TABLE 6.1 VOLUMES OF BODY FLUID COMPARTMENTSa | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||
is not a good marker of total body Na+. Blood pressure and physical signs of volume status such as the presence or absence of edema are much better markers of total body Na+.
TABLE 6.2 ELECTROLYTE CONCENTRATIONS IN EXTRACELLULAR AND INTRACELLULAR FLUIDS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
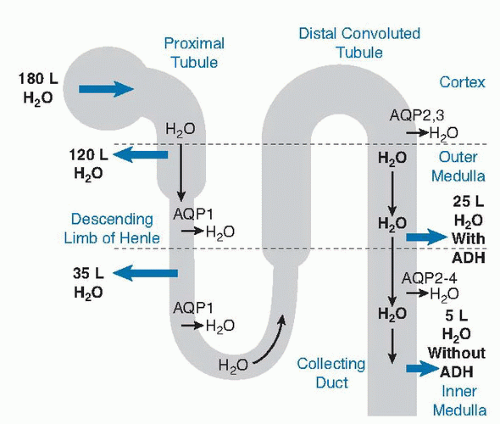
and volume (22). A low effective vascular volume (˜10% decrease) provokes thirst and ADH release (31, 32, 33). These effects are mediated through baroreceptors and some humoral factors released in response to reduced blood flow. This response explains the severe thirst despite hyponatremia seen in heart or liver failure. Other factors, including β-catecholamines, angiotensin II, and physical and emotional stress, enhance ADH output. Ethanol and catecholamines inhibit the output of ADH. Lithium, certain tetracycline antibiotics (demeclocycline), foscarnet, methoxyflurane, amphotericin B, and V-2 receptor antagonists (Vaptans) inhibit the effect of ADH on the kidney (22).
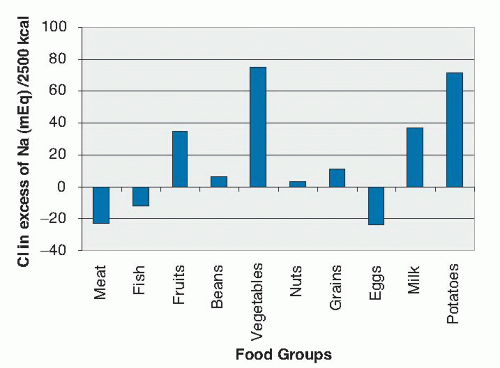
salt. Volume depletion describes the equal loss of salt and water. Salt in this case refers to NaCl, which is the main solute in the vascular space. Depending on the quantity of NaCl losses in relation to water losses, mixed forms of volume depletion and dehydration are encountered. In hypotonic dehydration, NaCl losses exceed water losses.
be seen. In the setting of a partial ADH deficiency, the urine osmolality is close to normal. ADH deficiency can be congenital or acquired (56, 57, 58, 59, 60, 61). During pregnancy, ADH deficiency may be caused by excessive production of vasopressinase (gestational DI) (65, 66). Causes of polyuria are listed in Table 6.3. Note that urine outputs of more than 2.5 L may be considered desirable in patients with kidney stone formation.
TABLE 6.3 CAUSES OF POLYURIA | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Na+ concentration resulting from a systematic error in measurement. Changes in methodology have largely reduced this problem in most clinical centers (75); however, the presence of a nonosmotic substance (in vitro hemolysis, hyperlipidemia, hyperproteinemia, and mannitol), may still cause pseudohyponatremia (76, 77, 78, 79).
Water can shift from the cell in response to an accumulation of extracellular solutes other than Na+ salts (78, 79, 80, 81, 82). Hyperglycemia causes hyponatremia by this mechanism, and the serum Na+ decreases by 1.6 mEq/L for every 100 mg/dL rise in the serum glucose. Serum osmolality is unchanged.
The body can retain excess water.
The body can fail to retain Na+ (83).
Na+ shifts into the cells.
TABLE 6.4 CAUSES OF HYPONATREMIA | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
consist mostly of triglycerides. This condition is obvious from the milky appearance of the serum. Substantial hyponatremia resulting from hyperlipidemia requires accumulation of more than 5 to 6 g/dL of lipids, and such a degree of hyperlipidemia does not occur with hypercholesterolemia alone.
administered as a 3% NaCl solution. In emergency situations, 100 mL or 2 mL/kg bolus infusions of 3% saline can be administered rapidly over minutes in the setting of seizures and can be repeated up to two times if necessary (109, 116). Very close serial monitoring of serum Na+ is required until blood levels are stable.
patients with a defective thirst mechanism, in those patients with continuous vomiting or who lack access to water, or in those patients with mechanical obstruction resulting from a condition such as an esophageal tumor.
TABLE 6.5 CAUSES OF HYPERNATREMIA | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree