BACTERIOLOGY
MORPHOLOGY AND STRUCTURE
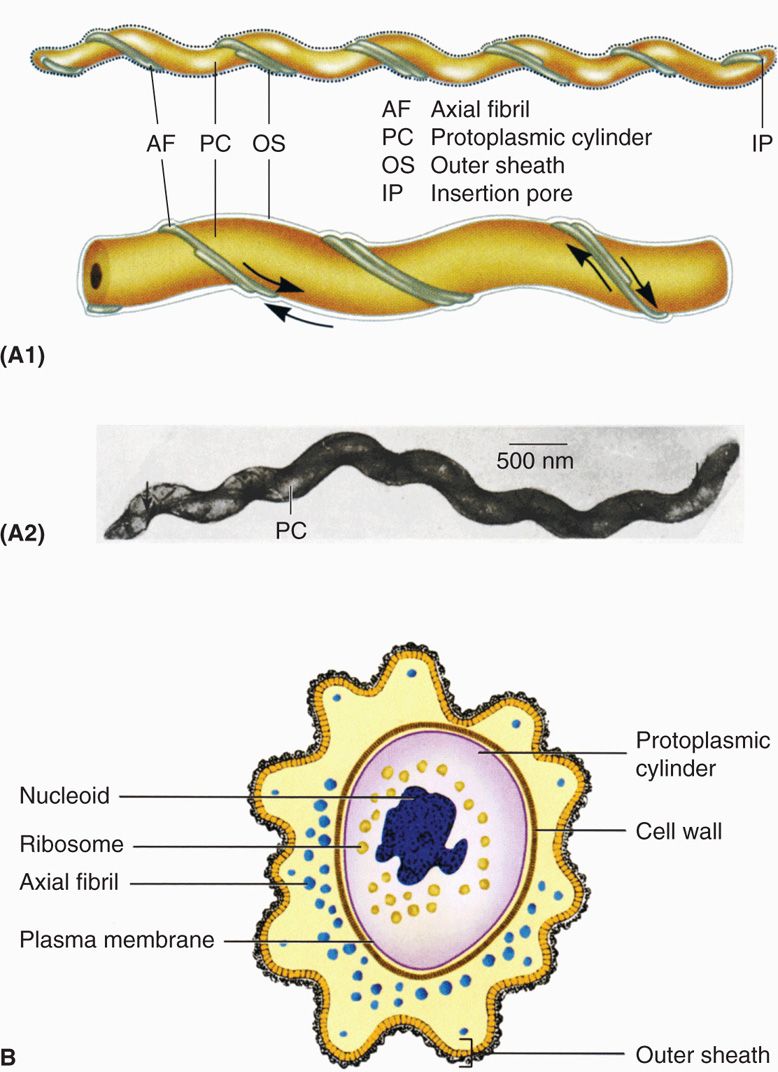
The spiral morphology of spirochetes (Figure 37–1) is produced by a flexible, peptidoglycan cell wall around which several axial fibrils are wound. The cell wall and axial fibrils are completely covered by an outer bilayered membrane similar to the outer membrane of other Gram-negative bacteria. In some species, a hyaluronic acid slime layer forms around the exterior of the organism and may contribute to its virulence. Spirochetes are motile, exhibiting rotation and flexion; this motility is believed to result from movement of the axial filaments, although the mechanism is not clear.
FIGURE 37–1. Spirochete morphology. A1. Longitudinal surface view of typical spirochete. A2. Electron micrograph of Treponema with axial filaments extending most of cell length. B. Cross-section of typical spirochete. (Reproduced with permission from Willey JM: Prescott, Harley, & Klein’s Microbiology, 7th edition. McGraw-Hill, 2008.)
Spiral structure is wound around endoflagella
Motility includes rotation and flexion
Many spirochetes are difficult to see by routine microscopy. Although they are Gram negative, many either take stains poorly or are too thin (0.15 μm or less) to fall within the resolving power of the light microscope. Only darkfield microscopy (Figure 37-2), immunofluorescence, or special staining techniques can demonstrate these spirochetes. Other spirochetes such as Borrelia are wider and readily visible in stained preparations, even routine blood smears.
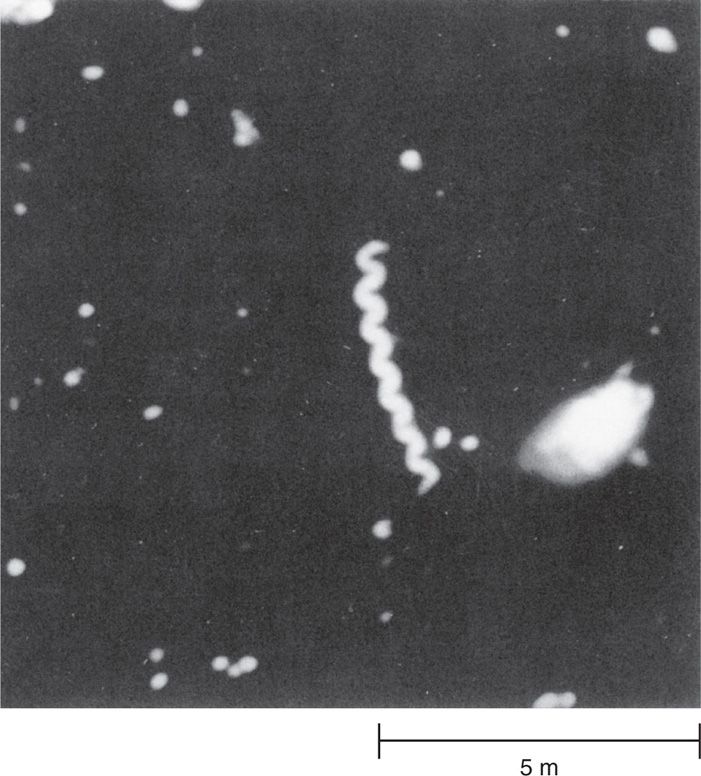
FIGURE 37–2. Treponema pallidum seen by darkfield microscopy. The darkfield method creates a bright halo around the corkscrew-shaped spirochetes. (Reproduced with permission from Nester EW: Microbiology: A Human Perspective, 6th edition. 2009.)
Many are thin and take stains poorly
Darkfield demonstrates spirochetes
GROWTH AND CLASSIFICATION
Parasitic spirochetes grow more slowly in vitro than most other disease-causing bacteria. Some species, including the causative agent of syphilis, have not been grown beyond a few generations in cell culture. Some are strict anaerobes, others require low concentrations of oxygen, and still others are aerobic. Compared with other bacterial groups, the taxonomy of the spirochetes is underdeveloped. Because spirochetes are difficult to grow, they are difficult to study; thus, there are relatively few phenotypic properties on which to base a classification. The medically important genera Treponema, Leptospira, and Borrelia have been distinguished primarily by morphologic characters such as the nature of their spiral shape and the arrangement of flagella. Modern DNA homology and ribosomal RNA analyses have supported these groupings.
Some have not been isolated in culture
May be aerobic or anaerobic
![]() SPIROCHETAL DISEASES
SPIROCHETAL DISEASES
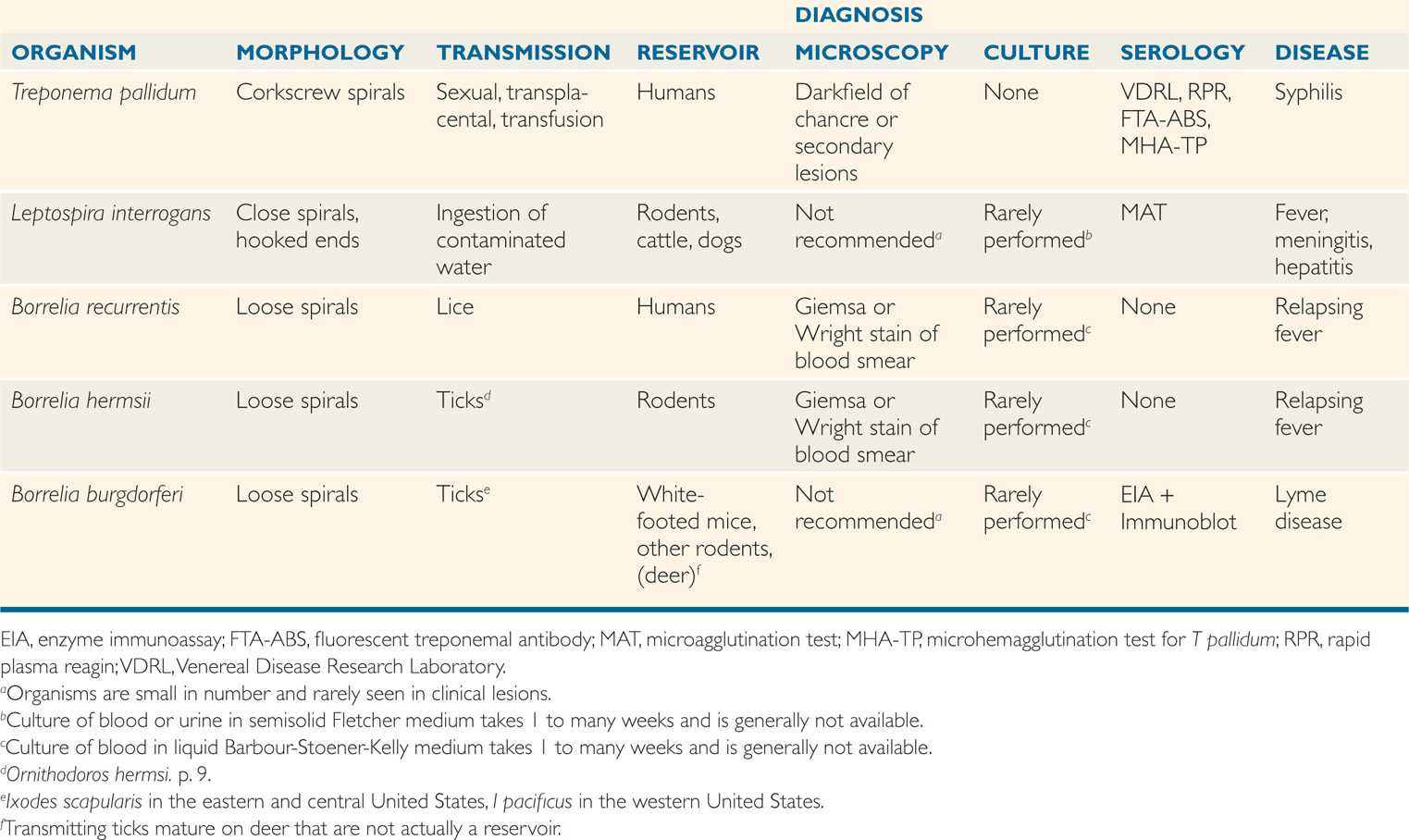
Some spirochetes are free living; some are members of the resident flora of humans and animals. The oral cavity, particularly the dental crevice, harbors a number of nonpathogenic species of Treponema and Borrelia as part of its flora. Under unusual conditions, these spirochetes, together with anaerobes in the flora, can cause necrotizing, ulcerative infection of the gums, oral cavity, or pharynx (Vincent infection, trench mouth). The pathogenesis of these opportunistic infections is not understood, but they are correlated with immunocompromise, severe malnutrition, and neglect of basic hygiene (Table 37-1). The term “trench mouth” refers to the occurrence of these infections in troops under the appalling conditions that existed in the trenches during World War I.
TABLE 37–1 Features of Spirochetal Diseases
Many are part of oropharyngeal flora
Overgrowth causes trench mouth
The major spirochetal diseases are caused by selected species of three genera that are not found in the resident flora, Treponema (T pallidum), Leptospira (L interrogans), and Borrelia (B recurrentis, B hermsii, and B burgdorferi). Most Borrelia and Leptospira infections are zoonoses transmitted from wild and domestic animals. Treponema pallidum is a strict human pathogen transmitted by sexual contact. Some rare nonvenereal treponemal diseases are summarized in Appendix 37-1.
APPENDIX 37–1 Nonvenereal Treponemes

Diseases are zoonoses or venereal
TREPONEMA PALLIDUM
Treponema pallidum is the causative agent of syphilis, a venereal disease first recognized in the 16th century as the “great pox,” which rapidly spread through Europe in association with urbanization and military campaigns. Some argue that it was brought back from the New World by the sailors with Christopher Columbus. Its extended course and the protean, often dramatic nature of its findings (genital ulcer, ataxia, dementia, ruptured aorta) are due to a state of balanced parasitism that spans decades. The cause of syphilis is actually a subspecies (T pallidum subsp. pallidum) closely related to other agents that cause rare nonvene-real treponematoses. Treponema pallidum is used here to indicate the pallidum subspecies.
Syphilis represents an extended balance of parasitism and disease
 BACTERIOLOGY
BACTERIOLOGY
Treponema pallidum is a slim spirochete 5 to 15 μm long with regular spirals whose wavelength and amplitude resemble a corkscrew (Figure 37-2). The organism is readily seen only by immunofluorescence, darkfield microscopy, or silver impregnation histologic techniques. Live cells show characteristic rotating motility with sudden 90-degree angle flexions, which suggest a gentleman quickly bowing at the waist. Treponema pallidum is extremely susceptible to any deviation from physiologic conditions. It dies rapidly on drying and is readily killed by a wide range of detergents and disinfectants. The lethal effect of even modest elevations of temperature (41-42°C) was the basis for the technique of fever therapy for syphilis introduced in Vienna a century ago (patients were infected with malaria parasites!).
Corkscrew spirals spin and bow
Heat, drying, and disinfectants kill quickly
Beyond these observations, the study of the biology and pathogenesis of T pallidum is severely impeded by our inability to grow the organism in culture. It multiplies for only a few generations in cell cultures and is difficult to subculture. Sustained growth is achieved only in animals (rabbit testes), which are the sole source of bacteria for diagnostic reagents and scientific study. The Tpallidum genome is amenable to study, and much of what follows is based on extrapolations comparing genes found there with those in other pathogenic bacteria. This genome, however, is among the smallest known and several times less than other bacterial pathogens discussed in this book. The unfolding picture of the syphilis spirochete is that it is a minimalist pathogen, growing very slowly and producing few definitive structures or products.
Prolonged growth only in animals
Genes compared to other pathogens
The sluggish growth (mean generation time more than 30 hours) of Tpallidum is felt to be due to lack of enzymes that detoxify reactive oxygen species (catalase, oxidase) and the absence of efficient energy (ATP)-producing pathways such as the tricarboxylic acid cycle and electron transport chain. Treponema pallidum shares the Gram-negative structural style of other spiro-chetes, but its outer membrane lacks lipopolysaccaride (LPS) and contains few proteins.
Lacks common enzymes
No LPS and few proteins in outer membrane
![]() SYPHILIS
SYPHILIS
EPIDEMIOLOGY
Treponema pallidum is an exclusively human pathogen under natural conditions. In most cases, infection is acquired from direct sexual contact with a person who has an active primary or secondary syphilitic lesion (Figure 37-3). Partner notification studies suggest transmission occurs in over 50% of sexual contacts in which a lesion is present. Less commonly, the disease may be spread by nongenital contact with a lesion (eg, of the lip), sharing of needles by intravenous drug users, or transplacental transmission to the fetus within approximately the first 3 years of the maternal infection. Late disease is not infectious. Modern screening procedures have essentially eliminated blood transfusion as a source of the disease. The incidence of new cases of primary and secondary syphilis in developed countries declined to an all time low at the end of the 20th century, but since then has risen more than 10%. Worldwide, syphilis remains a major public health problem, with an estimated 12 million new cases annually. There is evidence that syphilitic lesions are a portal for HIV transmission.
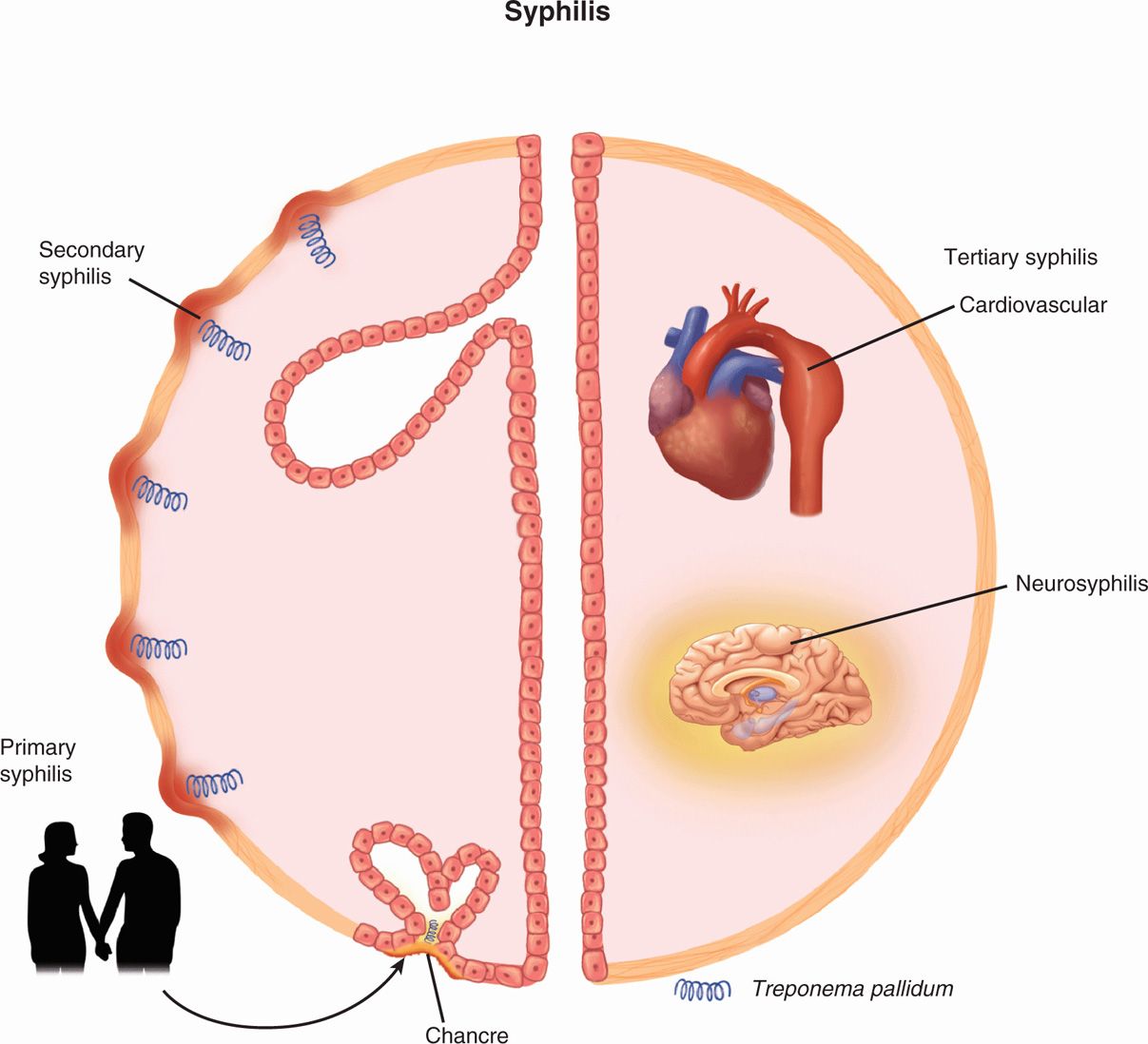
FIGURE 37–3. Syphilis overview. Infection is acquired by sexual contact, and the primary lesion is an ulcer on the genitalia called the chancre. The major feature of secondary syphilis is a maculopapular rash that is teeming with spirochetes. Tertiary syphilis (right) involves multiple organ systems. Shown are an aortic aneurysm as part of cardiovascular syphilis and inflammation of the brain in neurosyphilis.
Transmission is by contact with mucosal surfaces or blood
Congenital infection is transplacental
Tertiary syphilis is not infectious
PATHOGENESIS
The spirochete reaches the subepithelial tissues through unapparent breaks in the skin or possibly by passage between the epithelial cells of mucous membranes aided by at least one adhesin that binds to fibronectin and elements of the extracellular matrix. In the submucosa, it multiplies slowly stimulating little initial tissue reaction. This is probably due to the relative paucity of antigens in the Tpallidum outer membrane that would be exposed to the immune system. In experimental infections, the organisms spread from the primary site to the bloodstream within minutes and are established in distant tissues within hours. As lesions develop, the basic pathologic finding is an endarteritis. The small arterioles show swelling and proliferation of their endothelial cells. This reduces or obstructs local blood supply, probably accounting for the necrotic ulceration of the primary lesion and subsequent destruction at other sites (Figure 37-4A-C). Dense, granulomatous cuffs of lymphocytes, monocytes, and plasma cells surround the vessels. There is no evidence that this injury is due to any toxins or other classic virulence factors produced by T pallidum. Although the primary lesion heals spontaneously, the bacteria have already disseminated to other organs by way of local lymph nodes and the bloodstream.
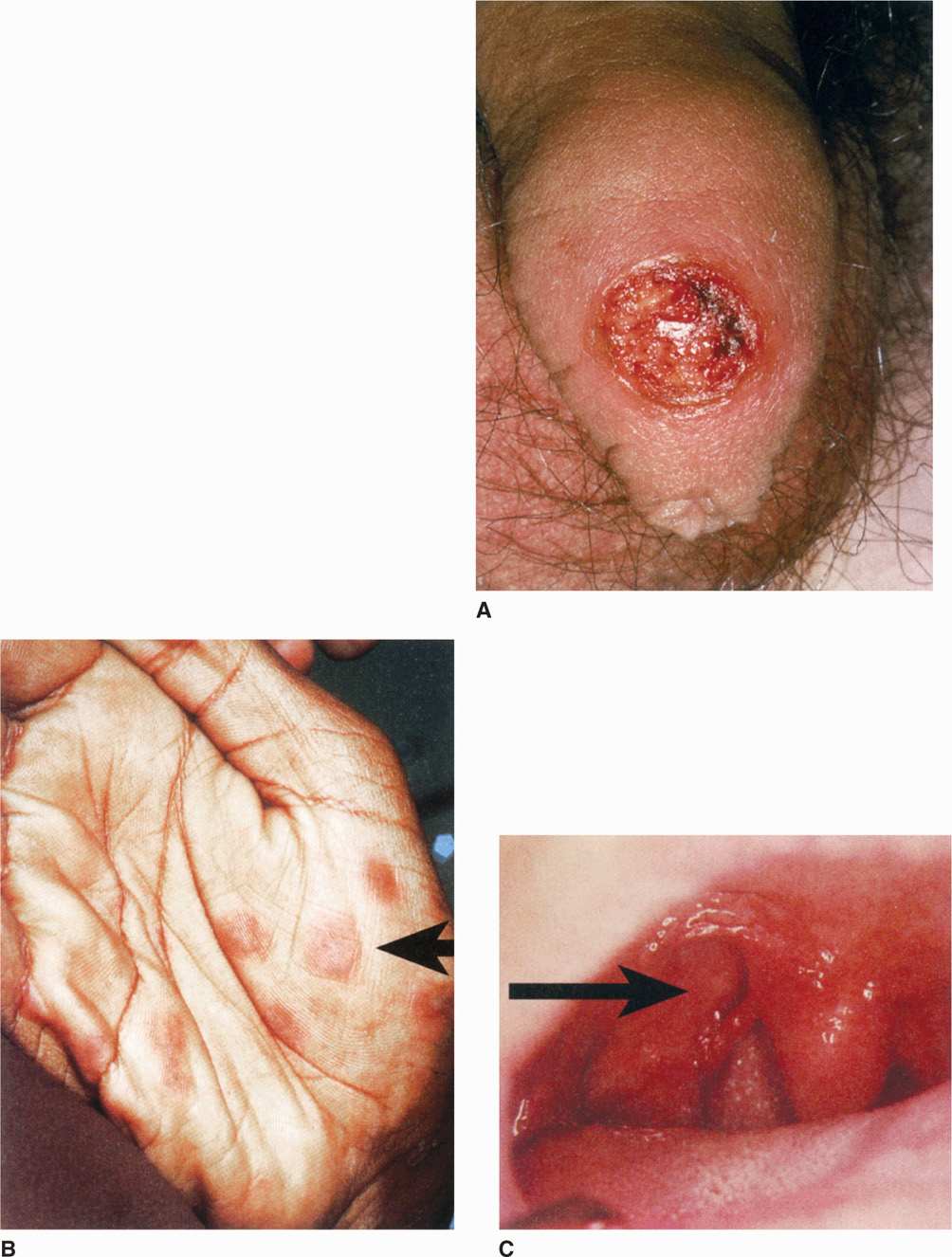
FIGURE 37–4. Syphilitic lesions. A. Primary syphilis. A syphilitic chancre is shown on the foreskin of the penis. Note the sharp edge and raw base of the ulcer. (Reproduced with permission from Nester EW: Microbiology: A Human Perspective, 6th edition. 2009.) B. Secondary syphilis. The maculopapular rash appears on the palm. C. Tertiary syphilis. A ruptured gumma appears as a lump and ulcer in the hard palate of the mouth. (B and C, Reproduced with permission from Willey JM: Prescott, Harley, & Klein’s Microbiology, 7th edition. McGraw-Hill, 2008.)
Spread from mucosal breaks to blood is rapid
Slow multiplication produces endarteritis, granulomas
Ulcer heals but spirochetes disseminate
The disease is clinically silent until the disseminated secondary stage develops and then is silent again with entry into latency. Although evasion of host defenses is clearly taking place, the mechanisms involved are unknown. Treponema pallidum strains found in secondary lesions have not been demonstrated to differ antigenically from those in the primary chancre. It may be that the combination of the low antigen content of its outer membrane combined with the extremely slow multiplication rate allows the organism to stay below whatever critical antigenic mass is required to trigger an effective immune response. Without virulence factors to explain the tissue destruction, we are left with injury due to a prolonged delayed-type hypersensitivity (DTH) response to the persistent bacteria.
Minimal triggers for immune response
Injury is due to prolonged hypersensitivity responses
IMMUNITY
Clinical observations suggest an immune response in syphilis that is slow and imperfect. Immunity to reinfection does not appear until early latency, and for at least one-third of those infected the subsequent host response is successful in clearing most but not all of the treponemes.
Immunity develops slowly and incompletely
The immune mechanisms involved are far from clear, but appear to involve both humoral and cell-mediated responses. Resistance to reinfection is correlated with appearance of antitreponemal antibody, which is able to immobilize and kill the organism. Exposed trepo-nemal outer membrane proteins (OMPs) are the most probable target of these antibodies. Cell-mediated responses appear to be dominant in syphilitic lesions with T lymphocytes (CD4+ and CD8+) and macrophages, the primary cell types, present. Activated macrophages play a major role in the clearance of T pallidum from early syphilitic lesions. The relapsing course of primary and secondary syphilis may reflect shifts in the balance between developing cellular immunity and suppression of T lymphocytes. Syphilis in immunocompromised patients such as those with acquired immunodeficiency syndrome (AIDS) may present with unusually aggressive or atypical manifestations.
Antibodies to OMP are associated with reinfection resistance
Development of cell-mediated immunity clears lesions
Variable T-lymphocyte suppression may link to stages
 SYPHILIS: CLINICAL ASPECTS
SYPHILIS: CLINICAL ASPECTS
MANIFESTATIONS
 Primary Syphilis
Primary Syphilis
The primary syphilitic lesion is a papule that evolves to an ulcer at the site of infection. This is usually the external genitalia or cervix, but could be in the anal or oral area depending on the nature of sexual contact. The lesion becomes indurated and ulcerates but remains painless, though slightly sensitive to touch. The fully developed ulcer with a firm base and raised margins is called the chancre (Figure 37-4A). Firm, nonsuppurative, painless enlargement of the regional lymph nodes usually develops within 1 week of the primary lesion and may persist for months. The median incubation period from contact until appearance of the primary lesion is about 3 weeks (range 3-90 days). It heals spontaneously after 4 to 6 weeks.
Painless, indurated ulcer starts the disease
Heals spontaneously after weeks
 Secondary Syphilis
Secondary Syphilis
Secondary or disseminated syphilis develops 2 to 8 weeks after the appearance of the chancre. The primary lesion has usually healed but may still be present. This most florid form of syphilis is characterized by a symmetric mucocutaneous maculopapular rash and generalized nontender lymph node enlargement with fever, malaise, and other manifestations of systemic infection. Skin lesions are distributed on the trunk and extremities, often including the palms (Figure 37-4B), soles, and face, and can mimic a variety of infectious and noninfectious skin eruptions. About one-third of patients develop painless mucosal warty erosions called condylomata lata. These erosions usually develop in warm, moist sites such as the genitals and perineum. All the lesions of secondary syphilis are teeming with spi-rochetes and are highly infectious. They resolve spontaneously after a few days to many weeks, but the infection itself has resolved in only one-third of patients. In the remaining two-thirds, the illness enters the latent state.
Lymphadenopathy and maculopapular rash are generalized
Spirochetes are abundant
Lesions resolve, but disease continues in one-third of patients
 Latent Syphilis
Latent Syphilis
Latent syphilis is by definition a stage in which no clinical manifestations are present, but continuing infection is evidenced by serologic tests. In the first few years, latency may be interrupted by progressively less severe relapses of secondary syphilis. In late latent syphilis (>4 years), relapses cease, and patients become resistant to reinfection. Transmission to others is possible from relapsing secondary lesions and by transfusion or other contact with blood products. Mothers may transmit T pallidum to their fetus throughout latency. About one-third of untreated cases do not progress beyond this stage.
Secondary relapses interrupt latency
Bloodborne transmission risk continues
 Tertiary Syphilis
Tertiary Syphilis
Another one-third of patients with untreated syphilis develop tertiary syphilis. The manifestations may appear as early as 5 years after infection but characteristically occur after 15 to 20 years. The manifestations depend on the body sites involved, the most important of which are the nervous and cardiovascular systems.
Neurosyphilis is due to the damage produced by a mixture of meningovasculitis and degenerative parenchymal changes in virtually any part of the nervous system. The most common entity is a chronic meningitis with fever, headache, focal neurologic findings, and increased cells and protein in the cerebrospinal fluid (CSF). Cortical degeneration of the brain causes mental changes ranging from decreased memory to hallucinations or frank psychosis. In the spinal cord, demyelination of the posterior columns, dorsal roots, and dorsal root ganglia produces a syndrome called tabes dorsalis (Figure 37-5), which includes ataxia, wide-based gait, foot slap, and loss of the sensation. The most advanced central nervous system (CNS) findings include a combination of neurologic deficits and behavioral disturbances called paresis, which is also a mnemonic (p ersonality, affect, reflexes, eyes, sensorium, intellect, speech) for the myriad of changes seen.
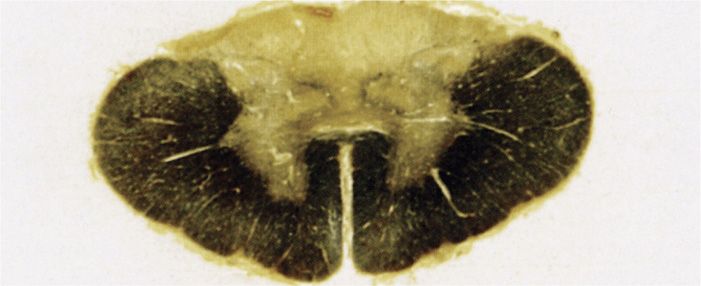
FIGURE 37–5. Tabes dorsalis. Loss of axons and myelin is evident in the posterior columns of the spinal cord (Woelke stain). (Reproduced with permission from Connor DH, Chandler FW, Schwartz DQ, et al: Pathology of Infectious Diseases. Stamford CT: Appleton & Lange, 1997.)
Chronic meningitis leads to degenerative changes and psychosis
Demylination causes peripheral neuropathies
Syphilitic paresis has many signs
Cardiovascular syphilis is due to arteritis involving the vasa vasorum of the aorta and causing a medial necrosis and loss of elastic fibers. The usual result is dilatation of the aorta and aortic valve ring. This in turn leads to aneurysms of the ascending and transverse segments of the aorta and/or aortic valve incompetence. The expanding aneurysm can produce pressure necrosis of adjacent structures or even rupture. A localized, granulomatous reaction to Tpallidum infection called a gumma (Figure 37-4C) may be found in skin, bones, joints, or other organ. Any clinical manifestations are related to the local destruction as with other mass-producing lesions, such as tumors.
Aortitis leads to aneurysm
Gummas are destructive, localized granulomas
 Congenital Syphilis
Congenital Syphilis
Fetuses are susceptible to syphilis only after the fourth month of gestation and adequate treatment of infected mothers before that time prevents fetal damage. Because active syphilitic infection is devastating to infants, routine serologic testing is performed in early pregnancy and should be repeated in the last trimester in women at high risk for acquiring syphilis. Untreated maternal infection may result in fetal loss or congenital syphilis, which is analogous to secondary syphilis in the adult. Although there may be no physical finding at all, the most common are rhinitis and a maculopapular rash. Bone involvement produces characteristic changes in the architecture of the entire skeletal system (saddle nose, saber shins). Anemia, thrombocytopenia, and liver failure are terminal events.
Rhinitis, rash, and bone changes are common
Serologic screening and treatment is preventive
DIAGNOSIS
 Microscopy
Microscopy
Treponema pallidum can be seen by darkfield microscopy in primary and secondary lesions, but the execution of this procedure requires experience and attention to detail. The suspect lesion must be cleaned and abraded to produce a serous transudate from below the surface of the ulcer base. This material can be captured in a capillary tube or placed directly on a microscope slide if a darkfield setup is close at hand. The microscopist must observe the corkscrew morphology and characteristic motility to make a diagnosis (Figure 37-2). A negative result from examination does not exclude syphilis; to be readily seen, the fluid must contain thousands of treponemes per milliliter. Darkfield microscopy of oral and anal lesions is not recommended because of the risk of misinterpretation of other spirochetes present in the resident flora. Direct fluorescent antibody methods have been developed but are available only in certain centers.
Darkfield requires experience and fluid from deep in lesion
May be negative owing to small numbers
 Serologic Tests
Serologic Tests
Most cases of syphilis are diagnosed serologically using serologic tests that detect antibodies directed at either lipid or specific treponemal antigens. The former are called nontrepone-mal tests, and the latter are referred to as treponemal tests. Their use in screening, diagnosis, and therapeutic evaluation of syphilis has been refined over many decades (Figure 37-6).
Tests may or may not use treponemes
 Nontreponemal Tests
Nontreponemal Tests
Nontreponemal tests measure antibody directed against cardiolipin, a lipid complex so called because one component was originally extracted from beef heart. Anticardiolipin antibody is called reagin, and the tests that detect it depend on immune flocculation of cardiolipin in the presence of other lipids. The most common nontreponemal tests are the rapid plasma reagin (RPR) and the Venereal Disease Research Laboratory (VDRL). The results become positive in the early stages of the primary lesion and, with the possible exception of some patients with advanced HIV infection, are uniformly positive during the secondary stage. They slowly wane in the later stages of the disease. In neurosyphilis, VDRL test results on CSF may be positive when the serum VDRL has reverted to negative. Nontreponemal tests are nonspecific; they may become positive in a variety of autoimmune diseases or in diseases involving substantial tissue or liver destruction, such as lupus erythe-matosus, viral hepatitis, infectious mononucleosis, and malaria. False-positive results can also occur occasionally in pregnancy and in patients with HIV infection.
Reagin antibody reacts with cardiolipin, a lipid complex
Antibody level peaks in secondary syphilis
Nonspecific reactions linked to autoimmune diseases
Sensitivity and low cost make nontreponemal tests preferred for screening, but positive results must be confirmed by one of the more specific treponemal tests described in the following text. The tests are also valuable for monitoring treatment because the height of the antibody titer is directly related to activity of disease. With successful antibiotic therapy, nontreponemal serologies slowly revert to negative.
Titer is used to follow therapy
 Treponemal Tests
Treponemal Tests
Treponemal tests detect antibody specific to T pallidum, such as an indirect immunoflu-orescent procedure called the fluorescent treponemal antibody (FTA-ABS), which uses spirochetes fixed to slides. ABS refers to an absorption step that removes nonspecific antispi-rochetal antibodies often found in normal serum. Another method, the microhemaggluti-nation test for Tpallidum (MHA-TP), uses antigens attached to the surface of erythrocytes, which then agglutinate in the presence of specific antibody.
T pallidum is used as the antigen
Treponemal tests are considerably more specific than the cardiolipin-based nontreponemal tests. Their primary role in diagnosis is to confirm positive RPR and VDRL results obtained in the evaluation of a patient suspected of having syphilis or in screening programs. These tests are not useful for screening or after therapy because, once positive, they usually remain so for life except for the immunocompromised. The basic approach is a two-step process. Initial screening is done with a nontreponemal test, and if positive, the result is confirmed with a treponemal test. The development of point-of-care and automated tests has been primarily with treponemal tests. These methods, if positive, still require confirmation of active disease with a nontreponemal test. The time course of serologic tests in the various stages of syphilis is illustrated in Figure 37-6.
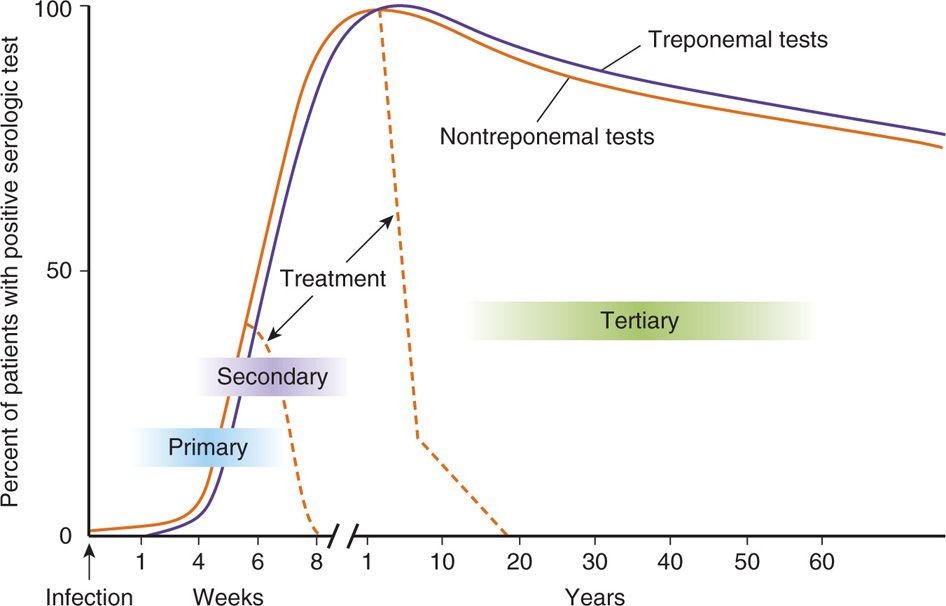
FIGURE 37–6. Syphilis serology. The time course of treponemal and nontreponemal tests in treated and untreated syphilis is shown. The nontreponemal test results (VDRL, RPR) rise during primary syphilis and reach their peak in secondary syphilis. They slowly decline with advancing age. With treatment, they revert to normal over a few weeks. The treponemal tests (FTA-ABS, MHA-TP) follow the same course but remain elevated even after successful treatment. FTA-ABS, fluorescent treponemal antibody; MHA-TP microhemagglutination test for T pallidum; RPR, rapid plasma reagin; VDRL, Venereal Disease Research Laboratory
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree