1 Introduction
Most of us have no notion of what life would be like without antibiotics. Penicillin was discovered in 1928 and after a lengthy developmental period became available for clinical use in the 1940s. Then followed what is often referred to as the golden era of antibiotic discovery which brought us the cephalosporins, the tetracyclines, the macrolides, the aminoglycosides, the 4-quinolones, etc. (Table 27.1). It is interesting to note that the impact of antibiotic discovery was sufficient to halt the development of virtually all other antimicrobial therapies being pursued at that time. The fact that Alexander Fleming in the early years had flagged up the probability of resistance development in bacterial pathogens was largely ignored because of the constant stream of new agents on to the market. There is anecdotal evidence that in 1967 the US Surgeon General stated ‘… it is time to close the book on infectious disease, declare the war against pestilence won…’. Whether or not he actually said this is debatable, but it represented a widely held view at the time that the stream of antibiotic discovery would never dry up. However, we now know that is precisely what has happened.
Table 27.1 Time line for the discovery and use of naturally occurring and synthetic antibiotics
| Time | Antibiotic |
| 1900-1920 | Salvarsan |
| 1920-1940 | Benzylpenicillin |
| 1940-1960 | Sulphanilamide |
| Cephalosporins | |
| Streptomycin | |
| Tetracycline | |
| Chloramphenicol | |
| Erythromycin | |
| Isoniazid | |
| Vancomycin | |
| Polymyxin | |
| Methicillin | |
| 1960-1980 | Ampicillin |
| Trimethoprim | |
| Sulphamethoxazole | |
| Fusidic acid | |
| Carbapenems | |
| Gentamicin | |
| Clindamycin | |
| Amoxycillin | |
| 4-Quinolones | |
| 1980-2000 | Mupirocin |
| Teicoplanin | |
| Clarithromycin | |
| Aztreonam | |
| Latamoxef | |
| Azithromycin | |
| Quinupristin/dalfopristin | |
| Ceftazidime | |
| Linezolid | |
| 2000 to date | Daptomycin |
| Doripenem | |
| Tigecycline |
At present we are faced with two major issues—we have seen an inexorable rise in the incidence of antibiotic resistance among the most important bacterial pathogens (dealt with in Chapter 13), while at the same time the development of novel antibacterial antibiotics has all but stopped. Some clinical isolates from species such as Pseudomonas aeruginosa, Acinetobacter baumannii, Enterococcus faecium and Mycobacterium tuberculosis are resistant to virtually every antibiotic we have at our disposal. It is not appropriate here to discuss the issue of antibiotic resistance, other than to say it is an almost inevitable event occurring as a consequence of the bacterial cell’s remarkable adaptability to environmental change. Without this ability to adapt to their environment bacteria would not still be thriving 3.5 billion years after first emerging on to what was at that time an extremely hostile earth. Suffice it to say there is very little we can do to stop this process.
The second issue of antibiotic discovery and development is, of course, under our control. We have the scientific ingenuity to devise novel approaches to circumvent the resistance mechanisms put in place by these bacteria. Unfortunately, the driving force behind drug discovery is not based on altruism but has a financial imperative. The cost of bringing a new chemical entity to the market has been estimated as $800 million or more, while the process of development takes about 8 years. Antibiotics simply do not generate sufficient revenue to justify this initial outlay of time and cost, and not surprisingly the focus for the large pharmaceutical companies has switched to those medicines which patients will take long-term such as statins, antidepressants and antihypertensives. The market for antiviral agents, particularly those to treat HIV/AIDS, remains buoyant simply because the patients will be taking the medicines for the remainder of their lives.
All this poses the question, ‘How in the near future will clinicians be able to treat patients with infections caused by multiresistant bacterial pathogens?’ In reality this is a question which is relevant today but will only become more pressing in the future. Some respected authorities in the field have already expressed the view that we are on the verge of entering a postantibiotic era.
This chapter examines a range of alternative therapeutic strategies available for the management of infectious disease (Table 27.2). It is not the intention to cover each of these in any depth but merely to give an overview of the general situation; the reader is referred to the further reading listed at the end of the chapter for more details. As we will see, a number of the techniques to be described have a very long history, some of them going back many centuries. Some of these have very little chance of ever moving from the fringes of medicine, some may have the potential to be useful but in reality suffer from the same developmental issues as conventional antibiotics, some are niche approaches which may find relevance in specific clinical areas, while a few may offer a realistic alternative, or at least an adjunct, to antibiotics.
Table 27.2 Potential alternatives to antibiotics for the treatment of bacterial infections
| Alternative therapeutic strategy | Selected references |
| Essential oils (tea tree oil) | Carsonet al. (2006) |
| Honey therapy | Moore et al. (2001) |
| Garlic | Amagase et al. (2001) |
| Probiotics | Mombelli & Gismondo (2000); Hedin et al. (2007) |
| Maggot therapy | Chan et al. (2007) |
| Photodynamic antimicrobial therapy | Wainwright (1998) |
| Vaccines and immunotherapy | BNF (2009); Döring & Pier (2008) |
| Silver | Edwards-Jones (2009) |
| Bacteriophages | Hanlon (2007) |
| Bacteriophage lysins | Fischetti (2008) |
Essential oils, sometimes termed ethereal or volatile oils, are contained within many plants and impart their characteristic odour. They can be obtained from the plant material by distillation, expression or solvent extraction and have been used throughout the centuries for medicinal and other purposes. In recent times attention has focused on a few of these oils, particularly tea tree oil, because of their reported antimicrobial activity and this will be used as an exemplar of the group as a whole. As the name suggests, they are hydrophobic, which makes them difficult to work with, and being natural products they frequently contain a large number of chemical components.
Tea tree oil (TTO) is the essential oil obtained from the Australian plant Melaleuca alternifolia (although other species may be used). Of its numerous chemical constituents (mainly monoterpenes, sesquiterpenes and their associated alcohols) terpinen-4-ol has been shown to have the major antimicrobial activity. It has general antimicrobial activity against viruses, bacteria and fungi but the main interest in this compound is centred on its activity against meticillin-resistant Staphylococcus aureus (MRSA). TTO has been suggested as a potential agent for the nasal and skin decolonization of MRSA in carriers. It has been formulated in a variety of topical preparations and is available over the counter for the treatment of a range of conditions.
One of the many issues surrounding the use of any natural material, especially multicomponent substances such as plant extracts, is that of batch variability and quality control. In the case of TTO there is a British Standard which regulates the composition of the 15 components of the oil and this is identical to ISO 4730:2004 (Table 27.3). The standard demands a lower limit of 30% for the main antimicrobial compound terpinen-4-ol, and other minor components are included in order to make the formulation of an artificial oil more difficult. Unless all preparations conform to the required standards of composition it is very difficult to judge the value of any in vitro or in vivo tests. The situation is further confused because the nature of the oil makes it problematic to evaluate. Oils are only sparingly soluble in the media used to test antimicrobial activity and when mixed with aqueous solutions result in turbid suspensions; consequently, end-points based on turbidity are difficult to determine, and the hydrophobic nature of the compounds limits their diffusion in agar. Thus in the literature a range of methodologies have been described to evaluate antimicrobial activity, and comparison between them is difficult.
Table 27.3 Composition of tea tree oil as determined by BS ISO 4730:2004
| Components | Minimum (%) | Maximum (%) |
| α-Pinene | 1 | 6 |
| Saninene | Trace | 3.5 |
| α-Terpinene | 5 | 13 |
| Limonene | 0.5 | 1.5 |
| p-Cymene | 0.5 | 8 |
| 1, 8-Cineole | Trace | 15 |
| γ-Terpinene | 10 | 28 |
| Terpinolene | 1.5 | 5 |
| Terpinen-4-ol | 30 | 48 |
| α-Terpineol | 1.5 | 8 |
| Aromadendrene | Trace | 3 |
| Ledene (syn.viridiflorene) | Trace | 3 |
| δ-Cadinene | Trace | 3 |
| Globulol | Trace | 1 |
| Viridiflorol | Trace | 1 |
Those in vitro studies which have been conducted indicate activity against a broad range of bacteria with similar minimum inhibitory concentrations (MICs) reported regardless of whether the isolate was antibiotic-sensitive or resistant. Generally bacteria are susceptible to concentrations below 1% (v/v) with the main exception being Ps.aeruginosawhich requires concentrations approaching 8% (v/v). The mechanism of action of TTO is thought to involve disruption of cellular membranes which will result in loss of intracellular constituents and inhibition of enzyme function. The more complex outer membranes of Ps. aeruginosa render that organism more resistant to the effects of TTO.
Clinical studies have evaluated TTO for the treatment of a range of bacterial and fungal infections. There is no doubt that it is effective, although in many cases the treatment was not found to be superior to conventional therapy. Despite being used for many years TTO has exhibited safety issues and is toxic if ingested. It has also been shown to have irritant effects on the skin, although these may be reduced if the oil is properly formulated. Essential oils such as TTO cannot be directly compared to conventional antibiotics since they are used primarily as topical antiseptics. However, in this regard they certainly have some value although more extensive, properly controlled trials are necessary to fully confirm their potential use.
Bees collect nectar (a weak natural sugar solution) and pollen from flowers in their locality, and in their hives it is ultimately transformed into honey. As a consequence of the processing in the hive, sucrose in the honey is converted into fructose and glucose and the enzyme glucose oxidase converts glucose into gluconic acid and hydrogen peroxide. As water evaporates from the honey the sugar concentration increases until it is of the order of 79% w/v. These features act to preserve the honey from microbial degradation. Other components of the honey are dependent on the geographical source of the nectar, and much interest has focused recently on honey collected in the vicinity of the Australian tea tree ( Melaleuca alternifolia) mentioned above, and the New Zealand manuka (Leptospermum scoparium). The honey collected from these areas has been studied widely because of its reported antibacterial and wound-healing properties. These have been attributed to a number of factors, including the following.
• pH. This ranges from 3.2 to 4.5, which is below the minimum pH values required for the growth of many common bacteria.
• Osmotic pressure and water activity. Honey is a supersaturated sugar solution with a water content of between 15 and 21% w/w. The water availability (Aw) ranges from 0.56 to 0.62, which again is below thatrequired for the growth of most common bacteria.
• Hydrogen peroxide. This is an effective antibacterial agent and, although it is rapidly neutralized, the presence of glucose oxidase ensures a constant replenishment.
• Non-peroxide antibacterial activity. Many workers have shown that the antimicrobial activity of honey persists even when the hydrogen peroxide has been removed.
The nature of the substance responsible for the non-peroxide activity has been elusive, but recent studies suggest that it may be methylglyoxal, found in some highly active forms of manuka honey. However, many other candidate materials are under investigation.
Results from animal studies, some clinical trials and much anecdotal evidence over centuries suggests that honey may have a role to play in the management of infected wounds due to its antibacterial and wound-healing properties. However, a recent Cochrane review examined 19 published clinical trials in which honey was evaluated in the treatment of acute or chronic wounds. The conclusion reached was that although honey may improve healing times in mild to moderate wounds there was insufficient evidence to support its use in other areas.
Garlic (Allium sativum) has been used as a culinary ingredient for thousands of years, but it has also been shown to possess a range of pharmacological activities leading to uses in cardiovascular disease and cancer. Its use in these conditions is outside the remit of this chapter; however, of interest here is the fact that it has also been reported to have antimicrobial activity against a broad spectrum of bacterial pathogens.
The chemistry of garlic is highly complex and like other plant extracts the material is made up of a large number of compounds. The main sulphur-containing components of intact garlic are γ-glutamyl-S-allyl cysteines and S-allyl-l-cysteine sulphoxides (of which the main one is known as alliin). When garlic is crushed or damaged, the enzyme alliinase, which is normally contained within vacuoles, comes into contact with the cytosolic cysteine sulphoxides and converts them to thiosulphinates such as allicin. This process is thought to represent a protective effect for the plant because assault by predators (worms, bacteria, etc.) would result in a localized high concentration of these toxic products. Allicin was the component that was first shown to be responsible for the marked antimicrobial activity of fresh garlic, and on the basis of this a number of garlic-containing products were marketed for their antibacterial effects. However, allicin was subsequently found to be highly unstable and to degrade rapidly within a matter of days to a variety of sulphides in both aqueous and alcoholic solutions. In the context of the plant’s protective strategy this makes sense, because having generated a toxic metabolite to counter an insult it is in the plant’s best interest to neutralize that toxin as rapidly as possible after the invader has been repelled.
Orally administered allicin is degraded in the stomach acid and is not absorbed from the gut, thus demonstrating that it cannot be responsible for any of the reported in vivo antibacterial effects. Attention has thus switched to other components, particularly the allicin breakdown products including diallyl sulphide and diallyl disulphide. Studies in mice have shown that oral dosing of these agents can reduce MRSA viability in blood, liver, kidney and spleen. They also provide immunological protective properties. However, the number of studies conducted is small and the methodologies variable. More comprehensive clinical trials are required together with a rigorous study of the role the individual components play in the overall biological activity.
Garlic suffers from many of the issues raised above for TTO in that the composition of the product varies mark-edly depending on the source of the raw material, how it is processed to extract the active ingredients, how it is formulated and how it is stored. Inconsistencies reported for the activity of garlic preparations are primarily due to a lack of standardization of the product. As things currently stand, garlic is unlikely to contribute significantly to the resolution of the issue of diminishing antibiotic availability.
Probiotics are defined as living microorganisms that confer some health benefits when utilized in sufficient numbers. The use of harmless bacteria to replace pathogenic ones (bacteriotherapy) is not a new concept but has gained renewed interest in recent years. Particularly, they have been strongly marketed as a prophylactic approach to the promotion of well-being. In addition, bacteriotherapy has been used in the treatment of a variety of gastrointestinal conditions (including diarrhoea), respiratory infections, recurrent bacterial vaginitis, recurrent otitis media, streptococcal tonsillitis and infected burns. The microorganisms most frequently used are normal components of the gut microflora and include the lactic acid bacteria such as lactobacilli, lactococci, bifidobacteria, enterococci and streptococci (see Table 27.4). Other bacteria such as Bacillus species and yeasts such as Saccharomyces may also be utilized. Since each of these organisms possesses different characteristics and requirements for growth it is likely that they do not have a common mechanism of action.
Table 27.4 Microorganisms commonly used as probiotics
| Bacillus subtilis |
| B. breve |
| B. infantis |
| B. lactis |
| B. longum |
| Enterococcus faecium |
| Escherichia coli Nissle 1917 |
| Lactobacillus acidophilus |
| L. casei immunitas |
| L. johnsonii |
| L. paracasei subsp paracasei 19 |
| L. plantarum |
| L. raffinolactis |
| L. reuteri |
| L. rhamnosus |
| L. salivarius |
| Leuconostoc spp. |
| Pediococcus pentoseceus |
| Streptococcus thermophilus |
| Saccharomyces boulardii |
| S. cerevisiae |
5.1 Gastrointestinal conditions
Probiotics for oral administration are marketed as dairy products such as yoghurt drinks, but also as powders, tablets or capsules. Irrespective of the nature of the product, there is a need to consume in the region of 109–1011 microorganisms in order to bring about any effect. A number of studies have highlighted issues of inconsistency in the content of viable microorganisms within various products. The precise manner in which probiotics bring about their health benefits is unclear, but a prerequisite property is the ability to colonize the gut. The orally administered organisms must first, therefore, be able to survive passage through the stomach acid and then compete with the indigenous flora in the intestine. A competitive edge will be provided by the ability to colonize the gut wall and the capacity to produce bacteriocins. Probiotics have been administered rectally to overcome the necessity for acid tolerance, but there is a risk of spreading faecal pathogens.
Probiotics have been found to be of benefit in the treatment of infectious diarrhoea in children. When used as a supplement to breast milk in premature infants they were found to reduce the incidence and severity of necrotizing enterocolitis. A recent review summarizing the available clinical trial data has suggested value in the treatment of ulcerative colitis, but the results for Crohn’ s disease were disappointing and the authors stated that the use of probiotics could not be recommended on the available evidence.
Clostridium difficile is a Gram positive, spore-forming anaerobic rod which, in certain conditions, can overgrow in the intestinal tract and produce toxins causing diarrhoea and damage to the epithelial lining of the gut. This condition is a particular problem in elderly hospitalized patients who have received courses of broad-spectrum antibiotics. Since the colitis is caused by overgrowth of a pathogenic bacterium in the intestinal tract it would seem to be potentially amenable to probiotic treatment. However, a recent Cochrane review of the current clinical trial data found that only treatment with Saccharomyces boulardii had any significant effect. Many hospitals use a range of commercially available probiotic yoghurt preparations but the effectiveness of these is currently unproven.
5.2 Infected burn wounds
Burn wounds are devoid of microorganisms immediately after the injury has been sustained, but fairly quickly thereafter they will become colonized with microflora from the adjacent intact skin and also by environmental microbes. These bacteria are mainly Gram-positive coagulase-negative staphylococci, but they may be gradually replaced by more harmful Gram-negative bacteria such as Ps. aeruginosa and Acinetobacter spp. over the following few days. These bacteria are notorious for being highly antibiotic resistant and they flourish, often forming biofilms, because the usual defensive properties of intact skin are no longer present. Colonization with these organisms delays healing, can inhibit grafting and may lead to systemic infection. Studies have shown that applications of Lactobacillus plantarum were as effective as silver sulphadiazine in the management of infected burn wounds. This may be of relevance in those countries where the costs of conventional antibiotic treatment are prohibitive or where there is a high level of antibiotic resistance.
5.3 Recurrent vaginitis
Many women suffer from vaginitis, caused by either bacteria or Candida albicans, which may lead on to urinary tract infections. In these cases the normal vaginal microflora which comprises mainly lactobacilli is overrun by various other, sometimes pathogenic, bacteria or fungi. The usual approach to treatment is the administration of antibiotics, but recurrence is commonplace. The vaginal administration of probiotic cultures has proved to be beneficial in these cases; however, strains of bacteria isolated from dairy products are not always suitable. The lactobacilli should be those that have the capacity to bind specifically to vaginal epithelial cells.
5.4 Acute otitis media
This is a very common condition in young children in which bacteria normally present in the nasopharynx transfer via the eustachian tube into the middle ear. The predominant bacteria are Streptococcus pneumoniae and Haemophilus influenzae. Antibiotic therapy is frequently unsuccessful and recurrences are common. As we have seen in the examples outlined above, the picture is once again one of undesirable bacteria replacing normal commensal flora and causing disease. Studies have been reported in which children were treated with suspensions of α-haemolytic streptococci sprayed into the ear to inhibit the growth of the invading pathogens and recolonize the infected site. The results showed that the treated patients had significantly better outcomes than the placebo group.
Some workers have highlighted the fact that probiotics should be used with caution in those patients whose immune system is impaired because there have been reported incidences of invasive infections occurring in immunocompromised adults. In addition, Lactobacillus bacteraemia has been reported following probiotic therapy in two children who had received multiple courses of antibiotics and who were suffering from other serious underlying conditions.
The currently available data suggest that probiotics may have some value in certain selected conditions, but a great deal more background scientific information is required before this can be moved forward substantially. The approach of using a single culture for a whole spectrum of conditions is untenable. It is clear that the probiotic bacteria will act in a variety ways, producing arange of interfering substances and having different abilities to adhere to epithelial cells. Individual cultures may therefore need to be developed for use in specific conditions and once this has been achieved large-scale trials are needed to verify their value.
Chronic infected wounds arising from conditions such as diabetes mellitus and peripheral vascular disease are a major problem, and their incidence is increasing. Whereas normal wounds go through well-defined phases of healing, chronic wounds do not heal because of the presence of necrotic tissue, slough, debris and infection. In order to allow the wound to begin to heal it is necessary to physically remove the necrotic burden, in a process known as debridement, and also deal with the infection. Clearing the wound of necrotic and devitalized tissue is normally carried out surgically before application of antimicrobial agents to remove the infecting pathogens. The mechanical techniques used in debridement are varied and can lead to pain and damage to underlying healthy tissue.
The use of maggots to facilitate wound debridement and subsequent healing has been known for several centuries, but its association with modern medicine goes back approximately 70 years. However, the clinical use of maggots dwindled with the advent of antibiotics and has only now experienced a resurgence of interest. The main focus of attention has been in the area of wounds infected with MRSA. The larvae of the blow fly Lucilia sericata are grown under sterile conditions and used when they are less than 8 hours old, at which point they are only 1–2 mm in length. Up to 1000 maggots are placed in the wound, covered to prevent escape and left for 1–3 days. The maggots require a moist environment for optimal activity and their secretions contain multiple proteolytic enzymes which degrade necrotic tissue. This digested material is subsequently consumed by the maggots as a source of nutrients, but it has been suggested that there is also a mechanical component to the clearing of necrotic tissue. One of the characteristics of the larvae is that they try to avoid light and as a result of their small size they tend to burrow into deep crevices which may have proved difficult to debride mechanically. The larval secretions contain ammonia which increases the local pH producing an antimicrobial effect, and in addition the maggots ingest the bacteria, thus clearing the site of infection. The presence of the maggots also leads to stimulation of granulation tissue formation and hence promotes healing.
There is now abundant literature which appears to affirm the view that maggot therapy can play an important role in the treatment of chronic infected wounds. However, a large (267 patients) multicentre trial was carried out in the UK recently comparing maggot therapy with a standard debridement technique (hydrogel) for the treatment of chronic infected leg ulcers. The authors found that there was no difference in healing times between the two groups and the larval group experienced more pain. Nonetheless, the larvae did reduce the time to debridement. Some workers have shown that the antimicrobial efficacy of the larvae is dependent on the species of bacterium under investigation. Grampositive bacteria such as Staph. aureus are very susceptible, whereas Gram-negative bacteria such as Ps. aeruginosa are more resistant. The precise reasons for this are unclear. Additional hurdles to the widespread use of maggot therapy include patient acceptability and resistance of medical staff to apparently ‘old-fashioned ’ techniques. Hence the case for maggot therapy is equivocal, and more basic research is required to understand why it appears to be effective in some circumstances and not in others.
7 Photodynamic therapy (photoactivated disinfection)
The therapeutic effects of light have been known for thousands of years and the combination of light with various chemicals again dates back to ancient times. It was only at the beginning of the 20th century, however, that the scientific basis of this phenomenon began to be explored. The use of photosensitizing agents to kill microorganisms was first shown in 1900 and it was shortly after this that the term photodynamic therapy (PDT) was coined. This chapter only discusses the use of this technique for the killing of microorganisms, although it should be pointed out that PDT has also been used to treat tumours using a combination of photosensitizing porphyrin compounds and laser light.
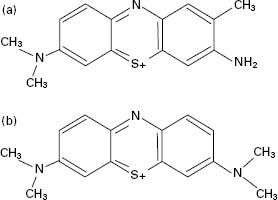
The most common photosensitizers used for their antimicrobial effects are the thiazine dyes methylene blue and toluidine blue O (also known as tolonium chloride; Figure 27.1). Both of these molecules carry a positive charge and this enables them to interact with the negatively charged outer surfaces of bacteria. In particular, they accumulate at the cytoplasmic membrane and when activated cause lethal damage to that target site. It has been found that Gram-positive bacteria are more sensitive to PDT than Gram-negative bacteria, because of the different nature of their cell walls (Chapter 3).
Figure 27.1 Structure of photosensitizers of the thiazine family of dyes: (a) toluidine blue O, (b) methylene blue.