33
Host Defenses
CHAPTER CONTENTS
INTRODUCTION
Host defenses against viruses fall into two major categories: (1) nonspecific, of which the most important are interferons and natural killer cells; and (2) specific, including both humoral and cell-mediated immunity. Interferons are an early, first-line defense, whereas humoral immunity and cell-mediated immunity are effective only later because it takes several days to induce the humoral and cell-mediated arms of the immune response.
A description of how viruses evade our host defenses appears in Chapter 32.
NONSPECIFIC DEFENSES
1. Alpha & Beta Interferons
Alpha and beta interferons are a group of proteins produced by human cells after viral infection (or after exposure to other inducers). They inhibit the growth of viruses by blocking the synthesis of viral proteins. They do so by two main mechanisms: One is a ribonuclease that degrades mRNA, and the other is a protein kinase that inhibits protein synthesis.
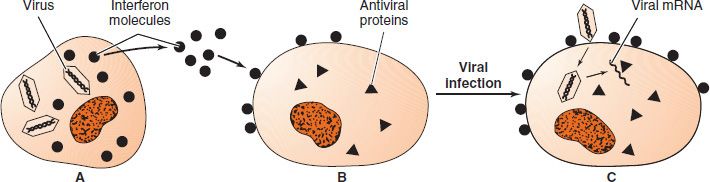
Interferons are divided into three groups based on the cell of origin, namely, leukocyte, fibroblast, and lymphocyte. They are also known as alpha, beta, and gamma interferons, respectively. Alpha and beta interferons are induced by viruses, whereas gamma (T cell, immune) interferon is induced by antigens and is one of the effectors of cell-mediated immunity (see Chapter 58). The following discussion of alpha and beta interferons focuses on the induction and action of their antiviral effect (Figure 33–1).
FIGURE 33–1 Induction and action of interferon. A: Virus infection induces the synthesis of interferon, which then leaves the infected cell. B: Interferon binds to the surface receptor of an uninfected cell and induces the synthesis of three new cell-encoded enzymes (antiviral proteins). C: A new virion enters the cell, but viral replication is inhibited by the interferon-induced antiviral proteins. One of these antiviral proteins is a ribonuclease that degrades mRNA, and another is a protein kinase that phosphorylates an initiation factor that inhibits protein synthesis. (Modified and reproduced with permission from Tortora G, Funk B, Case C. Microbiology: An Introduction. 5th ed. Benjamin/Cummings; 1995.)
Induction of Alpha & Beta Interferons
The strong inducers of these interferons are viruses and double-stranded RNAs. Induction is not specific for a particular virus; many DNA and RNA viruses are competent inducers, although they differ in effectiveness. The finding that double-stranded RNA, but not single-stranded RNA or DNA, is a good inducer has led to the conclusion that a double-stranded RNA is synthesized as part of the replicative cycle of all inducing viruses. The double-stranded RNA poly (rI-rC) is one of the strongest inducers and was under consideration as an antiviral agent, but toxic side effects prevented its clinical use. The weak inducers of microbiologic interest include a variety of intracellular bacteria and protozoa, as well as certain bacterial substances such as endotoxin.
This extensive list of inducers makes it clear that induction of these interferons is not specific. Similarly, their inhibitory action is not specific for any particular virus. However, they are typically specific in regard to the host species in which they act (i.e., interferons produced by human cells are active in human cells but are much less effective in cells of other species). It is clear, therefore, that other animals cannot be used as a source of interferons for human therapy. Rather, the genes for human interferons have been cloned, and interferon for medical use is now produced by genetic engineering techniques.
Action of Alpha & Beta Interferons
Interferons inhibit the intracellular replication of a wide variety of DNA and RNA viruses but have little effect on the metabolism of normal cells. The selectivity arises from the presence of double-stranded RNA in virus-infected cells, which is not present in uninfected cells.
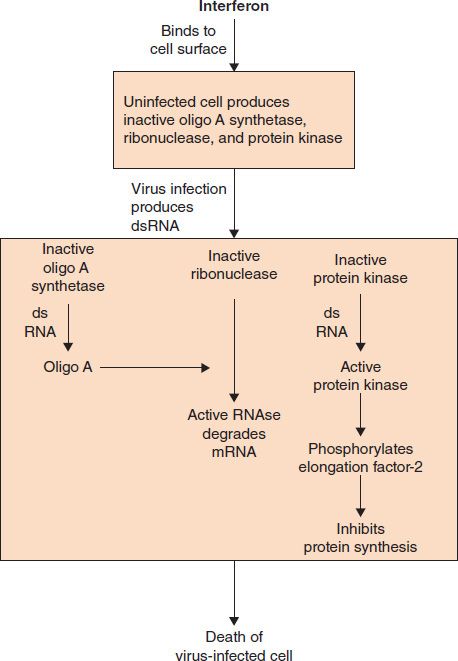
Interferons have no effect on extracellular virus particles. Interferons act by binding to a receptor on the cell surface that signals the cell to synthesize three proteins, thereby inducing the “antiviral state” (Figure 33–2). These three proteins are inactive precursor proteins until they are activated by double-stranded RNA synthesized during viral replication. As a result, these proteins are active in virus-infected cells but not in uninfected cells.
FIGURE 33–2 Interferon induces an antiviral state within an uninfected cell. Interferon binds to the surface of the uninfected cell and induces three proteins that remain inactive until a virus infects the cell. These proteins are oligo A synthetase, ribonuclease, and protein kinase. When a virus infects that cell, a double-stranded RNA (dsRNA) is synthesized as part of the viral replicative cycle. The dsRNA activates oligo A synthetase, which synthesizes oligo A that then activates the ribonuclease to degrade viral (and cell) mRNA. The dsRNA also activates the protein kinase that phosphorylates elongation factor-2. This inhibits both viral and cell protein synthesis. The cell dies without producing progeny virus, thereby limiting the spread of infection.
The three cellular proteins are (1) a 2,5-oligo A synthetase that synthesizes an adenine trinucleotide (2,5-oligo A), (2) a ribonuclease that is activated by 2,5-oligo A and degrades viral and cellular mRNAs, and (3) a protein kinase that phosphorylates an initiation factor (eIF-2) for protein synthesis, thereby inactivating it. The end result is that both viral and cellular protein synthesis is inhibited and the infected cell dies. No virus is produced by that cell, and the spread of the virus is reduced.
Because interferons are produced within a few hours of the initiation of viral replication, they act in the early phase of viral diseases to limit the spread of virus. In contrast, antibody begins to appear in the blood several days after infection.
Alpha interferon has been approved for use in patients with condyloma acuminatum and chronic active hepatitis caused by hepatitis B and C viruses. Beta interferon is used in the treatment of multiple sclerosis. Gamma interferon reduces recurrent infections in patients with chronic granulomatous disease (see Chapter 68). Interferons are also used clinically in patients with cancers such as Kaposi’s sarcoma and hairy cell leukemia.
2. Natural Killer Cells
Natural killer (NK) cells are an important part of the innate defenses against virus-infected cells. They are called “natural” killer cells because they are active without the necessity of being exposed to the virus previously and because they are not specific for any virus. NK cells are a type of T lymphocyte but do not have an antigen receptor. They recognize virus-infected cells by the absence of class I MHC (major histocompatibility complex) proteins on the surface of the virus-infected cell. They kill virus-infected cells by secreting perforins and granzymes, which cause apoptosis of the infected cells. (See page 501 for more information.)
3. Phagocytosis
Macrophages, particularly fixed macrophages of the reticuloendothelial system and alveolar macrophages, are the important cell types in limiting virus infection. In contrast, polymorphonuclear leukocytes are the predominant cellular defense in bacterial infections.
4. α-Defensins
α-Defensins are a family of positively charged peptides with antiviral activity. (They also have antibacterial activity; see Chapter 8.) They interfere with human immunodeficiency virus (HIV) binding to the CXCR4 receptor and block entry of the virus into the cell. The production of α-defensins may explain why some HIV-infected individuals are long-term “nonprogressors.”
5. Apolipoprotein B RNA-Editing Enzyme (APOBEC3G)
APOBEC3G is an important member of the innate host defenses against retroviral infection, especially against HIV. APOBEC3G is an enzyme that causes hypermutation in retroviral DNA by deaminating cytosines in both mRNA and retroviral DNA, thereby inactivating these molecules and reducing infectivity. HIV defends itself against this innate host defense by producing Vif (viral infectivity protein), which counteracts APOBEC3G, thereby preventing hypermutation from occurring.
6. Fever
Elevated body temperature may play a role in host defenses, but its importance is uncertain. Fever may act in two ways: (1) The higher body temperature may directly inactivate the virus particles, particularly enveloped viruses, which are more heat-sensitive than nonenveloped viruses; and (2) replication of some viruses is reduced at higher temperatures; therefore, fever may inhibit replication.
7. Mucociliary Clearance
The mucociliary clearance mechanism of the respiratory tract may protect the host. Its damage (e.g., from smoking) results in an increased frequency of viral respiratory tract infections, especially influenza.
8. Circumcision
There is evidence that circumcision prevents infection by three sexually transmitted viruses: HIV, human papillomavirus (HPV), and herpes simplex virus type 2 (HSV-2).
9. Factors That Modify Host Defenses
Several factors influence host defenses in a nonspecific or multifactorial way:
(1) Age is a significant variable in the outcome of viral infections. In general, infections are more severe in neonates and in the elderly than in older children and young adults. For example, influenza is typically more severe in older people than in younger adults, and herpes simplex virus infections are more severe in neonates than in adults.
(2) Increased corticosteroid levels predispose to more severe infections with some viruses, such as varicella-zoster virus; the use of topical cortisone in herpetic keratitis can exacerbate eye damage. It is not clear how these effects are mediated, because corticosteroids can cause a variety of pertinent effects, namely, lysis of lymphocytes, decreased recruitment of monocytes, inhibition of interferon production, and stabilization of lysosomes.
(3) Malnutrition leads to more severe viral infections (e.g., there is a much higher death rate from measles in developing countries than in developed ones). Poor nutrition causes decreased immunoglobulin production and phagocyte activity as well as reduced skin and mucous membrane integrity.
SPECIFIC DEFENSES
There is evidence for natural resistance to some viruses in certain species, which is probably based on the absence of receptors on the cells of the resistant species. For example, some people are resistant to HIV infection because they lack one of the chemokine receptors that mediate entry of the virus into the cell. However, by far the most important type of defense is acquired immunity, either actively acquired by exposure to the virus or passively acquired by the transfer of immune serum. Active immunity can be elicited by contracting the actual disease, by having an inapparent infection, or by being vaccinated.
1. Active Immunity
Active immunity, in the form of both antibodies and cytotoxic T cells, is very important in the prevention of viral diseases. The first exposure to a virus, whether it causes an inapparent infection or symptomatic disease, stimulates the production of antibodies and the activation of cytotoxic T cells. The role that antibodies and cytotoxic T cells play in the recovery from this first infection is uncertain and may vary from virus to virus, but it is clear that they play an essential role in protecting against disease when exposed to the same virus at some time in the future.
The duration of protection varies; disseminated viral infections such as measles and mumps confer lifelong immunity against recurrences, but localized infections such as the common cold usually impart only a brief immunity of several months. IgA confers protection against viruses that enter through the respiratory and gastrointestinal mucosa, and IgM and IgG protect against viruses that enter or are spread through the blood. The lifelong protection against systemic viral infections such as the childhood diseases measles, mumps, rubella, and chickenpox (varicella) is a function of the anamnestic (secondary) response of IgG. For certain respiratory viruses such as parainfluenza and respiratory syncytial viruses, the IgA titer in respiratory secretions correlates with protection, whereas the IgG titer does not. Unfortunately, protection by IgA against most respiratory tract viruses usually lasts less than 5 years.
The role of active immunity in recovery from a viral infection is uncertain. Because recovery usually precedes the appearance of detectable humoral antibody, immunoglobulins may not be important. Also, children with agammaglobulinemia recover from measles infections normally and can be immunized against measles successfully, indicating that cell-mediated immunity plays an important role. This is supported by the observation that children with congenital T-cell deficiency are vulnerable to severe infections with measles virus and herpesviruses. T cells are important in recovery from many but not all viral illnesses.
The protection offered by active immunity can be affected by the phenomenon of original antigenic sin. This term refers to the observation that when a person is exposed to a virus that cross-reacts with another virus to which that individual was previously exposed, more antibody may be produced against the original virus than against the current one. It appears that the immunologic memory cells can respond to the original antigenic exposure to a greater extent than to the subsequent one. This was observed in people with antibodies to the A1 type of influenza virus, who, when exposed to the A2 type, produced large amounts of antibody to A1 but very little antibody to the A2 virus. It is also the underlying cause of severe hemorrhagic dengue fever (see Chapter 42). This phenomenon has two practical consequences as well: (1) attempts to vaccinate people against the different influenza virus strains may be less effective than expected; and (2) epidemiologic studies based on measurement of antibody titers may yield misleading results.
How does antibody inhibit viruses? There are two main mechanisms. The first is neutralization of the infectivity of the virus by antibody binding to the proteins on the outer surface of the virus. This binding has two effects: (1) It can prevent the interaction of the virus with cell receptors, and (2) it can cross-link the viral proteins and stabilize the virus so that uncoating does not occur. The virus therefore cannot replicate. Furthermore, antibody-coated virus is more rapidly phagocytized than normal virus, a process similar to the opsonizing effect of antibody on bacteria. Antibody does not degrade the virus particle; fully infectious virus can be recovered by dissociating the virus–antibody complex. Incomplete, also called “blocking,” antibody can interfere with neutralization and form immune complexes, which are important in the pathogenesis of certain diseases. Some viruses, such as herpesviruses, can spread from cell to cell across intercellular bridges, eluding the neutralizing effect of antibody.
Antibodies that interfere with the adherence (adsorption and penetration) of viruses to cell surfaces are called neutralizing antibodies. Note that neutralizing antibody is directed against the surface proteins of the virus, typically the proteins involved with the interaction of the virus with receptors on the surface of the host cell. Antibodies formed against internal components of the virus (e.g., the core antigen of hepatitis B virus) do not neutralize the infectivity of the virus.
The second main mechanism is the lysis of virus-infected cells in the presence of antibody and complement. Antibody binds to new virus-specific antigens on the cell surface and then binds complement, which enzymatically degrades the cell membrane. Because the cell is killed before the full yield of virus is produced, the spread of virus is significantly reduced.
Lysis of virus-infected cells is also caused by cytotoxic T lymphocytes. These CD8-positive T cells recognize viral antigen only when it is presented in association with class I MHC proteins (see Chapter 58). They kill virus-infected cells by three methods: (1) by releasing perforins, which make holes in the cell membrane of the infected cells; (2) by releasing proteolytic enzymes called granzymes into the infected cell, which degrade the cell contents; and (3) by activating the FAS protein, which causes programmed cell death (apoptosis).
Not all virus infections induce antibodies. Tolerance to viral antigens can occur when the virus infection develops in a fetus or newborn infant. The model system in which tolerance has been demonstrated is lymphocytic choriomeningitis (LCM) infection in mice. If LCM virus is inoculated into a newborn mouse, the virus replicates widely, but no antibodies are formed during the lifetime of the animal. The virus is recognized as “self,” because it was present at the time of maturation of the immune system. If LCM virus is given to an adult mouse, antibodies are formed normally. There is no example of total tolerance to a virus in humans; even in congenital rubella syndrome, in which the virus infects the fetus, some antibody against rubella virus is made. However, virus production and shedding can go on for months or years.
Suppression of the cell-mediated response can occur during infection by certain viruses. The best-known example is the loss of tuberculin skin test reactivity during measles infection. Infection by cytomegalovirus or HIV can also cause suppression. Some viruses can “downregulate” (reduce) the amount of class I and class II MHC protein made by cells, which may be a mechanism by which these viruses suppress cell-mediated immunity.
2. Passive Immunity
Transfer of human serum containing the appropriate antibodies provides prompt short-term immunity for individuals exposed to certain viruses. The term passive refers to the administration of preformed antibodies. Two types of immune globulin preparations are used for this purpose. One has a high titer of antibody against a specific virus, and the other is a pooled sample from plasma donors that contains a heterogeneous mixture of antibodies with lower titers. The immune globulins are prepared by alcohol fractionation, which removes any viruses in the serum. The three most frequently used high-titer preparations are used after exposure to hepatitis B, rabies, and varicella-zoster viruses. Low-titer immune globulin is used mainly to prevent hepatitis A in people traveling to areas where this infection is hyperendemic.
Two specialized examples of passive immunity include the transfer of IgG from mother to fetus across the placenta and the transfer of IgA from mother to newborn in colostrum.
3. Herd Immunity
“Herd immunity” (also known as “community immunity”) is the protection of an individual from infection by virtue of the other members of the population (the herd) being incapable of transmitting the virus to that individual (Figure 33–3). Herd immunity can be achieved by immunizing a population with a vaccine that interrupts transmission, such as the live, attenuated polio vaccine, but not with a vaccine that does not interrupt transmission, such as the killed polio vaccine (even though it protects the immunized individual against disease). Note that herd immunity occurs with the live polio vaccine primarily because it induces secretory IgA in the gut, which inhibits infection by virulent virus, thereby preventing its transmission to others. In addition, the live virus in the vaccine can replicate in the immunized person and spread to other members of the population, thereby increasing the number of people protected. However, the important feature as far as herd immunity is concerned is the induction of IgA, which prevents transmission.
FIGURE 33–3 Herd immunity. Immunization of the nine people (tan color) can protect the one unimmunized person (red color) by interrupting transmission. Immunization levels of 90% are generally regarded as sufficient to protect the unimmunized individual.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree