57
Heavy Metal Intoxication & Chelators
CASE STUDY
A 48-year-old painter is referred for evaluation of recent onset of severe abdominal pains, headaches, and myalgias. For the last week, he has been removing old paint from an iron bridge using grinding tools and a blow torch. His employer states that all the bridge workers are provided with the equivalent of “hazmat” (hazardous materials) suits. What tests should be carried out? Assuming positive test results, what therapy would be appropriate?
Some metals such as iron are essential for life, whereas others such as lead are present in all organisms but serve no useful biologic purpose. Some of the oldest diseases of humans can be traced to heavy metal poisoning associated with metal mining, refining, and use. Even with the present recognition of the hazards of heavy metals, the incidence of intoxication remains significant, and the need for preventive strategies and effective therapy remains high. Toxic heavy metals interfere with the function of essential cations, cause enzyme inhibition, generate oxidative stress, and alter gene expression. As a result, multisystem signs and symptoms are a hallmark of heavy metal intoxication.
When intoxication occurs, chelator molecules (from chela “claw”), or their in vivo biotransformation products, may be used to bind the metal and facilitate its excretion from the body. Chelator drugs are discussed in the second part of this chapter.
 TOXICOLOGY OF HEAVY METALS
TOXICOLOGY OF HEAVY METALS
LEAD
Lead poisoning is one of the oldest occupational and environmental diseases in the world. Despite its recognized hazards, lead continues to have widespread commercial application, including production of storage batteries (nearly 90% of US consumption), ammunition, metal alloys, solder, glass, plastics, pigments, and ceramics. Corrosion of lead plumbing in older buildings or supply lines may increase the lead concentration of tap water. Environmental lead exposure, ubiquitous by virtue of the anthropogenic distribution of lead to air, water, and food, has declined considerably in the last three decades as a result of the elimination of lead as an additive in gasoline, as well as diminished contact with lead-based paint and other lead-containing consumer products, such as lead solder in cans used as food containers. Lead continues to be used in some formulations of aviation gasoline for piston-engine aircraft. The presence of lead in certain folk medicines (eg, the Mexican remedies azarcon and greta, and certain Ayurvedic preparations) and in cosmetics (eg, kohl utilized around the eyes in certain African and Asian communities) has contributed to lead exposure to children and adults. Although public health measures, together with improved workplace conditions, have decreased the incidence of serious overt lead poisoning, there remains considerable concern over the effects of low-level lead exposure. Extensive evidence indicates that low levels of lead exposure may have subtle subclinical adverse effects on neurocognitive function in children and may contribute to hypertension and cardiovascular disease in adults. Lead serves no useful purpose in the human body. In key target organs such as the developing central nervous system, no level of lead exposure has been shown to be without deleterious effects.
Pharmacokinetics
Inorganic lead is slowly but consistently absorbed via the respiratory and gastrointestinal tracts. It is poorly absorbed through the skin. Absorption of lead dust via the respiratory tract is the most common cause of industrial poisoning. The intestinal tract is the primary route of entry in nonindustrial exposure (Table 57–1). Absorption via the gastrointestinal tract varies with the nature of the lead compound, but in general, adults absorb about 10–15% of the ingested amount, whereas young children absorb up to 50%. Low dietary calcium, iron deficiency, and ingestion on an empty stomach all have been associated with increased lead absorption.
TABLE 57–1 Toxicology of selected arsenic, lead, and mercury compounds.
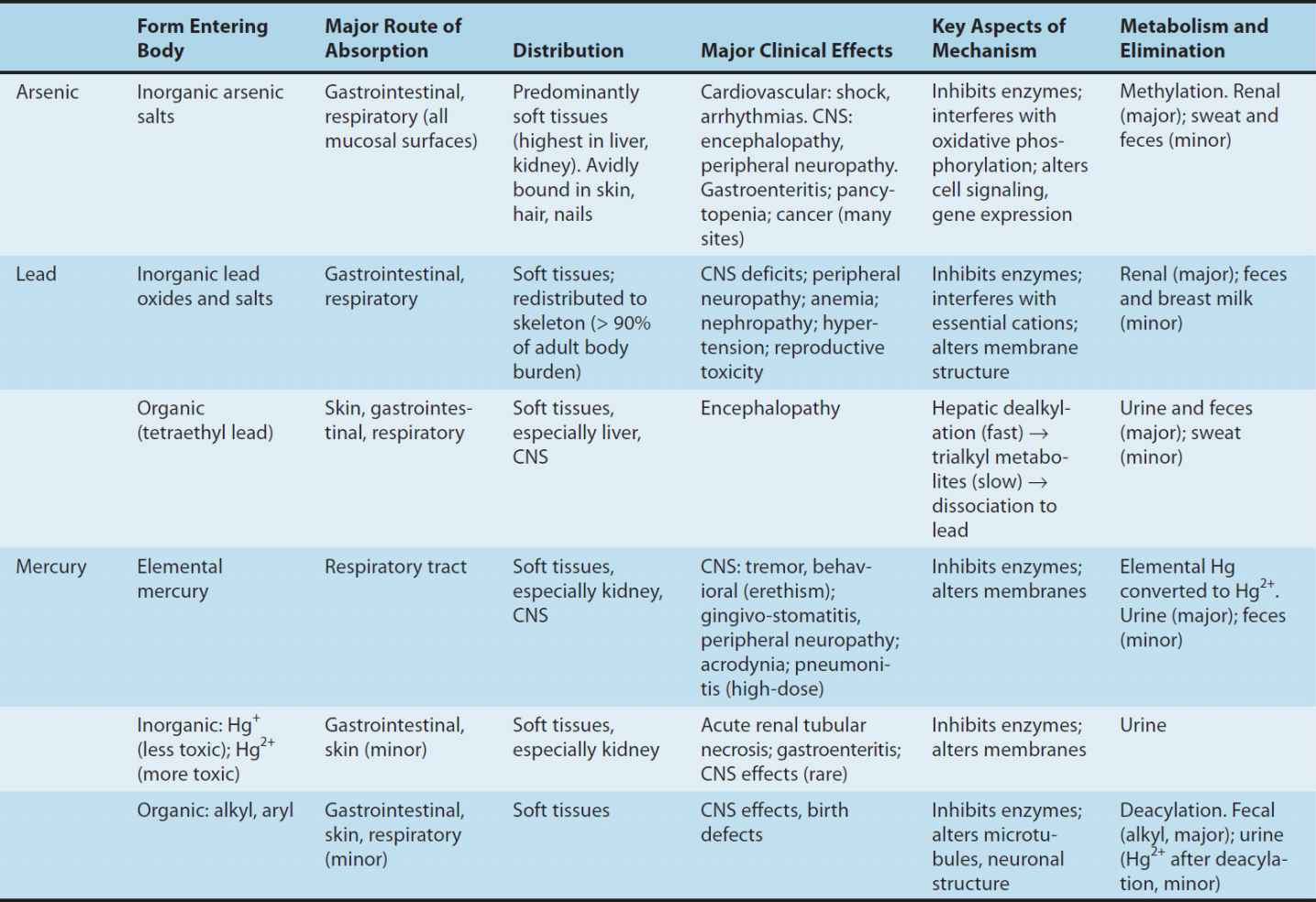
Once absorbed from the respiratory or gastrointestinal tract, lead enters the bloodstream, where approximately 99% is bound to erythrocytes and 1% is present in the plasma. Lead is subsequently distributed to soft tissues such as the bone marrow, brain, kidney, liver, muscle, and gonads; then to the subperiosteal surface of bone; and later to bone matrix. Lead also crosses the placenta and poses a potential hazard to the fetus. The kinetics of lead clearance from the body follows a multicompartment model, composed predominantly of the blood and soft tissues, with a half-life of 1–2 months; and the skeleton, with a half-life of years to decades. Approximately 70% of the lead that is eliminated appears in the urine, with lesser amounts excreted through the bile, skin, hair, nails, sweat, and breast milk. The fraction not undergoing prompt excretion, approximately half of the absorbed lead, may be incorporated into the skeleton, the repository of more than 90% of the body lead burden in most adults. In patients with high bone lead burdens, slow release from the skeleton may elevate blood lead concentrations for years after exposure ceases, and pathologic high bone turnover states such as hyperthyroidism or prolonged immobilization may result in frank lead intoxication. Migration of retained lead bullet fragments into a joint space or adjacent to bone has been associated with the development of lead poisoning signs and symptoms years or decades after an initial gunshot injury.
Pharmacodynamics
Lead exerts multisystemic toxic effects that are mediated by multiple modes of action, including inhibition of enzymatic function; interference with the action of essential cations, particularly calcium, iron, and zinc; generation of oxidative stress; changes in gene expression; alterations in cell signaling; and disruption of the integrity of membranes in cells and intracellular organelles.
A. Nervous System
The developing central nervous system of the fetus and young child is the most sensitive target organ for lead’s toxic effect. Epidemiologic studies suggest that blood lead concentrations less than 5 mcg/dL may result in subclinical deficits in neurocognitive function in lead-exposed young children, with no demonstrable threshold or “no effect” level. The dose response between low blood lead concentrations and cognitive function in young children is nonlinear, such that the decrement in intelligence associated with an increase in blood lead from less than 1 to 10 mcg/dL (6.2 IQ points) exceeds that associated with a change from 10 to 30 mcg/dL (3.0 IQ points).
Adults are less sensitive to the CNS effects of lead, but long-term exposure to blood lead concentrations in the range of 10–30 mcg/dL may be associated with subclinical effects on neurocognitive function. At blood lead concentrations higher than 30 mcg/dL, behavioral and neurocognitive signs or symptoms may gradually emerge, including irritability, fatigue, decreased libido, anorexia, sleep disturbance, impaired visual-motor coordination, and slowed reaction time. Headache, arthralgias, and myalgias are also common complaints. Tremor occurs but is less common. Lead encephalopathy, usually occurring at blood lead concentrations higher than 100 mcg/dL, is typically accompanied by increased intracranial pressure and may cause ataxia, stupor, coma, convulsions, and death. Recent epidemiological studies suggest that lead may accentuate an age-related decline in cognitive function in older adults. In experimental animals, developmental lead exposure, possibly acting through epigenetic mechanisms, has been associated with increased expression of beta-amyloid, increased phosphorylated tau protein, oxidative DNA damage, and Alzheimer’s-type pathology in the aging brain. There is wide interindividual variation in the magnitude of lead exposure required to cause overt lead-related signs and symptoms.
Overt peripheral neuropathy may appear after chronic high-dose lead exposure, usually following months to years of blood lead concentrations higher than 100 mcg/dL. Predominantly motor in character, the neuropathy may present clinically with painless weakness of the extensors, particularly in the upper extremity, resulting in classic wrist-drop. Preclinical signs of lead-induced peripheral nerve dysfunction may be detectable by electrodiagnostic testing.
B. Blood
Lead can induce an anemia that may be either normocytic or microcytic and hypochromic. Lead interferes with heme synthesis by blocking the incorporation of iron into protoporphyrin IX and by inhibiting the function of enzymes in the heme synthesis pathway, including aminolevulinic acid dehydratase and ferrochelatase. Within 2–8 weeks after an elevation in blood lead concentration (generally to 30–50 mcg/dL or greater), increases in heme precursors, notably free erythrocyte protoporphyrin or its zinc chelate, zinc protoporphyrin, may be detectable in whole blood. Lead also contributes to anemia by increasing erythrocyte membrane fragility and decreasing red cell survival time. Frank hemolysis may occur with high exposure. Basophilic stippling on the peripheral blood smear, thought to be a consequence of lead inhibition of the enzyme 3™,5™-pyrimidine nucleotidase, is sometimes a suggestive—albeit insensitive and nonspecific—diagnostic clue to the presence of lead intoxication.
C. Kidneys
Chronic high-dose lead exposure, usually associated with months to years of blood lead concentrations greater than 80 mcg/dL, may result in renal interstitial fibrosis and nephrosclerosis. Lead nephropathy may have a latency period of years. Lead may alter uric acid excretion by the kidney, resulting in recurrent bouts of gouty arthritis (“saturnine gout”). Acute high-dose lead exposure sometimes produces transient azotemia, possibly as a consequence of intrarenal vasoconstriction. Studies conducted in general population samples have documented an association between blood lead concentration and measures of renal function, including serum creatinine and creatinine clearance. The presence of other risk factors for renal insufficiency, including hypertension and diabetes, may increase susceptibility to lead-induced renal dysfunction.
D. Reproductive Organs
High-dose lead exposure is a recognized risk factor for stillbirth or spontaneous abortion. Epidemiologic studies of the impact of low-level lead exposure on reproductive outcome such as low birth weight, preterm delivery, or spontaneous abortion have yielded mixed results. However, a well-designed nested case-control study detected an odds ratio for spontaneous abortion of 1.8 (95% CI 1.1–3.1) for every 5 mcg/dL increase in maternal blood lead across an approximate range of 5–20 mcg/dL. Recent studies have linked prenatal exposure to low levels of lead (eg, maternal blood lead concentrations of 5–15 mcg/dL) to decrements in physical and cognitive development assessed during the neonatal period and early childhood. In males, blood lead concentrations higher than 40 mcg/dL have been associated with diminished or aberrant sperm production.
E. Gastrointestinal Tract
Moderate lead poisoning may cause loss of appetite, constipation, and, less commonly, diarrhea. At high dosage, intermittent bouts of severe colicky abdominal pain (“lead colic”) may occur. The mechanism of lead colic is unclear but is believed to involve spasmodic contraction of the smooth muscles of the intestinal wall, mediated by alteration in synaptic transmission at the smooth muscle-neuromuscular junction. In heavily exposed individuals with poor dental hygiene, the reaction of circulating lead with sulfur ions released by microbial action may produce dark deposits of lead sulfide at the gingival margin (“gingival lead lines”). Although frequently mentioned as a diagnostic clue in the past, in recent times this has been a relatively rare sign of lead exposure.
F. Cardiovascular System
Epidemiologic, experimental, and in vitro mechanistic data indicate that lead exposure elevates blood pressure in experimental animals and in susceptible humans. The pressor effect of lead may be mediated by an interaction with calcium-mediated contraction of vascular smooth muscle, as well as generation of oxidative stress and an associated interference in nitric oxide signaling pathways. In populations with environmental or occupational lead exposure, blood lead concentration is linked with increases in systolic and diastolic blood pressure. Studies of middle-aged and elderly men and women have identified relatively low levels of lead exposure sustained by the general population to be an independent risk factor for hypertension. Lead exposure has also been associated with prolongation of the QTc interval on the electrocardiogram. Of considerable public health concern, recent epidemiologic studies suggest that low to moderate levels of lead exposure are risk factors for increased cardiovascular mortality.
Major Forms of Lead Intoxication
A. Inorganic Lead Poisoning (Table 57–1)
1. Acute—Acute inorganic lead poisoning is uncommon today. It usually results from industrial inhalation of large quantities of lead oxide fumes or, in small children, from ingestion of a large oral dose of lead in the form of lead-based paint chips; small objects, eg, toys coated or fabricated from lead; or contaminated food or drink. The onset of severe symptoms usually requires several days or weeks of recurrent exposure and manifests as signs and symptoms of encephalopathy or colic. Evidence of hemolytic anemia (or anemia with basophilic stippling if exposure has been subacute) and elevated hepatic aminotransferases may be present.
The diagnosis of acute inorganic lead poisoning may be difficult, and depending on the presenting symptoms, the condition has sometimes been mistaken for appendicitis, peptic ulcer, biliary colic, pancreatitis, or infectious meningitis. Subacute presentation, featuring headache, fatigue, intermittent abdominal cramps, myalgias, and arthralgias, has often been mistaken for a flu-like viral illness. When there has been recent ingestion of lead-containing paint chips, glazes, pellets, or weights, radiopacities may be visible on abdominal radiographs.
2. Chronic—The patient with symptomatic chronic lead intoxication typically presents with multisystemic findings, including complaints of anorexia, fatigue, and malaise; neurologic complaints, including headache, difficulty in concentrating, and irritability or depressed mood; weakness, arthralgias, or myalgias; and gastrointestinal symptoms. Lead poisoning should be strongly suspected in any patient presenting with headache, abdominal pain, and anemia; and less commonly with motor neuropathy, gout, and renal insufficiency. Chronic lead intoxication should be considered in any child with neurocognitive deficits, growth retardation, or developmental delay. It is important to recognize that adverse effects of lead that are of considerable public health significance, such as subclinical decrements in neurodevelopment in children and hypertension in adults, are usually nonspecific and may not come to medical attention.
The diagnosis of lead intoxication is best confirmed by measuring lead in whole blood. Although this test reflects lead currently circulating in blood and soft tissues and is not a reliable marker of either recent or cumulative lead exposure, most patients with lead-related disease have blood lead concentrations higher than the normal range. Average background blood lead concentrations in North America and Europe have declined by 90% in recent decades, and the geometric mean blood lead concentration in the United States in 2009–2010 was estimated to be 1.12 mcg/dL. Though predominantly a research tool, the concentration of lead in bone assessed by noninvasive K X-ray fluorescence measurement of lead has been correlated with long-term cumulative lead exposure, and its relationship to numerous lead-related disorders is the subject of ongoing investigation. Measurement of lead excretion in the urine after a single dose of a chelating agent (sometimes called a “chelation challenge test”) primarily reflects the lead content of soft tissues and may not be a reliable marker of long-term lead exposure, remote past exposure, or skeletal lead burden. Accordingly, this test is rarely indicated in clinical practice. Because of the lag time associated with lead-induced elevations in circulating heme precursors, the finding of a blood lead concentration of 30 mcg/dL or more with no concurrent increase in zinc protoporphyrin suggests that the lead exposure was of recent onset.
B. Organolead Poisoning
Poisoning from organolead compounds is now very rare, in large part because of the worldwide phase-out of tetraethyl and tetra-methyl lead as antiknock additives in gasoline. However, organolead compounds such as lead stearate or lead naphthenate are still used in certain commercial processes. Because of their volatility or lipid solubility, organolead compounds tend to be well absorbed through either the respiratory tract or the skin. Organolead compounds predominantly target the CNS, producing dose-dependent effects that may include neurocognitive deficits, insomnia, delirium, hallucinations, tremor, convulsions, and death.
Treatment
A. Inorganic Lead Poisoning
Treatment of inorganic lead poisoning involves immediate termination of exposure, supportive care, and the judicious use of chelation therapy. (Chelation is discussed later in this chapter.) Lead encephalopathy is a medical emergency that requires intensive supportive care. Cerebral edema may improve with corticosteroids and mannitol, and anticonvulsants may be required to treat seizures. Radiopacities on abdominal radiographs may suggest the presence of retained lead objects requiring gastrointestinal decontamination. Adequate urine flow should be maintained, but overhydration should be avoided. Intravenous edetate calcium disodium (CaNa2EDTA) is administered at a dosage of 1000–1500 mg/m2/d (approximately 30–50 mg/kg/d) by continuous infusion for up to 5 days. Some clinicians advocate that chelation treatment for lead encephalopathy be initiated with an intramuscular injection of dimercaprol, followed in 4 hours by concurrent administration of dimercaprol and EDTA. Parenteral chelation is limited to 5 or fewer days, at which time oral treatment with another chelator, succimer, may be instituted. In symptomatic lead intoxication without encephalopathy, treatment may sometimes be initiated with succimer. The end point for chelation is usually resolution of symptoms or return of the blood lead concentration to the premorbid range. In patients with chronic exposure, cessation of chelation may be followed by an upward rebound in blood lead concentration as the lead re-equilibrates from bone lead stores.
Although most clinicians support chelation for symptomatic patients with elevated blood lead concentrations, the decision to chelate asymptomatic subjects is more controversial. Since 1991, the Centers for Disease Control and Prevention (CDC) has recommended chelation for all children with blood lead concentrations of 45 mcg/dL or greater. However, a randomized, double-blind, placebo-controlled clinical trial of succimer in children with blood lead concentrations between 25 mcg/dL and 44 mcg/dL found no benefit on neurocognitive function or long-term blood lead reduction. Prophylactic use of chelating agents in the workplace should never be a substitute for reduction or prevention of excessive exposure.
Management of elevated blood lead levels in children and adults should include a conscientious effort to identify and reduce all potential sources of future lead exposure. Many local, state, or national governmental agencies maintain lead poisoning prevention programs that can assist in case management. Blood lead screening of family members or coworkers of a lead poisoning patient is often indicated to assess the scope of the exposure. In 2012, the CDC adopted a new policy that recognized childhood blood lead concentrations at or exceeding a reference value of 5 mcg/dL to be elevated and to merit clinical follow-up and environmental investigation. Although the US Occupational Safety and Health Administration (OSHA) lead regulations introduced in the late 1970s mandate that workers be removed from lead exposure for blood lead levels higher than 50–60 mcg/dL, an expert panel in 2007 recommended that removal be initiated for a single blood lead level greater than 30 mcg/dL, or when two successive blood lead levels measured over a 4-week interval are 20 mcg/dL or more. The longer-term goal should be for workers to maintain blood lead levels less than 10 mcg/dL, and for pregnant women to avoid occupational or avocational exposure that would result in blood lead levels higher than 5 mcg/dL. Environmental Protection Agency (EPA) regulations effective since 2010 require that contractors who perform renovation, repair, and painting projects that disturb lead-based paint in pre-1978 residences and child-occupied facilities must be certified and must follow specific work practices to prevent lead contamination.
B. Organic Lead Poisoning
Initial treatment consists of decontaminating the skin and preventing further exposure. Treatment of seizures requires appropriate use of anticonvulsants. Empiric chelation may be attempted if high blood lead concentrations are present.
Arsenic
Arsenic is a naturally occurring element in the earth’s crust with a long history of use as a constituent of commercial and industrial products, as a component in pharmaceuticals, and as an agent of deliberate poisoning. Recent commercial applications of arsenic include its use in the manufacture of semiconductors, wood preservatives for industrial applications (eg, marine timbers or utility poles), nonferrous alloys, glass, herbicides, and nitarsone, an organoarsenical pharmaceutical used in certain poultry. In some regions of the world, groundwater may contain high levels of arsenic that has leached from natural mineral deposits. Arsenic in drinking water in the Ganges delta of India and Bangladesh is now recognized as one of the world’s most pressing environmental health problems. Environmental risk assessments have suggested that arsenic migrating from coal combustion wastes (eg, coal ash) deposited in unlined landfills may contaminate underlying groundwater. Arsine, an arsenous hydride (AsH3) gas with potent hemolytic effects, is manufactured predominantly for use in the semiconductor industry but may also be generated accidentally when arsenic-containing ores come in contact with acidic solutions.
It is of historical interest that Fowler’s solution, which contains 1% potassium arsenite, was widely used as a medicine for many conditions from the eighteenth century through the mid-twentieth century. Organic arsenicals were the first pharmaceutical antimicrobials* and were widely used for the first half of the twentieth century until supplanted by sulfonamides and other more effective and less toxic agents.
Other organoarsenicals, most notably lewisite (dichloro-[2-chlorovinyl]arsine), were developed in the early 20th century as chemical warfare agents. Arsenic trioxide was reintroduced into the United States Pharmacopeia in 2000 as an orphan drug for the treatment of relapsed acute promyelocytic leukemia and is finding expanded use in experimental cancer treatment protocols (see Chapter 54). Melarsoprol, another trivalent arsenical, is used in the treatment of advanced African trypanosomiasis (see Chapter 52).
Pharmacokinetics
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


