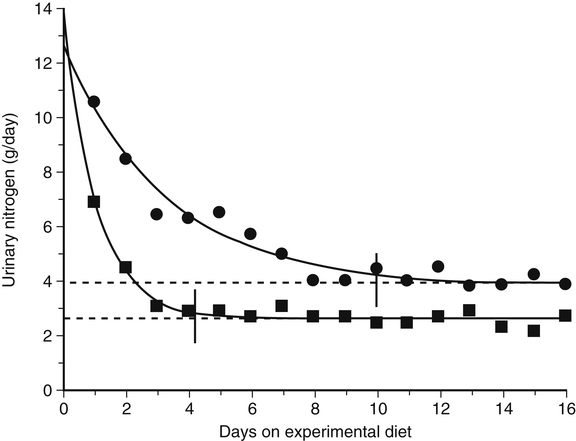
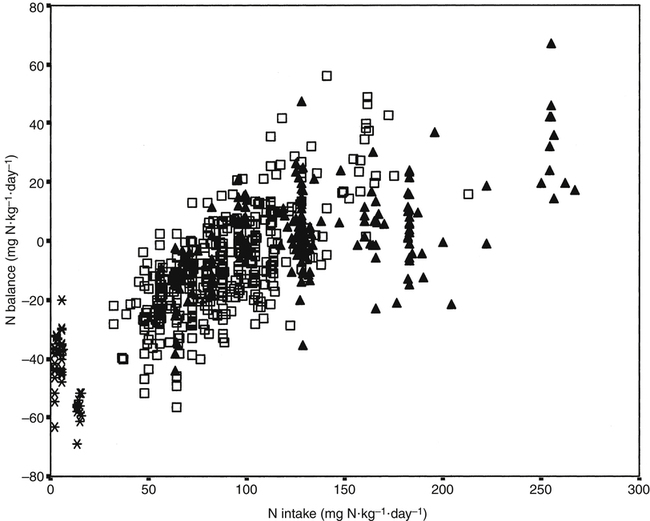
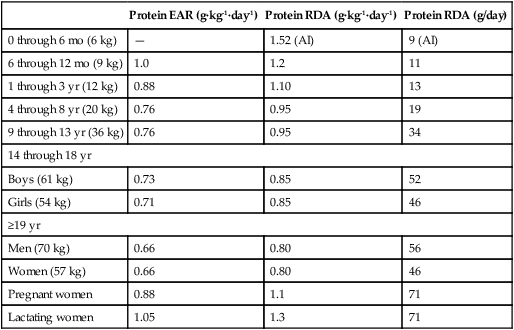
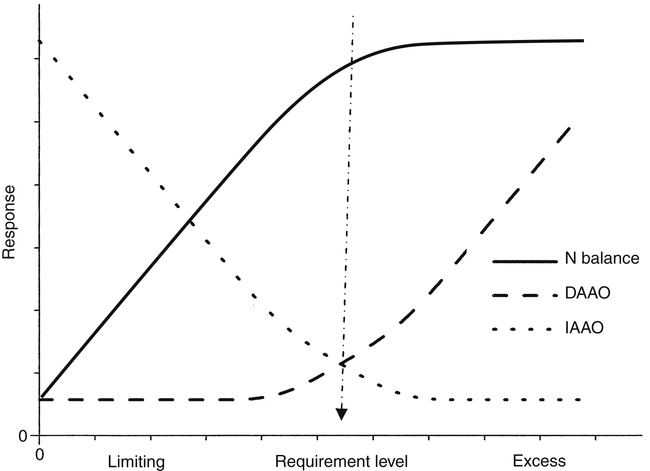
Crystal L. Levesque, PhD and Ronald O. Ball, PhD∗ Borman and colleagues (1946) defined an indispensable amino acid as “one which cannot be synthesized by the animal organism, out of materials ordinarily available to the cells, at a speed commensurate with the demands for normal growth” (p. 593). Humans do not possess the pathways for the synthesis of nine amino acids from compounds ordinarily available to cells: histidine, isoleucine, leucine, lysine, methionine, phenylalanine, threonine, tryptophan, and valine (Box 15-1). Thus these nine amino acids are always indispensable. The other eleven amino acids (alanine, arginine, asparagine, aspartate, cysteine, glutamate, glutamine, glycine, proline, serine, and tyrosine) can be synthesized by cells from materials ordinarily available to the cells. However, the extent of synthesis of some of these amino acids depends upon the dietary supply of particular precursor amino acids or the biosynthetic capacity of the organism. In addition, the amount of an amino acid needed may vary under specific circumstances such as injury or parenteral feeding. These factors have led to subclassifications of the dispensable amino acids and the use of the terms semiessential, conditionally indispensable, and truly dispensable (Reeds, 2000; Institute of Medicine [IOM], 2005). Arginine, proline, glutamine, and glycine are considered to be conditionally indispensable from a dietary viewpoint because the rate at which they are provided by endogenous synthesis may fall below the rate at which they are used. In a state of rapid growth, such as in the neonate, the essentiality of these amino acids is much more dependent on their rates of de novo synthesis. For example, in neonatal piglets, arginine is synthesized primarily in the small intestine from proline and citrulline, but the gut synthesis of arginine is insufficient to meet the whole body arginine demand (Urschel et al., 2007). Conditions that affect gut metabolism and function (i.e., intestinal disease, gut atrophy, or parenteral nutrition) can also affect the requirement for arginine as well as proline. Dramatic decreases in both arginine and proline synthesis occurred when piglets were fed parenterally, demonstrating the conditional essentiality of arginine and proline during total parenteral nutrition when amino acids bypass the gut, limiting synthesis of proline and citrulline (Bertolo et al., 2003). During critical illness, glutamine is used as a fuel for the immune system and gastrointestinal tract with increased uptake of glutamine by immune cells, the intestinal mucosa, and the kidney. Although large amounts of glutamine are released from the muscle, the increased demand for glutamine during critical illness may not be met sufficiently by an increase in synthesis, such that glutamine often becomes conditionally indispensable (Wischmeyer, 2008). Further contributing to a deficiency in glutamine during critical illness can be a decline in total body muscle tissue with progressing disease state. Glutamine-supplemented total parenteral nutrition reduced infection rate and incidence of pneumonia in critically ill patients in a large double-blind, randomized clinical trial in France (Déchelotte et al., 2006). The rate of endogenous glycine production during recovery (catch-up growth) from severe childhood malnutrition may be less than the rate of glycine utilization (Badaloo et al., 1999). There are suggestions that glycine may similarly behave as an indispensable amino acid in infants (especially premature infants), who have a limited capacity for glycine synthesis (Jackson et al., 1981). In preterm infants, cysteine, tyrosine, glutamine, arginine, proline, and glycine are typically considered conditionally indispensable because of their low endogenous synthesis. Courtney-Martin and colleagues (2010) demonstrated that the total sulfur amino acid requirement in preterm infants could be met by methionine alone. This suggests that cysteine may not be a conditionally indispensable amino acid in preterm infants. High plasma phenylalanine and excretion of abnormal metabolites of phenylalanine were observed when phenylalanine was added to commercial total parenteral nutrition solutions fed to neonatal piglets (Wykes et al., 1994), suggesting a low conversion of phenylalanine to tyrosine in neonatal piglets. Interestingly, the mean tyrosine requirement in parenterally fed neonates was three to four times higher than that supplied by commercial total parenteral nutrition solutions (Roberts et al., 2001). Some of these conditionally essential amino acids may need to be present in higher concentrations in formulations used for enteral or parenteral feeding, particularly of preterm infants. Maintenance is the state in which there is no net change in body protein mass. Although protein degradation returns amino acids to the free amino acid pool for use in protein synthesis, some irreversible loss of amino acids occurs such that there is a dietary requirement for amino acids at maintenance. Even when there is no change in body protein mass, amino acids or proteins are lost due to irreversible modification of amino acids, loss of proteins through the epithelia, loss of amino acids in the urine, use of amino acids for synthesis of nonprotein substances, and oxidation of amino acids as fuels (see Chapters 5, 9, 13, and 14). Except for the first months of life, maintenance requirements account for the major proportion of the dietary protein needs of children, adolescents, and adults. For example, the proportion of the total requirement due to maintenance is approximately 80% at 1 to 3 years and more than 98% by age 18 years. Estimation of the magnitude of obligatory amino acid and nitrogen losses is usually done by feeding protein-free diets to adults and measuring the loss of nitrogen from the body in the urine and feces, as well as cutaneous and miscellaneous losses. The association between obligatory nitrogen losses of individuals adapted to protein-free diets (i.e., ≤ 5 mg N·kg-1·day-1, or ≤ 2.2 g protein·kg-1·day-1) and the magnitude of the negative nitrogen balance experienced by these subjects is illustrated in Table 15-1. It should be noted, however, that obligatory nitrogen losses are expected to be somewhat higher in individuals consuming protein in their diets; this is especially true for losses due to amino acid catabolism. TABLE 15-1 Obligatory Nitrogen Losses in Healthy Adults on Protein-Free Diets Based on data summarized by Rand, W. M., Pellett, P. L., & Young, V. R. (2003). Meta-analysis of nitrogen balance studies for estimating protein requirements in healthy adults. The American Journal of Clinical Nutrition, 77, 109–127. Because dietary intake can vary enormously, the body must have mechanisms to adjust the rate of amino acid disposal according to the supply, and the major mechanism is the modulation of amino acid oxidation. This adaptive component changes only slowly with a sustained change in intake, as illustrated by the pattern of urinary nitrogen excretion by subjects switched from a diet containing adequate protein to one providing only a minimal level of protein; an example is shown in Figure 15-1. Thus obligatory losses will be affected by prior intake as well as by current intake unless a period of adaptation to the new intake is allowed before measurement of losses. Obligatory fecal nitrogen loss represents nitrogen released from the body into the gastrointestinal tract but not reabsorbed; the sources of obligatory nitrogen lost via the gut are proteins secreted into the gastrointestinal tract in the salivary, gastric, pancreatic, hepatic (bile) and intestinal secretions and proteins of cells shed from the gastrointestinal epithelium. Most of the endogenous proteins released into the gastrointestinal tract are digested and reabsorbed, but about 20% of the endogenous proteins (particularly proteins such as mucin that are inherently resistant to proteolysis) escape digestion and pass into the large intestine where they are used by the gastrointestinal microflora. Accurate measurement of obligatory losses of amino acids from the gastrointestinal tract has raised considerable debate, and although the protein-free diet method is most commonly used, no one method is without limitations (Stein et al., 2007). As summarized in Table 15-1, the sum of obligatory nitrogen losses in adults amounts to 48 mg N·kg-1·day-1. Using the general conversion factor of 6.25 g protein per g of nitrogen, the obligatory protein loss is 0.3 g protein·kg-1·day-1. The amount of protein required to maintain N equilibrium is much greater than the sum of the obligatory losses, however, because the efficiency with which absorbed amino acids are used for protein synthesis is not 100%. The efficiency of dietary protein utilization is about 50% (47% for adults and 58% for children), as determined from the slope of N balance response curves. This means that 0.5 g N is retained per 1 g N consumed. Thus (0.3/0.5) g protein·kg-1·day-1, or 0.6 g protein·kg-1·day-1, would be required to replace losses measured under the protein-free condition, and even more might be required in practice because obligatory losses are higher with normal diets than with protein-free diets. (With regard to this issue, note that the average protein requirement determined by nitrogen balance studies is about 0.66 g protein·kg-1·day-1.) • Supply of amino acid N to free amino acid pool = Removal of amino acid N from free amino acid pool • Amino acid N intake + Amino acid N from endogenous protein degradation = Amino acid N used for protein synthesis + Amino acid catabolism • Amino acid intake = Amino acid catabolism • Amino acids used for protein synthesis = Amino acids liberated by degradation of endogenous proteins Nitrogen balance data have been used by both the IOM (2005) and the World Health Organization (WHO, 2007) as well as many other groups for estimating protein requirements. For nonpregnant, nonlactating adults in nitrogen equilibrium, the nitrogen intake required for balance represents the requirement for maintenance. The average requirement of adults for protein was based on a meta-analysis of published nitrogen balance studies, as illustrated in Figure 15-2 (Rand et al., 2003). For each study, the lowest continuing intake of dietary protein that was sufficient to achieve body nitrogen equilibrium was considered as the individual requirement. The median nitrogen requirement derived from the meta-analysis was 105 mg·kg-1·day-1, and this value, along with the conversion factor of 6.25 g protein per g of nitrogen, was used to set the average requirement for protein at 0.66 g·kg-1·day-1 for men and women (≥19 years of age). Maintenance requirements of children aged 6 months to 18 years are similar to those of adults on a per unit body weight basis. The protein needs for growth are an important component of the protein requirement of infants, children, and adolescents. A newborn infant deposits about 0.55 g protein·kg-1·day-1; this rate of protein deposition gradually decreases to about 0.11 g·kg-1·day-1 by age 18 months, and then to less than 0.06 g·kg-1·day-1 by age 3 years (WHO, 2007). Using an efficiency of dietary protein utilization of 58%, the protein requirements for growth would be 0.95, 0.19, and 0.10 g·kg-1·day-1 at ages 1 month, 18 months, and 3 years, respectively. By adding the growth requirement to an assumed maintenance requirement of 0.66 g protein·kg-1·day-1, the total protein requirement would be 1.61, 0.83, and 0.76 g·kg-1·day-1 at age 1 month, 18 months, and 3 years, respectively. Thus during the first 2 months of life, an infant’s requirement for growth exceeds the infant’s requirement for maintenance with almost 60% of the newborn’s protein requirement being for growth. The proportion of the total protein requirement needed for growth decreases to about 22% at 18 months and then to 12% or less by 3 years, remaining in the range of 0% to 12% throughout the remainder of childhood and adolescence. Protein is deposited in fetal and maternal tissues during pregnancy. The Food and Agriculture Organization (FAO) of the United Nations, the WHO, and the United Nations University (UNU) arms of the United Nations system used estimates of 0, 1.9, and 7.4 g/day for amounts of protein deposited during the first, second, and third trimesters of pregnancy, based on a mean gestational weight gain of 13.8 kg (WHO, 2007). The efficiency of dietary protein intake for protein accretion during pregnancy was conservatively set at 0.42, yielding additional protein requirements of 4.5 and 17.7 g/day for the second and third trimesters, respectively. It should be noted that maintenance requirements also increase during pregnancy on a g/day basis due to the increase in total body mass. The IOM (2005) used a similar approach in estimating needs during pregnancy but averaged total protein accretion during gestation over the second and third trimesters (5.4 g/day). They used this average, along with an efficiency of 0.43, to set the need for additional protein at 12.6 g/day for protein accretion during pregnancy. Based on a reference weight of 57 kg, this corresponds to an increase of 0.22 g·kg-1·day-1 or a total protein requirement of 0.66 + 0.22 = 0.88 g·kg-1·day-1 during the later two trimesters. The additional amino acid requirements for lactation derive primarily from the quantity and composition of the protein and amino acids secreted in the milk. The IOM (2005) based the increase in protein requirement for lactating women on an average output of 10 g protein per day in the milk (g N × 6.25) and the adult efficiency of protein utilization of 0.47, yielding an increased protein requirement of 21 g protein per day, or 0.39 g·kg-1·day-1. Adding the lactation requirement to the maintenance requirement, the total protein requirement for lactating women is 0.39 + 0.66 = 1.05 g·kg-1·day-1. The FAO/WHO/UNU (WHO, 2007) used a similar approach but made separate recommendations for the first 6 months and second 6 months of breast-feeding. Based on estimates of milk protein content and milk volume, along with correction for an efficiency of 0.47, WHO set the average increase in requirement for protein at 15 g/day for the first 6 months and at 10 g/day for the second 6 months, assuming partial breast-feeding during the second 6 months (WHO, 2007). The protein requirements established by the IOM and the FAO/WHO/UNU are based on nitrogen balance studies in which subjects were fed high-quality proteins or protein mixtures. In setting the Dietary Reference Intakes (DRIs) for the populations of the United States and Canada, the IOM (2005) did not include an adjustment for protein quality, because it was assumed that these North American populations consume varied diets with both high-quality and complementary proteins, such that protein quality is not of concern for most of the population. However, recommended intakes would need to be higher for populations or individuals consuming diets in which the overall protein quality is not high. The FAO/WHO/UNU (WHO, 2007) made their recommendations with provisions for additional adjustments for protein quality when appropriate. The topic of protein quality is considered in more detail later in this chapter. The IOM (2005) set DRIs for protein intake of various age and sex groups. In the DRIs Across the Life Cycle box, the Estimated Average Requirement (EAR) and Recommended Dietary Allowance (RDA) are shown as g protein·kg-1·day-1, and RDAs are also given as g/day for reference size males and females (e.g., 70-kg man and 57-kg woman). Thus the RDA for protein is 0.8 g·kg-1·day-1 for adults, or 46 g/day for the 57-kg reference woman and 56 g/day for the 70-kg reference man. There is considerable debate whether the requirement for protein is higher in elderly adults. The IOM (2005) and the FAO/WHO/UNU (WHO, 2007) concluded that the lack of demonstrated improvement in biochemical indicators of protein sufficiency or measured nitrogen balance with higher protein intakes suggests that the requirement for protein does not increase with age. However, it was noted that the protein to energy ratio for diets of the elderly needs to be higher than for younger individuals because of the decreased energy expenditure of the elderly, despite no change in their protein requirement per se. For infants, the IOM (2005) did not determine an EAR/RDA, but an Adequate Intake (AI) was set based on the known adequacy of breast milk for infants. The AI for infants up to 6 months of age, set by the IOM, is based on the average volume of milk intake (0.78 L/day) and the average protein content of human milk during the first 6 months of lactation (11.7 g/L) and is 9 g/day. The AI can be stated as 1.5 g·kg-1·day-1, based on a reference weight of 6 kg for 2- to 6-month-old infants. A similar estimate based on milk plus complementary food intake is the basis of the AI for infants during the second 6 months of life. The FAO/WHO/UNU (WHO, 2007) has calculated safe protein intakes for infants based on estimates of their maintenance requirements (0.58 g protein·kg-1·day-1 from nitrogen balance studies) and growth requirements (protein deposition adjusted for an efficiency of 0.66), with the average requirement increased by 2 SD, to yield safe levels ranging from 1.8 g protein·kg-1·day-1 (for the 1-month-old) to 1.1 g protein·kg-1·day-1 (for the 6-month-old infant). Depending on the approach used to determine an indispensable amino acid requirement, three different patterns of response are possible; these are illustrated in Figure 15-3. One pattern is that seen when either nitrogen balance in the adult or growth in a growing animal or child is plotted against amino acid intake. Nitrogen balance or growth will increase as the intake of the limiting indispensable amino acid (i.e., test amino acid) increases up until the requirement amount is reached, at which point nitrogen balance or growth will plateau. A second pattern is seen for the indicator amino acid oxidation (IAAO) method where oxidation of the indicator amino acid is high when amino acids are not being used maximally for protein synthesis but decreases to a plateau once the requirement of the limiting amino acid for protein synthesis has been met. For nitrogen balance, growth, and IAAO, once the requirement is reached the measured response will plateau despite the addition of higher levels of protein or limiting amino acid. A third pattern occurs when oxidation of a test amino acid is measured directly, called the direct amino acid oxidation (DAAO) method. In this case, oxidation of the test amino acid remains low with little change until the requirement is approached or met, after which its oxidation increases progressively as intake is further increased. A similar pattern may be observed by following the plasma level of the test amino acid; the level of the test amino acid will begin to increase once intake exceeds the requirement. When lines are drawn for the slope of the two phases (change and plateau) of the response curve, the crossover point or breakpoint of the two lines is used to determine the requirement level as illustrated in Figure 15-3. In nitrogen balance experiments, the rate of body nitrogen retention is estimated as the difference between the dietary nitrogen intake and the sum of the losses in urine and feces and by other routes (integumental and miscellaneous). Nitrogen balance is based on the premise that protein or lean body tissue is the major nitrogen-containing component in the body; thus nitrogen loss or gain equates to protein loss or gain. It is assumed that body nitrogen increases in the growing child and remains constant in the adult (WHO, 2007). As described previously, it is generally presumed that the protein requirement of an adult is achieved when the individual is in zero nitrogen balance (i.e., nitrogen equilibrium). Infants, children, pregnant women, and individuals recovering from disease states in which lean body mass was lost should be in positive nitrogen balance, reflecting net deposition of protein. Nitrogen balance studies were used to obtain the first estimates of the amino acid requirements of human adults. Rose and colleagues, in a series of nitrogen balance studies in the 1940s, determined the amino acid requirements of young men (see Rose, 1957). First, men were given diets devoid of a single amino acid so as to establish which amino acids were dispensable and which were indispensable. Eight amino acids were determined to be indispensable. In these studies, removal of histidine from the diet did not lead to a negative nitrogen balance; this was interpreted to mean that histidine was dispensable, and this conclusion was only reversed much later by longer-term experiments. Rose and colleagues then conducted a series of quantitative studies in which they gave each subject a succession of diets with different concentrations of the amino acid under investigation. From the changes in nitrogen balance that ensued, they attempted to identify the intake of each amino acid that was required for nitrogen equilibrium to be achieved. Similar experiments were conducted on young women, but using more subjects and with several improvements in experimental procedures (Leverton et al., 1959). The results of these early experiments in determining the amino acid requirements of men and women formed the basis of the international estimates of adult amino acid requirements (WHO, 1985) that were widely used before 2000. One of the greatest challenges with the nitrogen balance method is accurate measurement of nitrogen losses. Due to difficulties in measuring integumental and miscellaneous losses, they are not always included in the calculation of nitrogen balance. Not accounting for integumental and miscellaneous losses results in underestimation of nitrogen loss. Urinary and fecal nitrogen can be measured relatively accurately; however, incomplete measurement of food spillage and food residues results in overestimation of nitrogen intake. The tendency to overestimate intake and to underestimate losses both contribute to the tendency of nitrogen balance studies to underestimate protein or amino acid requirements (WHO, 2007). This means that a subject apparently receiving sufficient amino acid (or protein) to maintain nitrogen equilibrium may actually be in negative nitrogen balance and need more of the amino acid (or protein) to maintain true nitrogen equilibrium. A further problem with the nitrogen balance approach, especially in short-term studies, may arise through the implicit assumption that a subject in nitrogen equilibrium must also be in amino acid equilibrium. This is not necessarily so. Histidine, for example, is stored in the form of the peptide carnosine (β-alanylhistidine). Carnosine, in experimental animals at least, is depleted during dietary histidine deficiency. This allows obligatory losses of histidine to be met for a time without loss of body protein, despite an inadequate histidine intake. There may also be a potential to adapt to an indispensable amino acid deficiency through modification of the amino acid composition of the body protein pool as a whole. Of course, it is not possible to modify the amino acid composition of any of the body’s proteins, but it is possible to vary the relative amounts of the various proteins. Because proteins have different amino acid compositions, there is the potential to adapt, at least temporarily, to a deficiency of one amino acid by depleting the body of proteins rich in that amino acid. This also happens in histidine deficiency, in which hemoglobin, which is very rich in histidine, can be gradually depleted. The mechanism of this adaptation is not known. However, this ability of the body to use endogenous sources of histidine explains why histidine was not identified as an indispensable amino acid in the early balance studies (Rose, 1957; Leverton, 1959). In the IAAO method, a separate indispensable amino acid is labeled (i.e., an indicator amino acid) and its oxidation is measured. The indicator amino acid is another indispensable amino acid present in adequate amounts while intake of the test amino acid is altered. When intake of the test amino acid is low and protein synthesis is limited by a lack of the test amino acid, the excess indicator amino acid (i.e., that not used for protein synthesis) will be oxidized. As the intake of the test amino acid is increased, incorporation of the indicator amino acid, as well as all other non-test amino acids, into protein will increase, such that oxidation of the indicator amino acid to 13CO2 will decrease. The IAAO method is not dependent upon release of the carboxyl carbon of the test amino acid and thus can be used to assess the requirement of any indispensable amino acid. Phenylalanine has been found to be the most responsive and accurate of the indispensable amino acids for use as the indicator amino acid in determining amino acid requirements (Levesque et al., 2010). There has been considerable debate over the most appropriate requirement estimates for indispensable amino acids in adults. However, extensive reviews (Young, 1986; Millward, 1988; Fuller and Garlick, 1994) of the original nitrogen balance studies by Rose (1957) and Irwin and Hegsted (1971) all conclude that previous estimates (WHO, 1985) based on nitrogen balance were too low. The greatest concern with these early studies was the lack of measurement of miscellaneous nitrogen losses, resulting in low estimates of the indispensable amino acid requirements for achieving nitrogen balance. The current FAO/WHO/UNU (WHO, 2007) and IOM (2005) estimates of indispensable amino acid requirements (mg amino acid·kg-1·day-1) are based primarily on amino acid oxidation studies. As shown in Table 15-2, the current estimates of requirements for 6 out of 10 of the indispensable amino acids are close to double the previous 1985 estimates. TABLE 15-2 ∗Calculated as the individual indispensable amino acid requirement (mg amino acid·kg-1·day-1) divided by the requirement for total protein (0.66 g protein·kg-1·day-1). †WHO. (2007). Protein and amino acid requirements in humans: Report of Joint FAO/WHO/UNU Expert Consultation. WHO Technical Series 935. Geneva: FAO. ‡IOM. (2005). Dietary Reference Intakes: Energy, carbohydrate, fiber, fat, fatty acids, cholesterol, protein, and amino acids. Washington, DC: The National Academies Press. §WHO. (1985). Energy and protein requirements: Report of a Joint FAO/WHO/UNU Expert Consultation. WHO Technical Report Series 724. Geneva: WHO. The estimates of amino acid requirements can be used to generate safe intakes for individual indispensable amino acids and for generation of the pattern or amounts of indispensable amino acids needed in a protein source for it to meet both the dietary protein and the indispensable amino acid requirements. In order for the required amount of dietary protein to meet all amino acid needs, it must contain the indispensable amino acid in amounts equal to or greater than the required pattern. The requirement patterns for indispensable amino acids are also shown in Table 15-2. Even based on the highest requirement pattern, only 27% of the total amino acids in the dietary protein need to be indispensable amino acids. If the pattern of the dietary protein mixture of a population has an inadequate amount of one or more indispensable amino acids, adjustments should be made to increase the total protein requirements of that population to allow intake of a sufficient amount of the limiting indispensable amino acids. Thus for lower-quality protein mixtures, higher total protein intakes will be needed. Another approach, of course, would be to improve the quality of the protein mixture by using a mixture of complementary proteins or adding a small amount of high-quality protein to the mixture. As with setting protein requirements, the requirements for indispensable amino acids in children, pregnant women, and lactating women were based on the factorial approach and include a component for both maintenance and growth or milk secretion. The factorial method is the approach in which the relevant components of the requirement (maintenance, growth, accretion of maternal tissues and growth of the fetus during pregnancy, and milk secretion during lactation) are estimated separately and then added to set the requirement. Although this approach ostensibly provides a logical system for determination of requirements, the direct estimation of the component requirements is difficult. This is particularly true during pregnancy, where direct estimates of protein and amino acid requirements for fetal growth are largely unknown. For example in school-aged children, the factorial estimate for total sulfur amino acids (15 mg·kg-1·day-1) and lysine (30 mg·kg-1·day-1) were similar to the mean requirement determined by IAAO (13 and 35 mg·kg-1·day-1, respectively) (Turner et al., 2006; Elango et al., 2007). However, the factorial estimate for total branched-chain amino acids (85 mg·kg-1·day-1) in school-aged children was significantly lower than the mean requirement based on IAAO (147 mg·kg-1) (Mager et al., 2003). To estimate the indispensable amino acid requirements of children, the IOM (2005) used the factorial method. The maintenance requirements for children were based on the EARs for indispensable amino acids in adults. To this was added the requirements for accretion of protein, which were based on the amino acid composition of body proteins and an estimated efficiency of utilization of amino acids derived from the diet (0.58). Safe intakes for infants were based on adequate intakes from breast milk during the first 6 months and from breast milk and complementary foods during the second 6 months. FAO/WHO/UNU (WHO, 2007) used a similar approach to estimate indispensable amino acid requirements of children and infants. However, to estimate the amino acid requirements of infants from 6 months to 1 years of age, the FAO/WHO/UNU applied the factorial method instead of simply relying on the amino acid composition and intake volume of human milk. Table 15-3 shows the indispensable amino acid intake of breast-fed infants and the indispensable amino acid requirements of children from 6 months to 18 years of age. TABLE 15-3 Average Requirements for Indispensable Amino Acids in Children and Adults
Protein and Amino Acid Requirements
Classification of Dispensable and Indispensable Amino Acids
Indispensable Amino Acids
Semidispensable, Conditionally Indispensable, and Dispensable Amino Acids
Conditionally Indispensable Amino Acids
Requirement for Protein (Amino Acids)
Protein (Amino Acid) Requirement for Maintenance
Obligatory Losses of Proteins and Amino Acids
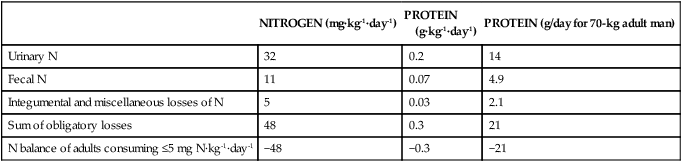
NITROGEN (mg·kg-1·day-1)
PROTEIN
(g·kg-1·day-1)
PROTEIN (g/day for 70-kg adult man)
Urinary N
32
0.2
14
Fecal N
11
0.07
4.9
Integumental and miscellaneous losses of N
5
0.03
2.1
Sum of obligatory losses
48
0.3
21
N balance of adults consuming ≤5 mg N·kg-1·day-1
−48
−0.3
−21
Protein (Amino Acid) Requirements for Nitrogen Equilibrium or Maintenance
Protein (Amino Acid) Requirements for Growth, Pregnancy, and Lactation
Consideration of Protein Quality
DRIs for Protein
Requirements for Individual Amino Acids
Amino Acid Requirements Based on Empirical Analysis of Dose Responses
Estimates of Indispensable Amino Acid Requirements by Nitrogen Balance Studies
Estimates of Indispensable Amino Acid Requirements by Amino Acid Oxidation Studies
Adult Requirements for Indispensable Amino Acids and Indispensable Amino Acid Requirement Patterns
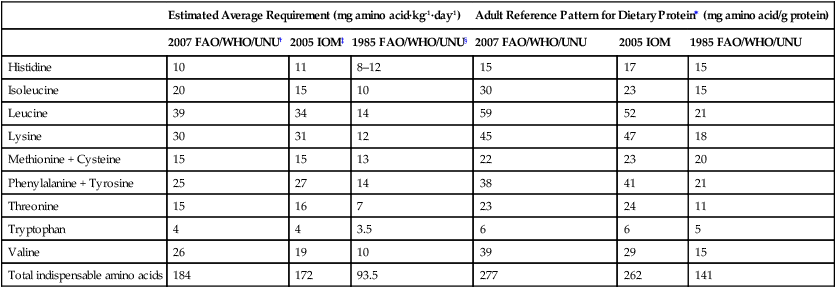
Estimated Average Requirement (mg amino acid·kg-1·day-1)
Adult Reference Pattern for Dietary Protein∗ (mg amino acid/g protein)
2007 FAO/WHO/UNU†
2005 IOM‡
1985 FAO/WHO/UNU§
2007 FAO/WHO/UNU
2005 IOM
1985 FAO/WHO/UNU
Histidine
10
11
8–12
15
17
15
Isoleucine
20
15
10
30
23
15
Leucine
39
34
14
59
52
21
Lysine
30
31
12
45
47
18
Methionine + Cysteine
15
15
13
22
23
20
Phenylalanine + Tyrosine
25
27
14
38
41
21
Threonine
15
16
7
23
24
11
Tryptophan
4
4
3.5
6
6
5
Valine
26
19
10
39
29
15
Total indispensable amino acids
184
172
93.5
277
262
141
Estimates of Indispensable Amino Acid Requirements of Children and of Pregnant and Lactating Women by the Factorial Method
Daily Intake of Indispensable Amino Acids in Breast-fed Infants
Amino Acid Requirement Determined by Factorial Method
Adult Requirement
Age in Years
BIRTH TO 6 MONTHS
0.5
1–2
3–10
11–14
15–18
>18
(mg amino acid·kg-1·day-1)
Histidine
20
22
15
12
12
11
10
Isoleucine
52
36
27
23
22
21
20
Leucine
90
73
54
44
44
42
39
Lysine
65
64
45
35
35
33
30
Methionine + Cysteine
31
31
22
18
17
16
15
Phenylalanine + Tyrosine
88
59
40
30
30
28
25
Threonine
41
34
23
18
18
17
15
Tryptophan
16
9.5
6.4
4.8
4.8
4.5
4.0
Valine
51
49
36
29
29
28
26 ![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree