection (Fig. 36.6). The H dimer of dimers (tetramer) associates with the prefusion F trimer in the secretory system of the host cell and exists as H-F hetero-oligomer on the virion surface (Fig. 36.3). The oligomers cooperate to induce fusion at neutral pH.516 Fusion requires H and F to be from compatible virus species and prior cleavage of F0 into F1 and F2.478 The MeV H protein stalk interacts directly with the MeV F protein head, suggesting that the metastable F trimer is shorter than the H tetramer, resulting in a staggered head domain arrangement on the virion surface (Figs. 36.3 and 36.6).385,527,547 The strength of H and F binding determines fusogenicity. More avid binding decreases fusion, indicating the need for H and F dissociation during the process of entry and productive infection.189 Separate regions on the H stalk have been identified for F interaction and for F triggering, suggesting that these are discrete functions.79,547 It is postulated that interaction of H with its receptor on the cell membrane induces a reorganization of the dimer–dimer head domains that transmit receptor binding to the F contact zone in the H stalk to trigger refolding of F and membrane fusion.277,499,624 This H reorganization may be represented in the crystal structure of H with SLAM that shows two forms of the H dimer with the orientation shifted with respect to each other277,547 (Fig. 36.7).
Cytopathic Effects
MeV replication in cell culture results in cytopathic changes of three varieties: multinucleated giant cells (syncytia), altered cell shape, and inclusion bodies.185 Cell-to-cell fusion occurs at neutral pH and syncytia formation occurs in vitro (Fig. 36.2) and in vivo184 presumably using fusion mechanisms similar to those for virus entry (Fig. 36.6). Syncytia formation is facilitated by basolateral expression of H and F and the actin filament–plasma membrane cross-linker moesin and is inhibited by cytochalasin B.167 Fusion of infected cells with uninfected cells may produce syncytia with 50 or more nuclei. Nuclei in the center of the syncytia have marginated chromatin184 and are often undergoing apoptotic cell death196 leading to plaque
formation in vitro. Infected cells may also change from a normal polygonal shape to a stellate, dendritic, or spindle shape with increased refractility to light. This type of “strand-forming” cytopathic effect appears after several passages and may be related to the production of DI particles.443
formation in vitro. Infected cells may also change from a normal polygonal shape to a stellate, dendritic, or spindle shape with increased refractility to light. This type of “strand-forming” cytopathic effect appears after several passages and may be related to the production of DI particles.443
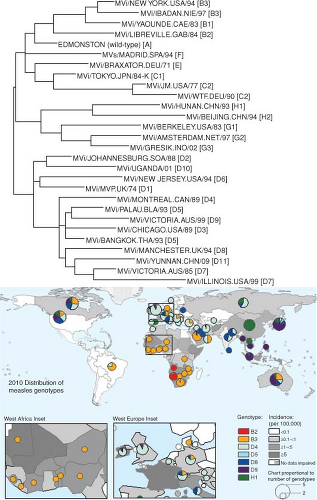 Figure 36.9. Genetic variation in wild-type measles viruses (MeV) and geographic distribution of MeV genotypes. The World Health Organization (WHO) currently recognizes 23 genotypes and one provisional genotype of wild-type MeV. The phylogenetic tree (top) is based on the sequences of the N genes of the WHO reference strains for each genotype775 and the provisional genotype.804 Map (bottom) shows the global distribution of MeV genotypes and measles incidence in 2010. Colored circles indicate MeV genotypes reported to the WHO database for the year 2010, and the size of the circles is proportional to the number of genotypes reported for the indicated areas. Two areas, Western Africa and Eastern Europe, are also shown as inserts to provide more resolution. The boundaries and names shown and the designations used on this map do not imply the expression of any opinion whatsoever on the part of the WHO concerning the legal status of any country, territory, city, or area or of its authorities, or concerning the delimitation of its frontiers of boundaries. Dotted lines on maps represent approximate borderlines for which there may not yet be full agreement. (Courtesy of David Featherstone, WHO, and Paul Rota, Centers for Disease Control and Prevention.) |
Both spindle-shaped cells and syncytial cells may contain intracytoplasmic and intranuclear inclusion bodies. Cytoplasmic inclusions are generally larger than nuclear inclusions and contain N-encapsidated RNA decorated with P, producing fuzzy or granular nucleocapsids.72 Intranuclear Cowdry type A inclusion bodies are characteristic of morbillivirus infections and occur late in infection. CDV intranuclear inclusion bodies are usually complex nuclear bodies derived from nucleoli that contain N and a cellular heat shock protein.511,513 MeV N, when expressed alone, migrates to the nucleus309,680 where it can assemble into nucleocapsids that lack P and viral RNA and appear “smooth” by electron microscopy.140,512 Because binding of P to assembled nucleocapsids leads to cytoplasmic retention,309 it has been
postulated that the amount of P may be limiting late in infection, allowing N to move into the nucleus.72,309
postulated that the amount of P may be limiting late in infection, allowing N to move into the nucleus.72,309
Budding
M plays a central role in virus assembly and release. In the absence of M, infectious particles are not released and expression of M alone leads to release of virus-like particles.102,109,556,611 To initiate virus assembly, M associates with the nucleocapsid, is co-transported to the plasma membrane,325 and interacts with the cytoplasmic domains of the F/H glycoprotein oligomers to promote virus budding.461,462,697 Proteins associate with detergent-resistant microdomains425,556,743 and budding is independent of the cellular endosomal sorting complex required for transport (ESCRT) system.618 In polarized epithelial cells, budding is directed to the apical surface by the M protein despite the intrinsic glycoprotein targeting to the basolateral surface,418,487 and loss of apical targeting by M enhances cell–cell fusion at the expense of virus production.102,103,610
Evolution, Antigenic Composition, and Strain Variation
Antigenically, MeV is a relatively stable virus. Antisera from individuals infected decades ago retain the ability to neutralize current wild-type strains of MeV and vice versa, although with varying efficiency.144,361,659 The observed rate of mutation of H in virus circulating in defined geographic locations is low, estimated at 5 × 10−4 per year for a given nucleotide,594 while the rate of mutation during growth in vitro is higher, estimated at 9 × 10−5 per replication for a nucleotide.644 Although historical accounts date the emergence of measles to approximately the 6th century (see History earlier), phylogenetic analysis of morbillivirus sequences suggests a more recent divergence from a common ancestor with RPV.222 However, this more recent estimate may reflect the effects on sequence evolution of population bottlenecks after outbreaks and purifying selection to maintain protein function.222,561,769 The structure of H with carbohydrates masking the top surface and exposed receptor-binding sites on the side (Fig. 36.8) is postulated to constrain acquisition of mutations.276,608 Evidence of vaccine-induced selective pressure on wild-type strains of MeV has been identified in the noose and receptor-binding regions of H.209,236,623,658,659,713
Nucleotide sequence variability, primarily in the N, P, and H genes, has been a useful tool for the MeV genotyping needed for molecular epidemiologic studies of transmission pathways.353,594,602 N genes differ by up to 7% in the C-terminal NTAIL region, the region most often used for strain identification82,236 (Fig. 36.4). The P gene is most variable in the shared PVNTD,45 and P gene sequencing has provided increased power to identify transmission routes when the NTAIL sequences are identical.353 The H gene nucleotide sequence is most variable between residues 167 and 241 where the N-linked glycosylation sites are located, but can become regionally fixed.353,602
Strains examined to date separate into eight different clades (A to H) and at least 24 different genotypes based on sequencing of the C-terminal 450 nucleotides of the N gene or the entire coding region of H590,605,606,775,804 (Fig. 36.9). New genotypes are designated if the nucleotide sequence differs from the closest reference sequence by more than 2.5% in N or 2.0% in H.776 Some genotypes are found in one geographic region, others are co-circulating, while others are inactive and may be extinct590 (Fig. 36.9). Live attenuated vaccines were all derived from genotype A wild-type strains and are quite similar.46
Pathogenesis and Pathology
Classic Measles
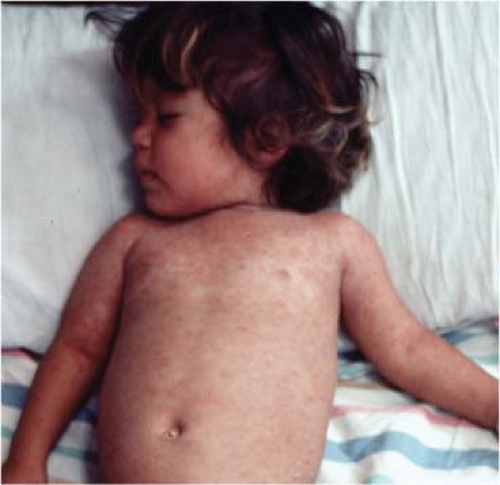
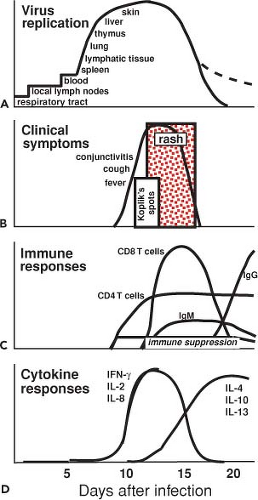
Measles is typically a childhood infection of humans spread by the respiratory route. Disease is characterized by a latent period of 10 to 14 days and a 2- to 3-day prodrome of fever, coryza, cough, and conjunctivitis followed by the appearance of a characteristic maculopapular rash (Fig. 36.10).362 The onset of the rash coincides with the appearance of the immune response and initiation of virus clearance (Fig. 36.11). Recovery is accompanied by lifelong immunity to reinfection.530 Macaques exposed to infected humans or experimentally infected with wild-type strains of MeV develop a similar disease, and much of our more detailed understanding of pathogenesis, immune responses, and sites of virus replication come from studies of nonhuman primates, often facilitated by the use of engineered reporter viruses.29,143,146,441,735
Entry and Sites of Primary Replication
MeV is efficiently transmitted over short distances by respiratory droplets and over longer distances by small-particle aerosols.119,617 High MeV infectivity suggests that the cellular sites of initial virus replication are very susceptible to infection. However, the nature of these cells is unclear because it has been difficult to identify MeV-positive cells in the respiratory tract at early times after infection.143,388 Although autopsy studies have shown abundant infection of respiratory epithelial cells,460,617 detailed studies of experimentally infected monkeys early after infection have only identified infected alveolar macrophages and subepithelial DCs.143,146,388 Because in vitro studies suggest that MeV infects epithelial cells from the basolateral
surface414,674 and MeV engineered not to infect respiratory epithelial cells can still initiate infection after intranasal inoculation,390 epithelial cell infection has been postulated to be a late, rather than an early, event. It is possible that pulmonary macrophages and DCs take up and transport MeV to local lymphoid tissue where virus is amplified, leading to viremia and subsequent systemic spread of infection to many tissues, including the lung.703
surface414,674 and MeV engineered not to infect respiratory epithelial cells can still initiate infection after intranasal inoculation,390 epithelial cell infection has been postulated to be a late, rather than an early, event. It is possible that pulmonary macrophages and DCs take up and transport MeV to local lymphoid tissue where virus is amplified, leading to viremia and subsequent systemic spread of infection to many tissues, including the lung.703
Spread
MeV interaction with DC-SIGN leads to up-regulation of SLAM on DCs, MeV entry,31 and likely transport from the respiratory tract to local lymphatic tissues in lung and draining lymph nodes.339,652,663 In vitro and in vivo studies suggest that DCs can transfer infection to susceptible T cells.143,148,663 Replication in lymphatic tissue is efficient, and infected CD150-expressing monocytes, T cells, and B cells are detected in peripheral blood within 4 to 7 days after infection.29,198,264,439,736 Only rarely has infectious virus been isolated from plasma,538 but viral RNA can be detected by reverse transcriptase-polymerase chain reaction (RT-PCR). MeV-infected mononuclear cells increase expression of the integrins LFA-1 (αLβ2) and VLA-4 (α4β1) and transmigratory cups that promote adherence to endothelial cells and cell-to-cell transmission of infection.26,166,313,413 These properties likely facilitate virus dissemination. The viremia is accompanied by leukopenia due either to death of infected cells or to changes in leukocyte trafficking.29,517,613
Target Cells and Tissues
From the blood, infection is spread to distal lymphoid tissue and to epithelial cells, endothelial cells, and macrophages in multiple organs.197,460 Transmigration across an endothelial barrier is impaired for MeV-infected lymphocytes,166 so entry of MeV into tissues may occur primarily from endothelial cells infected by circulating leukocytes or by movement of other types of infected cells, such as monocytes, across blood vessel walls.413 Once within tissue, spread is cell type and virus strain dependent and occurs by cell–cell fusion or by release of infectious virus. Tyrosine residues in the cytoplasmic tails of F (aa 549) and H (aa 12) are important for basolateral glycoprotein sorting and determine the fusogenic spread of MeV in epithelial cells.462,463 However, only the H sorting signal determines wild-type MeV release versus cell–cell fusion in lymphocytes.609,610
Lymphoid organs and tissues (e.g., thymus, spleen, lymph nodes, appendix, and tonsils) are prominent sites of virus replication617 where infection results in the appearance of lymphoid or reticuloendothelial giant cells first described by Warthin759 and Finkeldey.208 These cells can be 100 μm or more in diameter and contain up to 100 nuclei aggregated near the center. Inclusion bodies are not generally present. Warthin-Finkeldey cells tend to be located in or near germinal centers, in the thymus, and in submucosal lymphoid tissue.279,509 In the thymus, infection of epithelial cells and thymocyte apoptosis lead to a prolonged decrease in the size of the thymic cortex, while other lymphoid tissues recover promptly.772
MeV also spreads to the skin, conjunctivae, kidney, lung, gastrointestinal tract, respiratory mucosa, genital mucosa, and liver (Fig. 36.11). In these nonlymphoid sites the virus replicates primarily in endothelial cells, epithelial cells, and macrophages.197,198,460,699 Endothelial cell infection may be accompanied by vascular dilatation, increased vascular permeability, mononuclear cell infiltration, and infection of surrounding tissue.150 The histopathology of the measles rash suggests that the initial event is infection of dermal endothelial cells355 followed by spread of infection into the overlying epidermis with infection of keratinocytes in the stratum granulosum leading to focal keratosis and edema.699 Epithelial giant cells form and mononuclear cells accumulate around vessels.150 Koplik spots found on the oral mucosa are pathologically similar and involve the submucous glands.150
On rare occasions there is spread to the nervous system. In vitro studies have demonstrated infection of brain microvascular endothelial cells by adherent MeV-infected T lymphocytes,166 and infection of endothelial cells has been demonstrated in the brains of children dying of measles.197,358 Polarized endothelial cells can release virus from both the apical and basolateral cell surfaces, allowing access to the brain parenchyma, as well as the blood.166 If neurons become infected, virus can spread through the central nervous system (CNS) from neuron to neuron without the release of infectious particles.177 It has been suggested that the F protein interacts at the synapse with the substance P receptor neurokinin-1 to mediate transsynaptic spread.419
Immune Responses
The immune responses to MeV are important for clearance of virus and recovery from infection and are directly responsible for several of the clinical manifestations of measles. Although infectious virus cannot be isolated after the rash is cleared, viral RNA can be detected for many weeks, indicating that complete viral clearance is a prolonged process.529,542,734 The roles of various components of the immune response in recovery from infection have been deduced from experiments of nature in which the outcome of MeV infection in patients with deficiencies of immunologic function has been documented and from the studies of monkeys depleted of specific components of the immune system.244,539,540,542 In general, deficits in antibody production permit recovery, while deficits in cellular immune responses may lead to slowed clearance and progressive disease (see Measles in the Immunocompromised Host later). In immunologically normal individuals, the onset of clinically apparent disease coincides with the appearance of the MeV-specific adaptive immune response. There is also marked activation of the immune system that is coincident with the appearance of immune suppression (Fig. 36.11). Immune suppression and immune activation continue for many weeks after apparent recovery.
Early Innate Responses
Innate responses may contribute to control of virus replication during the incubation period, but determining the role and importance of specific components of the innate response in measles has been complicated. In vitro studies have shown that innate responses triggered by interaction of MeV RNA or proteins with pathogen recognition receptors at the cell surface or in the cytoplasm to activate signaling pathways involving transcription factors nuclear factor-κB (NFκB) and IRF-3 differ with the strain of virus, are cell type specific, and are highly regulated by the viral P, C, and V proteins.63,172,284,345,625,646,718 Epithelial cells show activation of NFκB and activator protein-1 (AP-1)320 and production of the chemokine CXCL8 (interleukin-8 [IL-8])629 after MeV infection. However, monocytes respond differently than epithelial cells, and interaction of H with TLR2 at the monocyte cell surface stimulates induction of IL-6 and increases surface expression of CD150,63 while interaction with CD46 inhibits IL-12 production.344 The NFκB pathway and tumor necrosis factor-α (TNF-α) production are suppressed in MeV-infected monocytes, potentially as a result of MeV P protein–induced up-regulation of the ubiquitin-modifying enzyme TNFAIP3 (A20), a negative regulator of NFκB.393,629,756
Some inflammatory cytokines and chemokines are induced in vivo during measles. Levels of IL-1β and IL-8 are increased in plasma of children during measles,629,807 and infected macaques show increases in IL-6 and IL-8.626 IL-1β mRNA and protein are increased in MeV-infected monocyte-derived cells and in PBMCs cultured from patients after rash onset.393,756 Transcriptional analysis of PBMCs from children with measles has shown increases in mRNAs for cytokines IL-1β and TNF-α and chemokines CCL4 (MIP-1β), CXCL2 (MIP-2α), and IL-8.807 The mRNA for CIAS-1 (NALP3), a component of the inflammasome responsible for processing proIL-1β to its active form, is also increased.807
Type I IFN is an important component of the innate response to many virus infections, and MeV replication is sensitive to the inhibitory effects of IFN-α/β.391,640,714 MeV replication is required for induction of IFN-β transcription in most responsive cells.288 Two induction mechanisms have been identified. In epithelial cells, MeV leader RNA can interact with and activate RIG-I and, to a lesser extent, MDA5,318,555 and N can interact with and activate IRF-3 in concert with an unidentified cellular co-factor.128,718 Induction of IFN by MeV may also occur at the cell surface through interaction of the virus with CD46 or TLR2.63,345 In vitro, MeV infection of epithelial cells and DCs leads to rapid production of IFN-β and many IFN-αs followed by induction of IFN-responsive genes.463,625,633,714,808 On the other hand, MeV infection of mitogen-stimulated PBMCs does not usually stimulate IFN production.498 In fact, MeV suppresses type I IFN production and signaling in CD4+ T cells625 and has a variable effect on plasmacytoid DC IFN production.169,633
Many of the reported effects of MeV on immune cell function in vitro are secondary to the effects of IFN. For instance, MeV induction of IFN inhibits development of DCs but stimulates maturation of immature DCs and terminal differentiation of cortical thymic epithelial cells.270,741 IFN also plays a role in suppressing proliferation of T cells in cultures of MeV-infected PBMCs.619 However, interpretation of investigations related to IFN induction and its role in measles pathogenesis has been confounded by the frequent presence of 5′ copy-back DI RNAs in the stocks of the virus strains studied.352,662 Vaccine strains are more likely to induce IFN-α/β than wild-type strains,498 but this may be related to the efficiency with which they generate DI RNAs,352,662,701 which are potent inducers of IFN through activation of MDA5.688,800
It is not clear whether IFN-α/β is induced during MeV infection in vivo. Transcriptional analysis of PBMCs during measles shows no evidence of up-regulation of IFN-induced genes.807 No IFN-α/β has been detected during natural infection in humans or experimental infection of macaques.259,262,476,664,801 Biologically active IFN has been detected occasionally, but IFN-γ produced by T cells is produced in response to infection, and the protein responsible for IFN activity (type I or II) was not identified.543,626 A recombinant wild-type MeV that cannot interfere with STAT1 translocation is attenuated in macaques, suggesting some role for this signaling pathway in the response to infection.156
Natural killer (NK) cells constitute another potentially important early defense mechanism, but studies of NK activity indicate that NK cell function is actually lower than normal during measles.262 These studies were performed using samples collected at, or after, the rash, so they do not exclude NK cell activation at earlier stages of infection.
MeV and MeV-infected cells activate the factor B–dependent alternative complement pathway, rendering the cells susceptible to complement-mediated lysis.675 This is a property of F1
and results in deposition of C3b on the virion and infected cell surface independent of virus use of the complement regulatory protein CD46 as a receptor.154 In infected cells, the complement regulators CD46 and CD55 are segregated into separate membrane microdomains from F.233
and results in deposition of C3b on the virion and infected cell surface independent of virus use of the complement regulatory protein CD46 as a receptor.154 In infected cells, the complement regulators CD46 and CD55 are segregated into separate membrane microdomains from F.233
Antibody
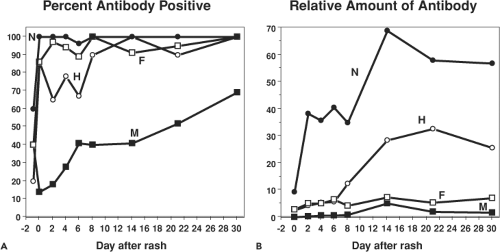
Antibodies are first detectable when the rash appears55,75,249 (Fig. 36.11). The isotype of MeV-specific antibody is initially IgM followed by a switch first to immunoglobulin G2 (IgG2) and IgG3 and then, in the memory phase, to IgG1 and IgG4.75,322 IgG is initially of low avidity and this improves steadily over several months.488,726 IgA, IgM, and IgG antibodies to MeV are found in secretions and sampling of saliva has provided a noninvasive method for determining immune status.81,317
Antibodies are eventually produced to most viral proteins (Fig. 36.12). The most abundant and most rapidly produced antibody is to N.249 Because of the abundance of anti-N antibody, absence of this antibody is an indicator of seronegativity. The M protein elicits only small amounts of antibody, except in atypical measles.249,415 Antibodies to H are the primary antibodies that neutralize virus infectivity.144,145,237 Neutralization is generally measured by plaque reduction of the Edmonston strain of MeV on CD46-expressing Vero cells,125 but this assay may not reflect neutralization of the infection of wild-type MeV strains on SLAM-expressing cells.559 Neutralizing epitopes have been mapped by competitive binding of monoclonal antibodies and by analysis of different strains and escape mutants (Fig. 36.8).307,312,396,658 Human convalescent sera show reactivity to linear epitopes, as well as to epitopes dependent on conformation and glycosylation.305,451,482 A highly conserved linear neutralizing epitope is in the H noose (aa 379 to 410).570 Major conformational epitopes have been localized to regions between amino acids 368 and 396 and in the SLAM-binding region.192,396 Essentially all of these epitopes are on exposed surfaces on the sides of H (Fig. 36.8).276,622 Antibodies to F induced by regions encompassing amino acid 73 and amino acids 388 to 402 contribute to virus neutralization, probably by preventing fusion of the virus membrane with the cell membrane at the time of virus entry.25,144,201,420,420,557 Human sera also recognize linear epitopes in six to seven regions spread over much of the F protein frequently close to T-cell epitopes.481,778
Antibody can protect from MeV infection, may contribute to recovery from infection, and may play a role in establishing persistent infection.9,186,577 Antibody-dependent cellular cytotoxicity correlates temporally with cessation of cell-associated viremia,212 and failure to mount an adequate antibody response carries a poor prognosis.770 However, in monkey studies transient depletion of B cells does not affect clearance of infectious virus.539 Antibody binding to infected cells alters intracellular virus replication and may contribute to control of infection.218,240,639 The role of antibody in protection from infection is discussed under Vaccination and the role in establishing persistence is discussed under Persistent Infection.
Cellular Immunity
The ability to recover from measles was postulated by Burnet to be an indication of the adequacy of T-lymphocyte–mediated immune responses,86 and depletion of CD8+ T cells in infected monkeys impairs control of virus replication and slows clearance.540 MeV-specific, proliferating, and clonally expanded CD8+ T cells are present in blood at the time of the rash and in bronchoalveolar lavage fluid during pneumonitis.331,464,476,485,732,757 IFN-γ, soluble CD8, and β2-microglobulin, a component of
the major histocompatibility complex (MHC) class I molecule, are increased in plasma.259,260,261,476 The H and M proteins can be processed and presented to CD8+ T cells in a transporter associated with antigen processing (TAP)-independent fashion.502 Cultures of PBMCs with autologous MeV-infected or MeV peptide-pulsed cells after recovery show expanded CD8+ T cells that are cytotoxic and produce IFN-γ, demonstrating that effector CD8+ T-cell memory is established by infection.331,332,492,732 CD8+ T-cell responses in humans show a broad pattern of reactivity with epitopes identified in all viral proteins, except V.289,330,331,523,737,738,739
the major histocompatibility complex (MHC) class I molecule, are increased in plasma.259,260,261,476 The H and M proteins can be processed and presented to CD8+ T cells in a transporter associated with antigen processing (TAP)-independent fashion.502 Cultures of PBMCs with autologous MeV-infected or MeV peptide-pulsed cells after recovery show expanded CD8+ T cells that are cytotoxic and produce IFN-γ, demonstrating that effector CD8+ T-cell memory is established by infection.331,332,492,732 CD8+ T-cell responses in humans show a broad pattern of reactivity with epitopes identified in all viral proteins, except V.289,330,331,523,737,738,739
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree