Chapter 36
Alterations of Pulmonary Function in Children
Valentina L. Brashers and Sue E. Huether
Structure and Function
Lower Airways and Lung Parenchyma
During fetal development the lung is transformed from a somewhat dense organ to one that is more delicately structured to facilitate air exchange. Beginning in the second trimester, there is loss of interstitial (mesenchymal) tissue with concomitant expansion of the future air spaces. Capillaries grow into the distal respiratory units that keep subdividing (alveolarization) to maximize surface area for gas exchange. The number of alveoli continues to increase during the first 5 to 8 years of life, after which the alveoli increase in size and complexity. In addition to the structural development of the lung in utero, there is accompanying functional maturation during which specialized cell types, such as type II cells, manifest. (Figure 36-1 contains a summary of alveolar development and stages of fetal lung development.)
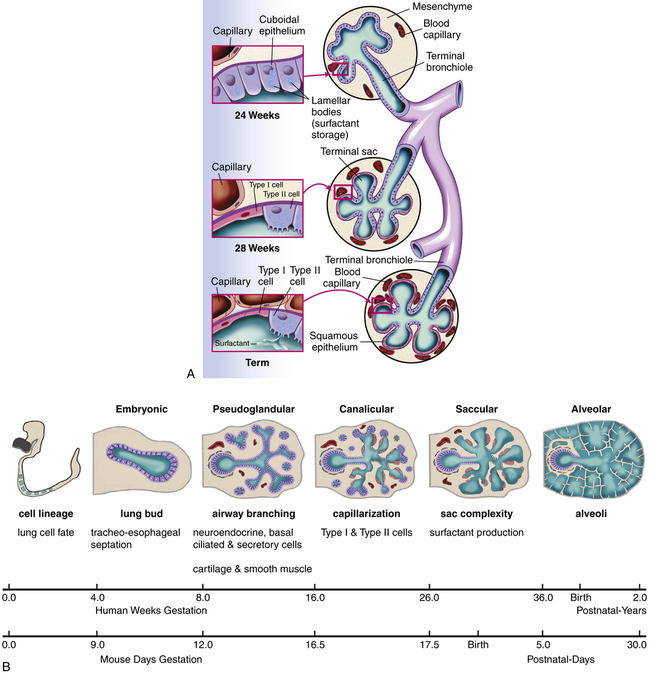
A, Epithelial cells differentiate into type II and type I cells. Mature type II cells are cuboidal, have apical microvilli, and contain lamellar bodies for surfactant storage and secretion. Type I cells are derived from type II cells and consist of flattened epithelium overlying capillaries, thus forming part of the desired thin air-blood barrier. During fetal development the pulmonary capillaries initially are randomly distributed in mesenchyme. They progressively arrange around the epithelial tubes and establish close contacts to the lining epithelium. Overall the volume of mesenchyme decreases and that of the potential air space increases. B, Stages of fetal lung development. (B adapted from Gleason CA, Devaskar S: Avery’s diseases of the newborn, ed 9, Philadelphia, 2012, Saunders.)
Chest Wall Dynamics
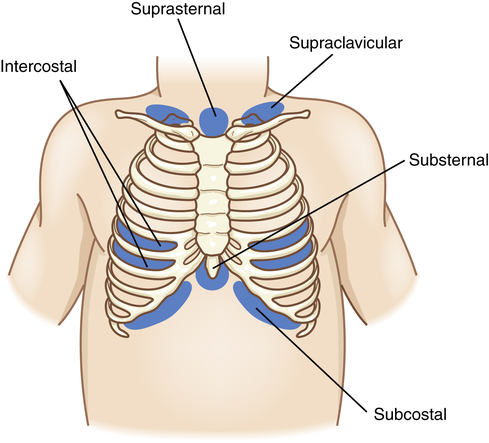
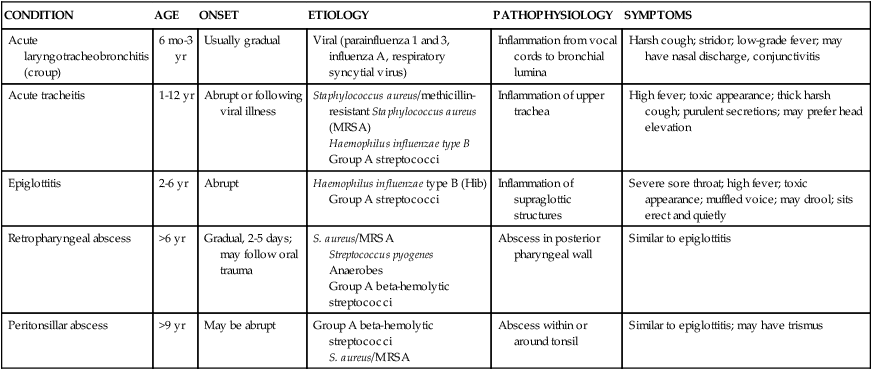
Chest wall compliance is high in infants, particularly premature infants. The cartilaginous structures of the thoracic cage are not yet well ossified (ossification continues to occur throughout childhood), and the chest wall is easily collapsible. During inspiration in the young child, air is drawn in by the downward movement of the diaphragm, but the resulting negative pressure causes the “soft” chest wall to be drawn inward (Figure 36-2); this produces so-called paradoxic breathing, or diaphragmatic breathing. Paradoxic breathing is especially seen during rapid eye movement (REM) sleep of premature infants. With pulmonary compromise the accessory muscles are drawn inward, creating retraction of the intercostal and supraclavicular spaces (Figure 36-3).
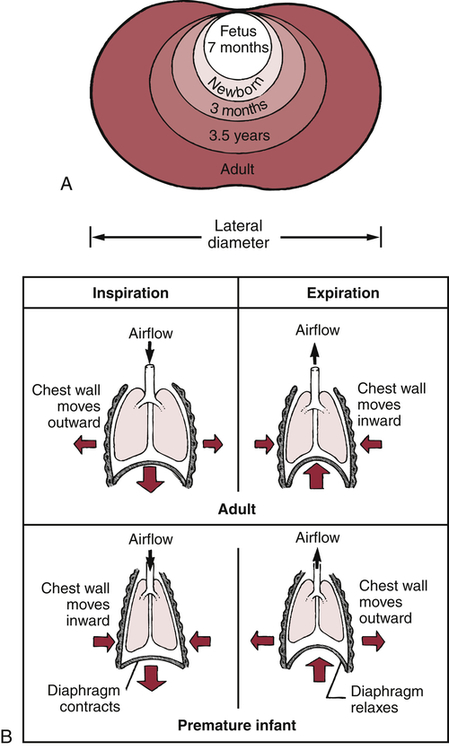
A, Changes in chest wall shape with age. B, Differences in lung mechanics caused by differences in chest wall compliance (degree of rigidity) in premature infants and adults. (Arrows indicate direction of airflow, chest wall movement, and diaphragm movement.)
Physiologic Control of Respiration
For up to 3 weeks of age, the newborn has a blunted ventilatory response to hypoxia compared with older children and adults. The mechanisms for this are not well understood but may reflect reduced activity of the peripheral chemoreceptors (in the carotid body) and nonadaptive responses in the respiratory center (in the brainstem). Ventilatory response to hypercarbia is normal in term infants but may be reduced in premature infants. Congenital or acquired lesions of the central nervous system may cause hypoventilation or apnea. Exposure to maternal smoking both during and after fetal development can have significant deleterious effects on lung development and subsequent susceptibility to pulmonary disorders (see What’s New? Maternal Smoking: Fetal Lung Development and Lung Disease).
Disorders of the Upper Airways
respiratory failure are the same regardless of etiology. These include the following:
• Increased respiratory effort with retractions (see Figure 36-3) or gasping (apnea in some conditions)
• Decreased level of consciousness
• Cardiovascular signs: tachycardia, mottled color, or bradycardia
• Physiologic compromise reflected by hemoglobin desaturation, hypoxemia, hypercarbia, and acidosis
Upper Airway Obstruction
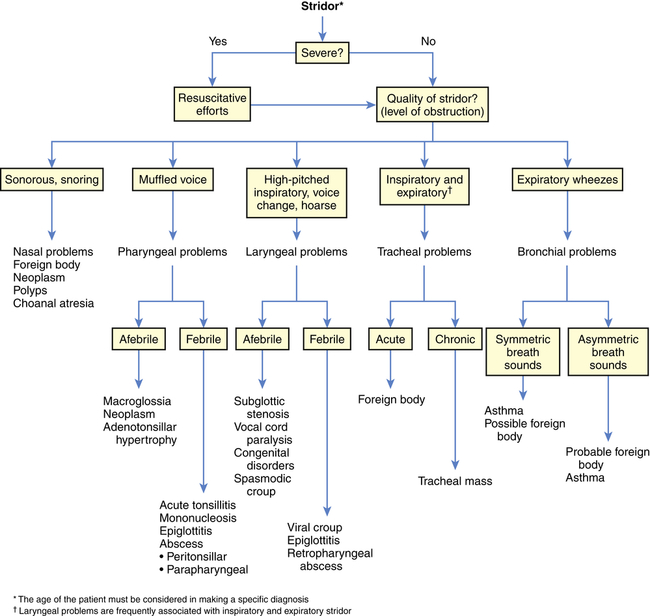
The crucial issue in the upper airways is patency. The most common causes of acute-onset upper airway obstruction (UAO) in children are infections, foreign body aspiration, angioedema, obstructive sleep apnea, and trauma. Chronic UAO has many etiologies, including congenital malformations affecting the airway, cartilaginous weakness, vocal cord paralysis, and subglottic stenosis. Chronic upper airway symptoms should prompt referral to a pediatric pulmonologist or an otolaryngologist because specialized diagnostic studies may be needed. A list of causes of pediatric UAO can be found in Box 36-1.
The sounds of the child’s breathing can provide key clues (Figure 36-4). A sonorous, snoring noise is typical for nasopharyngeal obstruction, such as adenotonsillar hypertrophy. A common sign of pediatric UAO is stridor, a harsh, vibratory sound of variable pitch caused by turbulent flow through the partially obstructed airway. A diagnostic approach to stridor is outlined in Figure 36-5. Whether it is present in inspiration, expiration, or both reflects the site of the problem. In general, inspiratory stridor is generated with obstruction of the extrathoracic airway (above the thoracic inlet), which includes the supraglottic structures, the larynx, the subglottic space, and the upper trachea. Expiratory stridor or a monophonic wheeze may be generated by an obstruction in the intrathoracic airway (the middle to lower trachea and central bronchi). Biphasic stridor typically reflects obstruction at the glottis (e.g., vocal cord paralysis) itself or a fixed rather than a dynamic lesion in the subglottic space (e.g., hemangioma or subglottic stenosis). Biphasic noise may sometimes mean abnormalities of both the extrathoracic and intrathoracic trachea (long-segment stenosis or malacia).
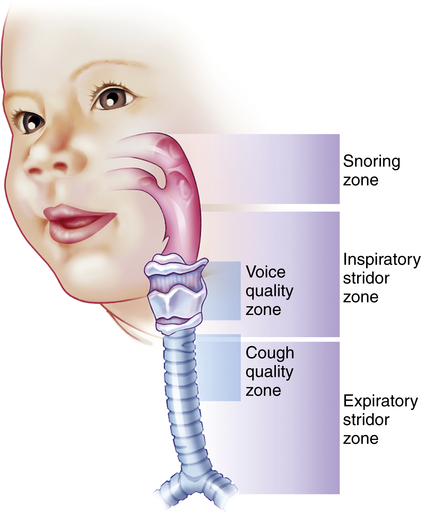
A loud, gasping snore suggests enlarged tonsils or adenoids. Stridor during inspiration suggests the airway is compromised at the level of the supralaryngeal structures (epiglottis and arytenoid cartilages), vocal cords, subglottic region, or upper trachea. With forced inspiration, intrathoracic pressure becomes quite negative and is less than atmospheric pressure, promoting collapse at or just above the site of obstruction. Expiratory stridor or central wheeze results from narrowing or collapse of the lower trachea or bronchi. During forced exhalation, rising pleural pressure may exceed intratracheal pressure. Airway noise during both inspiration and expiration often represents a fixed obstruction of the vocal cords or subglottic space. Hoarseness or a weak cry is a byproduct of obstruction at the vocal cords. If a cough is croupy or low pitched, suspect tracheal pathology. (Redrawn from Eavey RD: Contemp Pediatr 3[6]:78, 1986; used with permission; original illustration by Paul Singh-Roy.)
Infections
Infections of the upper airway (Table 36-1) are common in children; some have the potential to cause life-threatening emergencies. Recognition and rapid evaluation of these problems are crucial pediatric care skills.
TABLE 36-1
COMPARISON OF UPPER AIRWAY INFECTIONS
| CONDITION | AGE | ONSET | ETIOLOGY | PATHOPHYSIOLOGY | SYMPTOMS |
| Acute laryngotracheobronchitis (croup) | 6 mo-3 yr | Usually gradual | Viral (parainfluenza 1 and 3, influenza A, respiratory syncytial virus) | Inflammation from vocal cords to bronchial lumina | Harsh cough; stridor; low-grade fever; may have nasal discharge, conjunctivitis |
| Acute tracheitis | 1-12 yr | Abrupt or following viral illness | Staphylococcus aureus/methicillin-resistant Staphylococcus aureus (MRSA) Haemophilus influenzae type B Group A streptococci | Inflammation of upper trachea | High fever; toxic appearance; thick harsh cough; purulent secretions; may prefer head elevation |
| Epiglottitis | 2-6 yr | Abrupt | Haemophilus influenzae type B (Hib) Group A streptococci | Inflammation of supraglottic structures | Severe sore throat; high fever; toxic appearance; muffled voice; may drool; sits erect and quietly |
| Retropharyngeal abscess | >6 yr | Gradual, 2-5 days; may follow oral trauma | S. aureus/MRSA Streptococcus pyogenes Anaerobes Group A beta-hemolytic streptococci | Abscess in posterior pharyngeal wall | Similar to epiglottitis |
| Peritonsillar abscess | >9 yr | May be abrupt | Group A beta-hemolytic streptococci S. aureus/MRSA | Abscess within or around tonsil | Similar to epiglottitis; may have trismus |
Croup
Croup illnesses can be divided into two categories: (1) acute laryngotracheobronchitis (croup) and (2) spasmodic croup. Diphtheria can be considered a croup illness but is now rare because of vaccinations. Croup illnesses are all characterized by infection and obstruction of the upper airways.1
Croup is an acute laryngotracheobronchitis and most commonly occurs in children from 6 months to 3 years of age, with peak incidence at 2 years of age.2 In 85% of cases, croup is caused by a virus, most commonly parainfluenza; however, other viruses such as influenza A virus, respiratory syncytial virus (RSV), rhinovirus, adenovirus, and rubella virus (measles) as well as the atypical bacterium Mycoplasma pneumoniae has been associated with causation. The incidence of croup is highest in late autumn and winter, corresponding to the parainfluenza and RSV seasons, respectively. Croup is more common in boys than girls. In a significant portion of affected children, croup is a recurrent problem during childhood, and there is a family history of croup in about 15% of cases. Spasmodic croup is characterized by similar hoarseness, barking cough, and stridor, but is of sudden onset, usually at night and without viral prodrome. It often resolves as quickly as it develops and usually occurs in older children. The etiology is unknown although association with viruses, allergies, asthma, and gastroesophageal reflux disease (GERD) is being investigated.3
Pathophysiology
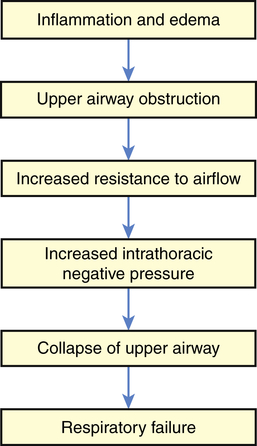
The pathophysiology of viral croup is caused primarily by subglottic edema from the infection. The mucous membranes of the larynx are tightly adherent to the underlying cartilage, whereas those of the subglottic space are looser and thus allow accumulation of mucosal and submucosal edema (Figure 36-6). Furthermore, the cricoid cartilage is structurally the narrowest point of the airway, making edema in this area critical. As illustrated in Figure 36-7, increased resistance to airflow leads to increased work of breathing, which generates more negative intrathoracic pressure, which in turn may exacerbate dynamic collapse of the upper airway.
A, Normal. B, Narrowing and obstruction from edema caused by croup. (From Hockenberry MJ, Wilson D: Wong’s nursing care of infants and children, ed 9, St Louis, 2011, Mosby.)
Evaluation and Treatment
The degree of symptoms determines the level of treatment. Most children have a barking cough and viral symptoms and may need no specific treatment. However, the presence of stridor (especially at rest), retractions, or agitation suggests a sicker child. The tool most often used for estimating croup severity is the Westley croup score, which provides a cumulative score for the degree of stridor, retractions, air entry, cyanosis, dyspnea, and level of consciousness in the child.4
Severity also is classified as mild, moderate, and severe. Most children with croup require no treatment. Inhalation of humidified air does not improve symptoms in mild to moderate croup.5 Glucocorticoids, either injected or oral (dexamethasone) or nebulized (budesonide), have been shown to improve symptoms within 6 hours. Less sleep is lost by children, less stress is experienced by parents, and fewer children have a need for return healthcare visits or hospitalization when corticosteroids are used.6 The use of nebulized racemic epinephrine improves outcomes with moderate to severe croup.2,7 Epinephrine stimulates α- and β-adrenergic receptors and is thought to decrease airway secretions and mucosal edema. It is a temporizing measure until concomitantly given corticosteroids begin to take effect. Thus children who are given nebulized epinephrine should be closely observed for 2 to 3 hours to ensure that they will remain stable. Oxygen also should be administered. Heliox (helium-oxygen mixture of 80:20 or 70:30) may be used for severe cases of croup, although there is lack of scientific evidence to establish use in routine clinical practice.8
Acute Epiglottitis
Historically, acute epiglottitis was caused by Haemophilus influenzae type B (Hib). Since the advent of Hib immunization, the overall incidence of acute epiglottitis in children has decreased significantly, although Hib still accounts for approximately 25% of the cases seen in children. Infants less than 1 year of age are at greater risk.9 Current pediatric cases usually represent Hib vaccine failures or are caused by alternative pathogens, such as groups A, B, C, F, and G streptococci, Streptococcus pneumoniae, Candida species, Staphylococcus aureus, and viral pathogens. Thermal injuries, trauma, and posttransplant lymphoproliferative disorder also have been reported as causes of epiglottitis.10
Pathophysiology
The epiglottis arises from the posterior tongue base and covers the laryngeal inlet during swallowing. It has a rich blood and lymphatic circulation. Bacterial invasion of the mucosa with associated inflammation leads to the rapid development of edema causing severe, life-threatening obstruction of the upper airway.10
Evaluation and Treatment
Acute epiglottitis is a life-threatening emergency. The essentials are early recognition, avoidance of disturbing the child (which could worsen the obstruction), and securing the airway. Examination of the throat should not be attempted because it may trigger laryngospasm and cause respiratory collapse. Tracheal intubation should be accomplished by the most experienced personnel (usually an anesthesiologist and/or otolaryngologist) using fiberoptic laryngoscopy. Subsequent culture of the airway is obtained and intravenous broad-spectrum antibiotics are administered promptly. Therapy is reevaluated after culture results return. Corticosteroids also are generally used in treatment regimens although there are no published randomized trials to support this practice.10–12 Despite the severe presentation of epiglottitis, resolution with treatment is usually rapid, with intubation rarely needed for more than a couple of days. When Hib epiglottitis is diagnosed, the American Academy of Pediatrics (AAP) recommends that postexposure prophylaxis with rifampin be administered to household contacts (specific to certain ages of children present).12 When caused by microorganisms other than H. influenzae, as is now the usual situation, epiglottitis may present in ages outside the typical range and with more gradual rather than fulminant onset, thus making diagnosis less obvious. Such cases also may respond more slowly to treatment.
Tonsillar Infections
Tonsillar infections (tonsillitis) are occasionally severe enough to cause UAO.13 As with other infections of the upper airway, the incidence of tonsillitis secondary to group A beta-hemolytic Streptococci (GABHS) and methicillin-resistant S. aureus (MRSA) has risen notably in the past 15 years.14 Significant swelling of the tonsils and pharynx occurs, and a tenacious membrane may cover the mucosa. UAO because of tonsillitis is a well-known complication of infectious mononucleosis, especially in a young child. The development of UAO in tonsillar infections requires the use of appropriately selected antibiotics and may require the use of corticosteroids, especially in the case of mononucleosis.15 Some children with recurrent tonsillitis benefit from tonsillectomy.
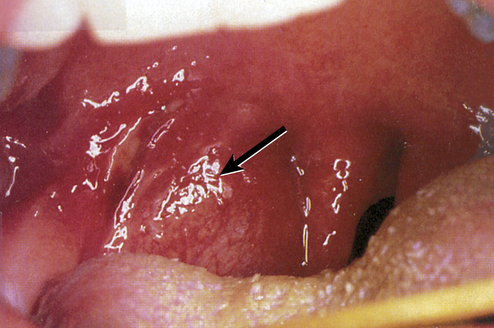
Peritonsillar abscess is usually unilateral and is most often a complication of acute tonsillitis.16 The most common causative microorganism is GABHS. Children have fever, sore throat, dysphagia, trismus, pooling of saliva, and muffled voice. Peritonsillar bulging (Figure 36-8) and cervical adenopathy on the same side are usually visible. The abscess must be drained and the child given antibiotics.17 Death can occur from spontaneous abscess rupture with aspiration or airway obstruction.18
Bacterial Tracheitis
Bacterial tracheitis is the most common potentially life-threatening upper airway infection in children. It is most often caused by Staphylococcus aureus (S. aureus) (including methicillin-resistant [MRSA] strains), H. influenzae, group A beta-hemolytic Streptococcus (GABHS [Streptococcus pyogenes]), or Moraxella catarrhalis.19 It accounts for 5% to 14% of UAOs in children requiring intensive care.20 A virus or a fungus is more likely to be seen as the source of tracheitis in immunocompromised children.21 Treatment of viral croup with corticosteroids has increased the risk for serious bacterial tracheitis (especially by GABHS), resulting in death rates between 18% and 40%.20 The presence of airway edema and copious purulent secretions leads to airway obstruction that can be worsened by the formation of a tracheal pseudomembrane and mucosal sloughing. Increased morbidity can occur because of respiratory and cardiopulmonary arrest, respiratory failure, pneumonia, septic shock, toxic shock syndrome, acute respiratory distress syndrome (ARDS), and multiple organ dysfunction syndrome (MODS). The onset of symptoms may be sudden or may be preceded by a pre-existing viral upper respiratory tract infection or croup. The acute clinical presentation frequently includes tachypnea, stridor, hoarse voice, fever, cough, and/or increased secretions from the mouth and nose. There may be evidence of concurrent infections, such as sinusitis, otitis, pneumonia, or pharyngitis.22 Children with chronic gastroesophageal reflux are more likely to experience difficulty with these types of concurrent infections. Management requires the rapid administration of broad-spectrum intravenous antibiotics. The majority of children with tracheitis require endotracheal intubation in order to prevent airway obstruction. Corticosteroids (parenteral and inhaled) are used to decrease tracheal inflammation. Many children recover adequately to be extubated within 72 to 96 hours.23
Retropharyngeal Abscess
Retropharyngeal abscess can be caused by aerobic, anaerobic, or polymicrobial infection. A decrease in retropharyngeal abscess has been noted in the past several years that is likely related to the use of corticosteroids in the treatment of influenza and croup. There also has been an increase in GABHS strains associated with this condition, and the emergence of MRSA as the offending microorganism is increasingly noted.24
Retropharyngeal abscess usually occurs more commonly in male children about 4 years of age and as a consequence of either nasopharyngeal infection or penetrating local injury.25 Clinical signs include fever, dysphagia, drooling, stridor, respiratory distress, and stiff neck. This condition requires intravenous antibiotics targeted at the suspected microorganism, and sometimes incision and drainage.26
Aspiration of Foreign Bodies
Most children who aspirate a foreign object (foreign body aspiration) are between 1 and 3 years of age. More than 100,000 cases occur each year.27 Often the aspiration either is not witnessed or does not seem significant to the parent; thus medical care is often not pursued until after the first 24 hours. At the time of aspiration, the child may cough, choke, gag, or wheeze, and stridor or cyanosis occasionally occurs. This may be followed by a quiescent interval of minutes to even weeks or months before symptoms reappear from resulting local irritation, granulation, bronchial obstruction, or infection (pneumonia or bronchiectasis). Pronounced inspiratory stridor, cough, and wheezing are typical symptoms that prompt the parents to seek medical attention. Examples of common aspirated objects include nuts, sunflower seeds, hot dog chunks, popcorn, coins, and small toys or toy parts. Meat or food impactions are more common in adolescents. Items of particular concern are batteries and multiple magnets. In general, foreign bodies require early intervention secondary to their propensity to cause respiratory symptoms and complications, including esophageal erosions or aortoesophageal fistula (rare).
The symptom history is often the most critical aid in diagnosis.28 Symptoms are determined by the size of the object and the site in which it is located, as well as the child’s age and size (see Figure 36-4). Foreign bodies lodged in the upper trachea typically produce inspiratory stridor, whereas those located in the lower intrathoracic airways more commonly produce wheezing. About 75% of aspirated foreign bodies lodge in a bronchus. Children with an unexplained persistent cough and refractory parenchymal infiltrates also should be considered for unrecognized foreign body aspiration.29 Many objects are not radiopaque; however, if the object has completely occluded a lung segment, atelectasis will be visible on a chest radiograph. Occasionally, air will accumulate distal to the obstruction if the object is causing a ball-valve effect. This effect can sometimes be documented by inspiratory and expiratory chest films (Figure 36-9). In a younger child, bilateral decubitus films may show failure to compress the obstructed lung when in the “down” position.
Inspiratory (left) and expiratory (right) chest radiographs of a child who aspirated a portion of a potato into the left mainstem bronchus. Left lung field is hyperaerated and the mediastinum is shifted to the right on expiration because of left-sided obstructive emphysema. (From Kenna MA, Bluestone CD: Pediatr Rev 10[1]:25, 1988.)
Most foreign bodies can be removed by bronchoscopy and only rarely is a pulmonary lobectomy required. Soft particles such as food, as well as hard objects, must be removed because infectious processes will otherwise occur.30 Objects lodged in the laryngeal or subglottic regions are particularly dangerous because of their potential for complete or near-complete airway occlusion.
Angioedema
Angioedema is a localized edema involving the deep, subcutaneous layers of skin or mucous membranes. Generally, angioedema causes facial swelling first, particularly around the eyes and lips, and may progress to airway swelling.31 Angioedema is usually secondary to allergic phenomena (e.g., allergy to peanuts, cow’s milk, chicken eggs).32 If airway compromise is apparent, standard treatment includes epinephrine (subcutaneous), antihistamines, and corticosteroids. An occasional cause of pediatric angioedema is use of angiotensin-converting enzyme inhibitors for treatment of hypertension or heart disease. Increased levels of bradykinin appear to mediate this adverse effect by causing vasodilation, increased vascular permeability, and histamine release.33
An inherited deficiency of the plasma protein C-1 inhibitor (C-1 INH) causes hereditary angioneurotic edema (HAE), a rare but serious problem in children. About 50% of cases occur during childhood.34 The mean age of onset of initial symptoms is 8 to 12 years but it also may occur as early as the first year of life. This condition is characterized by recurring attacks of angioedema involving subcutaneous tissues (especially limbs, genitalia, and face); abdominal and pelvic viscera; and, much less often, the airway. Laryngeal attacks in these individuals may be life threatening and do not respond reliably to standard measures for airway edema. Complement studies usually provide an accurate diagnosis.34 The mortality of undiagnosed HAE can be as high as 50%. The mainstays of supportive care are airway monitoring, hydration, pain relief, and control of nausea. Concentrates of C-1 INH appear to produce rapid improvement within 15 to 60 minutes. Short- and long-term prophylaxis can be instituted using antifibrinolytic agents, attenuated androgens, and C-1 INH concentrates.35
Congenital Malformations
Laryngomalacia and Tracheomalacia
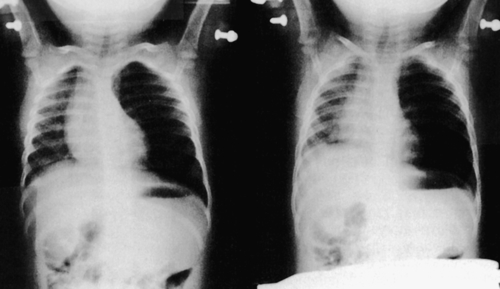
Laryngomalacia results from abnormally soft laryngeal cartilages and is the most common cause of chronic stridor in infants. Boys are twice as likely as girls to present with symptoms. In laryngomalacia, the epiglottis or arytenoids, or both, fold inward with inspiration, partially covering the glottis (Figure 36-10). The pathophysiology of these abnormalities is still not completely understood. Two primary hypotheses are anatomic or neuromuscular.36 Anatomically there may be foreshortened or tight aryepiglottic folds or there may be redundant soft tissue in the supraglottis.37 The neuromuscular hypothesis suggests that there is an abnormality of the sensorimotor integrative function of the brainstem and peripheral reflexes are responsible for laryngeal tone and airway patency.38 Laryngeal nerve hypertrophy also has been documented.39 Gastroesophageal reflux disease (GERD) and other comorbidities are associated with laryngomalacia.40
In the normal larynx (left), supralaryngeal structures maintain their upright orientation during inspiration. In contrast, in infants with laryngomalacia (right), there is inward prolapse of the arytenoid masses, which include the prominent cuneiform tubercles and the arytenoid cartilages. The glottis becomes partially covered, and airflow is impeded. Sometimes the edges of the epiglottis curl inward, further exacerbating the obstruction. In expiration, these structures are “blown” aside passively.
Typical signs of laryngomalacia include inspiratory stridor beginning in the first days or weeks of life, accentuated with activity, and sometimes with positional changes (worse in supine or head-flexed positions). Feeding difficulties may be noted, but they are usually mild. Cry is normal. Laryngoscopy is used to confirm the diagnosis. Laryngomalacia is usually mild and improves spontaneously over the first year of life as the supralaryngeal cartilage structures stiffen; thus most cases are managed with watchful waiting. A late-onset variant of this disease has been noted in the literature and should be suspected if the following occurs: potential cause of feeding difficulties in toddlers, sleep apnea in children, and exercise intolerance in teenagers.41,42
In tracheomalacia, or tracheobronchomalacia, the tracheobronchial cartilages are flaccid and tend to collapse during the expiratory cycle, causing stridor. This may be classified as primary (idiopathic) or secondary. When malacia is caused by a secondary source, it is usually related to extrinsic compression of the trachea from a vascular malformation (congenital tracheomalacia). Tracheobronchomalacia presents clinically as a spectrum of respiratory illnesses that range from life-threatening conditions to chronic cough and wheeze conditions. In some cases symptoms may be more subtle than in laryngomalacia. Low-pitched inspiratory stridor may be a sign of malacia of the upper trachea or centrally located, single-pitch (monophonic) wheeze may be present in malacia of the middle to distal trachea. Tracheomalacia can be suspected clinically and confirmed by bronchoscopy. Depending on the type and severity of the lesion, surgical approaches for repair may be indicated.43
Vocal Cord Paralysis
Vocal cord paralysis is second to laryngomalacias as a cause of stridor and is a relatively uncommon congenital disorder. The vocal cords should move apart to facilitate inspiration and move together to facilitate vocalization. Paralysis of one or both vocal cords may affect breathing, swallowing, and speech. The etiology of the congenital abnormality is unclear but may be caused by immaturity of the vagus nerve or brainstem, or both. Iatrogenic injury is frequently cited as the major secondary cause of vocal cord paralysis, such as surgical trauma to the recurrent laryngeal nerve during cardiac surgery. Other secondary causes include Arnold-Chiari malformation (the region of the brainstem in which the nucleus ambiguus acts as the “relay station” for laryngeal function), cerebral palsy, hydrocephalus, myelomeningocele, spina bifida, or hypoxia.44 Other associations include infectious and neoplastic causes, trauma, and inflammatory conditions.45 In older children and adolescents, exercise has been known to precipitate vocal cord dysfunction (VCD).46
Clinical findings of vocal cord paralysis in children less than 1 year include dysphonia, glottic incompetence, GERD, and stridor.45 It sometimes resolves spontaneously (most often during the first year of life) or with correction of the underlying problem. Flexible laryngoscopy and chest x-ray are common evaluative tools that may help determine the cause. In some individuals, VCD may be misdiagnosed as asthma.47 Medical therapy may include use of corticosteroids, proton pump inhibitors, and speech therapy. Recurrent pulmonary infections secondary to aspiration may occur and require treatment until the cords are repaired.48 Severe cases may necessitate endotracheal intubation and tracheostomy. Tracheostomy may be used until the vocal cords are surgically repaired or can be used as a permanent measure for bilateral vocal cord paralysis.48
Subglottic Stenosis
Congenital subglottic stenosis is the third most common laryngeal anomaly and is defined as a subglottic airway diameter of less than 4 mm at the cricoid region in a full-term infant, and less than 3 mm in a premature infant.48 Incomplete recanalization of the laryngotracheal tube during the third month of gestation results in this defect. Subglottic stenosis also is associated with eosinophilic esophagitis, Wegener granulomatosis, and neurofibromatosis.49–51 Traumatic injury to the upper airway with development of subglottic stenosis is a well-described complication of endotracheal intubation.52 Factors that contribute to subglottic stenosis include utilization of long-term assisted ventilation, use of an endotracheal tube that is too large, excessive movement of the tube, and susceptibility of the individual.53 Neonates can tolerate long periods of endotracheal intubation; the overall rate of symptomatic subglottic stenosis in neonates is 0.2%.53 The occurrence of subglottic stenosis can be minimized by ensuring that the tube size allows a small air leak during inspiration (at a peak inspiratory force of approximately 25 mmHg) and that the tube is securely taped. Sedation is generally required to reduce head movement for children who are intubated. Because of the rapid growth of the lumen of the trachea and cricoid cartilage in the first year (which triples in size), infants may outgrow the obstruction, particularly if mild or moderate.54 Serial balloon dilation can be effective.55 For severe subglottic stenosis, laryngotracheal reconstruction (LTR) along with cricotracheal resection and thyrotracheal anastomosis may be required.56
Other Congenital Malformations
Congenital malformations of the upper airways, trachea, and bronchial tree are rare and cause airway obstruction. Lesions include coanal atresia; laryngeal atresias, webs, cysts, clefts, and subglottic hemangiomas; and tracheal stenosis. Coanal atresia can be unilateral or bilateral and causes lack of patency of the nasal cavity within the nasopharynx. Bilateral coanal atresia can be life threatening in newborns because they are obligate nasal breathers.57 Laryngeal atresias and webs are caused by failure of the larynx to recanalize during embryogenesis. Most of these disorders are in the area of the glottis with extension into the subglottis.48 Tracheal stenosis is a rare malformation characterized by inspiratory and expiratory stridor within the first few weeks of birth. It is usually associated with pulmonary, cardiovascular, and gastrointestinal malformations.58 Structural abnormalities such as pectus excavatum or abnormalities involving the great vessels also can result in tracheal compression. For example, absent pulmonary valve syndrome dilates the pulmonary artery, which can compress the trachea and bronchi.59 Tracheal or bronchial compression results in airway symptoms or feeding difficulties, or both, ranging from dysphagia, recurrent respiratory tract infections, wheezing, and stridor to acute respiratory distress or “dying spells.” Many older children are first thought to have gastroesophageal reflux or asthma as the principal problem. Surgical management is usually required for these conditions, and some infants may require mechanical ventilation while awaiting surgery.59
Obstructive Sleep Apnea Syndrome
Obstructive sleep apnea syndrome (OSAS) is a breathing disorder defined by prolonged partial and/or intermittent complete UAO during sleep with disruption of normal ventilation and normal sleep patterns. Childhood OSAS is common with an estimated prevalence of 2% to 3% among middle-school children and as many as 13% of children ages 3 to 6 years. Prevalence is estimated to be two to four times higher in vulnerable populations (blacks, Hispanics, and preterm infants).60 Unlike adults, OSAS in children occurs equally among males and females. Infants are at risk because they have both anatomic and physiologic predispositions toward airway obstruction and gas-exchange abnormalities, including a superiorly placed larynx, increased chest wall compliance, ventilation-perfusion mismatching, and ventilatory control instability. Congenital abnormalities of the airway, as described previously in this chapter, may have adverse effects on airway patency. Neck flexion, airway secretions, gastroesophageal reflux, and sleep deprivation can be associated with airway obstruction. Obstructive sleep apnea in infants has been associated with failure to thrive, behavioral deficits, and sudden infant death.61
Pathophysiology
The pathophysiology of childhood OSAS is likely to be multifactorial in origin. The most common predisposing factor is adenotonsillar hypertrophy, which causes physical impingement on the nasopharyngeal airway. OSAS often occurs more frequently in overweight or obese children, as well as in those with orthodontic/craniofacial anomalies, neurologic disorders, allergy, asthma, abnormalities in the motor tone of the upper airways (frequently an issue in neurologically impaired children), abnormal arousal mechanisms, and genetic susceptibility.62 Recent studies have documented that children with sleep disordered breathing (SDB) have increased inflammation in the upper airway and elevated serum levels of C-reactive protein, particularly in obese children.63 Activation of inflammatory pathways contributes to end-organ injury with cardiovascular and metabolic morbidities.64
Clinical Manifestations
Common manifestations of OSAS include snoring and labored breathing, restlessness, and sweating during sleep, which can be continuous or intermittent. There may be episodes of increased respiratory effort, but no audible airflow, often terminated by snorting, gasping, repositioning, or arousal. Affected children are often chronic mouth breathers and have large tonsils. They also may exhibit nocturnal enuresis.65 It appears that there also may be a correlation between OSAS and elevated blood pressure similar to adults.66 This is further linked to increased body mass index (BMI) and episodes of desaturation and apnea/hypopnea.67
OSAS can result in chronic hypoxemia and hypercapnia affecting multiple organ systems. Significant morbidity is associated with OSAS including cognitive problems, neurobehavioral impairment (e.g., inattention, hyperactivity, aggression, conduct problems, attention-deficit/hyperactivity disorder [ADHD]/emotional [mood]), excessive daytime sleepiness, impaired school performance, and poor quality of life.68 Left untreated it also can cause cardiovascular disease, particularly left ventricular hypertrophy, and insulin resistance, as well as pulmonary complications (upper and lower respiratory tract infections) and reduced somatic growth.69–71
Evaluation and Treatment
All parents should be asked if their child exhibits snoring, a symptom that is often not spontaneously reported to a healthcare provider. The history and physical examination are the most effective means of diagnosis. Screening tools and sleep questionnaires may be helpful in evaluating the presence of SDB.72 Radiographic images of the upper airway may reveal upper airway narrowing caused by adenoidal hypertrophy, and magnetic resonance imaging (MRI) and acoustic reflectometry may detect reduced upper airway dimensions.73 The most definitive evaluation (“gold standard”) is the polysomnographic sleep study, which documents obstructed breathing and physiologic impairment and may be combined with nocturnal oximetry studies. If obstructive sleep apnea caused by tonsillar enlargement is documented or strongly suspected clinically, children are most often referred for tonsillectomy and adenoidectomy (T&A). For severely affected children who do not respond to T&A or who have different problems, such as obesity that cannot be rapidly remedied, continuous positive airway pressure (CPAP) delivered through a tight-fitting nasal mask may be used during sleep. Treatment is important to prevent associated morbidities. Successful medical or surgical treatment results in improvement in physical, behavioral, and emotional difficulties as well as quality of life.74–76
Disorders of the Lower Airways
Respiratory Distress Syndrome of the Newborn
Respiratory distress syndrome (RDS) of the newborn (previously known as hyaline membrane disease [HMD]) is a major cause of neonatal morbidity and mortality. It occurs almost exclusively in premature infants and the incidence has increased in the United States over the past two decades. Preterm births account for about 12% of all births. Black mothers have 1.5 times the risk of preterm birth and 3.4 times the risk of preterm-related mortality.77 RDS occurs in 50% to 60% of infants born at 29 weeks of gestation and decreases significantly by 36 weeks. Infants of diabetic mothers and those with cesarean delivery (especially elective C-section) are more likely to develop RDS because the labor-associated catecholamine and steroid surge do not occur, causing decreased pulmonary surfactant release.78 It is more common in boys than girls and in one cohort it was more common in black infants.79,80 Death rates have declined significantly since the introduction of antenatal steroid therapy and postnatal surfactant therapy. The major predisposing factor is prematurity because the immature lung is not well structured for gas exchange and has not yet developed adequate surfactant production and secretion. Death rates have declined significantly since the introduction of antenatal steroid therapy and postnatal surfactant therapy. The epidemiology, pathophysiology, and clinical presentation of RDS are outlined in Box 36-2.
Pathophysiology
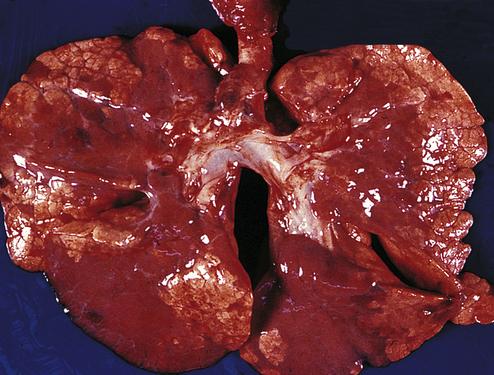
RDS is caused by surfactant deficiency, which decreases the alveolar surface area available for gas exchange. Surfactant lipoproteins have a detergent-like effect that separates the liquid molecules inside the alveoli, thereby decreasing alveolar surface tension. Without surfactant, alveoli collapse at the end of each exhalation.81 Surfactant normally is not secreted by the alveolar cells until approximately 20 to 24 weeks of gestation (see Figure 36-1). Premature infants also are born with underdeveloped and small alveoli that are difficult to inflate. Additionally, the alveoli have thick walls and an inadequate capillary blood supply such that gas exchange is significantly impaired. The infant’s chest wall is weak and highly compliant and, thus, the rib cage tends to collapse inward with respiratory effort. The net effect is atelectasis (Figure 36-11), resulting in significant hypoxemia. Atelectasis is difficult for the neonate to overcome because it requires a significant negative inspiratory pressure to open the alveoli with each breath. The increased work of breathing and a decrease in tidal volume cause alveolar hypoventilation and hypercapnia. Hypoxia and hypercapnia cause pulmonary vasoconstriction, which increases intrapulmonary resistance and shunting. Increased pulmonary vascular resistance causes a partial return to fetal circulation, with right-to-left shunting of blood through the ductus arteriosus and foramen ovale further compromising pulmonary perfusion. There is decreased surfactant production with inadequate pulmonary perfusion and alveolar ventilation.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree