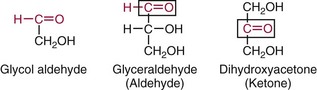
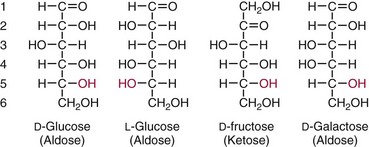
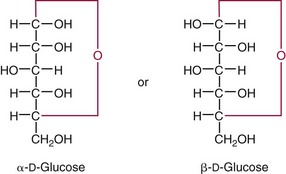
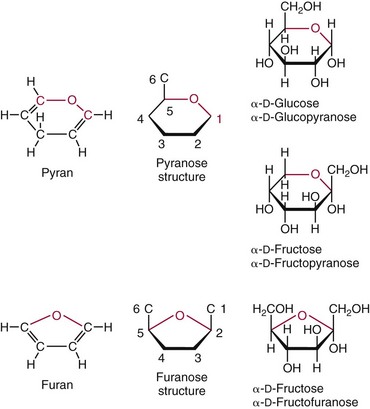
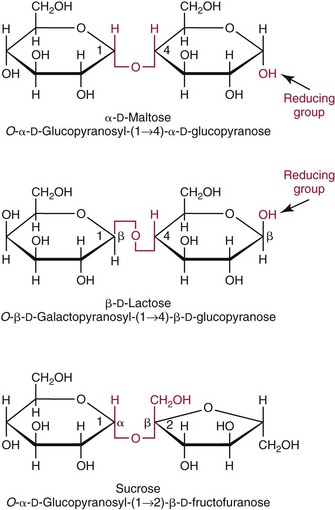
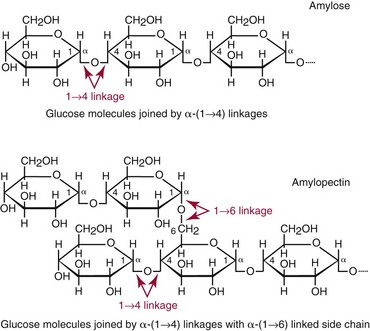
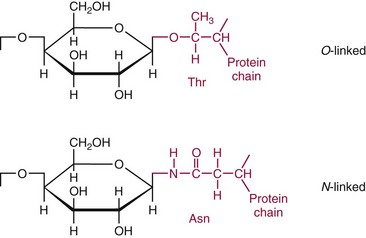
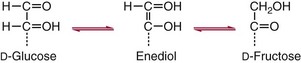
Chapter 26 Monosaccharides, or simple sugars, consist of a single polyhydroxy aldehyde or ketone unit and cannot be hydrolyzed to a simpler form. The backbone is made up of a number of carbon atoms. Sugars containing three, four, five, six, and seven carbon atoms are known as trioses, tetroses, pentoses, hexoses, and heptoses, respectively. One of the carbon atoms is double bonded to an oxygen atom to form a carbonyl group. An aldehyde has the carbonyl group at the end of the carbon chain, whereas if the carbonyl group is at any other position, a ketone is formed (Figure 26-1). The simplest carbohydrate is glycol aldehyde, the aldehyde derivative of ethylene glycol. The aldehyde and ketone derivatives of glycerol are, respectively, glyceraldehyde and dihydroxyacetone (see Figure 26-1). Monosaccharides are termed aldose or ketose, according to the position of the carbonyl group (Figure 26-2). Compounds that are identical in composition and differ only in spatial configuration are called stereoisomers. The carbon atoms in the unbranched chain are numbered, as shown by the numbers at the left of the formula for D-glucose (see Figure 26-2). The designation D or L refers to the position of the hydroxyl group on the carbon atom adjacent to the last (bottom) CH2OH group. In general, the designation of D or L for a sugar molecule refers to the stereoisomeric forms of the highest-numbered asymmetric carbon atom.* By convention, the D-sugars are written with the hydroxyl group on the right, and the L-sugars are written with the hydroxyl group on the left (see Figure 26-2). Most sugars in the human body are of the D-configuration. Several different structures exist, depending on the relative positions of the hydroxyl groups on the carbon atoms. Aldehyde and alcohol groups react to form hemiacetals. In the case of glucose, the aldehyde group reacts with the hydroxyl group on carbon 5 (Figure 26-3). Note that this ring structure contains an additional asymmetric carbon atom and exists in two stereoisomeric forms. By convention, the form with the hydroxyl group on the right of the first carbon atom is called α-D-glucose, and the form with the hydroxyl group on the left is called β-D-glucose. The common anhydrous crystalline glucose is in the α-D-form. The β-D-form is obtained by crystallization from acetic acid. The two forms differ with respect to optical rotation of polarized light. The specific rotation— From the ring structures shown in Figure 26-3, it is not apparent why the aldehyde group should react with the distant hydroxyl group on carbon 5. The spatial arrangement of the atoms is better represented by a symmetric ring structure, depicted by the Haworth formula, in which glucose is considered as having the same basic structure as pyran (Figure 26-4). In this formula, the plane of the ring is considered to be perpendicular to the plane of the paper, with heavy lines pointing toward the reader. Hydroxyl groups in position 1 are then below the plane (α-configuration) or above the plane (β-configuration). A six-member ring sugar, containing five carbons and one oxygen, is a derivative of pyran and is called a pyranose. When linkage occurs with formation of a five-member ring, containing four carbons and one oxygen, the sugar has the same basic structure as furan and is called a furanose. Representative formulas are shown in Figure 26-4. Fructose is shown in two cyclic forms. Fructopyranose is the configuration of free sugar, and fructofuranose occurs whenever fructose exists in combination in disaccharides and polysaccharides, as in sucrose and inulin. Several conventions are followed in the nomenclature of disaccharides (Figure 26-5). The compound is written with the nonreducing end to the left. An O precedes the name of the first (left) monosaccharide, emphasizing that the linkage occurs by an oxygen atom. The configuration of the anomeric (carbonyl) carbon is designated α or β. Five- (furanosyl) and six- (pyranosyl) membered rings are distinguished, and carbon atoms joined by the glycosidic bond are identified. Because sucrose has no reducing end, it is written as O-α-D-glucopyranosyl-(1 → 2)-β-D-fructofuranose or O-β-D-fructofuranosyl-(2 → 1)-α-D-glucopyranose. If the linkage between two monosaccharides is between the aldehyde or ketone group of one molecule and a hydroxyl group of another molecule (as in maltose and lactose), one potentially free ketone or aldehyde group remains on the second monosaccharide. Consequently, the second glucose residue can be oxidized (thus the disaccharide is a reducing sugar) and is capable of existing in α- or β-pyranose form. The reducing power, however, is only approximately 40% of the reducing power of the two single monosaccharides added together, primarily because one of the reducing groups is not available. On the other hand, if the linkage between two monosaccharides involves the aldehyde or ketone groups of both molecules (as in sucrose), a nonreducing sugar results because no free aldehyde or ketone group remains. Nearly all starches consist of a mixture of two types of glucosans called amyloses and amylopectins. The relative proportions of these two glucosans in a starch vary from approximately 20% amylose and 80% amylopectin in wheat, potato, and ordinary corn starch to nearly 100% amylopectin in the starch of waxy corn. On the other hand, a few corn starches are known to contain as much as 75% amylose. Both amylose and amylopectin consist of glucose residues, but their structures exhibit one significant difference. Amylose consists of one long unbranched chain of glucose units linked together by α-1,4-linkages with only the terminal aldehyde group free (Figure 26-6). In amylopectin, most of the units are similarly connected with α-1,4-links, but α-1,6-glycosidic bonds are present every 24 to 30 residues, producing sidechains (see Figure 26-6). Amylopectin contains up to 1 million glucose residues. The structure of glycogen is similar to that of amylopectin, but branching is more extensive in glycogen and occurs every 8 to 12 glucose residues. These branches enhance the solubility of glycogen and allow the glucose residues to be mobilized more readily. Glycogen is most abundant in liver and is found in skeletal muscle as well. The most favorable conformation for α-1,4-linked polymers of D-glucose, such as starch or glycogen, is a tightly coiled helical structure. The difference in structure between amylose and amylopectin is important when the appropriate starch substrate is selected for amylase determinations (see Chapter 22). The rate of hydrolysis is affected by structural differences in the starch. α-Amylase from the pancreas hydrolyzes internal α-1,4-glycosidic linkages. This hydrolysis results initially in the production of some maltose and a mixture of dextrins, which subsequently are hydrolyzed to maltose. The β-1,6-linkages are not attacked by α-amylase, and relatively large molecules of so-called residual (limit) dextrins are left after the action of the enzyme on amylopectin. Dextrins are the products of partial hydrolysis of starch. They are a complex mixture of molecules of different sizes. Those formed from amylose are unbranched chains, whereas amylopectins produce branched chains of glucose molecules. Cellulose, an important structural polysaccharide in plants, is an unbranched polymer of glucose residues joined by β-1,4-linkages. The β-configuration facilitates the formation of long straight chains, producing fibers of high tensile strength. The β-1,4-linkages are not hydrolyzed by α-amylases. Because humans do not have cellulases, they are unable to digest vegetable fiber. Glycosylation is one of the most frequent enzymatic modifications of proteins. Many integral membrane proteins have oligosaccharides covalently attached to the extracellular region, forming glycoproteins. In addition, most proteins that are secreted, such as antibodies, hormones, and coagulation factors, are glycoproteins. The number of attached carbohydrates varies among proteins and constitutes 1 to 70% of the weight of the glycoprotein. The oligosaccharides are attached by O-glycosidic linkages to the sidechain oxygen of serine and/or threonine residues or by N-glycosidic linkages to the sidechain nitrogen of asparagine residues (Figure 26-7). One of the biological functions of the carbohydrate chains is to regulate the life span of proteins. For example, removal of sialic acid residues from the end of oligosaccharide chains on erythrocytes results in the disappearance of red blood cells from the circulation. Carbohydrates have also been implicated in cell-cell recognition, and in secretion and targeting of proteins to specific subcellular domains. Defects in protein glycosylation have been linked to several forms of congenital muscular dystrophy that are associated with brain abnormalities.53 Ingested starch and glycogen are partially digested by the action of salivary amylase in the mouth to form intermediate dextrins and maltose (see Chapter 51). The acid pH of the stomach inhibits amylase activity, but alkaline pancreatic secretions increase the pH in the small intestine, allowing pancreatic amylase to complete digestion to oligosaccharides, preponderantly maltose. Maltose, along with any ingested lactose and sucrose, is hydrolyzed by the appropriate disaccharidase (maltase, lactase, or sucrase) from the intestinal mucosa to glucose, galactose, and fructose. These monosaccharides are absorbed across the wall of the duodenum and ileum by an active, energy-requiring, carrier-mediated transfer process. The rate of absorption for glucose and galactose is several times greater than for similar molecules absorbed by passive diffusion (e.g., xylose). Some conversion of fructose to glucose may occur during the process of absorption, and the interconversion can be visualized in terms of the enediol form common to both (Figure 26-8). Fructose is absorbed more slowly than glucose and galactose by a carrier-mediated process different from glucose and galactose transport mechanisms. The monosaccharides are then transported by the portal vein to the liver. Some steps in the intermediary metabolism of glycogen and hexoses are shown in Figure 26-9. Each step is catalyzed by enzymes. In some cases, different enzymes are responsible for the forward and reverse reactions. For example, the initial phosphorylation of glucose is mediated by glucokinase, but the reverse reaction depends on glucose-6-phosphatase. Figure 26-9 Major steps in the intermediary metabolism of carbohydrates. Numbers shown refer to specific enzymes. ( – – – – – – – – -), Multistep pathway; (—————), single-step pathway. Various inborn errors of metabolism (Table 26-1) result from deficiencies or absence of some of the enzymes listed in Figure 26-9. Some of these are discussed later in the chapter. The relationship of carbohydrate metabolism to the production of lactate, ketone bodies, and triglycerides is also depicted in Figure 26-9. The pentose phosphate pathway, also known as the hexose monophosphate shunt, is an alternative pathway for glucose metabolism that generates the reduced form of nicotinamide-adenine dinucleotide phosphate (NADPH), which is used in maintaining the integrity of red blood cell membranes, in lipid and steroid biosynthesis, in hydroxylation reactions, and in other anabolic reactions. The complete picture of intermediary metabolism of carbohydrates is complex and is interwoven with the metabolism of lipids and amino acids. For details, readers should consult a biochemistry textbook. TABLE 26-1 Inborn Errors of Carbohydrate Metabolism GSD, Glycogen storage disease. The concentration of glucose in the blood is regulated by the complex interplay of multiple pathways, modulated by a number of hormones. Glycogenesis is the conversion of glucose to glycogen. The reverse process, namely, the breakdown of glycogen to glucose and other intermediate products, is termed glycogenolysis. The formation of glucose from noncarbohydrate sources, such as amino acids, glycerol, or lactate, is termed gluconeogenesis. The conversion of glucose or other hexoses into lactate or pyruvate is called glycolysis. Further oxidation to carbon dioxide and water occurs through the Krebs (citric acid) cycle and the mitochondrial electron transport chain coupled to oxidative phosphorylation, generating energy in the form of adenosine triphosphate (ATP). Oxidation of glucose to carbon dioxide and water also occurs through the hexose monophosphate shunt pathway, which produces NADPH. Discussion of the hormones that regulate blood glucose is provided in Chapter 46. Hypoglycemia is a blood glucose concentration below the fasting value, but it is difficult to define a specific limit.73 The most widely suggested cutoff is 50 mg/dL, but some authors suggest 60 mg/dL.8 A transient decline may occur 1.5 to 2 hours after a meal, and it is not uncommon for a plasma glucose concentration as low as 40 mg/dL to be observed 2 hours after ingestion of an oral glucose load. Similarly, extremely low fasting blood glucose values may occasionally be noted without symptoms or evidence of underlying disease. Hypoglycemia is rare in patients who do not have drug-treated diabetes mellitus.19 Symptoms of hypoglycemia vary among individuals, and none is specific. Epinephrine produces the classic signs and symptoms of hypoglycemia, namely, (1) trembling, (2) sweating, (3) nausea, (4) rapid pulse, (5) lightheadedness, (6) hunger, and (7) epigastric discomfort. These autonomic (neurogenic) symptoms are nonspecific and may be noted in other conditions, such as hyperthyroidism, pheochromocytoma, or even anxiety. Although controversial, it has been proposed that a rapid decrease in blood glucose may trigger the symptoms even though the blood glucose itself may not reach hypoglycemic values, whereas gradual onset of hypoglycemia may not produce symptoms.25 The brain cannot store or produce glucose, and in resting adults the central nervous system (CNS) consumes approximately 50% of the glucose used by the body.31 Very low concentrations of plasma glucose (<20 or 30 mg/dL) cause severe CNS dysfunction. During prolonged fasting or hypoglycemia, ketones may be used as an energy source. The broad spectrum of symptoms and signs of CNS dysfunction range from headache, confusion, blurred vision, and dizziness to seizures, loss of consciousness, and even death; these symptoms are known as neuroglycopenia. Restoration of plasma glucose usually produces a prompt recovery, but irreversible damage may occur. The age of onset of hypoglycemia is a convenient way to classify the disorder (Box 26-1), but it should be borne in mind that some overlap occurs among the various groups. For example, some glycogen storage disorders may present in the third decade of life, and hormone deficiencies occur in childhood. Neonatal blood glucose concentrations are much lower than adult concentrations (mean <35 mg/dL) and decline shortly after birth when liver glycogen stores are depleted. Glucose concentrations as low as 30 mg/dL in a term infant and 20 mg/dL in a premature infant may occur without clinical evidence of hypoglycemia. The more common causes of hypoglycemia in the neonatal period include prematurity, maternal diabetes, gestational diabetes mellitus (GDM), and maternal eclampsia (see Box 26-1; for review, see Haymond).33 These are usually transient. Hypoglycemia with onset in early infancy is usually less transitory and may be due to inborn errors of metabolism or ketotic hypoglycemia; it usually occurs after fasting or a febrile illness. Hypoglycemia may result from a decreased rate of hepatic glucose production or an increased rate of glucose use. Symptoms suggestive of hypoglycemia are fairly common, but hypoglycemic disorders are rare. However, true hypoglycemia usually indicates serious underlying disease and may be life threatening. A precise threshold for establishing hypoglycemia is not always possible, and values as low as 30 mg/dL may be encountered in healthy premenopausal women after a 72-hour fast.54 Symptoms usually begin at plasma glucose concentrations below 55 mg/dL, and impairment of cerebral function begins when glucose is less than 50 mg/dL. More than 100 causes of hypoglycemia have been reported. Some of the more common conditions are listed in Box 26-1. Drugs are the most prevalent cause,72 and a wide variety, including pentamidine, gatifloxacin, and quinine, can produce hypoglycemia. Oral hypoglycemic agents, which have a long half-life (35 hours for chlorpropamide), are the most frequent cause of drug-induced hypoglycemia. Sulfonylureas stimulate secretion of insulin, proinsulin, and C-peptide, and may mimic an insulinoma. Differentiation is made by demonstration of the drug in blood or urine. Surreptitious administration of insulin can be detected by finding low C-peptide concentrations with increased insulin concentrations. Demonstration of a low plasma glucose concentration in the presence of an abnormally high plasma insulin value is highly suggestive of an insulin-producing pancreatic islet cell tumor.27 Because insulin concentrations exhibit a wide range in normal people, absolute hyperinsulinemia occurs in fewer than 50% of patients with insulinomas. Serum insulin concentrations inappropriately high for concurrent plasma glucose have been proposed to increase diagnostic accuracy. Critical diagnostic findings include a plasma insulin concentration of at least 18 pmol/L, plasma C-peptide concentrations greater than or equal to 0.2 nmol/L, and plasma proinsulin concentrations of at least 5.0 pmol/L when fasting plasma glucose is below 55 mg/dL. Ratios employing insulin and glucose have no diagnostic value. Provocative tests [glucagons,42 tolbutamide,74 calcium,40 or suppression tests (infusion of insulin and measurement of C-peptide)], although strongly recommended in the past, generally are not necessary. Intra-arterial calcium stimulation with right hepatic vein sampling for insulin gradients appears to be a sensitive preoperative test for localizing insulinoma, although advances in imaging are making this approach far less common.26 Spontaneous production of antibodies to insulin may produce hypoglycemia (these antibodies are distinct from those elicited by insulin therapy and from the antibodies detected in certain patients with type 1 diabetes). Anti-insulin antibodies causing hypoglycemia have been reported in Graves’ disease, multiple myeloma, systemic lupus erythematosus, and rheumatoid arthritis. This disorder is reported primarily among persons of Japanese or Korean ancestry and is less frequent among whites.19 Patients exhibit postprandial hyperglycemia and fasting hypoglycemia.61 Laboratory analysis demonstrates low plasma C-peptide and very high plasma insulin concentrations during hypoglycemia. The high insulin concentrations are believed to be an assay artifact caused by the antibody, and diagnosis is usually made by demonstrating high-titer serum insulin antibodies. Nonpancreatic neoplasms that cause hypoglycemia are often extremely large mesenchymal neoplasms that appear to overuse glucose but may also have an inhibitory effect on glucose mobilization. Tumors of epithelial origin may cause hypoglycemia, frequently by producing insulin-like growth factor (IGF) II.22,75 Hypoglycemia caused by septicemia should be relatively easy to diagnose.56 The mechanism is not well defined, but depleted glycogen stores, impaired gluconeogenesis, and increased peripheral use of glucose may be contributing factors. Glucose tolerance is commonly depressed in renal disease, and hypoglycemia may occur in end-stage renal failure. Some of the conditions producing fasting hypoglycemia are readily apparent, but others require a lengthy diagnostic work-up. Once hypoglycemia is demonstrated, specific tests should be performed to establish the underlying cause. The oral glucose tolerance test (OGTT) is not an appropriate study for evaluating a patient suspected of having hypoglycemia.19 A group of disorders may produce hypoglycemia in the postprandial (fed) state.34 These include drugs, antibodies to insulin or the insulin receptor, and inborn errors (e.g., fructose-1,6-diphosphatase deficiency). Also included is reactive hypoglycemia (referred to as functional hypoglycemia), which has been the subject of much debate.8 Many commentaries and editorials have been published regarding the existence of reactive hypoglycemia (Hofeldt34 and references listed therein). The general consensus is that no scientific evidence supports the existence of “functional hypoglycemia.” It has been proposed that for individuals with vague symptoms after food ingestion, the preferred terminology should be idiopathic reactive hypoglycemia34 or idiopathic postprandial syndrome.73 At the Third International Symposium on Hypoglycemia,46 reactive hypoglycemia was defined as a clinical disorder in which the patient has postprandial symptoms suggesting hypoglycemia that occur in everyday life and are accompanied by a blood glucose concentration less than 45 to 50 mg/dL as determined by a specific glucose measurement on arterialized venous or capillary blood, respectively. Patients complain of autonomic symptoms occurring approximately 1 to 3 hours after eating and seem to obtain relief, lasting 30 to 45 minutes, by food intake. These symptoms are rarely due to low blood glucose concentrations (e.g., diabetes mellitus, gastrointestinal dysfunction, hormonal deficiency states). Most of these individuals have postprandial autonomic symptoms without neuroglycopenia in the postprandial state. Some experts in the field state that no true hypoglycemic disorder is characterized solely by autonomic symptoms.73 A 5- or 6-hour glucose tolerance test was the standard procedure to establish the presence of postprandial hypoglycemia, but its use has been discredited.8 The test is not reproducible in any particular individual, and low values for plasma glucose may be noted in the absence of symptoms, whereas symptoms may occur with normal glucose concentrations.15 In addition, patients who have low blood glucose concentrations with autonomic symptoms 3 or 4 hours after an oral glucose load may have identical symptoms with normal blood glucose values after a mixed meal.15 This may be due in part to anxiety provoked by the stressful environment during the glucose tolerance test. Demonstration of increased plasma epinephrine concentrations at the glucose nadir during an OGTT was reported to differentiate patients with reactive hypoglycemia,12 but patients studied were identified on the basis of autonomic symptoms and signs, and only 25% demonstrated hypoglycemia. The OGTT should not be used in the diagnosis of reactive hypoglycemia.76 Postprandial hypoglycemia is infrequent, and demonstration of hypoglycemia during spontaneously occurring symptomatic episodes is necessary to establish the diagnosis.61 If this is not possible, a 5-hour meal tolerance test15 (which simulates the composition of a normal diet) or a hyperglucidic (high glucose) breakfast test8 has been proposed. A protocol for a mixed-meal test can be found in Cryer and associates.19 A diagnosis of hypoglycemia has been used to explain a wide variety of disorders that appear unrelated to blood glucose abnormalities.11 These nonspecific symptoms include fatigue, muscle spasms, palpitations, numbness, tingling, pain, sweating, mental dullness, sleepiness, weakness, and fainting. Behavior abnormalities, poor school performance, and delinquency have been incorrectly attributed to low blood glucose concentrations. Widespread use of the insensitive and nonspecific 5-hour glucose tolerance test caused overdiagnosis of hypoglycemia and led the American Diabetes Association (ADA) to publish a statement to discourage the inappropriate use of the OGTT for the diagnosis of hypoglycemia.76 Lay publications78 have supported this recommendation, but it is still important for the medical community to reassure such patients that low blood glucose is not the cause of their symptoms and to deal with specific abnormalities that might underlie patients’ complaints or problems. A diagnosis of hypoglycemia should not be made unless a patient meets the criteria of Whipple’s triad of low blood glucose concentration with typical symptoms alleviated by glucose administration. Demonstration of a plasma glucose concentration >70 mg/dL during a symptomatic episode indicates that the symptoms unequivocally are not the result of hypoglycemia.
Carbohydrates
Chemistry of Carbohydrates
Monosaccharides
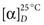 —for the α-D-form is +113°, and for the β-D-form is +19.7°. As a result of mutarotation, either form in aqueous solution gives rise to an equilibrium mixture that has a specific rotation of +52.5°. The equilibrium established at room temperature is such that about 36% of glucose exists in the α-form and 64% in the β-form; only a trace remains in the free aldehyde form. The enzyme glucose oxidase reacts only with β-D-glucose. For this reason, calibrating solutions to be used in glucose oxidase methods for glucose determinations should be permitted to stand at least 2 hours to obtain equilibrium comparable with that in the test samples to be analyzed.
—for the α-D-form is +113°, and for the β-D-form is +19.7°. As a result of mutarotation, either form in aqueous solution gives rise to an equilibrium mixture that has a specific rotation of +52.5°. The equilibrium established at room temperature is such that about 36% of glucose exists in the α-form and 64% in the β-form; only a trace remains in the free aldehyde form. The enzyme glucose oxidase reacts only with β-D-glucose. For this reason, calibrating solutions to be used in glucose oxidase methods for glucose determinations should be permitted to stand at least 2 hours to obtain equilibrium comparable with that in the test samples to be analyzed.
Disaccharides
Polysaccharides
Glycoproteins
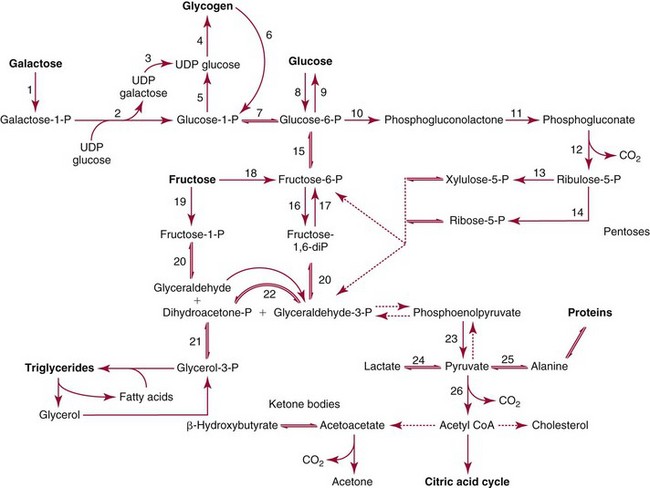
Metabolism of Carbohydrates
Digestion and Absorption
Intermediary Metabolism
Enzyme Deficiency
Disease State
Glucose-6-phosphatase (9)*
Type I GSD (von Gierke disease)
Muscle phosphorylase
Type V GSD (McArdle’s disease)
Liver phosphorylase
Type VI GSD (Hers’ disease)
Galactose-1-phosphate-uridyl transferase (2)
Galactosemia
Galactokinase (1)
Galactosemia
Uridine diphosphate-galactose-4-epimerase (3)
Galactosemia
Fructokinase (19)
Essential fructosuria
Fructose-1-phosphate aldolase (20)
Hereditary fructose intolerance
Pyruvate kinase (23)
Hemolytic anemia
Glucose-6-phosphate dehydrogenase (10)
Hemolytic disease
Regulation of Blood Glucose Concentration
Hypoglycemia
Hypoglycemia in Neonates and Infants
Fasting Hypoglycemia in Adults
Postprandial Hypoglycemia
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Basicmedical Key
Fastest Basicmedical Insight Engine