Source Control
4 Source control is defined as any physical intervention to remove or eliminate a focus of invasive infection (drainage, debridement, and device removal) and to restore optimal anatomic function.5 In surgical infections, antimicrobial agents are used in conjunction with adequate source control of the initial infection. Adequate source control can be accomplished by either surgical or percutaneous interventional radiologic techniques. A number of factors have been identified that predict failure of source control for intra-abdominal infections (Table 8-4).
Pathogen Identification
5 Pathogen identification is extremely important, in part related to increased prevalence of multidrug-resistant pathogens that are associated with surgical infections. Whenever possible, high-quality specimens should be obtained from source control procedures for Gram stain and culture in order to identify causative pathogens and determine antimicrobial susceptibility of the bacterial isolates identified. This will enable appropriate deescalation of antimicrobial therapy from broad-spectrum therapy to directed potential single antimicrobial agent therapy.
INTRA-ABDOMINAL INFECTIONS
Classification
6 Complicated intra-abdominal infection (cIAI) extends beyond the hollow viscus of origin into the peritoneal space and is associated with either abscess formation or peritonitis. “Uncomplicated” intra-abdominal infection involves intramural inflammation of the gastrointestinal tract and has a substantial probability of progressing to cIAI if not adequately treated.
Table 8-2 Spectrum of Activity of Specific Antimicrobials for Specific Pathogens

Table 8-3 Antimicrobial Therapy, Surviving Sepsis Guidelines 2012

Diagnosis
Patients with cIAI present with abdominal pain and gastrointestinal symptoms. A comprehensive abdominal physical examination must be performed in these patients. If peritonitis is present, evaluation for emergent laparotomy without diagnostic studies is considered. If peritonitis is not present, recent evidence-based guidelines recommend that computed tomography (CT) scan of the abdomen and pelvis is the diagnostic imaging modality of choice to evaluate for cIAI and its source.6,7
Treatment
7 Early empiric appropriate systemic antimicrobial therapy to cover all potential causative pathogens and early source control are the mainstays of treatment of cIAIs. In order to select appropriate empiric antimicrobial agents for use, cIAIs are further classified as “community-acquired” or “healthcare-associated.”
Community-Acquired Complicated Intra-Abdominal Infection
For patients who have “community-acquired” cIAI, determination of whether the disease is “mild/moderate (appendicitis, mild diverticulitis)” or “severe (fecal peritonitis, perforated diverticulitis)” facilitates appropriate antimicrobial treatment (Table 8-5). Patients with “severe” community-acquired cIAI should have antibiotics that cover Pseudomonas and Enterococcus pathogens, and both single-agent and combination-agent regimens are available for use. A number of antimicrobials are no longer recommended for use in community-acquired cIAI:
Table 8-4 Clinical Factors Predicting Failure of “Source Control” for Intra-Abdominal Infection

 Ampicillin/sulbactam is not recommended because of high resistance of Escherichia coli to this agent (B-II)
Ampicillin/sulbactam is not recommended because of high resistance of Escherichia coli to this agent (B-II)
 Cefotetan and clindamycin are not recommended for use because of increasing resistance of the Bacteroides fragilis group to these agents (B-II)
Cefotetan and clindamycin are not recommended for use because of increasing resistance of the Bacteroides fragilis group to these agents (B-II)
 Because of the availability of less toxic agents demonstrated to be of at least equal efficacy, aminoglycosides are not recommended for routine use in community-acquired IAI in adults (B-II).
Because of the availability of less toxic agents demonstrated to be of at least equal efficacy, aminoglycosides are not recommended for routine use in community-acquired IAI in adults (B-II).
Healthcare-Associated Complicated Intra-Abdominal Infection
For patients with “healthcare-associated” cIAI, separate evidence-based guideline recommendations for empiric antimicrobial treatment are provided (Algorithm 8-1). Specific antimicrobials are recommended based on whether the patient and/or institution have high risk for infection with multidrug-resistant pathogens. Quinolone-resistant E. coli have become common in many communities, and quinolones should not be used unless hospital antibiograms confirm greater than 90% susceptibility of E. coli to quinolones. In patients with healthcare-associated cIAI, carbapenems are an excellent single-agent regimen for empiric treatment.
Table 8-5 Empiric Antimicrobial Treatment of Extrabiliary cIAIs, Community Acquired

Duration of Antibiotics for Complicated Intra-Abdominal Infection
8 Duration of antibiotics for treatment of cIAI has long been controversial. A recent important randomized controlled trial examined the efficacy of short-course (4 days) antimicrobial therapy in patients with intra-abdominal infections who had undergone an adequate source control procedure. Fixed-duration antibiotic therapy for 4 days was noninferior to longer duration antmicrobial therapy until resolution of physiologic abnormalities, with no difference in the composite endpoints of SSI, recurrent intra-abdominal infection, or death.8
SPECIFIC INTRA-ABDOMINAL INFECTIONS
Appendicitis
Appendicitis is common with over 300,000 hospital discharges in the United States per year. Although appendectomy provides definitive source control for the treatment of acute nonperforated appendicitis, intravenous (IV) antibiotics should be administered preoperatively with a dose repeated intraoperatively if the surgery is prolonged, a number of controversies in diagnosis and management of appendicitis persist.9
Antibiotics versus Appendectomy
In patients with uncomplicated appendicitis (nonperforated, no abscess, or phlegmon), appendectomy is definitive source control. But a number of recent randomized trials have confirmed that acute nonperforated appendicitis can be treated successfully with antibiotics alone. A meta-analysis of 6 studies with 1,201 patients reported that in patients treated with antibiotics alone, 7 ± 4% failed to respond and required appendectomy, and acute appendicitis recurred in 14 ± 11%. A normal appendix was found in 7.3 ± 5.1% of patients at appendectomy. Complications were considerably less likely to occur with antibiotic treatment than with appendectomy. Major surgical complications included enterocutaneous fistula and reoperation.10

Algorithm 8-1. Empiric antimicrobial treatment of extrabiliary cIAIs, community acquired versus healthcare associated. (Adapted from Solomkin JS, Mazuski JE, Bradley JS, et al. Diagnosis and management of complicated intra-abdominal infection in adults and children: guidelines by the Surgical Infection Society and the Infectious Diseases Society of America. Surg Infect (Larchmt) 2010;11(1):79–109.)
A Cochrane meta-analysis of 5 trials with 901 patients reported that 73.4% of patients who were treated with antibiotics and 97.4% patients underwent appendectomy were cured within 2 weeks without major complications (including recurrence) within 1 year.11 Another meta-analysis included only four trials and reported that efficacy was significantly higher for surgery but rates of perforated appendicitis were not different, and complication rates were significantly higher for surgery.12
The most recent trial (APPAC) enrolled 530 patients in Finland with uncomplicated appendicitis confirmed by CT scan and randomized to early appendectomy or antibiotic treatment (ertapenem for 3 days, then oral levofloxacin and metronidazole for 7 days) with 1-year follow-up. In the antibiotic group, 70 patients (27.3%) underwent appendectomy within 1 year of initial presentation for appendicitis. Antibiotic treatment did not meet the prespecified criterion for noninferiority compared with appendectomy. But it was notable that 72.7% of patients with uncomplicated appendicitis treated with antibiotics alone did not require subsequent appendectomy.13,14
The trials published to date were primarily from European countries, used antibiotics not available in the United States and had minimal use of laparoscopic appendectomy or CT scan for diagnostic imaging.15 Further high-quality randomized trials are therefore needed to determine which patients are most likely to benefit from antibiotic therapy alone. Although appendectomy remains the standard treatment for acute appendicitis, antibiotic treatment alone may be used as an alternative treatment in patients with contraindication to surgery (or where surgery is high risk).
Laparoscopic versus Open Appendectomy
Appendectomy can be performed by laparoscopic or open approach. A large retrospective review (2005–2008) of laparoscopic versus open appendectomy using the American College of Surgeons National Surgical Quality Improvement Program (NSQIP) reported that 76.4% were performed laparoscopically and 23.6% performed open. Open appendectomy was associated with increased morbidity, SSI, and mortality.16
A Cochrane review of 67 trials was updated in 2010 and reported that laparoscopic appendectomy was associated with lower SSI rates but higher intra-abdominal abscess rate. Laparoscopic approach was associated with shorter hospital length of stay, faster return to normal activity, and decreased postoperative pain. This review concluded that laparoscopic appendectomy should be considered the preferred approach where surgical expertise and equipment are available.17
Time to Appendectomy and Perforation Risk
It has long been thought that appendiceal luminal obstruction leads to appendiceal perforation without timely surgical intervention. This belief led to attempts to decrease time from presentation and diagnosis of acute appendicitis to operating room start time. With the advent of early appropriate empiric systemic antimicrobial therapy, this fundamental concept is no longer true. As discussed previously, in patients with uncomplicated appendicitis, antibiotic treatment alone does not lead to appendiceal perforation. Furthermore, using data from the Washington State Surgical Care and Outcomes Assessment Program (SCOAP) with 9,048 adult patients who underwent appendectomy, there was no association between appendiceal perforation and in-hospital time prior to surgery. This study confirmed that appendiceal perforation is most commonly a prehospital occurrence, and timing of surgery should be determined based on all factors relevant to the surgeon and patient.18
Abscess or Phlegmon Management
In patients with periappendiceal abscess or phlegmon, current evidence (mainly retrospective studies) supports nonoperative management with IV transitioned to oral antibiotics and percutaneous drainage if possible. A meta-analysis of 19 retrospective studies from 1969 to 2005 reported that nonsurgical treatment failed in only 7.6% of patients and immediate appendectomy was associated with higher morbidity. Similar findings were confirmed in a more recent review.19 Routine interval appendectomy is not recommended since the risk of recurrence is low (7.4%). But either colonoscopy or repeat CT imaging is recommended due to the risk of a malignant disease (1.2%) or important benign disease (0.7%) identified during follow-up.20
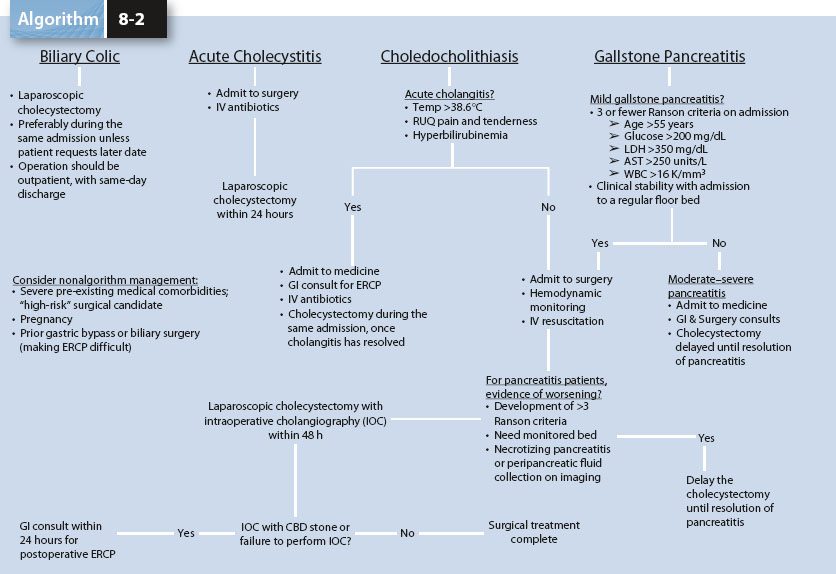
Algorithm 8-2. Biliary infections and algorithm for diagnosis and management. (From Demehri FR, Alam HB. Evidence-based management of common gallstone-related emergencies. J Intensive Care Med 2016;31(1):3–13.)
Interval Appendectomy after Initial Nonoperative Management
A large retrospective cohort study reported that 3% of patients were initially treated with nonoperative management. Of these, 15% had an interval appendectomy and the remaining 85% did not. Of the 864 patients that did not undergo interval appendectomy only 39 patients (5%) had recurrence of appendicitis after a median follow-up of 4 years.21 Routine interval appendectomy after initial successful nonoperative treatment is therefore not justified and should be abandoned. In a meta-analysis including 2,771 patients initially treated nonoperatively for appendiceal phlegmon or abscess, 31 patients were found to have a malignant diagnosis and a lower rate of inflammatory bowel disease. These data highlight the need for follow-up with either CT scan or colonoscopy after successful nonoperative management.20 Although a retrospective study in pediatric appendicitis reported a 2.8-fold higher rate of recurrent appendicitis in patients with an appendicolith, this study is limited by small sample size (n = 96) and a higher overall rate of recurrent appendicitis than all other published studies.22 See also chapter on Appendiceal Diseases.
Biliary Infections
Biliary infections include acute cholecystitis which may be calculous or acalculous, may include choledocholithiasis or not, and cholangitis.23 Acute cholecystitis and ascending cholangitis are primary inflammations of the gallbladder and bile ducts, respectively, which are caused by infection and possible biliary obstruction.
Acute Cholecystitis
Acute cholecystitis is inflammation of the gallbladder resulting from obstruction of the cystic duct and subsequent bacterial invasion and overgrowth. Cholelithiasis is the cause of cystic duct obstruction in more than 90% of cases of acute cholecystitis in the United States. Acalculous cholecystitis is more common in critically ill patients and the pathophysiology is transmural ischemia. Acalculous cholecystitis carries higher morbidity and mortality rates than calculous cholecystitis.
The diagnosis of acute cholecystitis is made based on the common constellation of right upper quadrant tenderness, leukocytosis, and fever. Initial diagnostic imaging is right upper quadrant ultrasound to evaluate for gallstones, gallbladder wall thickening, and pericholecystic inflammation (sensitivity 88% to 94%, specificity 78% to 80%). If ultrasound is inconclusive or is discordant with the clinical evaluation, then cholescintigraphy (99mTc-hepatobiliary iminodiacetic acid, HIDA scan) is recommended and nonvisualization of the gallbladder confirms acute cholecystitis with sensitivity of 96% and specificity of 90% (Algorithm 8-2).24
Early systemic antimicrobial treatment is indicated for acute cholecystitis to cover the causative pathogens, including aerobic, enteric, gram-negative bacilli (i.e., E. coli, Klebsiella, Enterobacter, and Proteus), and aerobic gram-positive organisms (i.e., Enterococcus and Streptococcus). Anaerobes are uncommon, identified in approximately 15% of isolates. Clostridial organisms can be identified in cases of emphysematous cholecystitis confirmed by identification of gas in the gallbladder wall.
Cholangitis
Cholangitis signs/symptoms include jaundice, fever, right upper quadrant tenderness, hyperbilirubinemia, and leukocytosis. Ultrasound confirms common bile duct dilation (>7 mm).
Appropriate evidence-based recommendations for diagnosis and treatment from the Tokyo Guidelines are provided in Tables 8-6 and 8-7. For treatment recommendations, urgent biliary drainage (<24 hours) is indicated when (1) obstructive biliary stones are associated with severe or moderate acute cholangitis OR (2) mild acute cholangitis is not responding to IV antibiotics and fluid resuscitation.
For patients with biliary infections, specific evidence-based guideline recommendations for antimicrobial treatment are classified into four patient populations (Table 8-8). For patients undergoing cholecystectomy for acute cholecystitis with complete source control (i.e., complete cholecystectomy), antimicrobial therapy should be discontinued within 24 hours of the operation unless there is evidence of infection outside the wall of the gallbladder. See also chapter on Calculous Biliary Disease.
Table 8-6 Diagnosis of Cholangitis: Tokyo Guidelines 2013

Table 8-7 Treatment of Acute Cholangitis by Severity Classification: Tokyo Guidelines 2013 Criteria

Diverticulitis
Diverticulitis is a common intra-abdominal infection. Its pathophysiology is associated with altered gut motility, increased luminal pressure, and an altered colonic microenvironment.
Uncomplicated diverticulitis is treated with systemic antibiotics and usually resolves. Antimicrobial treatment of diverticulitis is the same as for cIAI which is reviewed above. In a cohort study of 2,366 patients hospitalized in the Kaiser Permanente system and followed for 8.9 years, only 13.3% had a first recurrence and 3.9% had a second recurrence.25
Surgery for acute diverticulitis is indicated for patients who present with peritonitis and/or sepsis or who do not improve with medical management and/or percutaneous drainage of associated abscess/infection. Although less than 25% of patients will develop generalized peritonitis after colonic perforation, it is severe with high mortality. Surgical options include simple colostomy formation in the setting of profound inflammation (rarely performed), traditional sigmoid resection with colostomy (Hartmann procedure), and sigmoid resection with a primary colocolonic or colorectal anastomosis with or without a diverting loop ileostomy. Laparoscopic surgery is preferred to open surgery when possible.26
Table 8-8 Antimicrobials for Treatment of Biliary Infections


Figure 8-1. Hinchey classification scheme. Patients with stage 1 disease have small, confined, pericolic, or mesenteric abscesses, whereas those with stage 2 disease have larger abscesses, often confined to the pelvis. Stage 3 disease, or perforated diverticulitis, is present when a peridiverticular abscess has ruptured and caused purulent peritonitis. Rupture of an uninflamed and unobstructed diverticulum into the free peritoneal cavity with fetal contamination, the so-called free rupture, signifies stage 4 disease and carries the highest risk of an adverse outcome.
Since recurrence after recovery from an uncomplicated episodes of diverticulitis is rare and two or more recurrences are not associated with increased risk of complications, elective colectomy following two episodes of diverticulitis is no longer recommended. Decisions regarding surgical intervention and colectomy must therefore be based on individual potential risks and benefits of colon resection.27
For patients who require surgical intervention, the specific treatment that is associated with the best outcome is controversial. Hinchey proposed a practical clinical classification of diverticulitis based on the severity of peritoneal contamination (Fig. 8-1) identified at operation.28 The modified Hinchey classification includes Hinchey Ia (confined pericolic inflammation–phlegmon) and Ib (confined pericolic abscess) and considers classification based on radiologic imaging with CT scan as well. Increasing Hinchey classification is associated with increased mortality. The mortality for patients with Hinchey III is reported as 6% but increases greatly to 35% for fecal peritonitis (Hinchey IV). A systematic review comparing surgical treatments for Hinchey III or IV colonic diverticulitis confirmed that primary resection with anastomosis has a significant advantage with lower mortality compared to Hartmann procedure. Furthermore, laparoscopic peritoneal lavage with subsequent surgical resection if indicated had lower surgical morbidity and hospital length of stay compared to the primary resection and anastomosis group.29 See also chapter on Diverticular Disease.

Figure 8-2. Treatment of initial episode of Clostridium difficile infection.
Clostridium Difficile Colitis
The incidence and severity of Clostridium difficile infection (CDI) has increased, particularly in surgical patients who have been exposed to prior antimicrobial therapy. There has been a substantial change in the management of CDI related to the emergence of the NAP1/BI/027 strain which is associated with higher toxin production and more severe forms of disease. CDI may be associated with mild to moderate diarrhea or colonic ileus and obstipation to severe colitis.30
Early diagnosis of CDI is imperative. Recommended testing includes two steps, using a glutamine dehydrogenase assay for the Clostridium difficile antigen, followed by enzyme immunoassay toxin testing. Polymerase chain reaction (PCR) confirmatory testing is used if the antigen is positive and toxin is negative.31 Radiologic testing is helpful, and CT may show colonic mucosal edema (thumbprinting or target sign), ascites, inflammation, and fat stranding and the “accordion sign” which indicates alternating edematous colonic haustra separated by transverse mucosal ridges filled with enteral contrast.
Initial management of CDI is cessation of all antibiotics if possible. The 2010 SHEA/IDSA guidelines have evidence-based recommendations for CDI based on severity of disease (Fig. 8-2).32 Vancomycin and metronidazole are first-line therapy. Vancomycin is preferred for severe or complicated disease. Fidaxomicin (a macrocyclic antibiotic with a narrow antimicrobial spectrum against C. difficile, staphylococci, and enterococci) may be considered for recurrent CDI or where risk of recurrence is high. A multicenter multinational randomized trial confirmed decreased recurrence rates compared to oral vancomycin and it is the U.S. Food and Drug Administration (FDA) approved with a recommended treatment dose of 100 mg po bid.33 Fecal microbiota transplantation is associated with symptom resolution and may be effective in recurrent CDI, but has no role in primary CDI treatment.
9 CDI may warrant surgical intervention. Surgical consultation should occur very early in the course of disease in patients with severe and complicated CDI. The indications for surgical management of CDI patients are not clearly defined, but severe disease, worsening clinical condition despite appropriate treatment, and peritonitis or shock states are all potential indications. The two surgical procedures indicated for CDI treatment include (1) total abdominal colectomy (for peritonitis, colonic perforation, ischemia, necrosis, toxic megacolon with impending perforation, septic shock with organ failure) and (2) diverting loop ileostomy and intraoperative colonic lavage for toxin reduction while preserving the colon (Fig. 8-3). This new treatment strategy resulted in reduced mortality compared with the historical population who had undergone total abdominal colectomy (19% vs. 50%), and preservation of the colon was achieved in 93% of patients.34 No randomized studies have been published to date comparing these two options.
Pancreatitis
A new classification of acute pancreatitis has been implemented and is based on local disease (whether pancreatic necrosis is present or not, whether it is sterile or infected) and systemic determinants (whether organ failure is present or not, whether it is transient or persistent) of severity (Table 8-9). Early management requires goal-directed fluid resuscitation (with avoidance of over-resuscitation and abdominal compartment syndrome), assessment of severity of pancreatitis, diagnostic CT imaging to assess for necrotizing pancreatitis, consideration of endoscopic retrograde cholangiopancreatography (ERCP) for biliary pancreatitis, and early enteral nutrition support. Antibiotic prophylaxis for severe acute or necrotizing pancreatitis is not recommended.35

Figure 8-3. Diverting loop ileostomy with antegrade colonic irrigation for CDI. Before surgery is started:
1. Place the patient in lithotomy position with easy access to rectum
2. Place a fluid collection bag (commonly used by urology) under the patient’s rectum for drainage collection
3. Place a rectal drainage tube (large Malecot catheter, large foley catheter, #9 or #10 endotracheal tube, or other large catheter)
4. Have 8 L of warmed polyethylene glycol 3350/electrolyte solution (GoLytely; Braintree Laboratories) available in OR for intraoperative colonic irrigation, and dulcolax suppository
Intraoperatively:
1. Ensure that the colon is viable and without perforation
2. Create a loop ileostomy (laparoscopic or open)
3. Place a 24-French Malecot catheter into the efferent limb of the ileostomy and advance into the right colon through the ileocecal valve. Secure to skin with sutures.
4. Infuse 8 L of warmed polyethylene glycol 3350/electrolyte solution (GoLytely; Braintree Laboratories) into the 24-French Malecot catheter via the efferent limb of the ileostomy
5. Once colonic lavage completed, perform rectal examination to empty rectum, place dulcolax suppository in rectum to encourage colonic emptying
6. Instill antegrade colonic enema with vancomycin (500 mg in 500 mL) via ileostomy efferent limb, continue postoperatively
(Adapted from Neal MD, Alverdy JC, Hall DE, et al. Diverting loop ileostomy and colonic lavage. An alternative to total abdominal colectomy for the treatment of severe, complicated clostridium difficile-associated disease. Ann Surg 2011;254(3):423–429.)
Table 8-9 New International Classification of Acute Pancreatitis: Determinants of Severity of Acute Pancreatitis by the 2011 World Congress of the International Association of Pancreatology

Necrotizing pancreatitis is high risk for progression to necrotizing infected pancreatitis. Therapeutic antibiotics are required for treatment of documented infected pancreatic necrosis. Causative pathogens include gram-negative microbes and an increase in gram-positive organisms has been reported.36 Therefore broad-spectrum IV antimicrobials are recommended.
10 The initial treatment of infected pancreatic necrosis is percutaneous catheter or endoscopic (transgastric/transduodenal) drainage with a second drain placement as required. The “step-up” approach consisted of percutaneous drainage followed, if necessary, by minimally invasive retroperitoneal necrosectomy. The minimally invasive step-up approach, as compared with open necrosectomy, reduced the rate of the primary composite endpoint (12% vs. 50%, including major complications [new-onset multiple organ failure or multiple systemic complications, perforation of a visceral organ or enterocutaneous fistula, or bleeding] or death), but the mortality rate did not differ significantly between groups (19% vs. 16%).37,38
11 Lack of clinical improvement after these initial procedures warrants consideration of minimally invasive techniques for pancreatic necrosectomy including video-assisted retroperitoneal debridement (VARD), minimally invasive retroperitoneal pancreatectomy (MIRP), or transluminal direct endoscopic necrosectomy (DEN).39 VARD and MIRP use the tract created by percutaneous drainage catheters as a guide for placement of a laparoscope into the retroperitoneum so that debridement and lavage can be performed under direct visualization. Open necrosectomy is associated with substantial morbidity, but to date no randomized trial has documented superiority of either minimally invasive or open surgical technique. Additional trials are underway to address this (Algorithm 8-3). See also chapter on Acute Pancreatitis.
SKIN AND SOFT TISSUE INFECTIONS
Skin and soft tissue infections (SSTIs) span a broad spectrum of clinical entities from limited cellulitis to rapidly progressive necrotizing fasciitis, which may be associated with septic shock or toxic shock syndrome and associated organ failure.40,41 These SSTIs may result in critical illness and require management in the intensive care unit (ICU).42,43 Methicillin-resistant Staphylococcus aureus (MRSA) has emerged as the most common identifiable cause of severe SSTIs, therefore initiation of empiric anti-MRSA antimicrobials is warranted in all cases of severe SSTIs. In addition, appropriate critical care management, including fluid resuscitation, organ support, and nutritional support are necessary components of treatment of severe SSTIs.
The complex interplay of environment, host, and pathogen is important to consider when evaluating SSTIs and planning therapy. The key to a successful outcome in caring for patients with severe SSTIs is (1) early diagnosis and differentiation of necrotizing versus nonnecrotizing SSTI, (2) early initiation of appropriate empiric broad-spectrum antimicrobial therapy with consideration of risk factors for specific pathogens, (3) “source control,” that is, early aggressive surgical intervention for drainage of abscesses and debridement of necrotizing soft tissue infections (NSTIs), and (4) pathogen identification and appropriate deescalation of antimicrobial therapy.

Algorithm 8-3. Step-up approach to management of necrotizing infected pancreatitis. (Adapted from Besselink MG, van Santvoort HC, Nieuwenhuijs, et al.; Dutch Acute Pancreatitis Study Group. Minimally invasive ‘step-up approach’ versus maximal necrosectomy in patients with acute necrotizing pancreatitis (PANTER trial): design and rationale of a randomized controlled multicenter trial [ISRCTN13975868]. BMC Surg 2006;6:6.)
Table 8-10 Comparison of Old and New Classification of SSTIs by FDA

Classification of Skin and Soft Tissue Infections
The FDA previously classified SSTIs into two broad categories for the purpose of clinical trials evaluating new antimicrobials for the treatment of SSTIs: uncomplicated and complicated (Table 8-10). Uncomplicated SSTIs include superficial infections such as cellulitis, simple abscesses, impetigo, and furuncles. These infections can be treated by antibiotics and/or surgical incision for drainage of abscess alone. In contrast, complicated SSTIs include deep soft tissue infections that require significant surgical intervention, such as infected ulcers, infected burns, and major abscesses, and these patients also have significant underlying comorbidities, that is, disease states which complicate (and usually delay) response to treatment. Complicated SSTIs are a significant clinical problem, in part related to the increasing resistance of infecting bacteria to current antibiotic therapies.
Uncomplicated Skin and Soft Tissue Infections
Uncomplicated SSTIs are associated with low risk for life- or limb-threatening infection. These patients can be treated with empiric antibiotic therapy according to likely pathogen and local resistance patterns.
Complicated Skin and Soft Tissue Infections
Complicated SSTIs are associated with high risk for life- or limb-threatening infection. In these patients, it is of paramount importance to initiate appropriate and adequate broad-spectrum initial empiric antimicrobial therapy with coverage for MRSA and to consider the need for surgical intervention for abscess drainage or debridement.
Patients with complicated SSTIs require hospitalization for treatment. Specific circumstances that warrant hospitalization include the presence of tissue necrosis, sepsis, severe pain, altered mental status, immunocompromised state, and organ failure (respiratory, renal, hepatic). SSTIs can lead to serious potentially life-threatening local and systemic complications. The infections can progress rapidly and early recognition and proper medical and surgical management is cornerstone of therapy.
12 In October 2013, the FDA changed the SSTI terminology and issued final guidance for the treatment of acute bacterial skin and skin structure infections (ABSSSI).44 This guidance defined ABSSSI as cellulitis, erysipelas, wound infection, and major cutaneous abscess. An ABSSSI is defined as a bacterial infection of the skin with a lesion size area of at least 75 cm2 (lesion size measured by the area of redness, edema, or induration). The minimum area of involvement of 75 cm2 is chosen to select patients with acute bacterial skin infections for which a reliable control drug treatment effect can be estimated for the conduct of new antimicrobial treatment trials. While the FDA generally requires two phase III trials to support approval of drugs to treat ABSSSI, this guidance stated that a single phase III study that is supported by additional independent evidence may suffice.
Patients with the following infection types can be enrolled in ABSSSI clinical trials:
 Cellulitis/erysipelas: A diffuse skin infection characterized by spreading areas of redness, edema, and/or induration
Cellulitis/erysipelas: A diffuse skin infection characterized by spreading areas of redness, edema, and/or induration
 Wound infection: An infection characterized by purulent drainage from a wound with surrounding redness, edema, and/or induration
Wound infection: An infection characterized by purulent drainage from a wound with surrounding redness, edema, and/or induration
 Major cutaneous abscess: An infection characterized by a collection of pus within the dermis or deeper that is accompanied by redness, edema, and/or induration
Major cutaneous abscess: An infection characterized by a collection of pus within the dermis or deeper that is accompanied by redness, edema, and/or induration
Unfortunately, this new guidance does not address less serious skin infections, such as impetigo and minor cutaneous abscess, or more serious infections needing more complex treatment regimens, such as infections resulting from animal or human bites, NSTIs, diabetic foot infection, decubitus ulcer infection, myonecrosis, osteomyelitis, and ecthyma gangrenosum.
Early Diagnosis and Differentiation of Necrotizing versus Nonnecrotizing SSTI
Another classification for SSTIs that is commonly used is the differentiation of NSTIs from nonnecrotizing infections. This differentiation is critical since necrotizing infections warrant prompt aggressive surgical debridement. Clinical clues to the diagnosis of NSTIs are listed in Table 8-3. The differentiation of necrotizing infections from nonnecrotizing infections is critical to achieving adequate surgical therapy.45 A clear approach to these infections must allow rapid identification and treatment of NSTIs because they are limb threatening and life threatening.
When clinical “hard clinical signs” (bullae, crepitus, gas on x-ray, hypotension with SBP <90 mm Hg, or skin necrosis) of NSTI are present, establishing the diagnosis of NSTI is not difficult. However, hard signs of NSTIs are often absent on presentation, thus potentially delaying diagnosis and surgical intervention. Studies have documented that less than 50% of patients with a definitive diagnosis of NSTI presented with “hard clinical signs” of NSTI.46 Admission of white blood cell count >15,400 × 109/L and/or serum sodium <135 mEq/L was documented to help differentiate NSTI from non-NSTI and aided in early diagnosis.47,48 The Laboratory Risk Indicator for Necrotizing Fasciitis (LRINEC) score is also helpful as a laboratory aid in distinguishing necrotizing from nonnecrotizing SSTIs (see NSTI section below). In retrospective studies CT scan has been shown to have a negative predictive value of 100% and a positive predictive value of 76% in diagnosing NSTI.49
If there is any question regarding the possible diagnosis of an NSTI, it is imperative to proceed with surgical intervention and to be certain that the surgical incision is continued down to the fascial and muscle level to make a definitive diagnosis.
Early Initiation of Appropriate Empiric Broad-Spectrum Antimicrobial Therapy with Anti-MRSA Coverage and Consideration of Risk Factors for Specific Pathogens
Antimicrobial therapy is an essential element in the management of severe SSTIs. As in all serious life-threatening infections, it is important to initiate early and appropriate empiric antimicrobial therapy. It is well established that prompt appropriate treatment of hospitalized infections reduces mortality.2 Similar findings were reported in studies of patients with ventilator-associated pneumonia 50 and sepsis.51 A study of ICU patients found that the higher mortality rate associated with inappropriate initial therapy is still observed when antibiotics are switched from an inappropriate to an appropriate treatment.52
Furthermore, appropriate and timely antibiotic therapy improves treatment outcomes for SSTIs caused by MRSA.53 In a study of 492 patients with community-onset MRSA SSTIs, 95% of episodes treated with an active antibiotic within 48 hours were treated successfully, compared with an 87% rate of successful treatment in patients who did not receive an active antibiotic. In logistic regression analysis, failure to initiate active antimicrobial therapy within 48 hours of presentation was the only independent predictor of treatment failure. Similarly, in a study of patients admitted to the hospital with MRSA sterile-site infection, multivariate analysis found inappropriate antimicrobial treatment to be an independent risk factor for hospital mortality.54
An empiric treatment algorithm for SSTI directed against community-associated MRSA (CA-MRSA) in the emergency department that promotes both the use of antibiotics likely active against CA-MRSA and early incision and drainage of abscesses was examined. Clinical failure occurred in only 3% of cases treated according to the algorithm, compared with 62% of those not treated according to the algorithm. Furthermore, among cases that underwent immediate incision and drainage, initial treatment with antibiotics active in vitro against the MRSA isolate was associated with a decreased clinical failure rate when compared to those treated with inactive antibiotics (0% vs. 67%).55
Empiric antibiotic therapy should be initiated in all patients with SSTIs. IV broad-spectrum antimicrobial therapy should be initiated when an infection is severe or progresses rapidly, when there are signs of systemic illness, when the patient has comorbidities or is immunosuppressed, for very old or young patients, when an abscess cannot be completely drained, and when the infection does not respond to incision and drainage.56
Timely initiation of antimicrobial therapy is also important in the treatment of severe SSTIs, particularly if associated with septic shock. In a study of 2,731 adult patients with septic shock, a strong relationship between the delay in effective antimicrobial initiation and in-hospital mortality was noted.4 Administration of an antimicrobial effective for isolated or suspected pathogens within the first hour of documented hypotension was associated with a survival rate of 79.9%. Each hour of delay in antimicrobial administration over the ensuing 6 hours was associated with an average decrease in survival of 7.6%. By the second hour after onset of persistent/recurrent hypotension, in-hospital mortality rate was significantly increased relative to receiving therapy within the first hour. In multivariate analysis (including Acute Physiology and Chronic Health Evaluation II score and therapeutic variables), time to initiation of effective antimicrobial therapy was the single strongest predictor of outcome. Interestingly, only 50% of septic shock patients received effective antimicrobial therapy within 6 hours of documented hypotension.
Necrotizing Soft Tissue Infections
NSTIs are aggressive soft tissue infections that cause widespread necrosis, and can include necrotizing cellulitis, fasciitis, and myositis/myonecrosis.57,58 Establishing the diagnosis of NSTI can be the main challenge in treating patients with NSTI, and knowledge of all available tools is key for early and accurate diagnosis.59 There have been a number of recent advances in the definition, pathogenesis, diagnostic criteria, and treatment of NSTIs.60,61
Patients with NSTIs require prompt aggressive surgical debridement, appropriate IV antibiotics, and intensive support. Despite aggressive treatment, their mortality and morbidity rates remain high, with some series reporting mortality rates of 25% to 35%.62 A high index of suspicion should be used in conjunction with laboratory and imaging studies to establish the diagnosis as rapidly as possible. Successful treatment requires early, aggressive surgical debridement of all necrotic tissue, appropriate broad-spectrum systemic antibiotic therapy, and supportive care (fluid resuscitation, organ and critical care support) to maintain oxygenation and tissue perfusion. Delayed definitive debridement remains the single most important risk factor for death. Early operative debridement is the major determinant of outcome in NSTIs.
A recent single-institution series of 166 patients documented that the overall mortality rate was 17% and limb loss occurred in 26% of patients with extremity involvement.63 Independent predictors of mortality included white blood cell count greater than 30,000 × 103/μL, creatinine level greater than 2 mg/dL, and heart disease at hospital admission. Independent predictors of limb loss included heart disease and shock (systolic blood pressure <90 mm Hg) at hospital admission. Clostridial infection was an independent predictor for both limb loss and mortality and was highly associated with IV drug use and a high rate of leukocytosis on hospital admission.
A 30-day postoperative mortality risk calculator for NSTI was developed using the NSQIP which identified seven independent variables that correlated with mortality: age older than 60 years, functional status, requiring dialysis, American Society of Anesthesiologists (ASA) class 4 or higher, emergent surgery, septic shock, and low platelet count. The receiver operating characteristic area was 0.85, which indicated a strong predictive model that can aid physicians in the decision-making process.64
13 Early operative debridement is the major determinant of outcome in NSTIs. However, early recognition of NSTIs is difficult clinically. A novel diagnostic scoring system for distinguishing NSTIs from other severe soft tissue infections based on laboratory tests routinely performed for the evaluation of severe SSTIs is called the LRINEC score (Table 8-11).65 The LRINEC score was initially developed in a retrospective observational study including 145 patients with necrotizing fasciitis and 309 patients with severe cellulitis or abscesses admitted to the 2 tertiary care hospitals. The cutoff value for the LRINEC score was 6 points with a positive predictive value of 92% and negative predictive value of 96%. The LRINEC score is a robust score capable of detecting clinically early cases of necrotizing fasciitis. The variables used are routinely measured to assess severe soft tissue infections. Patients with a LRINEC score of ≥6 should be carefully evaluated for the presence of necrotizing fasciitis.
Table 8-11 The Laboratory Risk Indicator for Necrotizing Fasciitis (LRINEC) Score

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


