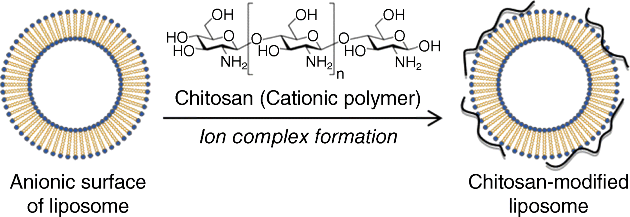
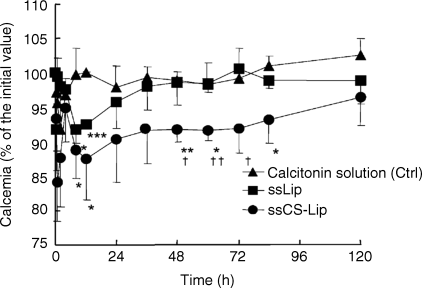
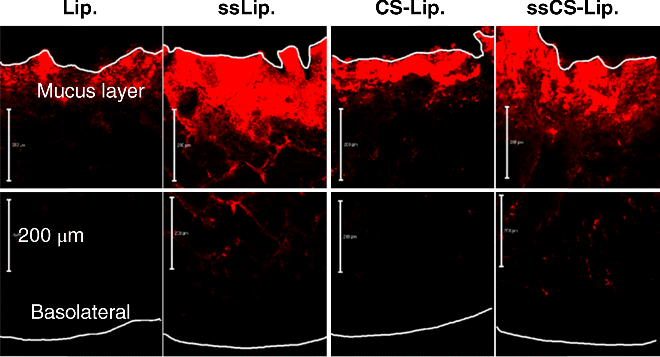
13 Kohei Tahara and Hirofumi Takeuchi Laboratory of Pharmaceutical Engineering Gifu Pharmaceutical University Japan Bioadhesion of the dosage forms of poorly absorbable drugs has received much attention; in the context of the gastrointestinal mucous membranes, this can be described in terms of mucoadhesion. Since mucoadhesion can prolong the residence time of drug carriers at absorption sites, improved drug absorption is expected from a combination of mucoadhesiveness and controlled drug release from such devices [1]. Colloidal drug carriers, such as liposomes or nanoparticles of biodegradable polymers, have received much attention for their ability to improve the absorption of poorly absorbable drugs, including peptides [2]. It has been reported that the mucoadhesive properties of these particulate systems can prolong their retention in the gastrointestinal tract, thus improving drug absorption. Liposomes are vesicles that comprise a phospholipid bilayer surrounding an aqueous compartment. Because of their biphasic characteristics and diversity in design, liposomes offer an adaptable function for improving drug absorption. Owing to the lipid domains of their bilayer membranes they may aid administration of lipophilic drugs. It has been reported that liposome encapsulation efficiency of lipophilic drugs depends on the physicochemical properties of the drug, such as lipophilicity [3], and on factors involving bilayer composition and the method of preparation. The research team at Gifu Pharmaceutical University has developed mucoadhesive liposomes for oral delivery of peptide drugs by modifying the anionic liposomal surface using the cationic mucoadhesive polymer chitosan (CS). The effectiveness of CS-modified liposomes (Lip) was confirmed with enhanced and prolonged pharmacological effects of insulin, which was orally administered to rats in polymer-coated liposomes [4]. Moreover, improved drug absorption was demonstrated using calcitonin as a model peptide drug. Carbopol (CP)-modified liposomes, which have mucoadhesive properties similar to that of CS–Lip, were as effective as CS–Lip in improving drug absorption [5]. In addition, the effects of particle size on the mucoadhesive properties of CS–Lip and on the pharmacological effect of entrapped calcitonin were evaluated. Recently, surface-modified liposomes for peptide delivery were applied to pulmonary administrations. For both local and systemic treatments, pulmonary drug delivery is one of the most promising noninvasive routes. It has several advantages over the other delivery routes, including large surface area, thin absorption barrier, low metabolic activity, avoidance of first-pass metabolism, decreased side effects and direct delivery of therapeutic agents to the site of action [6,7]. Local and systemic pulmonary delivery of several drugs, including small molecules, genes and protein/peptide drugs, has been investigated [8–10]. Many studies have been focused on local applications of small molecule and gene drugs to treat chronic respiratory diseases, such as lung cancer and asthma, and chronic obstructive pulmonary disease [11–13]. In addition, pulmonary application of protein/peptide drugs offers great potential for systemic drug delivery [14]. Subsequent therapeutic outcomes and pharmacodynamic effects are related to pulmonary bioavailability and lung deposition of inhaled therapeutic drugs [15]. Liposomes are one of the most extensively investigated systems for controlled delivery of drugs to the lung [16–18]. Inhaled liposomes protect drugs against the enzymatic degradation and result in significantly higher relative bioavailability compared with drug solutions [19]. Applications of surface-modified liposomes to oral and pulmonary delivery of peptide drugs are discussed in this chapter, focusing in particular on the pulmonary route. Oral administration is usually intended to deliver drugs to the systemic circulation or to exert local effects on mucosal membranes. A problem associated with the mucosal administration routes is the short residence time of the dosage form on mucosal membranes. Such limited contact times can lead to insufficient drug plasma levels and local effects. Chitosan is a mucoadhesive polysaccharide capable of opening the tight junctions between epithelial cells. Several properties of CS make it a good candidate for a mucoadhesive polymer coating, including nontoxicity, biocompatibility and biodegradability. Surface-modified liposomes with functional polymers are effective particulate drug carriers for transmucosal administration of drugs. They are easily prepared by mixing a liposomal suspension with polymers such as CP [5]. The polymer-modified liposomes were consecutively formed (Figure 13.1). The basic preparative mechanism for polymer-modified liposomes involves the formation of ion complexes on liposome surfaces. In the case of positively charged polymers, such as CS with the amino group, negatively charged liposomes are prepared and mixed with the CS solution. Subsequently, a coating layer is formed on the surface of the liposomes; this is confirmed and detected by measuring zeta potential. The zeta potential of liposomes was changed by increasing the concentration of oppositely charged coating polymers, which neutralize the surface charge of liposomes. In the case of the preparation of CP-modified liposomes, stearyl amine has been used to confer a positive charge to formulations of CP-modified liposomes. Figure 13.1 Preparation of chitosan-modified liposomes. Mucoadhesive liposomes were developed by coating anionic liposomal surfaces with the cationic mucoadhesive polymer, CS [2]. The effectiveness of the resulting CS–Lip was confirmed in the previous studies that revealed enhanced and prolonged pharmacological effects of insulin, which was orally administered to rats in the polymer-modified liposomal form. The effectiveness of mucoadhesive liposomes in drug absorption has been demonstrated using calcitonin as a model peptide drug. Carbopol-coated liposomes, with the mucoadhesive properties similar to that of CS–Lip, were as effective as CS–Lip. In addition, the effects of particle size on the mucoadhesive properties of CS-coated liposomes and on the absorption of entrapped calcitonin were evaluated. In these experiments, submicron-sized CS-coated liposomes (ssCS–Lip) exhibited excellent penetration into the intestinal mucosa and the pharmacological effect of calcitonin was sustained for up to 120 hours after oral administration to rats (Figure 13.2) [20]. Figure 13.2 Profiles of plasma calcium levels after intragastric administration of submicron-sized liposomes, ssLip and ssCS–Lip, containing calcitonin. Mean particles sizes of ssLip and ssCS–Lip were 196.4 and 473.4 nm, respectively. The formulation of liposomes was DSPC: DCP: Chol = 8 : 2 : 1. The concentration of chitosan for coating was 0.3%. Significant differences from calcitonin solution are indicated as follows: *p < 0.05, **p < 0.01 and ***p < 0.001; and from ssLip: †p < 0.05 and ††p < 0.01 (n = 3 in each case). Reprinted from [20] with permission from Elsevier. Characterization of the mucoadhesive properties of fine particulate systems in vivo is crucial. In a previous study, the team detected liposomes in mucosal layers of rat intestines using confocal laser scanning microscopy (CLSM) after administration of these particulate systems [20]. For this purpose, the fluorescence marker 1,1-dioctadecyl-3,3,3,3–tetramethylindo- carbocyanine perchlorate (DiI; LAMBDA, Austria) was formulated into liposome particles and the intestinal association and penetration of DiI-loaded liposomes was visualized using CLSM. After oral administration of DiI-loaded liposomes to rats, segments from duodenum, jejunum and ileum were isolated at the appropriate time and were cryofixed prior to mounting for CLSM imaging. Images were captured on both mucosal and basolateral sides of the intestinal membrane to evaluate the extent of mucosal penetration. Subsequently, mucoadhesion profiles of CS–Lip and ssCS–Lip in the intestinal tube were evaluated by determining residual liposomes on the mucosa; increased mucoadhesion of ssCS–Lip compared with CS–Lip was confirmed in comparisons of the resulting photographs (Figure 13.3). Although few CS–Lip particles were observed in the jejunum, large amounts of ssCS–Lip were detected there. In the ileum, retention of liposomal particles appeared almost the same compared to that of micro-sized CS–Lip regardless of their size, but ssCS–Lip tended to deeply penetrate into the mucosal part of the intestine. A similar size dependency was observed in measurements of the mucoadhesive properties of uncoated liposomes of various particle sizes (Lip and ssLip). Although the retained amount of uncoated liposomes was lower than that of CS–Lip, ssLip showed penetrative behaviour similar to that of ssCS–Lip. Thus, it was confirmed that ssLip had lower retention than ssCS–Lip, because only a small amount of ssLip was observed at the jejunum. The pharmacological effects of calcitonin administered with the liposomal formulations corresponded well with their intestinal retention profiles, as observed using CLSM. In fact, CLSM may be a promising method for characterization of the mucoadhesive properties of fine particle drug delivery systems, and may provide explanations for their effectiveness in oral administration of drugs. Figure 13.3 Mucopenetrative properties of various types of liposomes in the upper ileum at 60 min after intragastrical administration were indicated. The measured mean particle sizes of Lip, ssLip, CS–Lip, and ssCS–Lip were 7.56 μm, 224.7 nm, 3.58 μm and 281.2 nm, respectively. The formulation of liposomes was DSPC: DCP: Chol = 8 : 2 : 1 and the chitosan concentration for coating was 0.3%. Reprinted with permission from [20]. Copyright (2005) Elsevier Ltd. All rights reserved.
Liposome-Based Mucoadhesive Formulations
13.1 Introduction
13.2 Oral Administration of Surface-Modified Liposomes with the Mucoadhesive Properties
13.3 The Behaviour of Liposomes After Oral Administration
13.4 Pulmonary Administration of Peptide Drugs with Liposomal Formulations: Effective Surface Modification Using Chitosan or Poly(Vinyl Alcohol) with a Hydrophobic Anchor
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


