Chapter Ten Making it better
CHECK YOUR EXISTING KNOWLEDGE
There is no formal assessment of knowledge base for this section: either you do know how modalities function and how they achieve their physiological results or you don’t.
Ultrasound
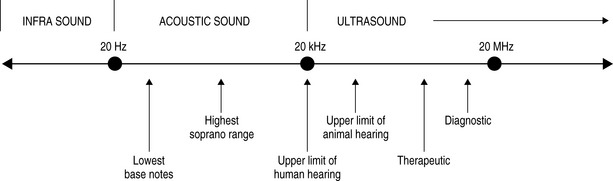
We have already discussed ultrasonography as a tool for diagnostic imaging; however, it is also the oldest and most widely used of the physical modalities. The term ultrasound refers to cyclic sound pressure with any frequency greater than that of the upper limit of human hearing. Although this limit varies between individuals and genders and decreases with age, it is approximately 20 kHz (20 000 hertz) in healthy young adults and this figure serves as a useful lower limit in describing ultrasound (Fig. 10.1).
As with all waves, the ultrasound wave has a wavelength and reciprocally related frequency. It also has amplitude and a velocity that will vary according to the density of the medium through which it is passing (Table 10.1).
Table 10.1 Velocity of ultrasound waves in selected human tissues
| Material | Velocity (ms−1) |
|---|---|
| Fat | 1450 |
| Water | 1480 |
| Soft tissue | 1540 |
| Bone | 4100 |
Once the beam enters the body, it can be absorbed, transmitted or reflected by the tissues therein. We have already dealt with this in regard to ultrasonographic imaging in Chapter 9; however, there are additional considerations for therapeutic usage.
In order to have a therapeutic effect, absorption of the applied energy is necessary; therefore the effectiveness of the modality will vary according to a tissue’s capacity to absorb the applied energy. The rate of absorption is related to protein content; tissues with a higher protein content (e.g. ligaments, tendons and scar tissue) will absorb ultrasound to a greater extent whilst tissues with high water and low protein content (e.g. blood and fat) absorb much less energy.