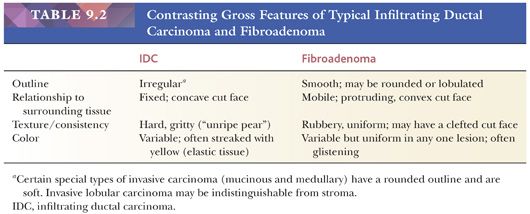
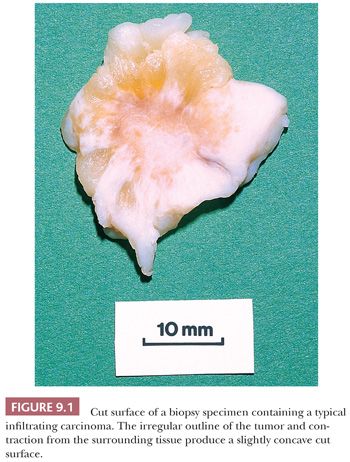

The number of blocks submitted from specimens varies depending on circumstances. It is important, however, that tumors be sampled thoroughly because their microscopic appearance may vary from area to area. The center and periphery of tumors should be sampled, as well as the surrounding tissue, because malignant change may be far more extensive than is suspected on gross examination. If possible, a section of the largest full-cut face of a tumor should be submitted in one block. Because noninvasive carcinoma may be an incidental finding, it is important to sample apparently normal tissue, even that around an obviously benign lesion, with concentration on the nonfatty component and, as is discussed later, areas of mammographic abnormality. The fatty component is unlikely to contain significant pathologic changes that are not also present in the nonfatty component of the specimen (2).
FROZEN SECTION
In recent years, the use of frozen section to diagnose mammary carcinoma has declined. Malignancy is usually diagnosed by either needle biopsy or open excisional biopsy of ambulatory patients in order to best consider alternatives prior to definitive treatment (2). Frozen sections are still used to confirm a cytologic diagnosis of carcinoma or to evaluate surgical margins. If frozen section is undertaken and doubt arises about the diagnosis, a final decision should be deferred until processed tissue is available. Difficulties with frozen sections are usually attributable to sampling errors, poor sections, or, occasionally, histologic misinterpretation. Errors in interpretation arise particularly with sclerosing lesions (sclerosing adenosis, radial scar, ductal adenoma), epithelial hyperplasia, papillary lesions, and fat necrosis. In situ carcinoma may be difficult to distinguish from benign epithelial hyperplasia and from invasive carcinoma in small samples.
NEEDLE BIOPSY
Core needle biopsy (CNB) has the advantage over fine-needle aspiration (FNA) of allowing identification of benign entities and localization of calcium deposits with the result that the proportion of indeterminate and inadequate specimens is far less. Distinction between in situ and invasive carcinoma may also be more readily accomplished, and predictive markers (estrogen receptor [ER], progesterone receptor [PR], HER2/neu, etc.) can be better evaluated. However, FNA may be done more quickly with a diagnosis of malignancy at the time of outpatient visit. In the evaluation of CNB, problems are encountered because of the limited size of the specimens and, not infrequently, the presence of crush artifact. Other problems include potential destruction of lesional tissue by hemorrhage or infarction and displacement of benign epithelium to simulate invasive carcinoma (3), although displacement of epithelium has also been reported following localization by guide-wire for excision of a mammographic abnormality (4). CNB of nonpalpable lesions requires radiographic guidance by either ultrasonography or stereotactic mammography. In CNB performed to evaluate microcalcification, it is important that the calcifications be identified on specimen radiographs (5) and confirmed in the histologic sections, even though calcifications imaged on radiographs are often larger than those seen histologically. Calcification is rarely seen in FNA. Uncertainty in the interpretation of a CNB or the finding of significant atypia should lead to open biopsy. In patients receiving neoadjuvant therapy, CNB specimens may be the only histologic evidence of cancer if there is a complete response.
OPEN BIOPSY
Open biopsy may take the form of either (a) an incisional biopsy in which only part of the lesion is sampled or (b) an excisional biopsy intended to remove the lesion in its entirety as part of definitive therapy (lumpectomy, partial mastectomy, or tylectomy) for conservation treatment. Therefore, histologic assessment of the surgical margins is important. During the operation, some surgeons rely on the gross appearance alone, whereas others ask for a frozen section evaluation of the margins. However, full evaluation of margins on frozen section may be too time-consuming to be useful. Assessment of specimen x-ray images has been used to note the proximity of a neoplasm to the surgical margins but is not reliable (6). Touch preps have been advocated to evaluate margins rapidly. Although the exact techniques used to assess neoplastic involvement of tumor margins vary, it is always important to ink the surface of the specimen before dissection. This may be facilitated, particularly in the case of fresh specimens, if the surface is thoroughly blotted dry, or dipped briefly into alcohol or acetone, both before and after application of the ink. This procedure helps fix the dye onto the surface of the specimen and limit its spread into the planes of the underlying tissue. The use of several different colored inks facilitates orientation of sections, but this is possible only if the specimen has been oriented by the surgeon with the use of clips, sutures of different lengths, or some other means at the time of excision. Identification of the location of the nipple allows orientation of the specimen in the direction of the ducts, which may be important to determine the extent of ductal carcinoma in situ (DCIS). When carcinoma involves the margin, it should be noted whether the involvement is focal or diffuse and by in situ or invasive carcinoma. When the margin is not involved, its distance from the carcinoma should be reported in millimeters. Each excision margin should be included on at least one section and, ideally, there should be two or three sections from the margin closest to the grossly identified neoplasm. If resection is found to be incomplete and the specimen has been oriented to allow a determination of which margin is involved, reexcision of only the appropriate area may sometimes be carried out. Some surgeons prefer to perform shave biopsies of the wall of the cavity that is left after the excision biopsy has been performed. Alternatively, the outer wall of the excision biopsy specimen can be shaved. Use of shaved margins may result in overestimation of the extent of tumor and the use of perpendicular margins may result in underestimation, but false positives and false negatives are associated with both methods (6).
MICRODOCHECTOMY
Microdochectomy is the removal of a diseased duct system in patients with nipple discharge. It is helpful if the surgeon marks the apex of the specimen with a suture and leaves a probe in the resected duct. The specimen may be dissected either by opening the duct with fine scissors or by making serial slices across the duct lumen. We favor the former method if a probe is left in the duct. It is important to report whether a cause of the discharge has been removed; if not, continued clinical follow-up may be required.
MASTECTOMY
Although the increased use of conservation therapy in the treatment of breast cancer has resulted in more limited specimens, mastectomy, when carried out, always necessitates a thorough gross description (7). Orientation of the specimen is facilitated if markers are placed at the time of operation at the 12 o’clock position on the skin ellipse and in the axilla. The axillary portion can be divided into three levels (Fig. 9.3) but only on radical mastectomy specimens, which are uncommon in current clinical practice. In the absence of resected muscles, levels must be identified intraoperatively. The size and appearance of the skin ellipse and nipple should be noted, and the site and size of any biopsy incision should be recorded. After ink has been applied to the deep margin, the breast should be sliced serially in the sagittal plane from its posterior aspect. If the skin is left intact, the specimen can later be reconstructed if examination of more tissue is necessary. Any abnormalities found within the breast tissue should be carefully described and localized, with particular attention paid to the features listed in Table 9.1. The general appearance of the remaining breast tissue should also be noted. Blocks should be obtained from any abnormal area, including any previous biopsy site, if present, and from the deep margin closest to the tumor or biopsy site. At least one block should be obtained from each quadrant and one parallel to the skin in the subareolar region to sample the major ducts.
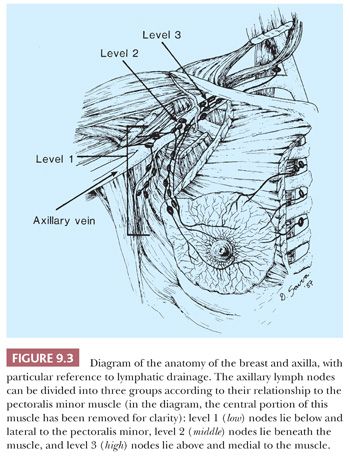
The number of lymph nodes found per axilla varies depending on the extent of the axillary dissection. Careful manual palpation of the tissue is the generally accepted practice for locating axillary lymph nodes. Special techniques, including radiographic imaging and clearing methods, may increase the yield of lymph nodes. Examination of multiple levels of each block of lymph nodes and immunohistochemical study with the use of epithelial markers may help detect small metastases, but the significance of discovery of micrometastases gained by these laborious techniques is of limited clinical consequence (8). Conservation treatment of mammary carcinoma often includes axillary dissection. The tissue containing the nodes may be removed in continuity with that containing the carcinoma; more often, it is separate. Careful orientation of the specimen, as previously described, may be important.
SENTINEL LYMPH NODE BIOPSY
Excision of the sentinel lymph node(s) is being used increasingly as an alternative to traditional axillary dissection, and the recent report of the National Surgical Adjuvant Breast and Bowel Project (NSABP) B-32 study showed that staging results in clinically node-negative cases were comparable to those of axillary dissection (9). The sentinel lymph node(s) is identified by the surgeon following injection of the affected breast with dye and/or radioactive-labeled tracer and then submitted to the pathologist. The sentinel node may be examined intraoperatively by frozen section or cytologic (touch preparation) examination. Sentinel lymph nodes often receive more extensive pathologic evaluation than nodes procured via standard axillary dissection. There is currently heterogeneity in the handling of sentinel lymph nodes, and several methods have been used to demonstrate small metastases. Perhaps the most important consideration is to make sure that the nodes are initially cut at intervals no greater than 2 mm to ensure finding macrometastases, defined as larger than 2 mm (10). Although immunohistochemical stains for cytokeratin and molecular techniques have been used to detect occult metastases in sentinel and nonsentinel lymph nodes, the clinical value of these adjunctive procedures has not been established (8).
SPECIAL TECHNIQUES
SPECIMEN RADIOGRAPHY AND LOCALIZATION BIOPSY
The mammographic appearance of carcinoma and benign breast lesions is described briefly in Table 9.4. The handling of mammographically detected lesions is time-consuming but, for an accurate assessment, consistent methods must be used (11). Radiologic signs of malignancy may or may not be accompanied by a palpable abnormality. In the absence of any clinical sign, a mammographically suspect lesion must be localized preoperatively. The technique most frequently used is the insertion of a fine hooked wire, which serves to guide the surgeon. After excision, the specimen should be radiographically examined and the findings compared with the preoperative mammogram (Fig. 9.4) to ensure that the lesional tissue and the wire have been removed. Specimen radiography should be performed while the patient is still in the operating room so that further tissue can be removed if necessary. An x-ray image of the specimen should be sent along with the specimen to the pathologist, with the area in question clearly indicated.
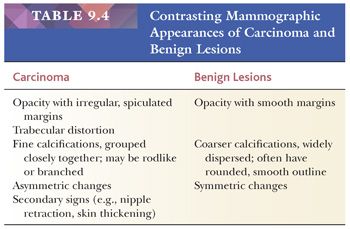
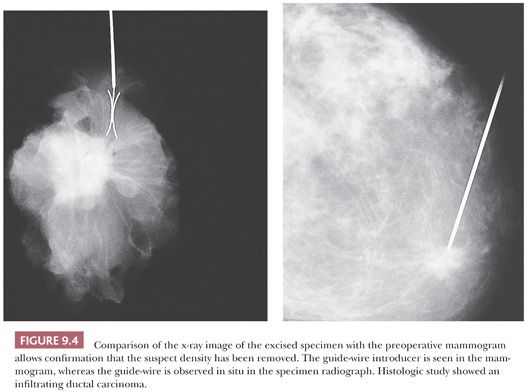
Microcalcification is the most frequent mammographic sign of a clinically occult tumor (Figs. 9.5 and 9.6), but it is usually confined to the DCIS of a carcinoma. Confirmation of the presence of calcium is not usually a problem, but it may require additional slides and even x-ray examination of the blocks. When extensive study is carried out, it should be remembered that calcifications seen on slides are significantly smaller than those visualized by the radiologist on x-ray film. Other mammographic signs of malignancy, however, such as architectural distortion, may be more difficult to match with those on the original mammogram. Compression of the excised specimen during the x-ray examination may help detect the abnormality. Occasionally, when the tissue has been excised, a palpable abnormality becomes apparent. Otherwise, frozen section is not recommended in this situation because (a) many mammographically detected abnormalities are small and (b) histologic interpretation is often difficult (2). The entire specimen should be sliced at 2- to 3-mm intervals. In the absence of an obvious gross lesion, all the slices should, if possible, be processed. If this is not feasible, blocks should include both the mammographic abnormality and the immediately surrounding tissue. All areas containing microcalcification should be examined. Selection of the involved areas is facilitated by further x-ray examination after the tissue has been finely sliced. If the specimen proves to contain carcinoma, additional sampling may be necessary. This is particularly important in cases of DCIS because it helps the examiner to assess the extent of disease, determine whether foci of infiltration exist, and demonstrate the relationships to the margins. If each slice is processed separately, the extent of the lesion can be deduced by counting the number of slices involved (11). If microscopic examination fails to confirm the presence of calcification, x-ray examination of the paraffin blocks may help to locate the exact site of the calcium within the tissue. Calcium oxalate crystals (weddellite), in contrast to the more common calcium phosphate, do not stain with hematoxylin and so are not easily seen in slides stained with hematoxylin and eosin (H&E) nor do they stain with von Kossa stain (12,13). The crystals, however, are readily seen by polarized light microscopy. Although either form of calcification may present as a mammographic abnormality, calcium oxalate crystals are usually associated with benign mammary lesions—in particular, cystic change and apocrine metaplasia. The histology report of all specimens removed as a result of a mammographic abnormality should include a comparison of the mammographic and gross findings, in addition to a comparison of the mammographic and histologic findings.
OTHER IMAGING TECHNIQUES
Both ultrasonography and magnetic resonance imaging may also provide useful information in the evaluation of patients with breast symptoms. The former is particularly helpful in distinguishing cystic from solid lesions, whereas the latter, in conjunction with mammography, may aid in the detection, diagnosis, and staging of breast cancer and in the differentiation of postsurgical scarring from tumor recurrence in patients whose breast carcinoma was previously treated with conservation therapy (14). Other imaging techniques, including scintimammography, positron emission tomography, and particularly digital mammography, may also be of value in the assessment of patients with breast disorders.
SUBGROSS EXAMINATION
Subgross examination, which is best suited to research laboratories, has greatly enhanced the knowledge and understanding of mammary tissue and its abnormalities, particularly by defining the site of origin of many pathologic entities. Samples are fixed, sliced, stained, dehydrated with alcohol, and then cleared with methyl salicylate (15). A dissecting or stereoscopic microscope can be used to examine the tissue slices, and abnormal areas may be selected for further histologic examination. Similar techniques for the stereoscopic examination of paraffin-embedded tissue blocks have also been reported and are used to evaluate the distribution of pathologic lesions in the mammary tree (16,17).
ANCILLARY STUDIES
ER and PR and HER2/neu status are part of the pathology report of carcinoma. Details of the procedures and reporting methods have been given by the American Society of Clinical Oncology (ASCO) and the College of American Pathologists (CAP) (18,19).
Electron microscopy is usually not required in the routine diagnosis of breast disease. It may occasionally be helpful in a problem case to distinguish among carcinoma, sarcoma, and lymphoma. However, in most situations, immunohistochemistry has largely supplanted the use of electron microscopy. A general outline of the use of immunohistochemical techniques appears in Table 9.5. Immunohistochemistry has largely supplanted histochemical stains, with the exception of stains for mucin and melanin. These studies should be applied in conjunction with good morphologic analysis and used as an adjunct to a histologic diagnosis and not to make a diagnosis in isolation.

There are currently five multigene expression–based prognostic tests for breast cancer offered as reference lab tests. However, there is as yet no consensus on which of these microarrays is the most useful in clinical application. Analysis of their current status is beyond the purview of this chapter, but the results of ongoing clinical trials should be watched closely.
THE PATHOLOGY REPORT
This vital component of the diagnostic process primarily records whether a lesion is benign or malignant. If malignant, it conveys data related to type and grade, extent, and prognosis and thus is the basis for treatment planning (7). The features listed in Table 9.6 should be recorded. The use of a synoptic or checklist-type format has been recommended to ensure a uniform system of reporting and increase the likelihood that all clinically important information is provided (20). In addition, it may be used for academic review in research and teaching.

THE NORMAL BREAST
Even though the normal mammary epithelium comprises only a small proportion of the total breast tissue mass, most of the diseases affecting the breast arise from it. The epithelium is arranged in the form of 10 to 15 segments, each consisting of a branching structure that has been likened to a flowering tree (21). The lobules represent the flowers, which drain into ductules and ducts (twigs and branches) (Fig. 9.7); these in turn drain into the collecting ducts (trunk) that open onto the surface of the nipple. Just below the nipple, the ducts are expanded to lactiferous sinuses. The epithelium throughout the duct system is bilayered, consisting of an inner epithelial layer and an outer myoepithelial layer. Evaluation of this double cell layer is one of the main guides to the distinction between benign and malignant lesions.
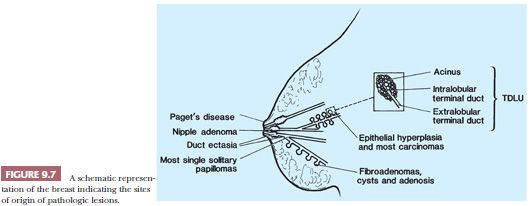
The terminal duct lobular unit (TDLU) is the physiologically active area of the breast and the site of origin of most pathologic lesions. It consists of a draining duct and a variable number of blind-ended terminal ductules, alternatively called acini, each of which has a typical double epithelial cell layer. The acini are surrounded by a specialized, hormonally responsive fibrovascular intralobular stroma, which contrasts with the denser and less cellular interlobular stroma (Fig. 9.8). The luminal epithelium of the resting breast is cuboidal or columnar. The outer basal or myoepithelial layer is anchored in the basement membrane and, although always present, is variably distinctive. In fresh tissue, myoepithelial cells display alkaline phosphatase activity, whereas in fixed tissue, they can be demonstrated by immunohistochemical methods with antibodies to actins, calponin, smooth muscle myosin heavy chain, high–molecular-weight cytokeratins, and p63. Epithelial cells are differentiated by their keratin profile and staining for gross cystic disease fluid protein-15 (GCDFP-15). The size of the mammary lobules and the number of acini are extremely variable. This may be partly related to the patient’s age, but also to the plane of the section.
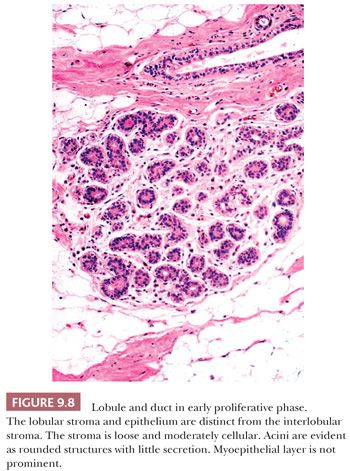
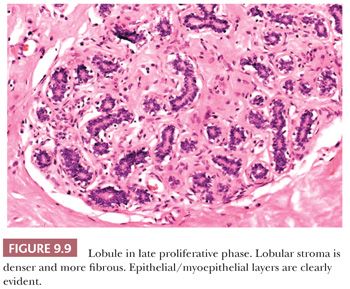
Familiarity with the cyclic changes of the normal lobule is basic to the interpretation of breast histology. Variation in the epithelial and stromal components of the mammary lobules occurs in association with the menstrual cycle but may not be observed synchronously in all lobules (22). In the early follicular phase, the stroma is dense and collagenized and the epithelium is columnar with only luminal cells apparent. The later follicular phase is characterized by distinct epithelial and myoepithelial cell types and a dense and fibrous lobular stroma (see Fig. 9.9). In the luteal phase, the lobular stroma is looser and more myxoid; both epithelial and myoepithelial cell layers are evident; and the acinar lumina are progressively distended with eosinophilic secretions.
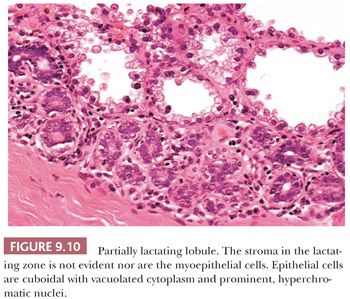
During pregnancy and lactation, the number of lobules and acini within each lobule increase at the expense of the interlobular and intralobular stroma. Luminal epithelial cells fill with cytoplasmic vacuoles, which often protrude into the lumen (Fig. 9.10). The florid changes in both nuclei and cytoplasm can be alarming to the inexperienced observer; areas of infarction, which occasionally develop in the breast during pregnancy, may compound the problem (23). When lactation ceases, the lobules involute and return to resting appearance, but involution usually proceeds unevenly and takes several months. Involuting lobules are irregular in contour and frequently infiltrated by lymphocytes and plasma cells. With increasing age, particularly after the menopause, gradual atrophy of the lobular acini occurs, together with a loss of specialized intralobular stroma. The postmenopausal breast eventually atrophies to consist largely of adipose tissue containing a few residual ducts and vessels in interlobular stroma. Occasionally, an isolated lobule showing secretory changes may be seen in the nonpregnant breast. Although this is often called a residual lactating lobule, it may occur in a nulliparous woman.
Most pathologic changes in the breast have been termed either lobular or ductal, in that lesions so designated were thought to arise from either the lobular acini or the larger ducts. In more recent years, however, this assumption has been questioned. Subgross studies (15,21) have shown that most lesions originally termed ductal (e.g., cysts, ductal epithelial hyperplasia, and DCIS) actually arise from the TDLU, which “unfolds” with coalescence of the acini to produce larger structures resembling ducts. Indeed, the only common benign lesion thought to arise from the large- or medium-sized duct rather than from the TDLU is the solitary intraductal papilloma. Also found in breast tissue are intramammary lymph nodes, which are more frequent than previously recognized and may be identified on routine mammograms (24).
The epidermis of the nipple contains occasional clear (Toker) cells that must not be confused with Paget cells and that are cytologically benign. Some of these cells are clear keratinocytes, whereas others are thought to be derived from epidermally located mammary acinar epithelium. Although the areola lacks pilosebaceous units, sebaceous glands are present, related to the lactiferous ducts. Sometimes, these glands drain via the lactiferous duct lumina and thus terminate in a common ostium, or they may terminate directly in the epidermis adjacent to the lactiferous ducts. During pregnancy, the glands become increasingly prominent, resulting in the formation of small elevations called Montgomery tubercles. Apocrine glands are also frequently present. Numerous bundles of smooth muscle are a distinctive feature of the subareolar region.
NIPPLE LESIONS
Lesions of the nipple present special diagnostic problems due in part to their location but also because of the special features they present.
PAGET DISEASE OF THE NIPPLE
Paget disease of the nipple is clinically seen as a red, weeping, often crusted lesion of the nipple which may be clinically indistinguishable from eczema. On microscopic examination, the epidermis is seen to be permeated by malignant cells arranged either singly, in groups, or, more rarely, in tubules (Fig. 9.11). The cells may be sparse or so numerous that they completely efface and destroy the epidermis. Paget cells have large nuclei, prominent nucleoli, and abundant pale-staining cytoplasm that often contains mucin. Because of shrinkage artifact, the cells sometimes appear to lie within a space. The underlying dermis shows variable degrees of telangiectasia and chronic inflammation. In almost every case of mammary Paget disease, an underlying carcinoma is found. This is nearly always of ductal type and may be either pure in situ carcinoma (usually of high-grade, poorly differentiated type) or a combination of in situ and infiltrating carcinoma.
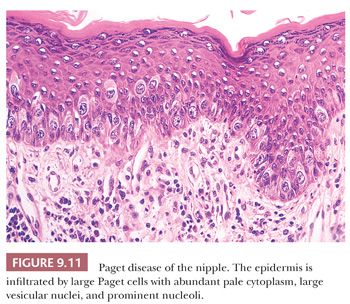
Paget disease occurs in 1% to 2% of all patients with mammary carcinoma. Although occasionally the in situ carcinoma is restricted to a single duct beneath the nipple, it is usually more widespread (25). A solitary focus of in situ carcinoma in a large duct beneath the nipple appears to be one instance in which carcinoma originates in a large duct rather than in the TDLU. The histogenesis of Paget cells has been a matter of controversy, but immunohistochemical studies have confirmed their glandular epithelial immunophenotype, consistent with migration from an origin in the duct system of the breast or possibly from intraepidermal mammary ductal epithelial cells (26). The microscopic appearance of Paget disease of the nipple can sometimes be confused with that of either superficial spreading melanoma or Bowen disease, both of which may rarely occur in the region of the nipple. Features helpful in the differential diagnosis include the presence of multinucleated giant cells and individual cell keratinization in Bowen disease. In malignant melanoma, tumor cells may come into direct contact with and invade the underlying dermis, whereas Paget cells remain within the epidermis. It should be noted that in some cases, Paget cells may contain melanin pigment; therefore, the presence of such pigment within pagetoid cells in the epidermis does not necessarily define the cells as those of malignant melanoma. Direct invasion of the epidermis by an underlying carcinoma is not Paget disease.
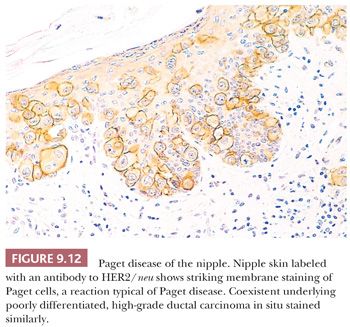
Histochemical and immunohistochemical stains that may be helpful in the differential diagnosis of Paget disease are listed in Table 9.7. The cells of Paget disease of the nipple, together with the underlying carcinoma, nearly always express the HER2/neu oncoprotein (Fig. 9.12). Antibodies to this protein, together with antibodies to low–molecular-weight cytokeratins, can help to make the diagnosis in difficult cases, especially when the Paget cells are few and far between (26). As discussed previously, cytologically benign clear cells, which do not express HER2/neu, occasionally occur in the epidermis of the normal nipple (Toker clear cells) and should not be mistaken for the cells of Paget disease.

NIPPLE ADENOMA (FLORID PAPILLOMATOSIS OF THE NIPPLE)
Nipple adenoma has also been called florid papillomatosis of the nipple, florid adenomatosis, subareolar duct papillomatosis, or erosive adenomatosis. It is a relatively uncommon entity that the unwary can mistake for carcinoma. It is most common in the fourth decade but may be seen in women of any age and also occurs in men. The soreness, ulceration, and swelling of the nipple, sometimes with discharge, may simulate Paget disease of the nipple (27).
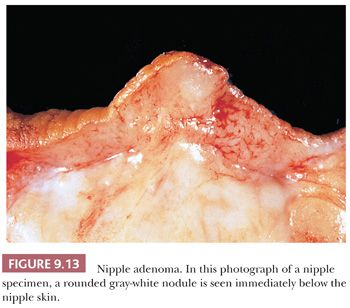
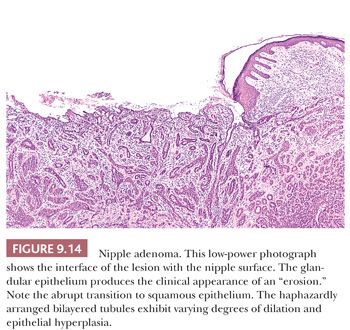
Grossly, it is usually an ill-defined nodule (Fig. 9.13). At low power, the lesion has a rounded outline and is composed of haphazardly arranged, proliferating, tubular structures surrounded by a varying amount of fibrous stroma (Fig. 9.14). The epithelial tubules, with their columnar cells, may extend to the surface of the nipple, in which case an abrupt transition to the stratified squamous epithelium is seen (Fig. 9.14). They are of various sizes and shapes, simulating infiltrating carcinoma. Lobular architecture is lacking, probably because the lesion arises from the larger ducts.
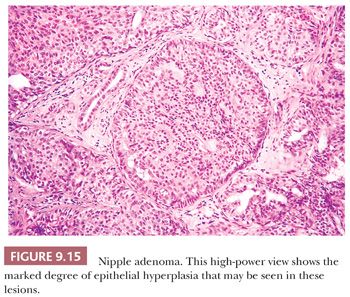
At high magnification, luminal and myoepithelial cells can be discerned, with different degrees of epithelial hyperplasia sometimes distending and occluding the glandular lumina (Fig. 9.15). The epithelial cells are usually cuboidal or columnar, but apocrine and squamous metaplasia both occur. As in many other lesions of the breast, stromal sclerosis sometimes distorts the glandular structures. Apparent involvement of muscle bundles may be seen, usually without reaction, and mitotic figures may be present, although not in abundance. Necrosis is rare. Rosen and Caicco (28) described three distinct growth patterns of nipple adenoma, but these do not appear to have prognostic significance. The lesion is generally considered entirely benign, but occasional cases of coexistent or subsequent carcinoma have been reported (28,29). The lesion may recur if it is incompletely excised. Nipple adenomas should be differentiated from syringomatous adenoma of the nipple (Table 9.8) and from an unusual subareolar papillary intraductal proliferative lesion called subareolar sclerosing duct hyperplasia (30). The latter abnormality is located beneath the areola without involvement of the surface of the nipple, but it may be associated with a nipple discharge. Although small ducts manifesting intraluminal papillary proliferation are present at the periphery of subareolar sclerosing duct hyperplasia, the lesion is distinctive because of the central sclerosis that results in entrapped and distorted ducts, analogous to the findings in a radial scar.

SYRINGOMATOUS ADENOMA OF THE NIPPLE
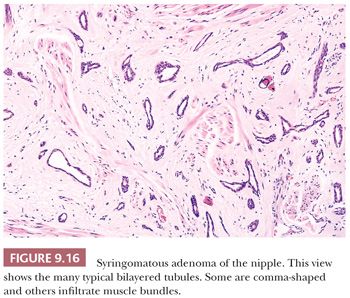
These rare tumors are composed of poorly circumscribed collections of small, bilayered epithelial tubular structures; solid islands of cells; and squamous cysts set within a fibrous stroma that can show myxoid or hyaline change. The tubular structures often have a comma-shaped extension similar to that seen in other syringomatous tumors (Fig. 9.16). Calcification may be present within the epithelial structures. Infiltration of stroma is characteristic and muscular and perineural invasion is common (31,32). This lesion has features similar to those of the low-grade adenosquamous carcinoma that occasionally arises within the parenchyma of the breast (see the section “Metaplastic Carcinoma” later in this chapter). It is also similar to microcystic adnexal carcinoma of the nonmammary skin. In the nipple region, syringomatous adenoma appears to behave in a benign fashion, but complete excision with a clear margin is advisable because recurrence of incompletely excised syringomas has been recorded (31,32). Syringomatous adenoma may be histologically confused with tubular carcinoma, which rarely occurs in the nipple. Features helpful in the differential diagnosis of nipple adenoma, syringomatous adenoma, and tubular carcinoma are listed in Table 9.8.
GENERAL GUIDELINES FOR THE HISTOPATHOLOGIC INTERPRETATION OF LESIONS OF BREAST TISSUE
A thorough understanding of the changes that occur in normal breast is essential to the assessment of pathologic lesions. As discussed previously, most lesions arise within the TDLU, and most benign lesions retain some lobular architecture, although it may be severely distorted, and frequently the specialized stroma is no longer discernible. With the exception of microglandular adenosis, benign lesions, however florid, contain epithelium and myoepithelium, but immunostaining may be needed to distinguish them.
Nonneoplastic benign epithelial changes affecting the TDLU can be divided broadly into four categories:
1. Metaplastic change within the epithelium (e.g., apocrine metaplasia)
2. Hyperplasia: an increased number of epithelial cells within preexisting glandular components, variously called epithelial hyperplasia, epitheliosis, and papillomatosis
3. An increased number of glandular components, termed adenosis
4. Distortion of preexisting glandular components by the stroma
In many benign lesions, a combination of these changes is seen, such as the coexistence of adenosis, sclerosis, elastosis, cyst formation, and epithelial hyperplasia in a radial scar. When breast tissue is examined, an initial low-power assessment is necessary to establish the presence or absence of an overall lobular architecture, and a high-power view is needed to confirm the presence or absence of the two cell types characteristic of benign lesions. Most benign lesions retain these two characteristics.
In contrast to benign changes, infiltrating carcinoma lacks a lobular pattern. The proliferating epithelial components are randomly arranged, variable in size, and consist of only one cell type (except in a few rare entities). Even when tubule formation is seen in well-differentiated carcinomas, the tubules are lined by a single type of cell without a myoepithelial component.
In many instances, one can avoid an overdiagnosis of malignancy by acquiring familiarity with the benign lesions that may mimic carcinoma. Lesions that commonly cause problems are the following:
1. Physiologic hyperplasia. Florid hyperplastic changes seen in the secretory breast (during pregnancy and lactation) may contain large nuclei with prominent nucleoli.
2. Reactive changes. Occasionally, chronic inflammation around a cyst or in an area of fat necrosis, when viewed at low power, infiltrates into the stroma and thus resembles infiltrating carcinoma.
3. Benign epithelial hyperplasia of ductal type, which may be mistaken for carcinoma in situ (see section “Epithelial Hyperplasia”).
4. Benign intraductal papilloma. Differentiation between benign and malignant papillary lesions is frequently difficult. Sclerosis within an intraductal papilloma further compounds the problem and may mimic infiltrating carcinoma. Similar problems can also occur with ductal adenomas (see sections “Papillary Lesions” and “Ductal Adenoma”).
5. Sclerosing lesions (sclerosing adenosis, radial scar, ductal adenoma), which may be mistaken for infiltrating carcinoma. The presence of apocrine metaplasia or coexisting cancerization by DCIS can cause particular problems (see sections “Sclerosing Adenosis,” “Radial Scar and Complex Sclerosing Lesion,” and “Ductal Carcinoma in Situ”).
6. Treatment-induced changes. Radiation and chemotherapy may induce distortion and striking nuclear atypia (see section “Changes Induced by Therapy”).
Before making a diagnosis of malignancy, particularly on frozen section or when dealing with a small tissue sample (e.g., CNB), the practitioner should always consider the benign lesions that mimic cancer.
BENIGN BREAST DISEASE
Every breast biopsy does not contain pathologic changes. Provided that the specimen has been carefully and thoroughly sampled, the pathologist should not be afraid of recording a diagnosis of normal breast tissue. A wide variety of physiologic changes affect the mammary parenchyma, especially the stroma, but they do not always occur uniformly throughout the tissue and, as a result, asymmetric features may be clinically observed.
Several all-embracing terms denote a wide spectrum of appearances. The term fibrocystic change is frequently used, but it should be accompanied by a full description of the specific changes found in the biopsy specimen. This has important prognostic significance because only some lesions have been shown to carry an increased risk for subsequent carcinoma. It is also important that the terms used to describe benign breast disease be clearly defined. Definitions of the most frequently used terms appear in Table 9.9.

METAPLASTIC EPITHELIAL CHANGE
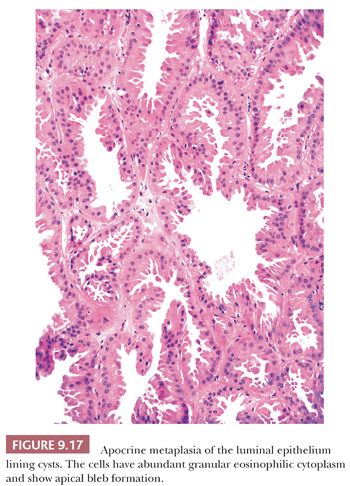
Apocrine metaplasia is common within breast epithelium. The luminal epithelial cells resemble those of apocrine glands. The cells are larger than normal, have abundant granular eosinophilic cytoplasm, and show apical luminal bleb formation (Fig. 9.17). Ill-defined supranuclear vacuoles are often present. The nuclei are of variable size and may be either hyperchromatic or vesicular, but they generally have prominent nucleoli. Sometimes, the nuclear features of apocrine epithelium can be worrisome, especially in the setting of other florid, benign epithelial changes, such as sclerosing adenosis and intraductal epithelial hyperplasia. Once the apocrine nature of the cells has been recognized, however, care should be exercised in their interpretation to avoid overdiagnosis. It has recently been reported that some benign apocrine lesions may partially or completely lack a myoepithelial layer (33).
Apocrine metaplasia in a background of adenosis has been termed apocrine adenosis. This term has also been used in other contexts—namely, apocrine metaplasia specifically within sclerosing adenosis (34) and a distinctive form of adenosis associated with adenomyoepithelioma (35).
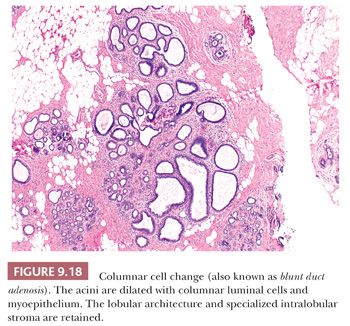
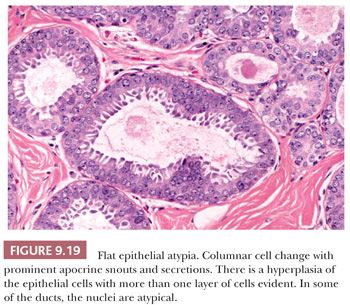
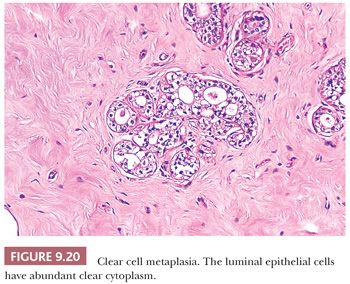
In columnar cell change, acini are variably dilated and lined by columnar epithelial cells with oval to elongated nuclei. Apical cytoplasmic snouts are often present at the luminal surface of these cells (Fig. 9.18). The term columnar alteration with prominent apical snouts and secretions (CAPSS) has been used to describe this appearance (36). Atypical hyperplasia in this setting has been described as flat epithelial atypia (FEA) (37) (see Fig. 9.19). Sometimes the luminal epithelial cells exhibit abundant clear cytoplasm, and it has been postulated that this clear cell metaplasia is a form of eccrine differentiation rather than a response to hormonal stimulation, as was previously suggested (38) (Fig. 9.20). This change has been noted in benign lesions and otherwise normal breast epithelium. Columnar cell changes have been found in association with neoplastic lesions, which are generally regarded as of low grade, but a cause-and-effect relationship has not been established (39).
Squamous metaplasia occasionally occurs in benign lesions, most often in duct papillomas and adenomas and in benign phyllodes tumors; more rarely, it occurs in radial sclerosing lesions and fibroadenomas, and it is also seen infrequently in otherwise normal structures traumatized by previous surgery. In at least some instances, the squamous cells appear to arise from myoepithelium rather than luminal epithelium (40). Except that their nature should be recognized, apocrine, columnar, clear cell, and squamous cell changes are of no known clinical significance.
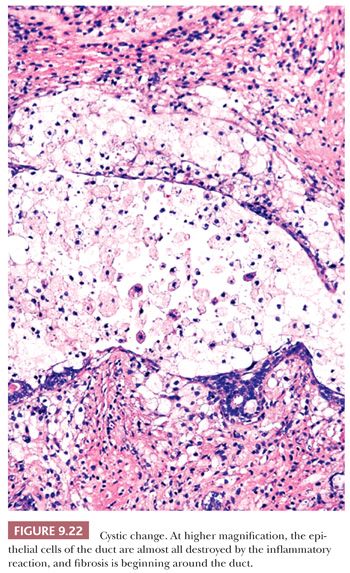
CYSTS
Cyst formation is one of the most common changes seen in breast tissue and is frequently seen in combination with other benign lesions. As described in the section “The Normal Breast,” it is generally accepted that cysts arise from lobular acini that coalesce (Figs. 9.21 and 9.22). The cysts are lined by a double cell layer, and the luminal epithelium frequently shows apocrine metaplasia. Cystic change does not usually cause major diagnostic problems; however, minor problems in interpretation may be encountered in the following situations:
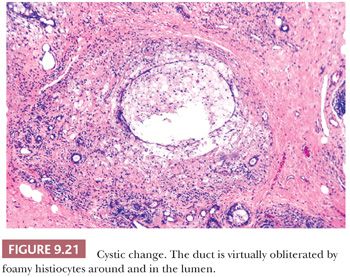
1. In large cysts, the epithelial lining may be partially, if not entirely, lost.
2. In duct ectasia, an inflammatory reaction around a cyst is accompanied by fibrosis and plasma cells, which can be mistaken for infiltrating carcinoma on low-power examination.
3. An intense inflammatory reaction consisting mainly of foamy macrophages may be all that remains at the site of a cyst and may be mistaken for fat necrosis.
4. Elastic tissue can be demonstrated around ectatic ducts.
5. Benign cysts must be differentiated from cystic hypersecretory DCIS.
ADENOSIS
The term adenosis, defined in Table 9.9, indicates an increase in the number of glandular elements. Younger women normally have many more lobules than the older women who are more often biopsied. There is wide, partly age-dependent variation in the density of lobular units. The term simple adenosis implies an increase in the size and number of lobules. The lobular proliferation seen in pregnancy is a “physiologic” form of adenosis.
Sclerosing Adenosis
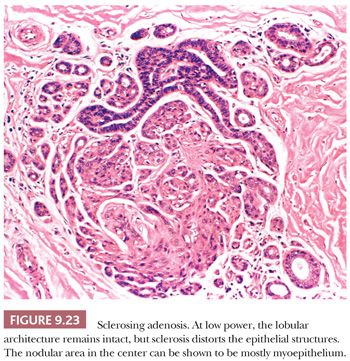
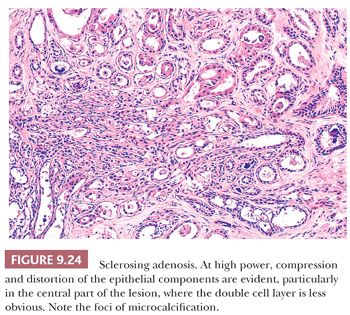
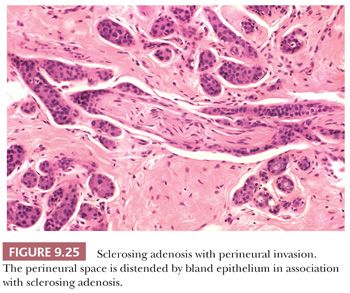
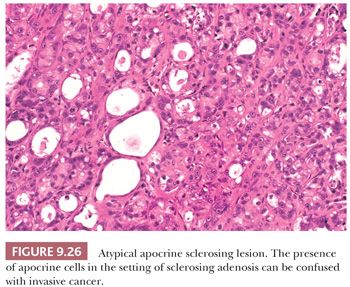
Sclerosing adenosis has the size and shape of an expanded lobule with a numeric increase in the glandular elements, accompanied by stromal proliferation, which produces glandular compression and distortion. It is especially important to be familiar with this most commonly seen form of adenosis because it can mimic carcinoma mammographically, and histologically, it may mimic invasive lobular or tubular carcinoma. Particular difficulty can occur with frozen sections and small tissue samples, but problems will be reduced if attention is paid to the aforementioned general guidelines. In cases of sclerosing adenosis, low-power examination reveals multiple nodular areas with retention of the overall rounded and defined lobular architecture (Fig. 9.23). The numerous tubules, although frequently compressed and distorted, retain their two cell layers, but this feature may not be evident in each individual tubule (Fig. 9.24). The compression of the glandular components is more marked at the center of the lesion, whereas the caliber of those at the periphery is greater. Myoepithelial proliferation is often a major component of the lesion, and sheets of these cells can be confirmed as myoepithelial by immunohistochemistry. Microcalcification is frequently seen within the glandular lumina (Fig. 9.24). The fibrous stroma is usually dense and often hyalinized, and elastic tissue may be prominent. Compression of the tubules occludes their lumina in many areas, resulting in an infiltrative growth pattern. Actual invasion of perineural spaces (Fig. 9.25) and vessels, although uncommon, is a well-recognized phenomenon associated with sclerosing adenosis and is not, by itself, an indication of malignancy. The proliferating epithelial cells lack atypical features, but a particularly worrisome picture may be seen when apocrine metaplasia is also present. Recognition of the metaplastic nature of the epithelial cells is therefore especially important in the context of sclerosing adenosis. As discussed previously, the term apocrine adenosis has been used to describe this phenomenon (34). The term atypical apocrine adenosis (Fig. 9.26) has been used when the apocrine cells show nuclear atypia (41,42). The difficulty of distinguishing the latter, which may show alarming cytology, from simple apocrine epithelium on the one hand and from apocrine carcinoma in situ on the other makes the study of so-called apocrine adenosis in relation to the incidence of subsequent carcinoma a somewhat controversial subject. When sclerosing adenosis contains in situ carcinoma, the differential diagnosis from infiltrating carcinoma may be difficult. Features that distinguish sclerosing adenosis from tubular carcinoma are listed in Table 9.10. In difficult cases, immunohistochemistry may help identify myoepithelial cells and basement membranes (Table 9.5). The term nodular adenosis, or adenosis tumor, has been applied to florid examples of sclerosing adenosis that enlarge to form palpable lesions, sometimes seen as rounded, pink, granular areas on gross examination.
Radial Scar and Complex Sclerosing Lesion
A variety of terms, including radial scar, complex sclerosing lesion, radial sclerosing lesion, scleroelastotic scar, stellate scar, nonencapsulated sclerosing lesion, indurative mastopathy, benign sclerosing ductal proliferation, and sclerosing papillary proliferation, have been used for this histologically complex lesion, which typically contains adenosis, epithelial hyperplasia, and, frequently, cyst formation. The term radial scar has been reserved by some for individual lesions of less than 1 cm; larger, more complex lesions, frequently with more pronounced architectural disturbance (possibly resulting from the convergence of several adjacent radial scars), have been termed complex sclerosing lesions. The number of synonyms for this lesion reflects the controversy surrounding its genesis and its variable microscopic appearance.
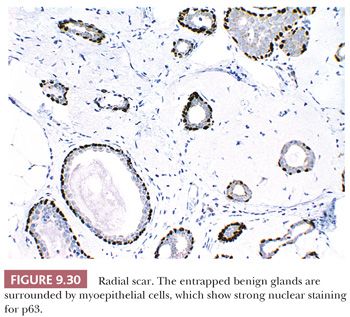
Radial scar can mimic carcinoma mammographically and grossly because of its stellate appearance and central sclerosis and elastosis (Fig. 9.27). The distinction from infiltrating carcinoma can also be difficult on histologic examination. For a summary of those features helpful in the differential diagnosis of benign sclerosing lesions and infiltrating carcinoma, see Table 9.10. Radial scars are generally nonpalpable, often multiple, and frequently bilateral. They are often discovered as an incidental finding in breast specimens, but may be detected mammographically. They appear grossly as firm, chalky white lesions with an irregular outline. The stellate appearance is caused by bands of dense, fibrous connective tissue extending outward from the central core ending in dilated ducts and giving the appearance of a daisy (Fig. 9.28). The characteristic feature is the central elastotic scar containing a few small, entrapped glandular structures, which may simulate tubular carcinoma. However, the entrapped ducts are usually relatively few in number and, at least some of them, can be shown to contain epithelial and myoepithelial layers (Figs. 9.29 and 9.30). They are surrounded by dense collagen and elastosis, which may be a dominant feature. Surrounding the central scar there may be lobules, sometimes distorted by sclerosis, and/or ducts with epithelial hyperplasia, which may be atypical or, rarely, contain intraductal carcinoma. In the outer epithelial components, apocrine metaplasia and calcification may be present. Radial scars are often associated with other benign lesions, particularly sclerosing adenosis. Although the clinical significance of radial scars remains controversial (43,44), most authorities consider them to be benign mimics of cancer (45). However, an increased incidence of carcinoma and atypical ductal hyperplasia (ADH) appears to be associated with large radial scars, particularly in women older than 50 years (46). In addition, one clinical follow-up study suggested that radial scars are associated with an increased risk for subsequent breast cancer, particularly when found in breasts that exhibit other benign proliferative lesions (47). Therefore, the size and histologic features of radial scar should be recorded with that diagnosis.
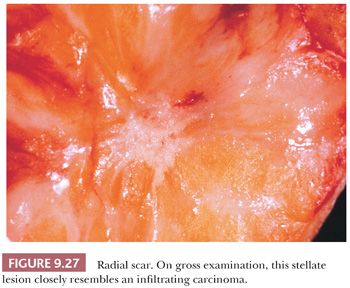
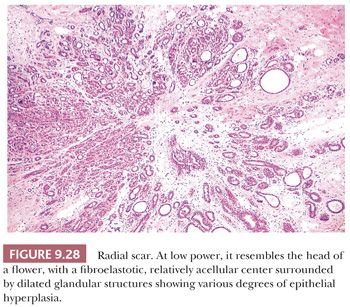
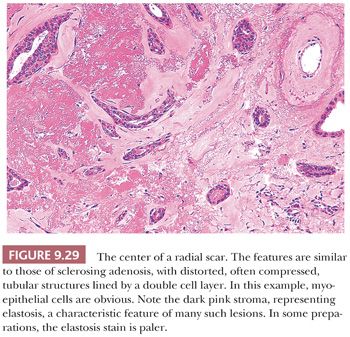
Radial scars are being reported with increasing frequency on screening mammograms, because they produce mammographic abnormalities that may be impossible for the radiologist to distinguish from those of infiltrating carcinoma. The possible confusion of radial scars with carcinoma is accentuated in CNB specimens because appreciation of the overall architecture may not be possible in a small sample, and the central scar more closely mimics cancer unless the context is noticed.
Tubular Adenosis
Tubular adenosis is an unusual, benign lesion that may also be misinterpreted as carcinoma (48). Microscopic examination reveals a haphazard proliferation of uniform, elongated, sinuous tubules with a bilayered structure of both luminal epithelium and myoepithelium. This lesion lacks the circumscription characteristic of most other types of adenosis. Luminal secretions are usually present and microcalcification is frequent. The rare occurrence of cancerization of this lesion by DCIS adds to the difficulties of differential diagnosis (48).
Microglandular Adenosis
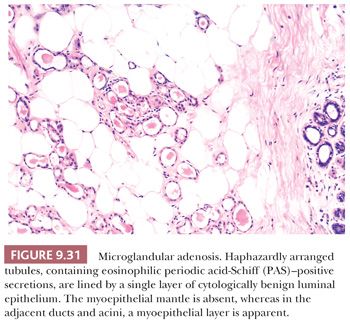
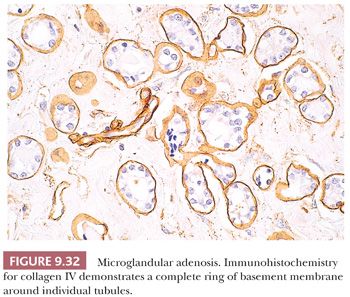
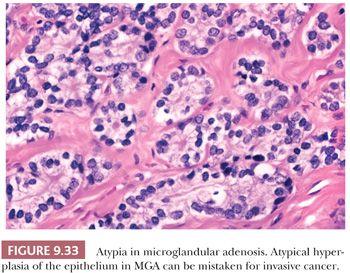
In microglandular adenosis, the proliferating tubules are lined by a single epithelial layer and lack an outer myoepithelial mantle (49–51). The arrangement of the tubules is haphazard; they are small, regular, and rounded without angulation (Fig. 9.31). Eosinophilic, periodic acid-Schiff (PAS)–positive, diastase-resistant material is frequently seen within the cytoplasm and the lumina, as are calcium deposits. The epithelial cells are occasionally vacuolated, cuboidal or flattened without apical snouts, and are cytologically benign. Immunohistochemistry with basement membrane markers, such as laminin and collagen IV (Fig. 9.32), in addition to electron microscopy, confirms the presence of an intact basement membrane around the epithelial structures and the absence of myoepithelial cells. An important feature in the recognition of microglandular adenosis is the absence of a desmoplastic reaction around the tubules. Most reports of microglandular adenosis indicate that it is benign (49–51), but atypical forms have been described (Fig. 9.33) (atypical microglandular adenosis) (52), and, rarely, carcinoma can arise in or in conjunction with microglandular adenosis (52–53).
The term secretory adenosis has been used to describe a lesion architecturally similar to microglandular adenosis but in which myoepithelial cells are evident around the tubular structures (54).
Blunt Duct Adenosis (Columnar Cell Change and Columnar Cell Hyperplasia)
The term blunt duct adenosis has been used by different authors to describe different lesions. Most often, this term has been applied to the histologic appearance illustrated in Figure 9.18. The lobular architecture is well maintained, usually with retention of the specialized stroma. It is debated whether acinar formation occurs at all or whether, as the name implies, the process results from the proliferation of terminal ducts without the development of true acini. “Acinar” structures may be increased in number and are distorted and variably dilated. The luminal cells lining these spaces are typically columnar and often show apical snouts. It has been proposed that lesions with this constellation of histologic features be termed columnar cell change when only one or two layers of epithelial cells are present and columnar cell hyperplasia when more than two epithelial cell layers are seen (55). Included within this category are lesions designated as columnar alteration with prominent apical snouts and secretions (CAPSS).
FLAT EPITHELIAL ATYPIA
Flat epithelial atypia is a descriptive term that encompasses changes in the breast TDLUs in which variably dilated acini are lined by one to several layers of epithelial cells, columnar in shape, which display low-grade monomorphic cytologic atypia (Fig. 9.19). There is no architectural pattern, the cells lack polarity, and their nuclei overlap those of low-grade DCIS. If a papillary or cribriform pattern were present, the lesion would merit consideration for ADH/DCIS. Observational studies have suggested that at least some FEAs may represent either a precursor of DCIS or early DCIS. However, the few follow-up studies of FEA suggest that the risk of local recurrence or progression to invasive carcinoma is low (37) and substantially less than for ADH or lobular carcinoma in situ. As a consequence, recommendations for clinical management are still evolving.
EPITHELIAL HYPERPLASIA
The term epithelial hyperplasia indicates a proliferation of epithelial cells within preexisting glandular structures. Ductal epithelial hyperplasia is synonymous with intraductal hyperplasia, epitheliosis, hyperplasia of usual type, and usual ductal hyperplasia (UDH), which is the term in current usage. No universally recognized entity is termed lobular hyperplasia. The distinction between UDH, ADH, and DCIS is sometimes problematic and raises some of the most frequent and difficult diagnostic dilemmas in breast pathology (Table 9.11). There are two borderlines between UDH and ADH and between ADH and DCIS, and even the most experienced pathologists do not always agree on individual cases. This has been emphasized by a study showing considerable disagreement in the evaluation of a small group of proliferative ductal lesions by pathologists (56). Reproducibility can be improved if carefully defined histologic criteria are used (57). Although ancillary techniques, including assessment of proliferative activity, morphometry, and immunohistochemistry with various antibodies (e.g., cytokeratins 5 and 6), have shown some overall differences between the groups, they do not allow clear separation of entities in individual cases.

Usual Ductal Hyperplasia
UDH varies in extent from a few foci to widespread involvement of large areas of glandular tissue and in degree of hyperplasia within each glandular structure. Several systems for grading the degree of hyperplasia have been described on the assumption that a spectrum of changes exists, ranging from normal to hyperplasia to atypia to carcinoma in situ (58). However, the concept that progression through these stages, histologically and biologically, occurs is not accepted by all (59). Certain architectural and cytologic features are characteristic of DCIS, and these hallmarks must be present before a diagnosis of carcinoma in situ can be made. However, it is probable that even though some proliferating cells are committed to eventuate in invasive carcinoma without manifesting diagnostic features of DCIS, some DCIS lesions are merely markers of an increased risk for the subsequent development of invasive malignancy and never actually progress themselves.
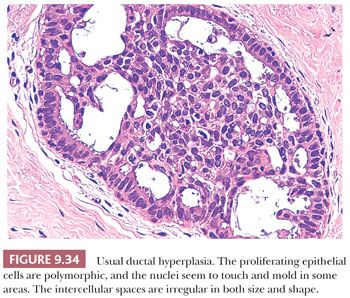
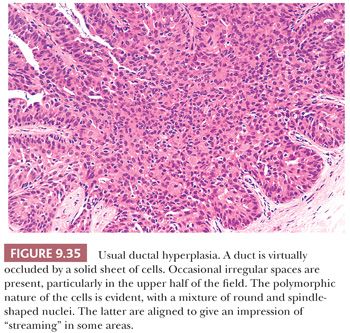
The features of UDH are best evaluated in ducts, although they may extend into acini, where the degree of irregularity is limited by the size of the lumens. UDH ranges from mild hyperplasia, in which the proliferating epithelium is more than two cells but no more than four cells thick, to moderate hyperplasia, marked by considerable luminal filling and bridging (Fig. 9.34), to florid hyperplasia, with distended ducts with lumens partially obscured by large and actively proliferating epithelial cells (Fig. 9.35). The differentiation of UDH from carcinoma in situ rests on several features listed in Table 9.12. The presence of outright malignant cells is definitive, but these are only seen in high-grade DCIS, which is much more readily recognized. It is the distinction of the overlapping features of UDH and low-grade DCIS which is much more problematic.
Low-power features characteristic of UDH include the following:
1. Luminal spaces between the proliferating cells are irregular and often slitlike (Fig. 9.34). They are frequently concentrated around the periphery of the duct. Micropapillary projections, if present, are elongated (often tapering) or tuftlike and may resemble the pattern of hyperplasia seen in gynecomastia (60). The occasional presence of stromal cores results in true papillary structures. Islands of stroma, separated by a myoepithelial layer, may be seen within a solid proliferation of epithelium.
2. More importantly, cellular arrangement is also irregular. Nuclei are densely crowded, overlap, and appear to “touch.”
3. Necrosis and hemorrhage are rare, except in larger papillary lesions.
4. Periductal stromal changes, such as fibrosis, elastosis, and chronic inflammation, are uncommon.
High-power features characteristic of benign hyperplasia include the following:
1. Cell and nuclear size, shape, and placement vary. A polymorphic cell population consisting of epithelium and spindle-shaped cells is present, sometimes with varying numbers of lymphocytes and histiocytes.
2. Apocrine metaplasia is often present. This is usually focal, but hyperplastic lesions composed entirely of apocrine cells also occur. As discussed previously, the cytologic appearance of apocrine cells may be worrisome, and a diagnosis of apocrine DCIS carcinoma should not be made without unequivocal evidence of malignancy.
3. The nuclei of proliferating cells are often parallel to one another, creating the impression of “streaming” (Fig. 9.35).
4. When epithelial bridges form, the nuclei of the proliferating cells lie parallel to the line of the bridge, and the bridges often taper in the center.
5. Nuclear features diagnostic of malignancy are absent. In particular, nucleoli are relatively inconspicuous, mitotic figures are infrequent, and abnormal mitotic figures are not seen.
Microcalcification and foam cells are seen in both benign and malignant lesions and are of little value in distinguishing between the two.
Atypical Ductal Hyperplasia
The concept of ADH has gained wide acceptance as a predictor for risk of subsequent carcinoma, but its recognition in an individual case continues to present a challenge. Efforts have been made to resolve the problem by more clearly defining the histologic appearance of atypical hyperplasia (61,62). In the original description, it was an unusual entity, but it is probably currently overdiagnosed. The diagnosis should be made only when lesions have some of the features of DCIS but fall short of the fully developed picture. Both architectural and cytologic features should be taken into account. However, the diagnostic criteria for atypical hyperplasia of ductal type are not yet precisely defined. This is partly because neither DCIS nor ADH is a single histologic entity; rather, each encompasses a variety of patterns so that strict histologic criteria for numerous borderlines are difficult to define. It also remains to be established whether ADH is a step in a progression between hyperplasia and carcinoma in situ or whether it merely comprises a variety of patterns in which benignity and malignancy cannot be distinguished with certainty. This may be because the features of in situ carcinoma are not fully developed within individual glandular components. Follow-up studies indicate that some patients with ADH will progress to develop lesions recognizable as DCIS or invasive cancer. The use of the atypical ductal hyperplasia term to describe DCIS lesions of limited quantity remains controversial as well. Page and Rodgers (62) stated that for a diagnosis of DCIS, diagnostic features of DCIS must be seen in at least two glandular spaces; otherwise, they were classified as ADH. On the other hand, Tavassoli and Norris (63) wrote that for a diagnosis of DCIS, the area involved should be at least 2 mm in extent, regardless of the number of ducts involved; if less than 2 mm in extent, the term atypical ductal hyperplasia was used. Follow-up data are consistent with the notion that minimal areas of DCIS, however defined, have the same prognosis as larger DCIS lesions.
The consistent finding that the risk of carcinoma subsequent to ADH applies to both breasts is compelling evidence that ADH is indeed different from DCIS, in which the subsequent risk is highest in the contiguous area (64).
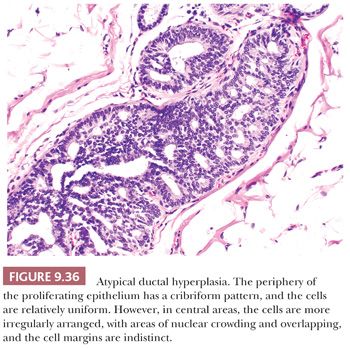
To summarize, the term atypical ductal hyperplasia should be applied only to lesions in which the diagnosis of carcinoma in situ is seriously considered but in which the features are insufficiently developed for a definite diagnosis of DCIS to be made (Figs. 9.36 and 9.37). Much work remains to be done in this area.
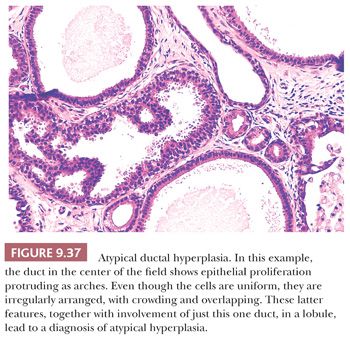
IN SITU CARCINOMA
The term in situ carcinoma was invented to describe a proliferation of presumably malignant epithelial cells that remain confined by a basement membrane. Because no lymphatics or blood vessels are present in the epithelial layer, metastatic spread cannot occur until malignant cells cross the basement membrane. This classic view has been modified in the light of electron microscopic and immunohistochemical findings, which have shown both a discontinuous basement membrane and the occasional extension of cells across the membrane (65,66). Nevertheless, the division into in situ and infiltrating carcinoma on the basis of light microscopy studies continues to be of significant clinical value. When the diagnosis of carcinoma in situ is made, it is important to exclude the coexistence of small foci of invasive carcinoma by examining multiple blocks of tissue and, if any suspect areas are found, multiple levels. Most in situ carcinomas are detected either by mammography or as an incidental finding in tissue adjacent to a palpable benign lesion. Sometimes, however, DCIS may produce a mass or present as Paget disease of the nipple or a nipple discharge. In situ carcinoma is divided into ductal and lobular types, based on the architectural and cytologic features of the proliferation rather than on its anatomic location within the ductal–lobular system because it is thought that both types arise from the TDLU.
DUCTAL CARCINOMA IN SITU
There is increasing recognition of the heterogeneous nature of DCIS in terms of clinical features, histology, biology, and, most importantly, clinical behavior. Traditionally, DCIS has been subclassified on the basis of architectural patterns, but this classification has proven unsatisfactory for several reasons. Although small lesions may show only one pattern, larger lesions frequently show considerable variation from slide to slide, within one slide, and even sometimes within a single involved TDLU or individual glandular component. Furthermore, the criteria are subjective and poorly defined, and interobserver variation is considerable. These facts are reflected in the varying incidence rates for the different subtypes in reported series of DCIS, and they may account for the differences in the incidence rates of subsequent invasive carcinoma reported for the various subtypes of DCIS in patients undergoing treatment with less than mastectomy. Because DCIS is being diagnosed more frequently, largely as a consequence of the increased use of mammography in breast cancer screening programs (67,68) and because of increased use of conservation therapy, more attention should be paid to the different histologic features of DCIS, both to understand its biology and to devise a more reproducible classification.
Although several studies suggest that recurrence following DCIS treated by conservation therapy is more frequent in patients with high-grade, poorly differentiated lesions (69–72), some reports indicate that recurrence in patients with low-grade, well-differentiated DCIS increases with a longer follow-up (73,74).
Within the existing classification, clear differences are found between the major patterns. The most notable of these are nuclear grade, the presence or absence of necrosis, and the presence or absence of architectural differentiation (cell polarization). In addition, the evaluation of several biologic markers, such as proliferative activity, ER and PR status, and oncoprotein production, suggests that biologic differences correlate with the histologic features (75).
Initial attempts to improve the classification divided DCIS into two broad groups, termed variously comedo and noncomedo, high-grade and low-grade, and large cell or small cell. However, evidence is increasing that division into two categories is an oversimplification, and several proposed systems divide DCIS into three categories (Table 9.12). Most of these are based on cytonuclear grade and the presence or absence of necrosis, but in one classification the secondary feature is the presence or absence of architectural differentiation, as manifested by the polarization of cells. Thus, general agreement has been reached in regard to the category that includes lesions with the least differentiated nuclei. Debate centers around whether division of the better differentiated lesions should be based on the presence or absence of necrosis or cell polarization or only on cytonuclear features. Lagios (76) was the first to emphasize the value of nuclear grade in the classification of DCIS, and he used a combination of nuclear grade, architectural growth pattern, and necrosis to define four categories, later contracted to three (76). In a selected group of patients, the risk for local recurrence following lumpectomy was significantly greater after the excision of high-grade lesions than after the excision of low-grade lesions. In Nottingham, DCIS was divided into pure comedo DCIS, DCIS with necrosis (non–pure comedo), and DCIS without necrosis (71). These two systems were used in the development of the Van Nuys classification (Table 9.13), which includes all cases with poorly differentiated nuclei in a high-grade group; the remaining cases of non–high-grade DCIS are divided according to the presence or absence of necrosis (69). A group of European pathologists proposed classifying DCIS into poorly, intermediately, and well-differentiated groups (77). Division is based primarily on cytonuclear differentiation and secondarily on architectural differentiation—which refers to the polarization of cells around intercellular spaces within the ductal lumen and over the surface of papillae. It has yet to be ascertained which of the aforementioned classifications will be the most widely accepted.
At a 1997 consensus conference (78), it was suggested that because all the major classifications reflect nuclear grade, DCIS should be categorized primarily according to nuclear grade, but that other features (particularly the presence or absence of necrosis and cellular polarization and the architectural growth pattern) should also be recorded. In addition, margins, lesion size, microcalcification associated with DCIS, correlation of DCIS with specimen x-ray and mammographic findings, and the presence of any microinvasion should all be documented in the pathology report.
The nuclear grade is usually homogeneous within a lesion but, when it is not, the proportion of each grade and the size and amount of the highest grade or percentage relevant to the entire lesion should be stated. High-grade nuclei are pleomorphic, with marked variation in size and shape, and they are usually large (more than 2.5 times the size of normal duct epithelial nuclei). The contours are irregular, the chromatin is coarse and irregularly distributed or vesicular, and the nucleoli are prominent and often multiple. Mitoses are usually present and often numerous. Low-grade nuclei are rounded and uniform in size and shape, and they are monotonous and monomorphic in appearance. Even though they are larger than those of the normal breast (1.5 to 2 times the size of normal duct epithelial nuclei), they are usually smaller than high-grade nuclei. The nuclear contour is smooth and rounded or oval, the chromatin is diffuse and fine, and the nucleoli are inconspicuous or absent. These features, together with the uniform spacing of the cells, give the lesions a strikingly monotonous appearance. Mitoses are few or absent. Intermediate-grade nuclei are the most difficult to define. Although they are pleomorphic, they do not show such marked variation in shape, size, and outline as high-grade nuclei. The size is usually intermediate; the chromatin is fine to coarse; nucleoli, although evident, are not prominent; and mitoses, although often present, are not frequent.
Necrosis should be carefully assessed and not confused with secretion or apoptosis. By definition, ghost cells and karyorrhectic debris should be present. Comedo-type necrosis consists of a central zone of necrosis within a duct. Punctate necrosis is not zonal.
Architectural differentiation (cell polarization) is the feature used in the DCIS classification described by Holland et al. (77). It is defined as polarization or radial orientation of the apices of the tumor cells toward intercellular spaces. A series of such formations results in a true cribriform pattern. Sometimes the intercellular spaces, or lumina, are very small, or cellular orientation is present without the formation of a true lumen, so that a rosette-like appearance results that may be seen in the solid pattern of DCIS. Polarization over cellular protuberances and bridges and over micropapillae is seen in the true micropapillary and clinging patterns of DCIS. Although the apices of the cells covering the cellular protrusions are oriented toward the duct lumen, polarization is not always as easy to see in these latter patterns as it is in the cribriform and solid patterns. Architectural differentiation usually parallels nuclear differentiation; it is minimal or absent in DCIS composed of cells of high nuclear grade but marked in DCIS composed of cells of low nuclear grade.
The architectural growth pattern should be described as comedo, solid, cribriform, papillary, micropapillary, or clinging. According to the criteria of Holland et al. (77), a true cribriform or micropapillary pattern is seen only when cell polarization is present. The appearance resembling a cribriform or micropapillary pattern, sometimes seen in DCIS of high nuclear grade, is usually a pseudopattern because cell polarization is absent.
The assessment of surgical margins and extent of disease is also of extreme importance.
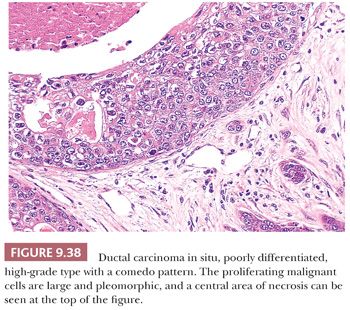
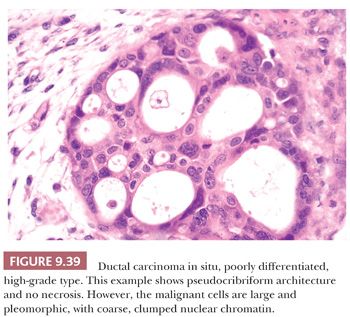
High-Grade Ductal Carcinoma in Situ, Poorly Differentiated Ductal Carcinoma in Situ, Comedo Ductal Carcinoma in Situ
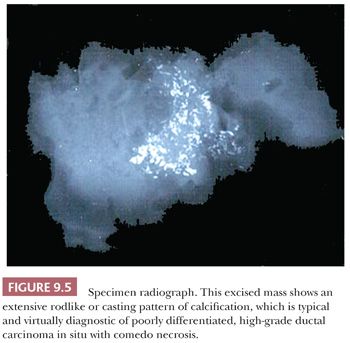
The distinction between DCIS with cells of high nuclear grade and benign disease does not usually present a problem because the component cells of DCIS have overt malignant features. Architectural differentiation (cell polarization), as described by Holland et al. (77), is usually absent or minimal, although it may be present around a few intercellular spaces. Central comedo-type necrosis is generally associated with high-grade nuclei and is a defining feature in some classifications, but not others (Table 9.12, Fig. 9.38). Necrosis is occasionally so extensive that only a few cells are seen at the periphery of the glandular lumen. Such lesions form part of the spectrum of DCIS with a clinging pattern. Some classifications accept that the malignant cells occasionally produce a solid sheet filling the duct lumen without central necrosis. Alternatively, the cells may grow in a pseudocribriform or pseudomicropapillary pattern (without cell polarization) (Fig. 9.39). The calcification, so often found within the central necrotic material, is amorphous and generally produces a typical branching, linear, or casting pattern on mammograms that is usually diagnostic of malignancy (79) (Fig. 9.5). The involved ducts may be very distended, and this type of DCIS is often quite large and may result in a palpable abnormality in the breast. This is partly a consequence of the surrounding stromal reaction, which is usually more evident in this pattern of DCIS than in others. Cancerization of the lobules is frequent. When present, Paget disease of the nipple is almost invariably associated with DCIS composed of cells with high-grade nuclei.
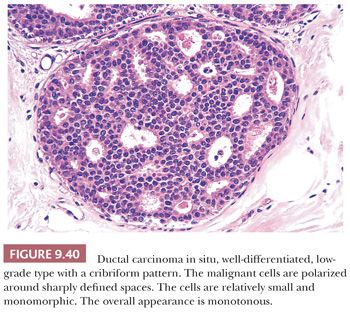
Low-Grade Ductal Carcinoma in Situ, Well-Differentiated Ductal Carcinoma in Situ
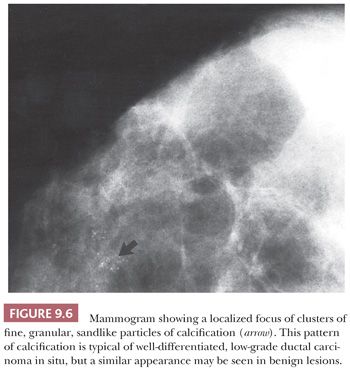
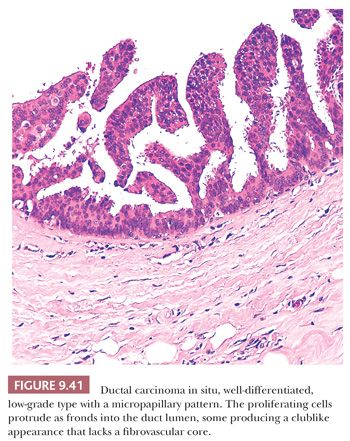
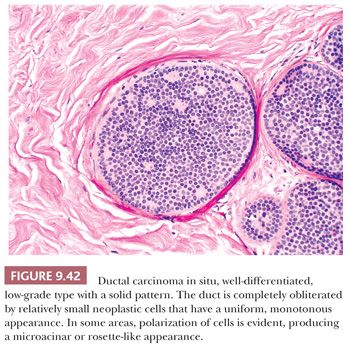
This type of DCIS is at the other end of the spectrum of classifications based primarily on nuclear grade. Most cases of the Van Nuys non–high-grade DCIS without necrosis and the Nottingham DCIS without necrosis probably fall into this category. DCIS composed of cells of low nuclear grade is sometimes difficult to differentiate from ductal epithelial hyperplasia. The most striking feature is the monotonous appearance of the cells. The growth pattern of most lesions of this group is true cribriform (Fig. 9.40), micropapillary (Fig. 9.41), or, less frequently, solid (Fig. 9.42). Prominent, uniform cell polarization (architectural differentiation) is seen. In the cribriform pattern, lumina between the proliferating cells are geometric, punched out, rigid, and rounded (Fig. 9.40), and they are usually evenly distributed within the cell masses. The cells within the center of the proliferating strands forming the bridges and arcades are arranged regularly or lie at a right angle to the plane of the cellular strands. In the micropapillary pattern, proliferating cells extend into the lumen of the glandular structures without a fibrovascular stalk. The papillae frequently have a clublike appearance, and cells are evenly distributed within them (Fig. 9.41), but the micropapillae may join to form arcuate structures around lumens, which are much less regular in size and shape and more slitlike than in a true cribriform pattern. Occasionally, true papillae with fibrovascular cores are associated with the micropapillae. Small rosettes of cells with surface polarization, apparently separated from the papillae, are often seen floating free within the duct lumen. When the growth pattern is solid, a pattern of small, incompletely formed intercellular spaces with polarization of the surrounding cells usually results in a rosette-like appearance (Fig. 9.42). Necrosis is rarely associated with cells of low nuclear grade, and its absence is one of the defining features of some classifications, but the presence of small, focal areas of necrosis is allowed in others (Table 9.12). However, secretions are frequently seen within the duct lumen and should not be mistaken for necrosis. Calcification is not as frequent as in poorly differentiated DCIS and, when present, it is usually rounded and laminated or psammomatous and deposited within the secretions. Such calcification produces clusters of granular, sandlike particles on mammograms (Fig. 9.6), but this pattern is not as distinctive as that of the calcification seen in poorly differentiated, high-grade DCIS, in that similar patterns may also occur in benign lesions (70). Some patterns of well-differentiated DCIS are difficult to distinguish from lobular carcinoma in situ (LCIS). Furthermore, some in situ carcinomas may show mixed ductal and lobular features.
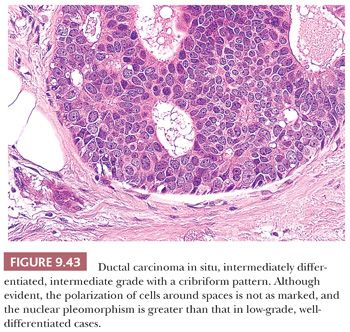
Intermediate-Grade Ductal Carcinoma in Situ, Intermediately Differentiated Ductal Carcinoma in Situ
The third type of DCIS in classifications based primarily on a three-tier nuclear grade system is composed of cells with intermediate-grade nuclei. Its features are intermediate between those of high-grade, poorly differentiated types and low-grade, well-differentiated types. In the classification of Holland et al. (77), the presence of cell polarization is important, although this is not as prominent and uniform as in the well-differentiated type (Fig. 9.43). The growth pattern varies and may be solid, cribriform (Fig. 9.43), micropapillary, or clinging. Necrosis may or may not be present. Calcification, when present, may be laminated or amorphous (77).
In the Van Nuys and Nottingham classifications, the presence or absence of necrosis is the defining feature that divides the non–high-grade types of DCIS.
Clinging Ductal Carcinoma in Situ
The terms clinging DCIS and clinging carcinoma have been applied to two histologically distinct lesions, both characterized by one or two cell layers confined to the periphery of the involved spaces. In the first lesion, the cells lining the involved spaces have high-grade nuclei. In these cases, central necrosis may be evident, and such lesions are examples of poorly differentiated DCIS (80,81). Other lesions included in the category of clinging DCIS by some authorities (80,81) are composed of smaller, more uniform cells with low-grade nuclei, characteristic of well-differentiated DCIS. In many such lesions, the cells are columnar. Although lesions of this type are sometimes seen in conjunction with low-grade DCIS that has a cribriform or micropapillary pattern, they are being encountered with increasing frequency as isolated lesions in breast biopsy specimens obtained to assess mammographic microcalcifications. This pattern is not universally recognized as representing fully developed DCIS, and some consider it to be part of the spectrum of ADH (61,62) or columnar cell hyperplasia with atypia (45). Such lesions do not appear to be associated with as high a risk for breast cancer as fully developed forms of low-grade DCIS (81,82).
Apocrine Ductal Carcinoma in Situ
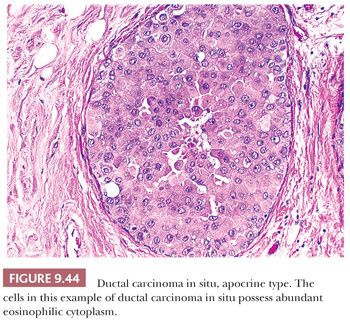
Apocrine change within DCIS is not unusual and is probably underrecognized in lesions diagnosed as DCIS. The same criteria applied in the diagnosis of nonapocrine DCIS should be used, and in most cases, this does not present a problem (Fig. 9.44). Occasionally, however, benign apocrine hyperplasia may have a worrisome appearance, and the differential diagnosis with DCIS is difficult. In our view, DCIS should be diagnosed only when the characteristic cytologic and architectural features are present.
An intermediate category of intraductal apocrine lesions has been suggested. O’Malley et al. (83) used nuclear features and lesion extent to define a borderline category between usual apocrine hyperplasia and well-differentiated, low-grade apocrine DCIS. Tavassoli and Norris (84) also used quantitative criteria to measure nuclear size, degree of hyperplasia, and lesion extent to define four groups of lesions: atypical apocrine metaplasia; atypical apocrine hyperplasia; poorly differentiated, high-grade (necrotic) DCIS; and well-differentiated, low-grade (nonnecrotic) DCIS.
Neuroendocrine (Argyrophilic) Ductal Carcinoma in Situ
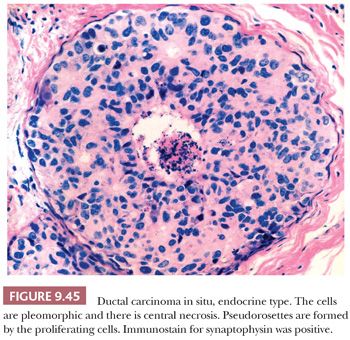
A solid pattern of DCIS with neuroendocrine histologic features has been recognized (85,86). They are usually seen in elderly patients, who often present with a blood-stained nipple discharge. The involved glandular elements are often markedly distended. The proliferating cells have a polygonal, oval, or spindle morphologic appearance and granular eosinophilic cytoplasm with intervening fibrovascular cores and septa. Rosettes and ribbons may be evident, as may mucin production and microglandular spaces (87). In some examples, the component cells are a mixture of spindle cells and argyrophilic signet ring cells (87) (Fig. 9.45). Because of the frequent lack of overt nuclear atypia, this type of DCIS can easily be misdiagnosed as benign. Small foci of invasive carcinoma are present in a significant number of cases, and in one series, neuroendocrine DCIS was associated with invasion in 20 of 34 cases. In another 18 cases, nearby papillomas were colonized by neuroendocrine DCIS (78). “Solid papillary carcinoma” of the breast would appear to be related to, if not identical with, neuroendocrine DCIS (88).
Signet Ring Cell Ductal Carcinoma in Situ
In situ ductal carcinomas consisting predominantly of signet ring cells have been described (88). However, signet ring cell formation is probably more commonly seen in in situ lobular carcinomas.
Cystic Hypersecretory Ductal Carcinoma in Situ
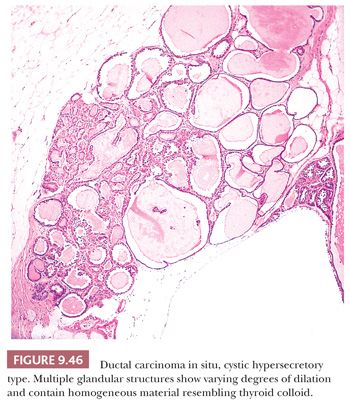
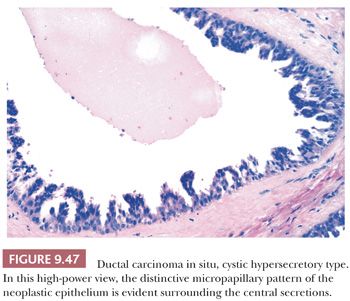
This is an uncommon pattern of DCIS (89). The gross appearance is characteristic, consisting of cysts filled with viscid material. Microscopic examination reveals multiple cyst-like structures containing homogeneous eosinophilic material that resembles thyroid colloid. Some are lined by histologically benign, flat, or columnar epithelium. Others, unlike simple cysts, have a lining epithelium that shows various degrees of hyperplasia, with foci of DCIS that have a micropapillary or cribriform pattern. The lining cells may exhibit secretory activity, with features reminiscent of the lactating breast (Figs. 9.46 and 9.47). Stains for mucin show focal positivity within the epithelial cells; most of the cyst contents are negative. This lesion can easily be overlooked and should be borne in mind when apparently benign cysts are examined. Occasionally, hyperplasia is seen without fully developed carcinoma in situ. Such lesions are termed cystic hypersecretory hyperplasia (90).
DUCTAL CARCINOMA IN SITU WITH BASAL-LIKE PHENOTYPE
Bryan et al. (91) identified a group of DCIS cases that were negative for ER, PR, and HER2/neu (triple-negative) and expressed basal cytokeratins, epidermal growth factor receptor (EGFR), or both, and suggested that they might represent precursors to invasive basal-like carcinomas. Livasy et al. (92) studied 245 cases of pure DCIS and found 61% to be luminal A type (ER+, HER2−), 9% luminal B (ER+, HER2+), 16% HER2 (HER2+, ER−) and 8% to be basal-like (ER−, HER2−, EGFR+, and/or cytokeratin 5/6+). The basal-like lesions were associated with unfavorable prognostic markers, including high-grade nuclei, p53 overexpression, and elevated Ki-67 index.
DUCTAL CARCINOMA IN SITU INVOLVING LOBULES (CANCERIZATION OF LOBULES)
When DCIS is extensive, it is frequently seen within obviously lobular structures (Fig. 9.48). The term cancerization of lobules has been used to describe this finding. It is nearly always associated with one of the typical patterns of DCIS previously described, but very occasionally it occurs alone. The appearance should not be misinterpreted as invasive carcinoma, a possibility that is enhanced when the process is further complicated by the presence of sclerosing adenosis (93,94). The differentiation from invasive carcinoma can be aided with the use of immunostaining for antibodies to myoepithelial cells (Fig. 9.49). Differentiation of lobular cancerization by DCIS from LCIS is discussed later.
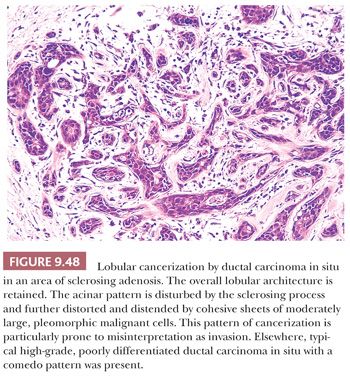
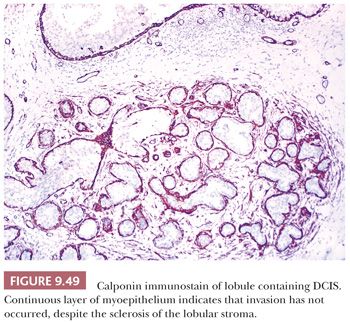
EXCLUDING INVASIVE CARCINOMA
In all cases of in situ carcinoma, it is important that the lesional tissue be examined thoroughly to exclude small foci of invasion, but invasion should not be diagnosed unless the evidence is unequivocal. Some authorities report invasion only when the interlobular stroma is involved and not when the only apparent invasion is basement membrane disruption within lobular stroma (85). The immunohistochemical demonstration of myoepithelial cells or basement membrane components may be of help in evaluating possible foci of invasion. Small foci of invasion have been found more frequently in extensive DCIS lesions and in those of high nuclear grade (70,76). The presence of microinvasion—invasive foci less than 1 mm in diameter—has little effect on prognosis.
OTHER IMPORTANT FEATURES OF DUCTAL CARCINOMA IN SITU
Following excision, DCIS most often recurs at the site of the previous lesion. This observation indicates that recurrence represents residual disease left behind at the time of initial surgery, and underscores the importance of excision margins in the treatment of DCIS. What comprises a clear margin, however, has not been well defined. The elegant three-dimensional studies of the distribution of DCIS in the mammary tree carried out by Faverly et al. (16) have a significant bearing on the surgical approach to DCIS. These studies have shown that poorly differentiated DCIS nearly always (90% of cases) exhibits a continuous growth pattern, with no gaps between the foci of DCIS. In well-differentiated DCIS, on the other hand, gaps are frequent, with normal epithelium seen between foci of DCIS. The growth pattern is defined as multifocal when the gaps are smaller than 40 mm and multicentric when they are larger than 40 mm. In only one case of DCIS, among 80 studied, were the gaps larger than 40 mm. In most (92%) of the cases of well-differentiated DCIS, the gaps were smaller than 10 mm. This means that a 10-mm surgical margin will theoretically guarantee clearance in most cases. These and other studies (95,96) negate the previously held view that DCIS is a multicentric process in most cases. When cases of DCIS are examined, the width of the nearest surgical margin should be recorded by the pathologist.
In addition to type of DCIS and the clearance margins, the size of a lesion may play a major role in dictating the most appropriate therapy for DCIS. These features are interrelated to some extent, in that low-grade, well-differentiated DCIS is more likely to be small and circumscribed, so that excision is easier, whereas high-grade, poorly differentiated DCIS is more often extensive and, in practice, more often incompletely excised. One exception to this general rule is DCIS with a micropapillary architectural pattern; studies have found that lesions with this pattern, regardless of nuclear grade, are frequently large (72,97). Furthermore, well-differentiated, low-grade DCIS is more likely to lack calcification, so the extent of the lesion on a mammogram is more difficult to define. Also, as mentioned, three-dimensional studies have shown that the growth pattern is usually discontinuous, so the assessment of excision is more difficult. When calcification is present within DCIS, mammographic assessment of the extent of the lesion can be helpful in planning surgery. In earlier studies, Holland et al. (86) found that the mammographic extent and histologic extent were more consistent in poorly differentiated than in well-differentiated lesions. With “state-of-the-art” mammography, however, such discrepancies have become less apparent (79). Evaluation of the extent of DCIS can be difficult because several adjacent sections may be involved. Careful correlation of the histologic with the gross examination findings and, when available, the results of specimen radiology is essential. Advocates of large, whole-mount sections claim that this is an area in which the technique can be of particular value. The evaluation of margins can also raise major problems in that no definition of a negative, close, or involved margin has been accepted. From a practical point of view, it is best to consider a margin positive when DCIS touches the surgical margin. In other situations, the distance between the edge of the DCIS and the margins should be recorded in millimeters or fractions thereof.
Silverstein et al. (98) proposed a prognostic index for DCIS based on histologic type, excision margin, and tumor size (Van Nuys prognostic index) (Table 9.13). They found that all three features were related independently to recurrence, but when the scores for each feature were combined, a stronger prognostic indicator was obtained. Each feature is scored from 1 through 3, and the scores are added together to give a combined score from 3 through 9. However, in a more recent study from this group, neither lesion size nor histology provided additional prognostic information in patients with a margin width of more than 10 mm, suggesting that margin status is the most important of these three factors (99).
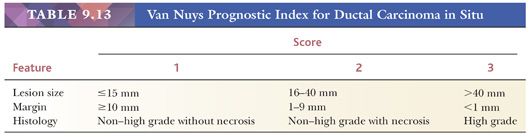
Through the years, many studies of biologic marker expression in DCIS have been carried out. The most recent studies have used immunohistochemical methods to demonstrate marker expression. Although the application of histologic classification, antibodies, and scoring methods has not been consistent, the results have shown general agreement (75). Poorly differentiated, high-grade DCIS usually has a high proliferation index; frequently expresses the c-erb-B2 oncoprotein; and more often expresses p53 protein than other types, but it is more likely to be negative for ER and PR. Low-grade, well-differentiated DCIS usually has a low proliferative index; rarely expresses either c-erb-B2 oncoprotein or p53 protein; and is nearly always positive for ER and PR. Intermediate types of DCIS show intermediate patterns of marker expression.
Angiogenesis is another feature that has been studied in DCIS. An increase in stromal microvessels has been reported, particularly in high-grade DCIS (100,101). The aforementioned associations underscore the biologic heterogeneity of DCIS. Moreover, the results of one study have indicated that, when used in conjunction with local excision and radiation therapy, tamoxifen significantly reduces the risk for local recurrence only among patients with ER-positive DCIS (102). Therefore, determination of the ER status of DCIS by immunohistochemistry is now being used with increasing frequency to guide patient management. At the present time, none of the other biologic markers has a clinical role.
Research on genetic alterations in DCIS has also provided new insights into these lesions. Some studies have indicated that the number and type of chromosomal alterations differ according to DCIS pattern and grade (103,104). However, a recent review noted that the in situ and invasive carcinoma, in a given case, show similar genetic changes and that progression to invasion may be the result of selection of a subset of heterogeneous clones in DCIS (105). The particular chromosomal loci that are altered appear to vary according to the grade of DCIS, with losses of material on chromosomes 17p and 16q particularly common in low-grade DCIS, gains of 1q and losses of 11q common in intermediate-grade DCIS, and gains of 17q and 11q common in high-grade DCIS (106). It is hoped that further understanding of the genetic and molecular alterations will lead to a better understanding of the progression from DCIS to invasive carcinoma.
DISTINGUISHING DUCTAL CARCINOMA IN SITU FROM EPITHELIAL HYPERPLASIA
In most cases of DCIS, no diagnostic problem arises; however, on occasion, differentiation from benign intraductal epithelial hyperplasia can be extremely difficult. The distinguishing features have been discussed and are listed in Table 9.11.
To summarize, low-power features suggestive of DCIS are the following: Intercellular spaces and luminal bridges are often regularly spaced; necrosis is often seen; and most significantly, the cells are larger, more evenly distributed, and either homogeneous or obviously malignant. High-power features suggestive of DCIS are the monomorphic makeup of the cells, lack of apocrine metaplasia or a streaming pattern, and the regular distribution of the nuclei and cytologic features suggestive of malignancy are usually discernible. Although in well-differentiated, low-grade DCIS nuclear pleomorphism may be minimal, the cells are usually hyperchromatic with at least one prominent nucleolus, and the nuclear-to-cytoplasmic ratio is increased. Mitotic figures are variable. They are frequent in high-grade, poorly differentiated DCIS; they are difficult to find in well-differentiated, low-grade DCIS.
The exception to these rules comes primarily in the low-grade arcuate pattern, which is less regular in its appearance.
Use of the term atypical ductal hyperplasia should be reserved for cases showing some of the aforementioned features but not the full picture of DCIS.
PAPILLARY LESIONS
Table 9.14 lists the breast lesions that have a papillary structure—a fibrovascular core covered by epithelium.
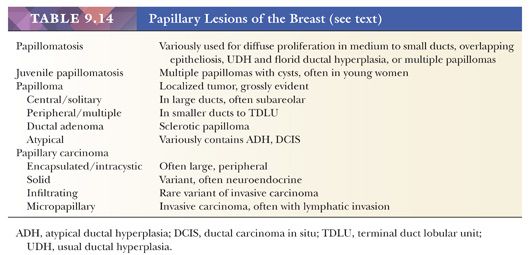
BENIGN PAPILLARY LESIONS
The most common papillary lesion is a benign diffuse, intraductal lesion, recognized microscopically as having a papillary and/or micropapillary (papillary configuration without the fibrovascular core) proliferation as well as an intact myoepithelial layer. It overlaps much of what is referred to as ductal hyperplasia, discussed earlier.
INTRADUCTAL PAPILLOMAS
Intraductal papillomas are localized intraductal lesions that present as grossly evident tumorous masses and include solitary or central papillomas, multiple or peripheral papillomas, and sclerotic papillomas, which are sometimes referred to as ductal adenomas. Papillomas occur in all age groups and in all areas of the breast. Central papillomas characteristically present as a subareolar mass with bloody nipple discharge and may be found by dissection of the ductal system, as discussed earlier under “Microdochectomy.” Grossly, they may be associated with large- or medium-size ducts to which they are confined (Fig. 9.50). Microscopically, they usually have prominent fibrovascular cores, but they may be quite cellular and irregular (Fig. 9.51) and may have all the patterns shown as ductal hyperplasia.
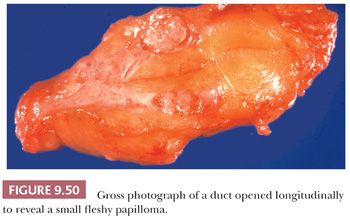
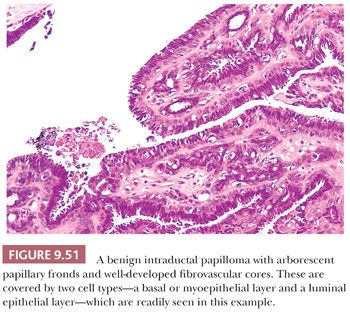
The following are the features most helpful in distinguishing between benign and malignant papillary lesions:
1. Benign papillomas have well-developed fibrovascular cores. In the classic form, these consist of arborescent fronds (Fig. 9.51), but often the papillae are broad and clublike, containing numerous components that result in a secondary adenomatous pattern.
2. The cardinal feature of a benign papillary lesion is the presence of both a luminal epithelial layer, which is usually proliferative, and an intact myoepithelial layer separating the epithelium from the basement membrane, which lies on the fibrovascular core. The myoepithelial layer is variably distinctive on H&E but is always present and can be highlighted by immunostaining for calponin, p63, and smooth muscle myosin heavy chain (Fig. 9.52).
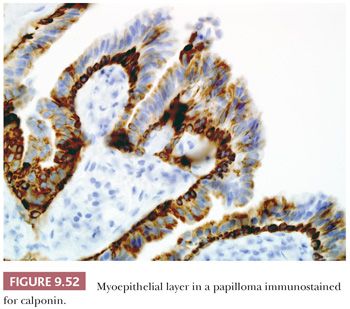
3. The luminal epithelium of a benign papilloma may show a variety of changes; epithelial hyperplasia is often present, apocrine metaplasia is frequent, and, rarely, squamous metaplasia occurs.
Infarction of benign papillomas is not uncommon and may account for the blood-stained nipple discharge (107). The presence of infarction and hemorrhage is, however, not helpful in distinguishing between benign and malignant lesions.
Some of the most alarming appearances in benign papillomas are produced by intense fibrosis in a central scar and especially around the duct with entrapment of epithelial elements (Fig. 9.53). This results in a pattern similar to that previously described in other sclerosing lesions and may raise a suspicion of infiltrating carcinoma. The presence of myoepithelium, luminal epithelium, and basement membrane in the papilloma and in its entrapped components indicate that the lesion is benign. Papillomas in which sclerosis is extreme, especially with obliteration of the ductal lumen, have been termed ductal adenomas.
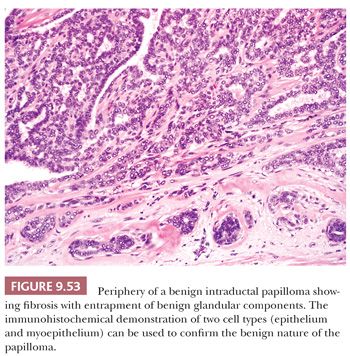
DUCTAL ADENOMA
The lesions termed ductal adenomas are thought to arise in medium-sized ducts rather than from the large ducts immediately below the nipple, and therefore they usually present as a palpable lump rather than as a nipple discharge. They may also present as a mammographic abnormality. Clinically and radiologically, the lesions can mimic carcinoma; on gross examination, although they usually consist of single or multiple rounded lesions, their gritty texture and elastic streaks may be strongly suggestive of malignancy. As with all sclerosing lesions, histologic diagnosis may be particularly difficult on frozen section and tissue core biopsy.
At low magnification, ductal adenomas are rounded and frequently bounded by the dense, fibrous wall of the distended duct within which they arise (Figs. 9.54 and 9.55), but the duct may be partially obliterated by fibrosis. The stroma is often hyalinized collagen reminiscent of that found in desmoid tumors or it may also show myxoid change or, rarely, cartilaginous metaplasia. Within the nodule, proliferating epithelial structures are seen without a papillary growth pattern. Although tubule formation is recognized in some areas, many of these are compressed and distorted by sclerosis of the stroma, resulting in a pseudoinfiltrative pattern. Focal areas of tubular dilation, epithelial hyperplasia, and cyst formation may also be seen. In some ductal adenomas, the surrounding fibrous wall is less apparent, and direct growth into the adjacent tissues appears to occur. The margin, however, remains well demarcated, and the outline is rounded and pushing. Ductal adenomas with marked central fibrosis and elastosis are strikingly similar to radial scars. High-power examination confirms the presence of two cell types lining the epithelial components. The myoepithelial cells are sometimes difficult to recognize but are often markedly hyperplastic. The luminal cells frequently show apocrine metaplasia and, sometimes, squamous metaplasia. Calcification may be present.
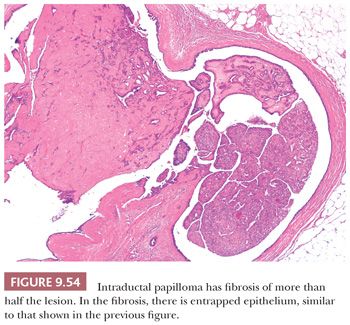
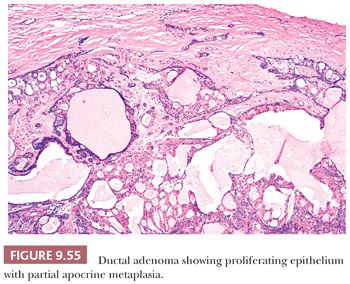
It is difficult to know how best to classify this lesion, and although the term ductal adenoma has gained wide acceptance, it is a misnomer in the strict sense of the word adenoma. Some of the lesions described by Azzopardi and Salm (108) almost certainly evolve from severe fibrosis within a benign ductal papilloma (109); some resemble pleomorphic adenomas of salivary glands; some have been included in the category of adenomyoepitheliomas; and others are placed under the heading of complex sclerosing lesions. Ductal adenomas may also occur as part of the inheritable complex of myxomas, spotty pigmentation, endocrine overactivity, and schwannomas, as described by Carney and Toorkey (110). Ductal adenomas are frequently accompanied by other benign changes, including cysts, sclerosing adenosis, and ductal epithelial hyperplasia.
In the differentiation of benign from malignant papillary lesions, careful attention to low- and high-power views is essential. The low-power view indicates the papillary nature of the lesion and the size of the fibrovascular cores. Usually, the outline of an encompassing duct wall can be discerned, even in cases with sclerosis and entrapment of epithelial structures. High-power examination permits an assessment of the other epithelial changes previously described, the most important of which is the presence of two cell types covering the fronds and lining the contained epithelial elements. The generally benign appearance of the nuclei precludes the diagnosis of carcinoma.
Three other points related to the diagnosis of benign papillomas merit mention. First, in the presence of a heavy, blood-stained discharge, organization can occasionally lead to the formation of a so-called granulation tissue polyp, which should not be mistaken for a true papilloma. Second, in sections from the region of the lactiferous sinus, tangential cuts of the tortuous ducts may present a pleated contour and give the false impression of a papilloma. Third, partial involvement of a benign papilloma by DCIS does sometimes occur.
In addition to the difficulties and controversy over the diagnosis of intraductal papilloma, and perhaps in part because of these difficulties, uncertainty persists over the precancerous significance of intraductal papillomas. Most authors believe that intraductal papillomas of the breast are rarely associated with invasive carcinoma during the follow-up period. Kraus and Neubecker (111), Hendrick (112), Haagensen et al. (113), Hart (114), and Lewison and Lyons (115) reported a total of 427 cases of intraductal papillomas of the breast in which carcinoma developed during the follow-up period in only two patients (0.4%) during a follow-up that ranged from 5 to 22 years. Three other series of papillomas—by Buhl-Jorgensen et al. (116), Moore et al. (117), and Kilgore et al. (118)—showed the development of cancer in 19 of 235 cases during a follow-up period of 4 months to 16 years. Carter (119) reported on 64 cases of intraductal papillomas of the breast followed for a period of 5 to 17 years. Six recurred as papillomas, and six patients went on to develop breast cancer. Two of the patients who developed carcinoma of the breast were among six with multiple papillomas. In 14 patients, there was distinct hyperplasia or papillomatosis of the associated ducts. Two of these patients developed breast cancer. Among 44 patients in whom there were only nonproliferative cystic changes or fibroadenomas, only 5% developed carcinoma. Rosen (120) reviewed the literature of 612 solitary papillomas treated by excision and found 11 ipsilateral carcinomas (2%), 10 contralateral carcinomas (2%), and 3 bilateral carcinomas (0.3%) in follow-up. He concluded that solitary papillomas were not precancerous. Page et al. (121) reported a case control study of women with papillomas that contained areas of atypical hyperplasia and found them to have a fourfold increase in risk of cancer compared to women after excision of papillomas that lacked atypia. Table 9.15 lists the features that distinguish papilloma from intracystic papillary carcinoma.

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


