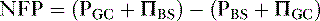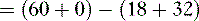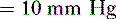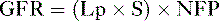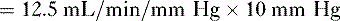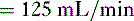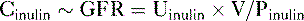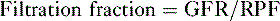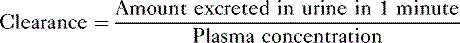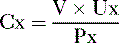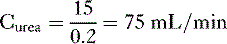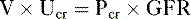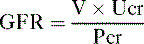Chapter 6 Renal Physiology
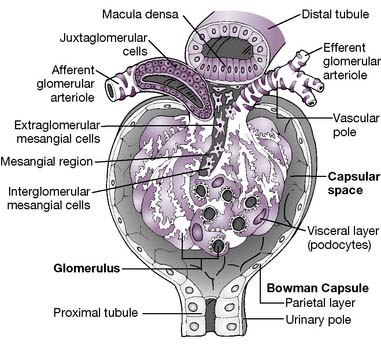
6-3 Anatomy of the glomerulus.
(From Bargmann W: Histologie und Mikronscopische Anatomie des Menschen. Stuttgart, Germany, Georg Thieme, 1977, p 86.)
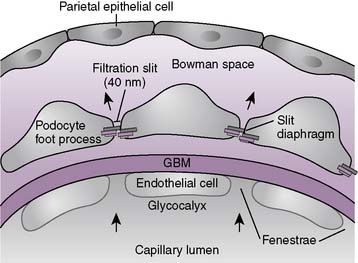
6-4 Layers of the glomerular filtration barrier. GBM, Glomerular basement membrane.
(From Mount DB, Pollak MR: Molecular and Genetic Basis of Renal Disease. Philadelphia, Saunders, 2008, Fig. 21-2B.)
TABLE 6-1 Effect of Changes in Starling Forces on Renal Plasma Flow, Glomerular Filtration Rate, and the Filtration Fraction
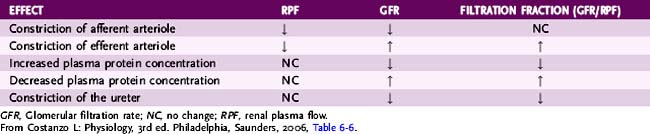
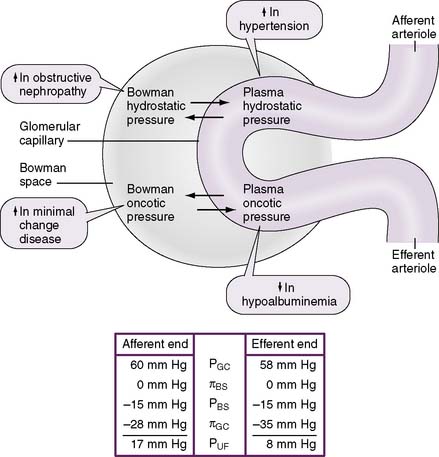
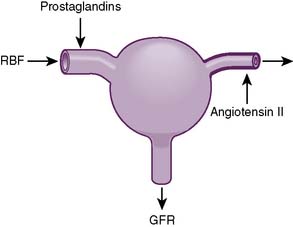
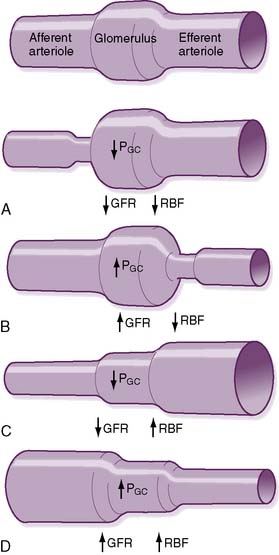
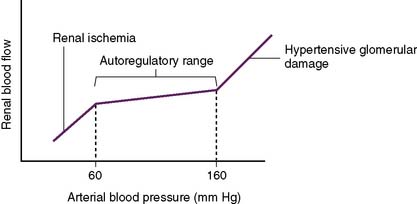
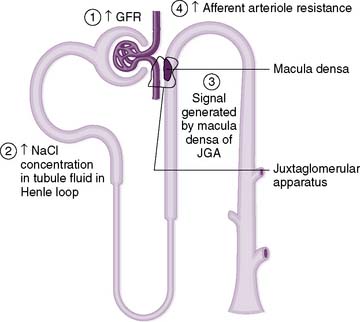
Clinical note: Narrowing of the renal arteries (renal artery stenosis) most commonly occurs as a result of atherosclerosis or fibromuscular hyperplasia. In unilateral renal artery stenosis, hypertension may occur because decreased perfusion of the affected kidney is incorrectly “interpreted” as intravascular volume depletion, which triggers a neurohormonal cascade response (the renin-angiotensin-aldosterone system and antidiuretic hormone [ADH]; see Chapter 3), causing fluid retention and vasoconstriction resulting in hypertension. When both renal arteries are affected (bilateral renal artery stenosis), renal blood flow may become so compromised that the kidneys are unable to perform their normal recycling functions, resulting in the toxic accumulation of metabolic byproducts.
so that
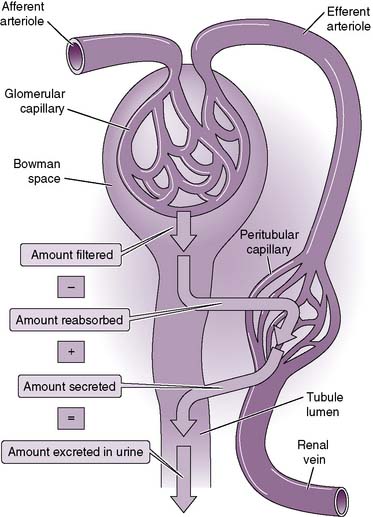
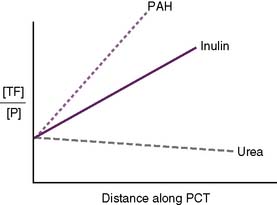
6-11 Filtration, reabsorption, and secretion along the nephron.
(From Boron W, Boulpaep E: Medical Physiology, 2nd ed. Philadelphia, Saunders, 2009, Fig. 33-8.)
TABLE 6-2 Summary of Important Clearance Values
| Substance | Approximate Clearance Rate (as % of GFR) |
|---|---|
| Urea | 50 |
| Inulin | 100 |
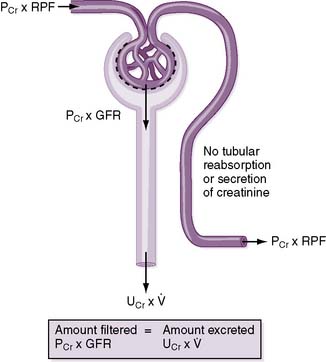
| Creatinine | 100 |
| Para-aminohippuric acid | >>100 |
| Sodium | 1 |
| Potassium | 10 |
| Glucose | 0 |
| Amino acids | 0 |
GFR, Glomerular filtration rate.
< div class='tao-gold-member'>
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree