CANDIDA: GENERAL CHARACTERISTICS
Candida species grow as typical 4 to 6 μm, budding, round, or oval yeast cells (Figure 46–1) under most conditions and at most temperatures. Under certain conditions, including those found in infection, they can form hyphae. The Candida cell wall contains the same chitin and carbohydrate elements found in other fungi. Species identification is based on a combination of biochemical, enzymatic, and morphologic characteristics, such as carbohydrate assimilation; fermentation; and the ability to produce hyphae, germ tubes, and chlamydoconidia. Of the over 150 Candida species, fewer than 10 appear in human disease. Particular attention is given to the differentiation of C albicans from other species, because it is by far the most common cause of disease.
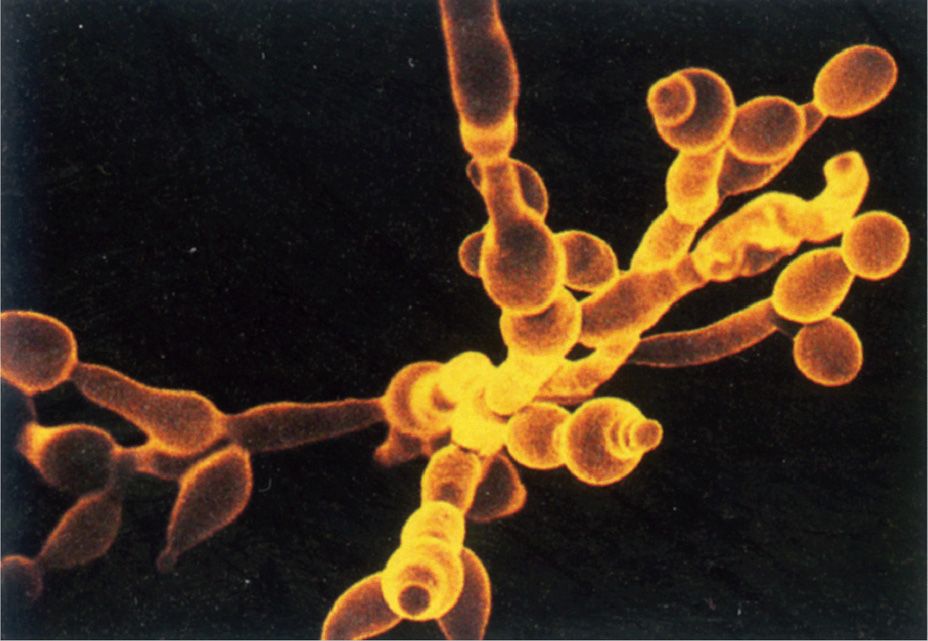
FIGURE 46–1. Candida albicans. This scanning electron micrograph demonstrates dimorphism with both blastoconidia and hyphae. (Reproduced with permission from Willey JM: Prescott, Harley, & Klein’s Microbiology, 7th edition. McGraw-Hill, 2008.)
Formation of hyphae and chlamydoconidia are distinguishing features
Carbohydrate assimilation and fermentation determine species
Most Candida species grow rapidly on Sabouraud’s agar and on enriched bacteriologic media such as blood agar. Smooth, white, 2 to 4 mm colonies resembling those of staphylococci are produced on blood agar after overnight incubation. Aeration of cultures favors their isolation. The primary identification procedure involves presumptive differentiation of C albicans from the other Candida species with the germ tube test. Germ tube–negative strains may be further identified biochemically or reported as “yeast not C albicans” depending on their apparent clinical significance.
Rapidly produce colonies resembling staphylococci
Calbicans produces germ tubes
Candida albicans
 MYCOLOGY
MYCOLOGY
Candida albicans grows in multiple morphologic forms, most often as a yeast with budding by formation of blastoconidia. Candida albicans is also able to form hyphae triggered by changes in conditions such as temperature, pH, and available nutrients. When observed in their initial stages while still attached to the yeast cell, these hyphae resemble sprouts and are called germ tubes (Figure 46–2A). Other elongated forms with restrictions at intervals are called pseudohyphae because they lack the parallel walls and septation of the true hyphae. There is evidence that these three forms have distinct stimuli and genetic regulation, making C albicans a polymorphic fungus. Unless otherwise specified, the term hyphae is used here to encompass both the true and pseudohyphal forms. The hyphal form also develops characteristic terminal thick-walled chlamydoconidia under certain cultural conditions (Figure 46–2B).
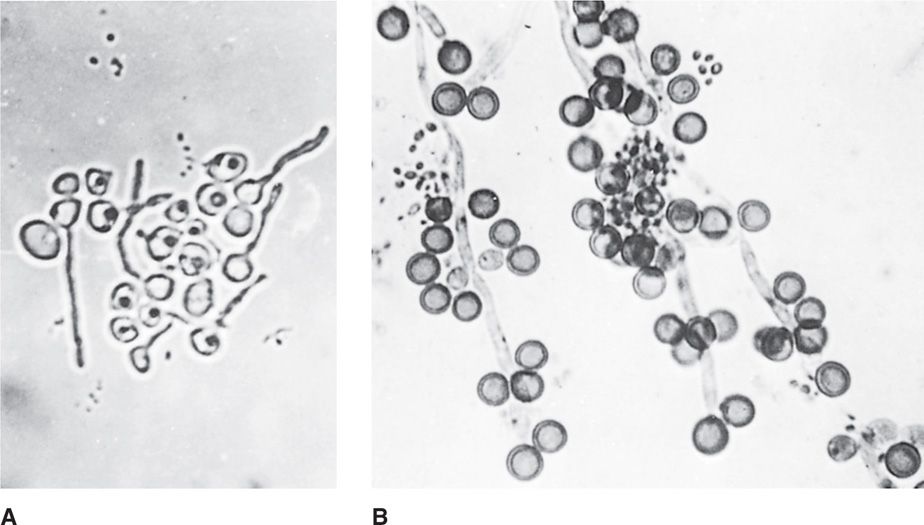
FIGURE 46–2. Candida albicans. A. When incubated at 37°C, C albicans rapidly forms elongated hyphae called germ tubes. B. On specialized media, C albicans forms thick-walled chlamydoconidia, which differentiate it from other Candida species. (Reprinted with permission from Dr E. S. Beneke and the Upjohn Company: Scope Publications, Human Mycoses.)
Yeast, hyphae, and pseudohyphae are formed
Chlamydoconidia develop from hyphae in culture
The C albicans cell wall is made up of a mixture of the polysaccharides mannan, glucan, and chitin alone or in complexes with protein. A fibrillar outer layer extending to the surface contains several glycoproteins and complexes of mannan with protein called mannoproteins. The exact composition of the cell wall and surface components varies under different growth conditions.
Cell wall includes surface mannoproteins
![]() CANDIDIASIS
CANDIDIASIS
EPIDEMIOLOGY
Candida albicans is present in 30% to 50% of the oropharyngeal, gastrointestinal, and female genital microbiota of healthy persons. Infections are endogenous except in cases of direct mucosal contact with lesions in others (eg, through sexual intercourse). Although C albicans is a common cause of nosocomial infections, the strains involved are usually derived from the patient’s own flora than from cross-infection. Invasive procedures and indwelling devices may provide the portal of entry, and the number of available Candida may be enhanced by the use of antibacterial agents.
Infections are from endogenous flora
PATHOGENESIS
Because C albicans is regularly present on mucosal surfaces, disease implies a change in the organism, the host, or both. The change from the yeast to the hyphal form is strongly associated with enhanced pathogenic potential of C albicans. In histologic preparations, hyphae are seen only when Candida starts to invade, either superficially or in deep tissues. This switch can be controlled in vitro by the manipulation of a wide variety of environmental conditions (serum, pH, temperature, amino acids). Various sensors and signaling pathways have been described including one in which C albicans induces its own change by altering the local pH. It is still not known what triggers these changes in human disease. What is known is that the morphologic change is also associated with the appearance of various factors associated with tissue adherence and digestion.
Shift from yeast to hyphae is associated with invasion
Switch is triggered by environmental conditions
Candida albicans hyphae have the capacity to form strong attachments to human epithelial cells. One mediator of this binding is a surface hyphal wall protein (Hwp1), which is found only on the surface of germ tubes and hyphae. Other mannoproteins that have similarities to vertebrate integrins may also mediate binding to components of the extracellular matrix (ECM), such as fibronectin, collagen, and laminin. Hyphae also secrete proteinases and phospholipases that are able to digest epithelial cells and probably facilitate invasion (Figures 46–3 and 46–4A and B). One family of hyphal enzymes, the secreted aspartic proteinases (Saps), is able to digest keratin and collagen, which would facilitate deep tissue invasion. The pattern of Sap production may be tissue-specific with those invading gastrointestinal and vaginal epithelium producing a different sets of Saps. Candida albicans is also able to form biofilms which include yeast and hyphal forms together with polymers which adhere to the ECM and plastics. Taken together, these factors represent a rich armamentarium of virulence factors all linked to the change from yeast to hyphal growth.
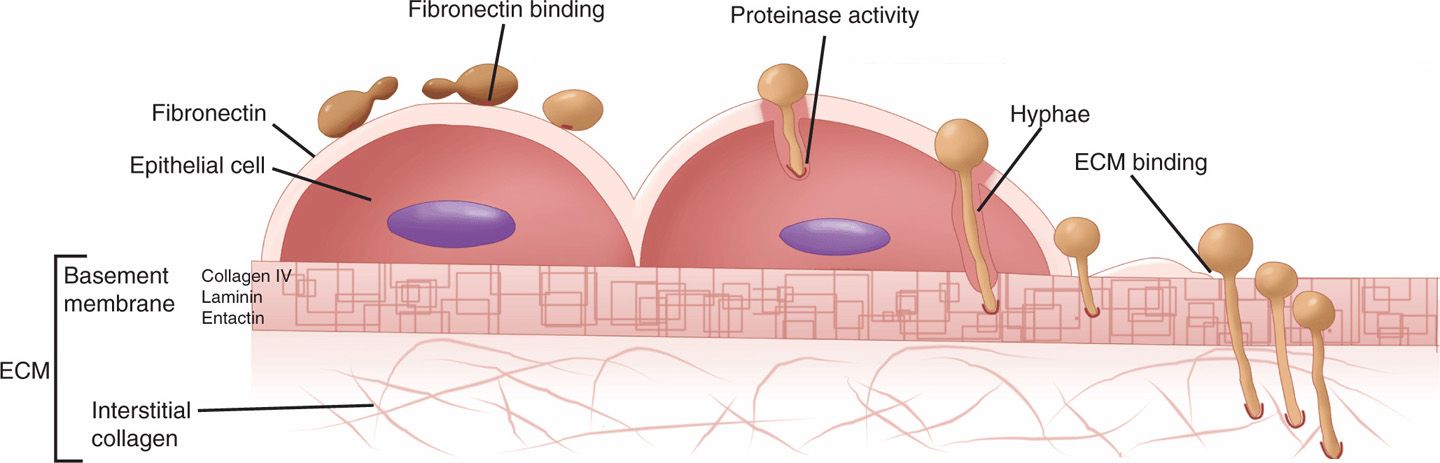
FIGURE 46–3. Pathogenesis of Candida albicans infections. Proposed mechanisms of C albicans attachment and invasion are shown. Surface glucomannan receptor(s) on the yeast may bind to fibronectin covering the epithelial cell or to elements of the extracellular matrix (ECM) when the epithelial surface is lost or when the Candida have invaded beyond it. Invasion is associated with formation of hyphae and production of proteinases, which may digest tissue elements.
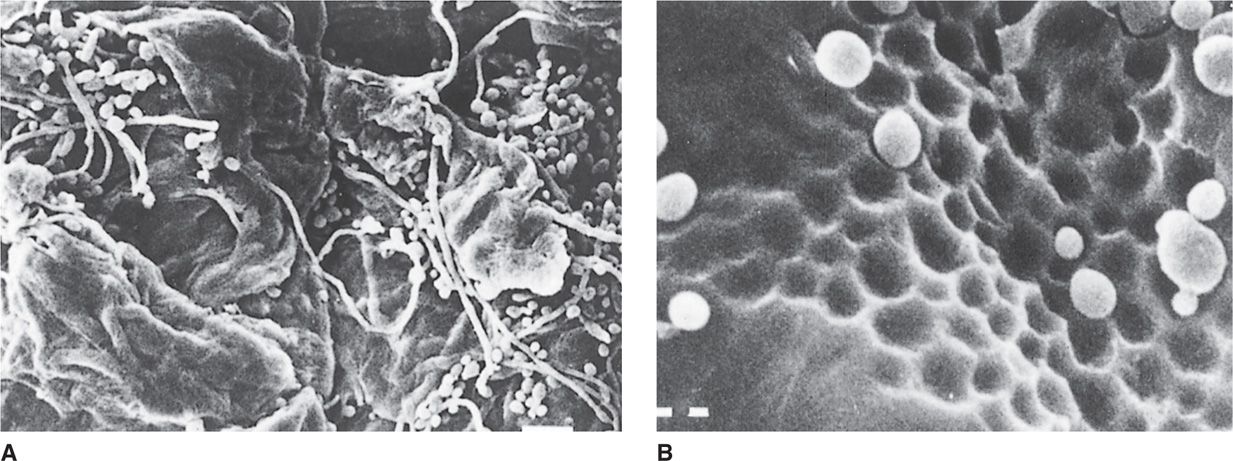
FIGURE 46–4. Invasiveness of Candida albicans. Two features of invasiveness are seen in these scanning electron micrographs taken from experiments with murine corneocytes. A. Both blastoconidia and mycelial elements are present. The mycelial elements spread over the surface and invade the cell cuticle. B. A C albicans strain that produces a protease is seen producing cavity-like depressions in the cell surface. This action could play a role in invasion of the cell. (Reprinted with permission of Thomas L. Ray and Candia D. Payne. Infect Immunol 1988;56:1945-1947, Figures 4,6B. Copyright American Society for Microbiology.)
Hwp1 binds to epithelial cells
Mannoproteins bind to ECM
Hyphae produce Saps, other enzymes
Candida albicans has various mechanisms which facilitate evasion of innate immune mechanisms. These include the masking of surface structures from Toll-like receptors (TLRs) and the accelerated degradation of surface complement C3b. The latter can be accomplished by binding of serum factor H or by secretion of its own protease. Hyphae also have surface proteins that resemble the complement receptors (CR2, CR3) on phagocytes. This seems likely to confuse the phagocyte’s ability to recognize C3b bound to the candidal surface. Enhanced production of these receptors under various conditions, such as elevated glucose concentration, is associated with resistance to phagocytosis by neutrophils. If phagocytosed, hyphal growth interferes with lysosomal fusion and leads to the death of macrophages.
Surface proteins resemble complement receptors
Factors that allow C albicans to increase its relative proportion of the flora (antibacterial therapy), that compromise the general immune capacity of the host (leukopenia or corticosteroid therapy), or that interfere with T-lymphocyte function (AIDS) are often associated with local and invasive infection. The disruptions of the mucosa associated with chronic disease and their treatments (indwelling devices, cancer chemotherapy) may enhance the invasion process by exposing Candida binding sites in the ECM. Biofilm formation on the plastics used in medical devices also contributes. Diabetes mellitus predisposes to C albicans infection, possibly because of the known greater production of the surface mannoproteins in the presence of high glucose concentrations.
Antimicrobials and immunosuppression increase risk
Mechanical disruptions may provide access to ECM
IMMUNITY
Both humoral immunity and cell-mediated immunity (CMI) are involved in defense against Candida infections. Neutrophils are the primary first-line defense. Yeast forms of C albicans are readily phagocytosed and killed when opsonized by antibody and complement. In the absence of specific antibody, the process is less efficient, but a naturally occurring antimannan IgG is able to activate the classical complement pathway and facilitate the alternate pathway. Hyphal forms may be too large to be ingested by polymorphonuclear neutrophils (PMNs), but they can still kill the fungi by attaching to the hyphae and discharging metabolites generated by the oxidative burst. A deficit in neutrophils or neutrophilic function is the most common correlate of serious C albicans infection.
Opsonized yeast forms are killed by PMNs
Antimannan IgG activates complement
The association of chronic mucocutaneous candidiasis with a number of T-lymphocyte immunodeficiencies emphasizes the importance of this arm of the immune system in defense against Candida infections. The increased frequency of oral and vaginal candidiasis in AIDS patients suggests that even superficial infections involve T-lymphocyte–mediated TH1 immune responses. In animal studies, Candida cell wall mannan has been shown to play an immunoregulatory function by downregulating CMI responses. A possible explanation for the association between AIDS and Candida infection is the upregulation of CD4 receptors on monocytes by Candida products. As with other fungi, cytokine activation of macrophages enhances their ability to kill C albicans. A favorable outcome appears to require the proper balance between TH1- and TH2-mediated cytokine responses. The cytokines associated with TH1 (interleukin-2 [IL-2], IL-12, interferon-γ, tumor necrosis factor-α) are correlated with enhanced resistance against infection in which TH2 responses (IL-4, IL-6, and IL-10) are associated with chronic disease.
Compromised CMI is associated with progressive infection
Candida mannan may downregulate CMI responses
Balance between TH1 and TH2 cytokines is necessary
 CANDIDIASIS: CLINICAL ASPECTS
CANDIDIASIS: CLINICAL ASPECTS
MANIFESTATIONS
Superficial invasion of the mucous membranes by C albicans produces a white, cheesy plaque that is loosely adherent to the mucosal surface. The lesion is usually painless, unless the plaque is torn away and the raw, weeping, invaded surface is exposed. Oral lesions, called thrush, occur on the tongue, palate, and other mucosal surfaces as single or multiple, ragged white patches (Figure 46–5). A similar infection in the vagina, vaginal candidiasis, produces a thick, curd-like discharge and itching of the vulva. Although most women have at least one episode of vaginal candidiasis in a lifetime, a small proportion suffers chronic, recurrent infections. No general or specific immune defect has yet been linked to this syndrome.
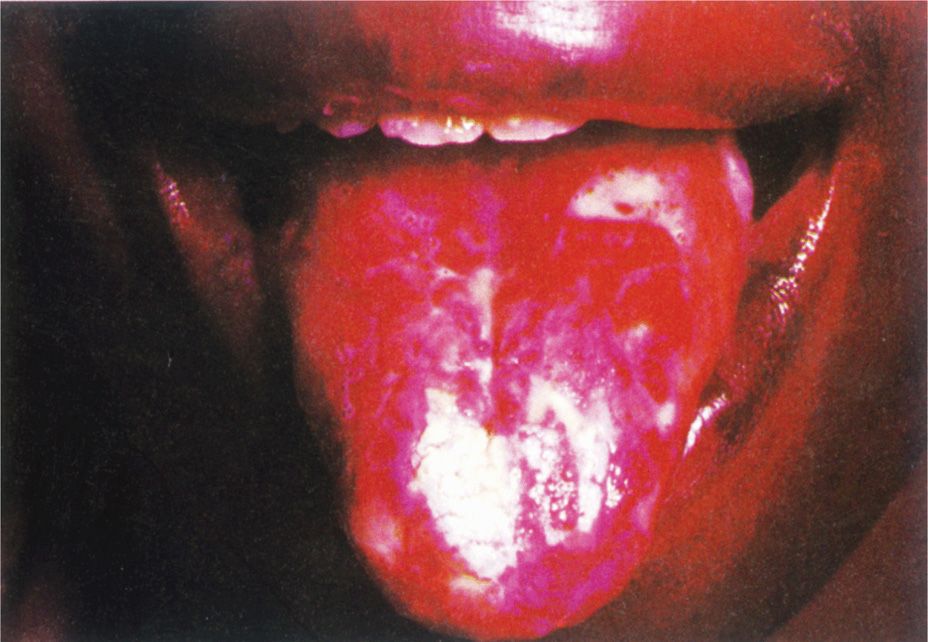
FIGURE 46–5. Trush. The white plaques on this AIDS patient’s tongue are caused by Candida albicans. (Reproduced with permission from Willey JM: Prescott, Harley, & Klein’s Microbiology, 7th edition. McGraw-Hill, 2008.)
White mucosal plaque is called thrush
Vaginitis may be recurrent
Candida albicans skin infections occur in crural folds and other areas in which wet, macerated skin surfaces are opposed. For example, one type of diaper rash is caused by C albicans (Figure 46–6A). Other infections of the skinfolds and appendages occur in association with recurrent immersion in water (eg, dishwashers). The initial lesions are erythematous papules or confluent areas associated with tenderness, erythema, and fissures of the skin. Infection usually remains confined to the chronically irritated area, but may spread beyond it, particularly in infants.
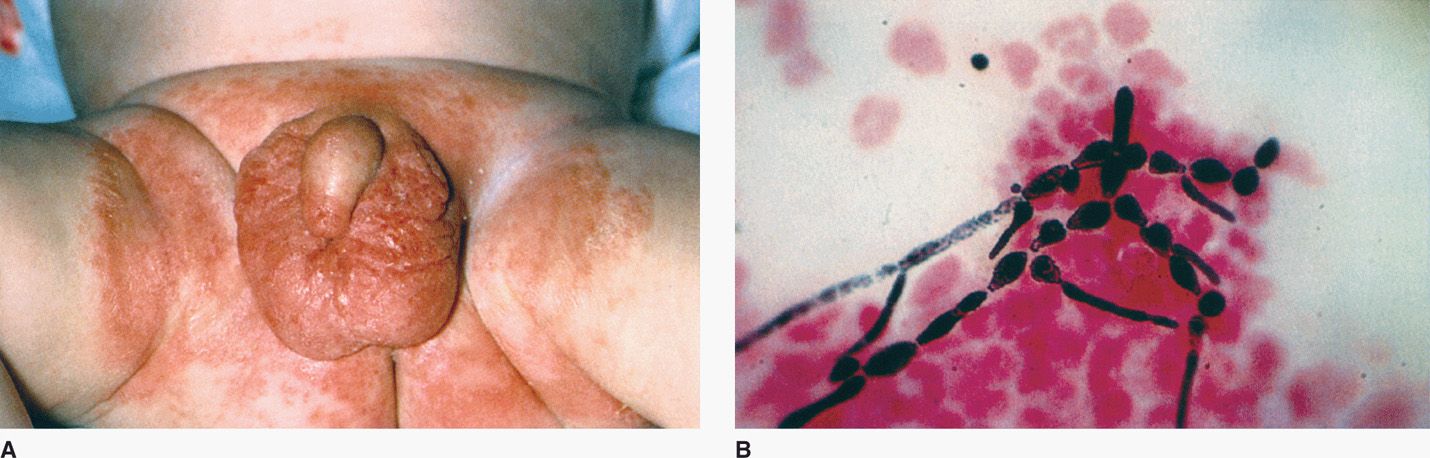
FIGURE 46–6. Candida albicans skin infection. A. This rash is preceded by chronically damp skin in the diaper area. B. This Gram stain demonstrates yeast cells and pseudohyphae. (Reproduced with permission from Nester EW: Microbiology: A Human Perspective, 6th edition. 2009.)
Macerated skin is a common site
In rare persons with specific defects in TH1 immune defenses against Candida, a chronic, relapsing form of candidiasis known as chronic mucocutaneous candidiasis develops. Infections of the skin, hair, and mucocutaneous junctions fail to resolve with adequate therapy and management. There is considerable disfigurement and discomfort, particularly when the disease is accompanied by a granulomatous inflammatory response. Although lesions may become extensive, they usually do not disseminate. To some degree, candidiasis may represent a clinical example of immunologic tolerance. Cutaneous anergy to C albicans antigens is commonly seen in these patients and is often reversed during antifungal chemotherapy, suggesting that it is due to chronic antigen excess.
Chronic mucocutaneous candidiasis is associated with specific T-cell defects
Inflammatory patches similar to those in thrush may develop in the esophagus with or without associated oral candidiasis. Painful swallowing and substernal chest pain are the most common symptoms. Extensive ulcerations, deformity, and occasionally perforation of the esophagus may ensue. In immunocompromised patients, similar lesions may also develop in the stomach, together with deep ulcerative lesions of the small and large intestine.
Esophagitis and intestinal candidiasis are similar to thrush
Infection of the urinary tract via the hematogenous or ascending routes may produce cystitis, pyelonephritis, abscesses, or expanding fungus ball lesions in the renal pelvis. The clinical findings in disseminated infections of the kidneys, brain, and heart are generally not sufficiently characteristic to suggest C albicans over the bacterial pathogens, which more commonly produce infection of deep organs. Candida endophthalmitis has the characteristic funduscopic appearance of a white cotton ball expanding on the retina or floating free in the vitreous humor. Endophthalmitis and infections of other eye structures can lead to blindness.
Urinary tract infections are ascending or hematogenous
Endophthalmitis appears as white cotton on retina
DIAGNOSIS
Superficial C albicans infections provide ready access to diagnostic material. Exudate or epithelial scrapings examined by KOH preparations or Gram smear (Figure 46–6B) demonstrate abundant budding yeast cells; if associated hyphae are present, the infection is almost certainly caused by C albicans. Candida albicans is readily isolated from clinical specimens including blood. Cultures from specimens, such as sputum, run the risk of contamination from the normal flora or a superficial mucous membrane lesion. A direct aspirate, biopsy, or bronchoalveolar lavage is often required to establish the diagnosis.
KOH and Gram smears of superficial lesions show yeast and hyphae
Lung involvement requires bronchoalveolar lavage
Deep organ involvement is difficult to prove without a direct aspirate or biopsy. Even positive blood cultures must be interpreted with caution if they could represent colonization of intravenous catheters. Candida endocarditis represents a special diagnostic problem because the yeasts seeding the blood from the valve may be filtered in the capillary beds due to their large size. Arterial blood cultures may be required in this situation.
Endocarditis may require arterial cultures
TREATMENT
Candida albicans is usually susceptible to amphotericin B, nystatin, flucytosine, caspofungin, and the azoles. Superficial infections are generally treated with topical nystatin or azole preparations. Measures to decrease moisture and chronic trauma are important adjuncts in treating Candida skin infections. Deeper C albicans infections may resolve spontaneously with elimination or control of predisposing conditions. Removal of an infected catheter, control of diabetes, or an increase in peripheral leukocyte counts is often associated with recovery without antifungal therapy. Persistent relapsing or disseminated candidiasis is treated with various combinations of fluconazole, amphotericin B, and caspofungin. Fluconazole has been the most effective treatment for chronic mucocutaneous candidiasis.
Topical nystatin or azoles for superficial lesions
Amphotericin B, fluconazole, and caspofungin reserved for invasive disease
Other Candida Species
Species of Candida other than C albicans produce infections in circumstances similar to those described above, but do so less frequently. When contamination of an indwelling device is the portal of entry, the probability of infection by these other species increases. Little is known of the pathogenesis of these species with the exception of Candida tropicalis. Both experimental and clinical evidence indicate that C tropicalis has virulence at least equal to that of C albicans. Candida tropicalis produces extracellular proteinases similar to those of C albicans, which may enhance its invasiveness.
Candida tropicalis is highly virulent
Candida glabrata is another common species. This species is very small for a yeast (2-4 μm) and does not produce hyphae. It is often a member of the gastrointestinal and genital micro-biota. The most common infections are in the urinary tract, but deep tissue involvement and fungemia occur. The organisms are small enough to be confused with Histoplasma capsulatum (Chapter 47) in histologic preparations. Therapy is similar to that for C albicans, although C glabrata is more likely to be resistant to fluconazole. Other species of Candida, which lack any distinguishing morphologic or clinical characteristics, may produce disease. Some of these fungi are inherently resistant to the antifungal azoles.
Candida glabrata is a very small yeast
ASPERGILLUS
 MYCOLOGY
MYCOLOGY
Aspergillus species are rapidly growing molds with branching septate hyphae and characteristic arrangement of conidia on the conidiophore (Figure 46–7A–C). Fluffy colonies appear in 1 to 2 days; by 5 days, they may cover an entire plate with pigmented growth. Species are defined on the basis of differences in the structure of the conidiophore and the arrangement of the conidia. The most common infections in humans are A fumigatus and Aflavus, but others, such as A niger and A terreus may be involved.
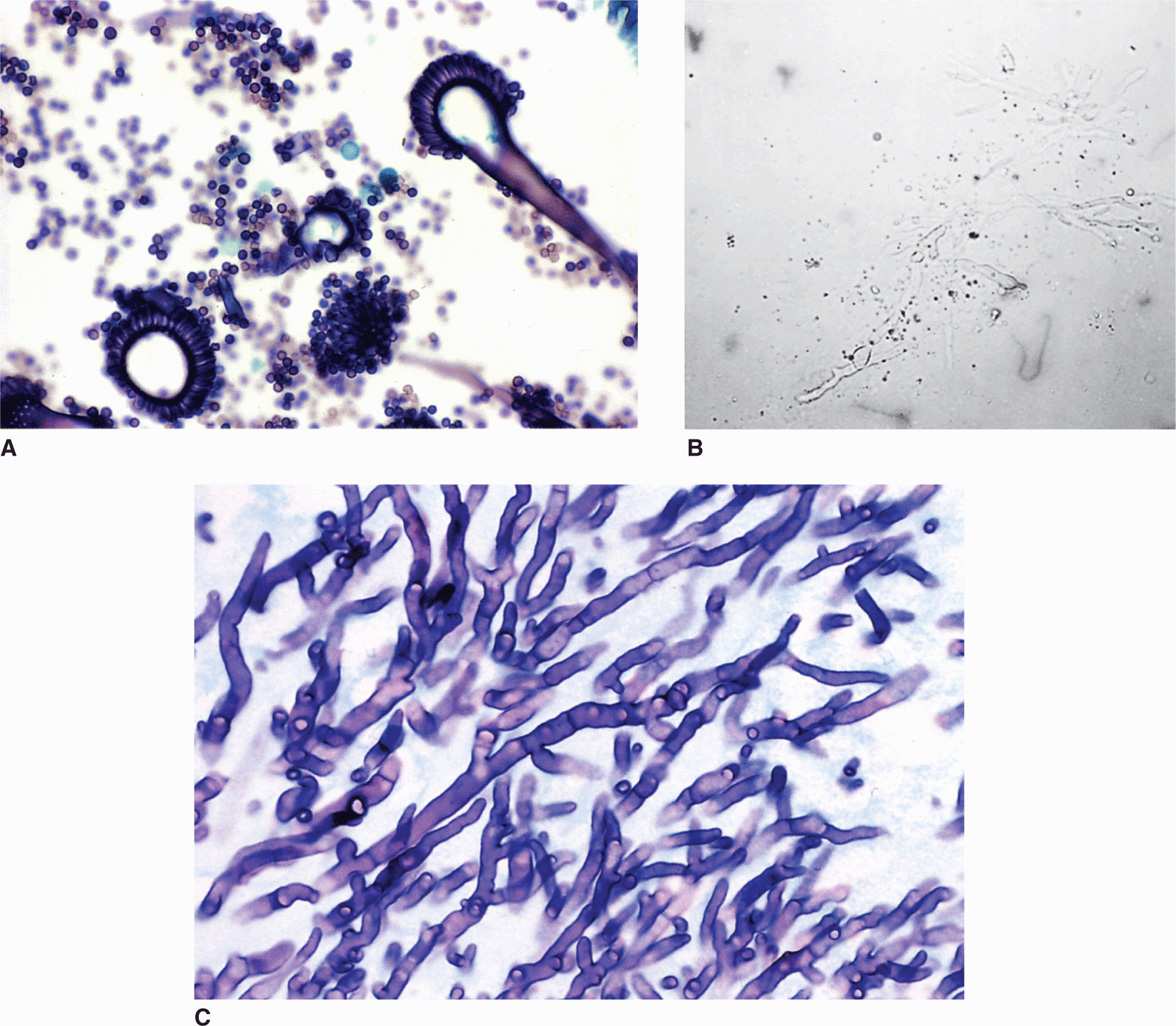
FIGURE 46–7. Aspergillus. A. This asexual conidium-forming structure is characteristic of Aspergillus species. The conidia are borne at the end of the finger-like extensions at the end of the conidiophore. These structures are rarely produced in vivo. B. This tissue aspirate mixed with KOH shows branching, septate hyphae. C. Histologic sections also show branching, septate hyphae, but because the conidia shown in A are not seen the findings are not diagnostic of Aspergillus. (A and C, Reproduced with permission from Connor DH, Chandler FW, Schwartz DQ, et al: Pathology of Infectious Diseases. Stamford CT: Appleton & Lange, 1997.)
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


