Figure 95-1. Thoracoabdominal aortic aneurysm: comparison of survival rates in untreated patients versus surgically treated patients.
Many TAAAs result from chronic ascending and descending aortic dissections. Familial clustering of aortic dissections is evident because up to 20% of patients with ascending thoracic aortic aneurysms that predispose to aortic dissections have one or more first-degree relatives with the same affliction.27–29 Marfan syndrome, characterized by skeletal, ocular, and cardiovascular abnormalities, is the most common inherited connective tissue disorder related to aortic aneurysm and dissection. Marfan syndrome occurs at a frequency of 1:5 to 10,000 worldwide. Aortic dilatation observed in Marfan patients has been linked to mutations in fibrillin-1 protein, encoded by the FBN1 gene. Other known genetic syndromes that predispose individuals to TAAA and dissection include Loeys–Dietz syndrome, Ehlers–Danlos syndrome, Turner syndrome, and polycystic kidney disease.30–33 In addition, families with multiple members who have thoracic aortic aneurysms and dissections have been reported in the literature.34 In most of these families, the phenotype for TAAA and dissection is inherited in an autosomal dominant manner with marked variability in the age at onset of aortic disease and decreased penetrance.34 Four genes have been identified for familial thoracic aortic aneurysms and dissections that account for 20% of this family condition. Mutations in the smooth muscle isoform of alpha-actin, encoded by ACTA2, are responsible for 15% of familial aortic disease. Other genes responsible for less than 2% include MYH11, TGFBR2, and TGFBR1.35–38
A small percentage of TAAAs are the result of infection (mycotic aneurysms) or trauma (pseudoaneurysms). An infected aneurysm frequently results from bacterial or septic emboli that seed an atherosclerotic aorta. Another mechanism is contiguous spread from empyema or adjacent infected lymph nodes. Although any organism can infect the aortic wall, Salmonella, Haemophilus influenza, Staphylococcus, Mycobacterium tuberculosis, and Treponema pallidum spirochetes species are the most common.39,40 Infected aortic aneurysms are usually saccular and thought to be at greater risk for rupture. Chronic traumatic pseudoaneurysms of the aorta related to previously unrecognized traumatic transection are also prone to rupture, and surgical repair is warranted at the time of diagnosis.
Approximately 25% of TAAAs are associated with chronic aortic dissection. An estimated 20% to 40% of patients will develop aneurysms in the thoracoabdominal aorta within 2 to 5 years following acute aortic dissection.41–43 Persistent patency of the false aortic lumen is reported to be a significant predictor of aneurysm formation.42,44 However, the presence of chronic aortic dissection or patent false lumen has not been linked to a higher risk of aortic rupture.45 Aneurysm disease occurs in more than one part of the aorta in approximately 20% of cases. The so-called “mega” aorta is an “extensive” aortic aneurysm involving the ascending, transverse arch, and the entire thoracoabdominal aorta. Although associated factors include Marfan syndrome and chronic aortic dissection, the cause of extensive aortic aneurysm remains unknown.
CLINICAL MANIFESTATIONS
Aortic aneurysms can cause compressive symptoms, although most do not until they reach a large size. The most frequent complaint is ill-defined chronic back pain, although pain can also occur in the chest, flank, or epigastrium. Acute changes in the characteristics and severity of pain can indicate sudden expansion or impending aortic rupture. Hoarseness, resulting from vocal cord paralysis caused by compression of the left recurrent laryngeal or vagus nerves, is frequently seen in patients with large aneurysms of the proximal descending thoracic aorta. Patients may also experience dyspnea related to compression of the tracheobronchial tree. A large aneurysm can exert pressure on the adjacent esophagus or duodenum, causing dysphagia or weight loss related to obstruction or early satiety. Direct erosion of the aneurysm into the adjacent tracheobronchial tree, esophagus, or both can cause exsanguination, presenting as massive hemoptysis or hematemesis, respectively. Less frequently, direct erosion can cause slow intermittent blood loss. Rarely, paraplegia or paraparesis can occur in patients with TAAA as a result of acute occlusion of the intercostal or spinal arteries. These findings are usually associated with acute aortic dissection, but can also result from thromboembolization. Although most aneurysms have a varying amount of mural thrombus, distal embolization causing acute mesenteric, renal, or lower extremity ischemia is infrequent.
Rupture is thought to be the first clinical manifestation of a TAAA in as many as 10% to 20% of patients (Fig. 95-2). The acute onset of severe chest, abdominal, or back pain associated with hypotension must raise the suspicion of a ruptured aneurysm. A pulsatile mass may be palpable in the abdomen. However, if the larger part of the TAAA is positioned deep within the thoracic cage, the aneurysm may not be apparent on physical examination. Although most ruptured aneurysms are fatal unless treated emergently, the ruptured arterial wall may temporarily seal for several hours or days before free rupture. In patients who are brought to the hospital alive, rupture is usually contained within the pleura or retroperitoneum. Free rupture is accompanied by severe hypotension and patients are more likely to die before reaching the hospital.

Figure 95-2. Computed tomography scan of a ruptured thoracoabdominal aortic aneurysm. Note stranding in the retroperitoneum denoting extravasation of blood.
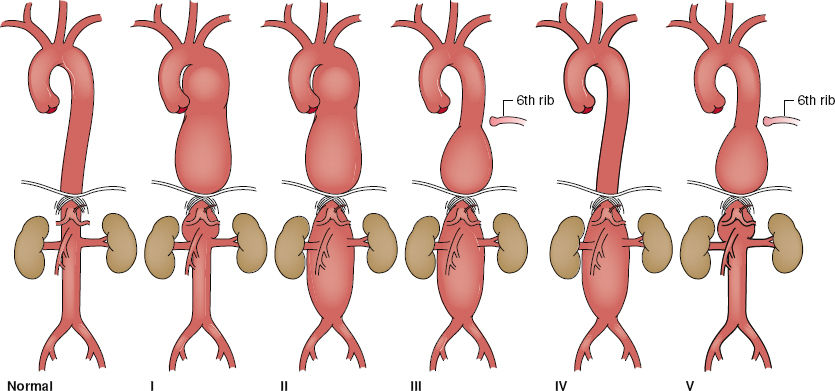
Figure 95-3. Thoracoabdominal aortic aneurysm classification. Extent I, distal to the left subclavian artery to above the renal arteries. Extent II, distal to the left subclavian artery to below the renal arteries. Extent III, from the sixth intercostal space to below the renal arteries. Extent IV, the twelfth intercostal space to the iliac bifurcation (total abdominal aortic aneurysm). Extent V, below the sixth intercostal space to just above the renal arteries.
DIAGNOSTIC IMAGING
The diagnosis of TAAA can be confirmed by various imaging modalities. Currently, computed tomography angiography (CTA) is the imaging modality of choice in defining the extent of TAAA as per the modified Crawford Classification (Fig. 95-3) and for planning operative strategy. The diameter of the entire aorta, from the ascending segment to the bifurcation, can be accurately measured at various levels on axial images assuming the centerline determination is made. The distinction between the false and true lumens in aortic dissection can be shown on CTA. CTA can also detect thrombus or inflammatory changes in the aortic wall. Furthermore, the presence of free (or contained) fluid or blood can indicate free (or contained) rupture. Thin-slice CTA image acquisition can also identify patent intercostal arteries. Coronal reformatting or three-dimensional reconstruction of axial CT images provide additional views of TAAA (Fig. 95-4). These views are often helpful for surgical planning as well as determining adjacent organ involvement. Intravenous iodinated contrast can be omitted in patients with impaired renal function because it is not required for simple sizing of TAAA.
Magnetic resonance angiography (MRA) has become widely available and is frequently used as a screening test to detect diseases of the aorta and its branches. The principal advantage of MRA over CTA is that MRA does not require intravenous iodinated contrast and, thus, can be performed safely in patients with impaired renal function. In addition, MRA avoids the radiation exposure required for CT especially when serial follow-up examinations are required. Although MRA provides better contrast resolution, its spatial resolution is poorer than that of spiral CT. In addition, CT can better demonstrate aortic calcification and intramural thrombus compared with MRA. The time required to acquire images, claustrophobia, internal metallic hardware (e.g., pacemakers or orthopedic implants), and higher cost are other limiting factors of MRA.

Figure 95-4. Somatosensory and motor evoked potentials are recorded at three sites: the popliteal fossa (A, B), C5 (C) and the vertex (D, E).
Transesophageal echocardiography (TEE) can provide excellent imaging of the ascending and descending thoracic aorta. TEE can be performed at the bedside or in the operating room. TEE can be used in patients who are too unstable to be transported to the CT scanner. TEE can show aortic wall disease, differentiating arteriosclerotic plaque from intimal tear due to dissection. In the operating room, TEE can also be used to locate the optimal area for aortic cross-clamping and to assess cardiac function. However, TEE is an invasive modality and requires an experienced operator for optimal visualization and interpretation. Examination of the aorta by TEE is limited to the supradiaphragmatic aorta because the ultrasound probe loses contact with the aorta as it crosses the gastroesophageal junction.
Intravascular ultrasound has become an important diagnostic modality in thoracic aortic disease as it allows very good characterization of the intraluminal relationships, especially in aortic dissection. Although relatively invasive, requiring arterial sheath access, this is becoming an important modality for planning endovascular therapies in aortic dissection. Epiaortic ultrasound has also been applied to TAAA treatment. Epiaortic ultrasound can allow better characterization of the thoracic aorta for cannulation or clamping.
PREOPERATIVE EVALUATION
The initial consultation with the TAAA patient focuses on a thorough history and physical examination, primarily to detect comorbidities, because there are generally few symptoms or physical signs related to the aneurysm itself. The extent of the TAAA is determined from imaging studies. Further evaluation of associated risk factors is performed, and consultation with a cardiologist, pulmonologist, or nephrologist is often necessary to aid in the stratification of risks.
Ischemic heart disease is prevalent in this population and is the most common cause of death in patients with a TAAA. TEE provides an excellent estimate of cardiac function. Coronary artery revascularization for critically stenosed coronary artery disease, using either percutaneous intervention (balloon angioplasty or stent) or surgical bypass, may be indicated prior to TAAA surgery, but the risk of rupture of the aneurysm must be weighed against the risk of coronary intervention and the delay caused by intervention. For patients who must undergo coronary artery bypass prior to TAAA repair, the conduit of choice is the saphenous vein graft. Use of the left internal mammary artery is avoided to prevent potential cardiac ischemia if aortic cross-clamping proximal to the left subclavian artery is required during the TAAA repair. Moreover, the internal mammary artery may be an important collateral blood supply to the spinal cord and chest wall.
Although percutaneous coronary intervention may be considered, the use of either bare metal stents or just balloon angioplasty is preferred over drug-eluting stents since drug-eluting stents require long-term administration of clopidogrel to prevent stent thrombosis. Fortunately, medical management can be adopted for most cases of mild-to-moderate coronary artery disease and in cases of severe disease that is asymptomatic with preserved ejection fraction. Patients who develop symptoms from critical disease, for example, left main or ostial left anterior descending coronary occlusive disease, should probably have this addressed prior to aneurysm repair. In very rare situations, combined and simultaneous coronary artery bypass and TAAA repair may be considered.
OPERATIVE TECHNIQUES
The patient is brought to the operating room and placed in the supine position on the operating table and prepared for surgery. The right radial artery is cannulated for continuous arterial pressure monitoring. General anesthesia is induced. Endotracheal intubation is established using a double-lumen tube for selective one-lung ventilation during surgery. A sheath is inserted in the internal jugular vein, and a pulmonary balloon-tipped catheter is floated into the pulmonary artery for continuous monitoring of the central venous and pulmonary artery pressures. Large-bore central and peripheral venous lines are established for fluid and blood replacement therapy. Temperature probes are placed in the patient’s nasopharynx and bladder. Electrodes are attached to the scalp for electroencephalography and along the spinal cord for both motor- and somatosensory-evoked potential (MEP and SSEP) to assess the central nervous system and spinal cord function, respectively (Fig. 95-5). The patient is then positioned on the right side with the hips and knees flexed to open the intervertebral spaces. A lumbar catheter is placed in the third or fourth lumbar space to provide cerebrospinal fluid (CSF) pressure monitoring and drainage (Fig. 95-6). The CSF pressure is kept at 10 mm Hg or less by gravity drainage of CSF throughout the procedure. The patient is then repositioned in the right lateral decubitus position with the hips slightly turned to allow access to both groins. The operative field is scrubbed using standard aseptic solution and draped.

Figure 95-5. Sagittal computed tomography image of an extent II TAAA.

Figure 95-6. Placement of the lumbar catheter in the third or fourth lumbar space to provide cerebrospinal fluid drainage and pressure monitoring.
The incision is tailored to complement the extent of the aneurysm (Fig. 95-7). The full thoracoabdominal incision begins posteriorly between the tip of the scapula and the spinous process, curving along the sixth intercostal space to the costal cartilage, and then obliquely to the umbilicus. The latissimus dorsi muscle is divided and the insertion of the serratus anterior muscle is mobilized. The left lung is deflated and the left thoracic cavity is entered. Usually, a full thoracoabdominal exploration is necessary for extent II, III, and IV TAAAs. A modified thoracoabdominal incision begins similar to the full thoracoabdominal incision, but ends at the costal cartilage. The modified thoracoabdominal incision provides excellent exposure for surgery involving the descending thoracic aorta, extents I and V TAAA when the aneurysm ends above the renal arteries. A self-retaining retractor is placed firmly on the edges of the incision to maintain full thoracic and abdominal exposure during the procedure.
The dissection begins at the level of the hilum of the lung cephalad to the proximal descending thoracic aorta. The ligamentum arteriosum is identified and transected, taking care to avoid injury to the left recurrent laryngeal nerve. The extent of the distal abdominal aneurysm is assessed. Only the muscular portion of the diaphragm is divided and the left phrenic nerve preserved (Fig. 95-8). A retroperitoneal plane is then developed, mobilizing the spleen, bowel, and left kidney to the right side of the abdominal aorta (medial visceral rotation). To prepare for distal aortic perfusion, the patient is anticoagulated using intravenous heparin (0.5 to 1 mg/kg body weight). The pericardium is opened posterior to the left phrenic nerve to allow direct visualization of the pulmonary veins and left atrium. The left inferior pulmonary vein is cannulated. A centrifugal pump with an inline heat exchanger is attached to the outflow cannula and the arterial inflow is established through the left common femoral artery via a Dacron graft sutured to the common femoral artery in an end-to-side fashion, or the descending thoracic aorta, if the femoral artery is not accessible. The end-to-side fashion prevents ischemia to the left leg that may be associated with renal dysfunction. Distal aortic perfusion is begun (Fig. 95-9).
Padded clamps are applied to the proximal descending thoracic aorta just distal to the left subclavian artery and the midthoracic aorta. When the proximal extent of the aneurysm is too close to the left subclavian artery, the aorta between the left common and left subclavian arteries is clamped. The left subclavian artery is clamped separately. Because of the danger of graft–esophageal fistula, the inclusion technique for the proximal anastomosis is no longer used. Instead, the aorta is transected to separate it from the underlying esophagus (Fig. 95-10A). A woven Dacron graft impregnated with collagen or gelatin for replacement is preferred. The graft is sutured in an end-to-end fashion to the descending thoracic aorta, using a running 2-0 or 3-0 monofilament polypropylene suture. The anastomosis is checked for bleeding. Pledgeted polypropylene sutures for reinforcement are placed, if necessary. Sequential clamping is used for all TAAAs. After completion of the proximal anastomosis, the middescending aortic clamp is moved distally onto the abdominal aorta at the celiac axis to accommodate intercostal reattachment. Reattachment of patent, lower intercostal arteries (T8 to T12) is performed, except in cases of occluded arteries, heavily calcified aorta, or acute aortic dissection, guided by the neuromonitoring, that is, MEPs and SSEPs. If no changes are noted in the neuromonitoring of the spinal cord, then patent intercostal arteries can be ligated without fear of spinal cord injury. If intercostal reattachment is performed, the proximal clamp is released from the aorta and reapplied on the aortic graft beyond the intercostal patch, restoring pulsatile flow to the reattached intercostal arteries (Fig. 95-10B). The distal clamp is moved onto the infrarenal aorta, the abdominal aorta is opened, and the graft is passed through the aortic hiatus. The celiac, superior mesenteric, and renal arteries are identified and perfused using 9- or 12-Fr balloon-tipped catheters, depending on the size of the ostia (Fig. 95-10C). The delivery of cold perfusate (4°C) to the viscera depends on the proximal aortic pressure, which is maintained between 300 and 600 mL/min. Renal temperature is directly monitored and kept at approximately 15°C. The visceral vessels are usually reattached using the inclusion technique. On completion of this anastomosis, the proximal clamp is moved beyond the visceral patch, restoring pulsatile flow to the viscera and kidneys (Fig. 95-10D). The final graft anastomosis is then completed at the aortic bifurcation. In most cases, an island patch accommodates reattachment of the celiac, superior mesenteric, and both renal arteries. If the right or left renal artery is located at too great a distance from other arteries, its reattachment usually requires a separate interposition bypass graft. A visceral patch is no longer used for patients with connective tissue disease (Marfan) and in patients younger than 60 years because of the high incidence of recurrent patch aneurysms in such cases. Instead, a woven Dacron commercially available graft is used with side-arm grafts of 10 mm and 12 mm for separate attachment of the celiac, superior mesenteric, and the left and right renal arteries. Similarly, in these patients, a loop graft has replaced the island patch for reattachment of the intercostal arteries.
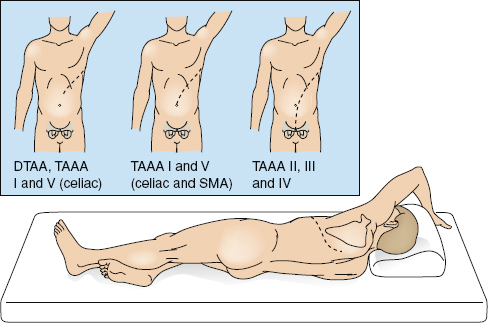
Figure 95-7. Tailoring of thoracoabdominal incisions for aneurysm extent. DTAA, descending thoracic aortic aneurysms; TAAA, thoracoabdominal aortic aneurysm; SMA, superior mesenteric artery.

Figure 95-8. In previous surgical practice, the diaphragm was divided (A); currently, only the muscular portion of the diaphragm is cut (B).

Figure 95-9. Distal aortic perfusion from the left inferior pulmonary vein to the left common femoral artery.

Figure 95-10. Sequential clamping and graft replacement. Padded clamps are placed on the proximal and middistal descending thoracic aorta. A: The proximal part of the aneurysm is opened. The aortic neck is completely transected and separated from the esophagus. The proximal anastomosis is fashioned. Subsequently, the patent lower intercostal arteries are reattached via an elliptical hole in the graft. B: The proximal clamp is then moved onto the graft to restore pulsatile flow to the intercostal arteries, and the graft is pulled through the hiatus into the abdomen. The distal clamp is reapplied onto the infrarenal aorta. The remainder of the aneurysm is opened. Balloon-tipped catheters are inserted into the celiac, superior mesenteric, and renal arteries to permit perfusion. C: An elliptical hole is made in the graft for reimplantation of the visceral and renal arteries.
The patient is weaned from partial bypass once the core body or nasopharyngeal temperature reaches 36°C. Protamine is administered (1 mg/1 mg heparin), and the atrial and femoral cannulae are removed. Once hemostasis is achieved, two or three 36-Fr chest tubes are placed in the pleural cavity for drainage. The diaphragm is reapproximated using running 1-0 polypropylene suture. The left lung is reinflated. Closure of the incision is done in a standard fashion. The patient is placed in the supine position and a single-lumen endotracheal tube is exchanged for the double-lumen tube. If the vocal cords are swollen, the double-lumen tube is kept in place until the swelling resolves. The patient is then transferred to the intensive care unit (ICU). Figure 95-11 shows an extent II TAAA before and after surgery.
Elephant Trunk Technique for Extensive Aortic Aneurysms
Single-stage repair of extensive aneurysms involving the ascending, arch, and thoracoabdominal aorta greatly increases operative risks. The patient undergoes a lengthy procedure that requires multiple incisions, a daunting array of protective surgical adjuncts, protracted clamp times, and considerable blood loss. Staged repair is a practical solution. Prior to the introduction of the elephant trunk technique by Borst46 in 1983, staged repair was fraught with complications, particularly excessive bleeding from the pulmonary artery and thoracic aorta in the second-stage repair of the thoracic or thoracoabdominal aorta. The elephant trunk technique resolves this problem because it allows the surgeon to avoid surgical manipulation and cross-clamping the proximal native descending thoracic aorta in the second stage.
The first stage of the elephant trunk technique is performed in a similar fashion to standard surgery of the ascending aorta and transverse arch, with the exception that either an inverted distal graft or a commercially available collared graft is inserted distally (Fig. 95-12A). The folded edge of the inverted graft or the collared portion of the elephant trunk graft is sutured to the descending thoracic aorta just distal to the left subclavian artery. When the distal anastomosis is completed, the inner portion of the inverted graft is retrieved. A side hole is made in the graft, and the aortic island containing the great vessels is reimplanted. The proximal anastomosis to the ascending aorta is completed, and the distal portion of the graft, or “elephant trunk,” is left dangling in the proximal descending aorta. In the first stage, cardiopulmonary bypass, profound hypothermia, circulatory arrest, and retrograde cerebral perfusion provide protection to the brain and guard against stroke.
The second stage of the elephant trunk technique is much like standard TAAA repair, using the adjuncts distal aortic perfusion and CSF drainage. After initiation of the pump, the distal clamp is applied at the mid-descending thoracic aorta. The proximal third of the descending thoracic aorta is opened without a proximal clamp. The elephant trunk portion of the graft, inserted in the descending thoracic aorta during stage 1, is grasped quickly and clamped (Fig. 95-12B and C). The new graft is sutured to the “elephant trunk” to replace the remaining aneurysm.

Figure 95-11. Example of TAAA extent II repair. (A) Preoperative computed tomography showing proximal part of the aneurysm with chronic dissection. Intraoperative photograph of the thoracoabdominal aortic aneurysm before (B) and after (C) graft replacement. The proximal anastomosis was just distal to the left subclavian artery; the patent lower intercostal arteries were reattached; the celiac, superior mesenteric and right renal arteries were reimplanted together; the left renal artery was reimplanted via an interposition bypass graft; and the distal anastomosis was to the aorta just above the bifurcation. The separate left renal artery graft was necessary because it was located far away from the remaining visceral arteries.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


