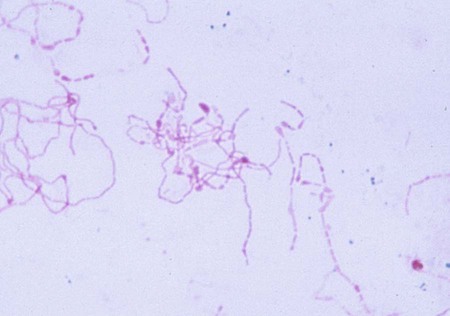
1. Describe the natural habitats of Streptobacillus moniliformis and Spirillum minus. 2. List the two ways in which S. moniliformis is transmitted to humans. 3. Define Haverhill fever, rat-bite fever, and sodoku. 4. List the symptoms of rat-bite fever. 5. Describe the optimal conditions for culturing S. moniliformis, including media, supplements, atmospheric conditions, and length of incubation. 6. Describe the different appearances of S. moniliformis colonial morphology when grown on various media. 7. Describe how S. minus is detected in the laboratory. 8. Compare and contrast the microscopic appearance of S. minus and S. moniliformis in Gram-stained or other smears. Unfortunately, the diagnosis of rat-bite fever caused by S. moniliformis is often delayed because of lack of exposure history, an atypical clinical presentation, and the unusual microbiologic characteristics of the organism. Organisms may be cultured from blood or aspirates from infected joints, lymph nodes, or lesions. No special requirements have been established for the collection, transport, and processing of these specimens except for blood. Because recovering S. moniliformis from blood cultures is impeded by concentrations of sodium polyanethol sulfonate (SPS) used in blood culture bottles, an alternative to most commercially available bottles must be used. After collection by routine procedures (described in Chapter 68), blood and joint fluids are mixed with equal volumes of 2.5% citrate to prevent clotting and are then inoculated to brain-heart infusion cysteine broth supplemented with heated horse serum and yeast extract, commercially available fastidious anaerobe broth without SPS, or thiol broth. Pus or exudates should be smeared, stained with Gram or Giemsa stain, and examined microscopically (Figure 39-1). S. moniliformis is a pleomorphic, gram-negative rod. Cells may appear straight of variable size or as long tangled chains and filaments with bulbar swellings. The cells may also appear spiral shaped and resemble a string of pearls. Direct detection of the 16sRNA gene sequence for S. moniliformis using polymerase chain reaction (PCF) analysis has been described. As previously stated, Gram-stained organisms from standard colonies show extreme pleomorphism, with long, looped, filamentous forms, chains, and swollen cells. The club-shaped cells can be 2 to 5 times the diameter of the filament. Carbolfuchsin counterstain or Giemsa stain may be necessary for visualization (see Figure 39-1).
Streptobacillus moniliformis and Spirillum minus
Streptobacillus Moniliformis
General Characteristics
Laboratory Diagnosis
Specimen Collection, Transport, and Processing
Direct Detection Methods
Cultivation
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Streptobacillus moniliformis and Spirillum minus