MITRAL REGURGITATION
ETIOLOGY
MR may result from an abnormality or disease process that affects any one or more of the five functional components of the mitral valve apparatus (leaflets, annulus, chordae tendineae, papillary muscles, and subjacent myocardium) (Table 284-1). Acute MR can occur in the setting of acute myocardial infarction (MI) with papillary muscle rupture (Chap. 295), following blunt chest wall trauma, or during the course of infective endocarditis. With acute MI, the posteromedial papillary muscle is involved much more frequently than the anterolateral papillary muscle because of its singular blood supply. Transient, acute MR can occur during periods of active ischemia and bouts of angina pectoris. Rupture of chordae tendineae can result in “acute-on-chronic MR” in patients with myxomatous degeneration of the valve apparatus.
Chronic MR can result from rheumatic disease, mitral valve prolapse (MVP), extensive mitral annular calcification, congenital valve defects, hypertrophic obstructive cardiomyopathy (HOCM), and dilated cardiomyopathy (Chap. 287). Distinction also should be drawn between primary (degenerative, organic) MR, in which the leaflets and/or chordae tendineae are primarily responsible for abnormal valve function, and functional (secondary) MR, in which the leaflets and chordae tendineae are structurally normal but the regurgitation is caused by annular enlargement, papillary muscle displacement, leaflet tethering, or their combination. The rheumatic process produces rigidity, deformity, and retraction of the valve cusps and commissural fusion, as well as shortening, contraction, and fusion of the chordae tendineae. The MR associated with both MVP and HOCM is usually dynamic in nature. MR in HOCM occurs as a consequence of anterior papillary muscle displacement and systolic anterior motion of the anterior mitral valve leaflet into the narrowed LV outflow tract. Annular calcification is especially prevalent among patients with advanced renal disease and is commonly observed in women >65 years of age with hypertension and diabetes. MR may occur as a congenital anomaly (Chap. 282), most commonly as a defect of the endocardial cushions (atrioventricular cushion defects). A cleft anterior mitral valve leaflet accompanies primum atrial septal defect. Chronic MR is frequently secondary to ischemia and may occur as a consequence of ventricular remodeling, papillary muscle displacement, and leaflet tethering, or with fibrosis of a papillary muscle, in patients with healed MI(s) and ischemic cardiomyopathy. Similar mechanisms of annular dilation and ventricular remodeling contribute to the MR that occurs among patients with nonischemic forms of dilated cardiomyopathy once the LV end-diastolic dimension reaches 6 cm.
Irrespective of cause, chronic severe MR is often progressive, because enlargement of the LA places tension on the posterior mitral leaflet, pulling it away from the mitral orifice and thereby aggravating the valvular dysfunction. Similarly, LV dilation increases the regurgitation, which, in turn, enlarges the LA and LV further, resulting in a vicious circle; hence the aphorism, “mitral regurgitation begets mitral regurgitation.”
PATHOPHYSIOLOGY
The resistance to LV emptying (LV afterload) is reduced in patients with MR. As a consequence, the LV is decompressed into the LA during ejection, and with the reduction in LV size during systole, there is a rapid decline in LV tension. The initial compensation to MR is more complete LV emptying. However, LV volume increases progressively with time as the severity of the regurgitation increases and as LV contractile function deteriorates. This increase in LV volume is often accompanied by a reduced forward CO. LV compliance is often increased, and thus, LV diastolic pressure does not increase until late in the course. The regurgitant volume varies directly with the LV systolic pressure and the size of the regurgitant orifice; the latter, in turn, is influenced by the extent of LV and mitral annular dilation. Because EF rises in severe MR in the presence of normal LV function, even a modest reduction in this parameter (<60%) reflects significant dysfunction.
During early diastole, as the distended LA empties, there is a particularly rapid y descent in the absence of accompanying MS. A brief, early diastolic LA-LV pressure gradient (often generating a rapid filling sound [S3] and mid-diastolic murmur masquerading as MS) may occur in patients with pure, severe MR as a result of the very rapid flow of blood across a normal-sized mitral orifice.
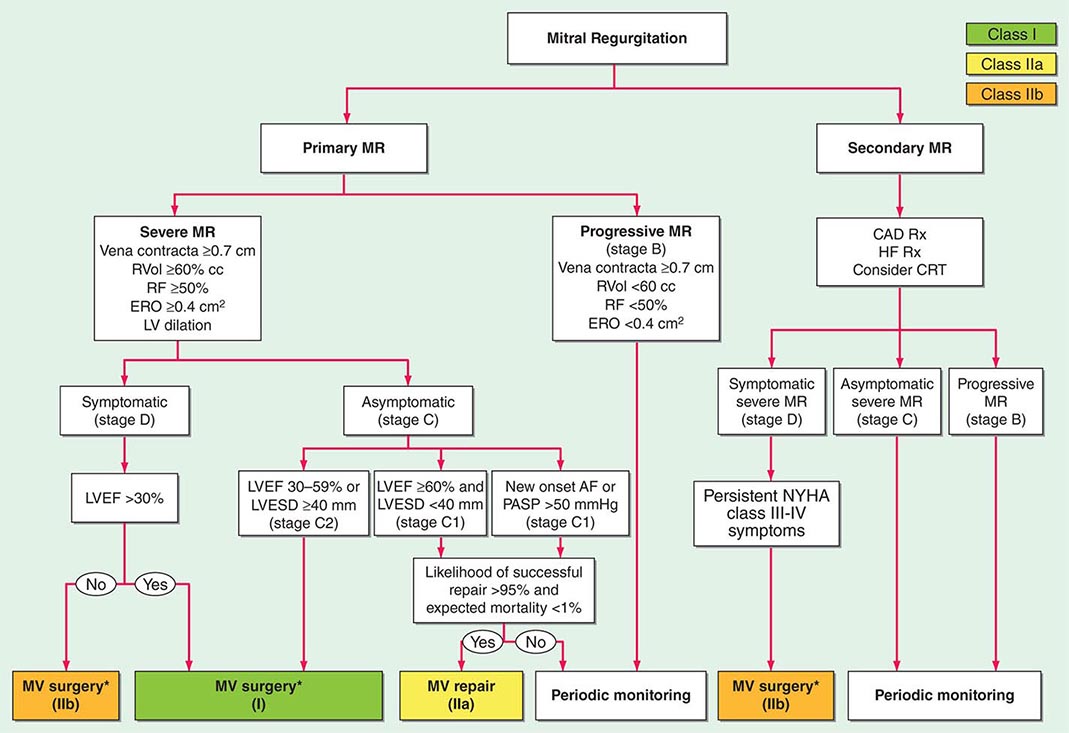
Semiquantitative estimates of LV ejection fraction (LVEF), CO, PA systolic pressure, regurgitant volume, regurgitant fraction (RF), and the effective regurgitant orifice area can be obtained during a careful Doppler echocardiographic examination. These measurements can also be obtained accurately with cardiac magnetic resonance (CMR) imaging, although this technology is not widely available. Left and right heart catheterization with contrast ventriculography is used less frequently. Severe, nonischemic MR is defined by a regurgitant volume ≥60 mL/beat, RF ≥50%, and effective regurgitant orifice area ≥0.40 cm2. Severe ischemic MR, however, is usually associated with an effective regurgitant orifice area of >0.2 cm2. In the latter instance, lesser degrees of MR carry relatively greater prognostic weight.
LA Compliance In acute severe MR, the regurgitant volume is delivered into a normal-sized LA having normal or reduced compliance. As a result, LA pressures rise markedly for any increase in LA volume. The v wave in the LA pressure pulse is usually prominent, LA and pulmonary venous pressures are markedly elevated, and pulmonary edema is common. Because of the rapid rise in LA pressures during ventricular systole, the murmur of acute MR is early in timing and decrescendo in configuration ending well before S2, as a reflection of the progressive diminution in the LV-LA pressure gradient. LV systolic function in acute MR may be normal, hyperdynamic, or reduced, depending on the clinical context.
Patients with chronic severe MR, on the other hand, develop marked LA enlargement and increased LA compliance with little if any increase in LA and pulmonary venous pressures for any increase in LA volume. The LA v wave is relatively less prominent. The murmur of chronic MR is classically holosystolic in timing and plateau in configuration, as a reflection of the near-constant LV-LA pressure gradient. These patients usually complain of severe fatigue and exhaustion secondary to a low forward CO, whereas symptoms resulting from pulmonary congestion are less prominent initially; AF is almost invariably present once the LA dilates significantly.
SYMPTOMS
Patients with chronic mild-to-moderate, isolated MR are usually asymptomatic. This form of LV volume overload is well tolerated. Fatigue, exertional dyspnea, and orthopnea are the most prominent complaints in patients with chronic severe MR. Palpitations are common and may signify the onset of AF. Right-sided heart failure, with painful hepatic congestion, ankle edema, distended neck veins, ascites, and secondary TR, occurs in patients with MR who have associated pulmonary vascular disease and pulmonary hypertension. Acute pulmonary edema is common in patients with acute severe MR.
PHYSICAL FINDINGS
In patients with chronic severe MR, the arterial pressure is usually normal, although the carotid arterial pulse may show a sharp, low-volume upstroke owing to the reduced forward CO. A systolic thrill is often palpable at the cardiac apex, the LV is hyperdynamic with a brisk systolic impulse and a palpable rapid-filling wave (S3), and the apex beat is often displaced laterally.
In patients with acute severe MR, the arterial pressure may be reduced with a narrow pulse pressure, the jugular venous pressure and wave forms may be normal or increased and exaggerated, the apical impulse is not displaced, and signs of pulmonary congestion are prominent.
Auscultation S1 is generally absent, soft, or buried in the holosystolic murmur of chronic, severe MR. In patients with severe MR, the aortic valve may close prematurely, resulting in wide but physiologic splitting of S2. A low-pitched S3 occurring 0.12–0.17 s after the aortic valve closure sound, i.e., at the completion of the rapid-filling phase of the LV, is believed to be caused by the sudden tensing of the papillary muscles, chordae tendineae, and valve leaflets. It may be followed by a short, rumbling, mid-diastolic murmur, even in the absence of structural MS. A fourth heart sound is often audible in patients with acute severe MR who are in sinus rhythm. A presystolic murmur is not ordinarily heard with isolated MR.
A systolic murmur of at least grade III/VI intensity is the most characteristic auscultatory finding in chronic severe MR. It is usually holosystolic (see Fig. 267-5A), but as previously noted, it is decrescendo and ceases in mid to late systole in patients with acute severe MR. The systolic murmur of chronic MR is usually most prominent at the apex and radiates to the axilla. However, in patients with ruptured chordae tendineae or primary involvement of the posterior mitral leaflet with prolapse or flail, the regurgitant jet is eccentric, directed anteriorly, and strikes the LA wall adjacent to the aortic root. In this situation, the systolic murmur is transmitted to the base of the heart and, therefore, may be confused with the murmur of AS. In patients with ruptured chordae tendineae, the systolic murmur may have a cooing or “seagull” quality, whereas a flail leaflet may produce a murmur with a musical quality. The systolic murmur of chronic MR not due to MVP is intensified by isometric exercise (handgrip) but is reduced during the strain phase of the Valsalva maneuver because of the associated decrease in LV preload.
LABORATORY EXAMINATION
ECG In patients with sinus rhythm, there is evidence of LA enlargement, but RA enlargement also may be present when pulmonary hypertension is significant and affects RV function. Chronic severe MR is frequently associated with AF. In many patients, there is no clear-cut ECG evidence of enlargement of either ventricle. In others, the signs of eccentric LV hypertrophy are present.
Echocardiogram TTE is indicated to assess the mechanism of the MR and its hemodynamic severity. LV function can be assessed from LV end-diastolic and end-systolic volumes and EF. Observations can be made regarding leaflet structure and function, chordal integrity, LA and LV size, annular calcification, and regional and global LV systolic function. Doppler imaging should demonstrate the width or area of the color flow MR jet within the LA, the duration and intensity of the continuous wave Doppler signal, the pulmonary venous flow contour, the early peak mitral inflow velocity, and quantitative measures of regurgitant volume, RF, and effective regurgitant orifice area. In addition, the PAPs can be estimated from the TR jet velocity. TTE is also indicated to follow the course of patients with chronic MR and to provide rapid assessment for any clinical change. The echocardiogram in patients with MVP is described in the next section. TEE provides greater anatomic detail than TTE (see Fig. 270e-5). Exercise testing with TTE can be useful to assess exercise capacity as well as any dynamic change in MR severity, PA systolic pressures, and biventricular function, for patients in whom there is a discrepancy between clinical findings and the results of functional testing performed at rest.
Chest X-Ray The LA and LV are the dominant chambers in chronic MR. Late in the course of the disease, the LA may be massively enlarged and forms the right border of the cardiac silhouette. Pulmonary venous congestion, interstitial edema, and Kerley B lines are sometimes noted. Marked calcification of the mitral leaflets occurs commonly in patients with long-standing, combined rheumatic MR and MS. Calcification of the mitral annulus may be visualized, particularly on the lateral view of the chest. Patients with acute severe MR may have asymmetric pulmonary edema if the regurgitant jet is directed predominantly to the orifice of an upper lobe pulmonary vein.
MITRAL VALVE PROLAPSE
MVP, also variously termed the systolic click-murmur syndrome, Barlow’s syndrome, floppy-valve syndrome, and billowing mitral leaflet syndrome, is a relatively common but highly variable clinical syndrome resulting from diverse pathologic mechanisms of the mitral valve apparatus. Among these are excessive or redundant mitral leaflet tissue, which is commonly associated with myxomatous degeneration and greatly increased concentrations of certain glycosaminoglycans.
In most patients with MVP, the cause is unknown, but in some, it appears to be genetically determined. A reduction in the production of type III collagen has been incriminated, and electron microscopy has revealed fragmentation of collagen fibrils.
MVP is a frequent finding in patients with heritable disorders of connective tissue, including Marfan’s syndrome (Chap. 427), osteogenesis imperfecta, and Ehlers-Danlos syndrome. MVP may be associated with thoracic skeletal deformities similar to but not as severe as those in Marfan’s syndrome, such as a high-arched palate and alterations of the chest and thoracic spine, including the so-called straight back syndrome.
In most patients with MVP, myxomatous degeneration is confined to the mitral valve, although the tricuspid and aortic valves may also be affected. The posterior mitral leaflet is usually more affected than the anterior, and the mitral valve annulus is often dilated. In many patients, elongated, redundant, or ruptured chordae tendineae cause or contribute to the regurgitation.
MVP also may occur rarely as a sequel to acute rheumatic fever, in ischemic heart disease, and in various cardiomyopathies, as well as in 20% of patients with ostium secundum atrial septal defect.
MVP may lead to excessive stress on the papillary muscles, which, in turn, leads to dysfunction and ischemia of the papillary muscles and the subjacent ventricular myocardium. Rupture of chordae tendineae and progressive annular dilation and calcification contribute to valvular regurgitation, which then places more stress on the diseased mitral valve apparatus, thereby creating a vicious circle. ECG changes (see below) and ventricular arrhythmias described in some patients with MVP appear to result from regional ventricular dysfunction related to the increased stress placed on the papillary muscles.
CLINICAL FEATURES
MVP is more common in women and occurs most frequently between the ages of 15 and 30 years; the clinical course is most often benign. MVP may also be observed in older (>50 years) patients, often men, in whom MR is often more severe and requires surgical treatment. There is an increased familial incidence for some patients, suggesting an autosomal dominant form of inheritance with incomplete penetrance. MVP varies in its clinical expression, ranging from only a systolic click and murmur with mild prolapse of the posterior leaflet to severe MR due to chordal rupture and leaflet flail. The degree of myxomatous change of the leaflets can also vary widely. In many patients, the condition progresses over years or decades; in others, it worsens rapidly as a result of chordal rupture or endocarditis.
Most patients are asymptomatic and remain so for their entire lives. However, in North America, MVP is now the most common cause of isolated severe MR requiring surgical treatment. Arrhythmias, most commonly ventricular premature contractions and paroxysmal supraventricular and ventricular tachycardia, as well as AF, have been reported and may cause palpitations, light-headedness, and syncope. Sudden death is a very rare complication and occurs most often in patients with severe MR and depressed LV systolic function. There may be an excess risk of sudden death among patients with a flail leaflet. Many patients have chest pain that is difficult to evaluate; it is often substernal, prolonged, and not related to exertion, but may rarely resemble angina pectoris. Transient cerebral ischemic attacks secondary to emboli from the mitral valve due to endothelial disruption have been reported. Infective endocarditis may occur in patients with MR and/or leaflet thickening.
Auscultation A frequent finding is the mid or late (nonejection) systolic click, which occurs 0.14 s or more after S1 and is thought to be generated by the sudden tensing of slack, elongated chordae tendineae or by the prolapsing mitral leaflet when it reaches its maximal excursion. Systolic clicks may be multiple and may be followed by a high-pitched, mid-late systolic crescendo-decrescendo murmur, which occasionally is “whooping” or “honking” and is heard best at the apex. The click and murmur occur earlier with standing, during the strain phase of the Valsalva maneuver, and with any intervention that decreases LV volume, exaggerating the propensity of mitral leaflet prolapse. Conversely, squatting and isometric exercises, which increase LV volume, diminish MVP; the click-murmur complex is delayed, moves away from S1, and may even disappear. Some patients have a mid-systolic click without a murmur; others have a murmur without a click. Still others have both sounds at different times.
LABORATORY EXAMINATION
The ECG most commonly is normal but may show biphasic or inverted T waves in leads II, III, and aVF, and occasionally supraventricular or ventricular premature beats. TTE is particularly effective in identifying the abnormal position and prolapse of the mitral valve leaflets. A useful echocardiographic definition of MVP is systolic displacement (in the parasternal long axis view) of the mitral valve leaflets by at least 2 mm into the LA superior to the plane of the mitral annulus. Color flow and continuous wave Doppler imaging is helpful to evaluate the associated MR and provide semiquantitative estimates of severity. The jet lesion of MR due to MVP is most often eccentric, and assessment of RF and effective regurgitant orifice area can be difficult. TEE is indicated when more accurate information is required and is performed routinely for intraoperative guidance for valve repair. Invasive left ventriculography is rarely necessary but can also show prolapse of the posterior and sometimes of both mitral valve leaflets.
285 | Tricuspid and Pulmonic Valve Disease |
TRICUSPID STENOSIS
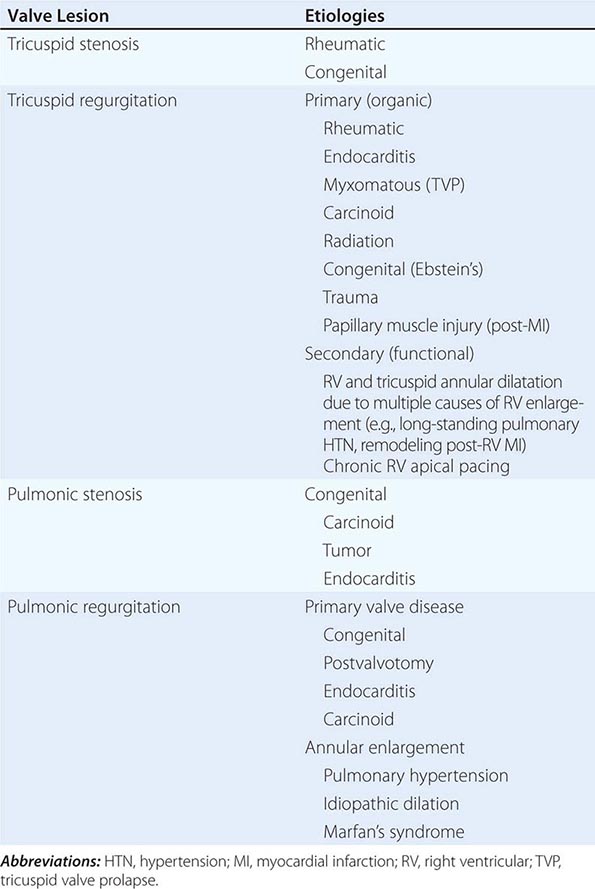
Tricuspid stenosis (TS), which is much less prevalent than mitral stenosis (MS) in North America and Western Europe, is generally rheumatic in origin, and is more common in women than men (Table 285-1). It does not occur as an isolated lesion and is usually associated with MS. Hemodynamically significant TS occurs in 5–10% of patients with severe MS; rheumatic TS is commonly associated with some degree of tricuspid regurgitation (TR). Nonrheumatic causes of TS are rare.
CAUSES OF TRICUSPID AND PULMONIC VALVE DISEASES |
PATHOPHYSIOLOGY
A diastolic pressure gradient between the right atrium (RA) and right ventricle (RV) defines TS. It is augmented when the transvalvular blood flow increases during inspiration and declines during expiration. A mean diastolic pressure gradient of 4 mmHg is usually sufficient to elevate the mean RA pressure to levels that result in systemic venous congestion. Unless sodium intake has been restricted and diuretics administered, this venous congestion is associated with hepatomegaly, ascites, and edema, sometimes severe. In patients with sinus rhythm, the RA a wave may be extremely tall and may even approach the level of the RV systolic pressure. The y descent is prolonged. The cardiac output (CO) at rest is usually depressed, and it fails to rise during exercise. The low CO is responsible for the normal or only slightly elevated left atrial (LA), pulmonary artery (PA), and RV systolic pressures despite the presence of MS. Thus, the presence of TS can mask the hemodynamic and clinical features of any associated MS.
SYMPTOMS
Because the development of MS generally precedes that of TS, many patients initially have symptoms of pulmonary congestion and fatigue. Characteristically, patients with severe TS complain of relatively little dyspnea for the degree of hepatomegaly, ascites, and edema that they have. However, fatigue secondary to a low CO and discomfort due to refractory edema, ascites, and marked hepatomegaly are common in patients with advanced TS and/or TR. In some patients, TS may be suspected for the first time when symptoms of right-sided failure persist after an adequate mitral valvotomy.
PHYSICAL FINDINGS
Because TS usually occurs in the presence of other obvious valvular disease, the diagnosis may be missed unless it is considered. Severe TS is associated with marked hepatic congestion, often resulting in cirrhosis, jaundice, serious malnutrition, anasarca, and ascites. Congestive hepatomegaly and, in cases of severe tricuspid valve disease, splenomegaly are present. The jugular veins are distended, and in patients with sinus rhythm, there may be giant a waves. The v waves are less conspicuous, and because tricuspid obstruction impedes RA emptying during diastole, there is a slow y descent. In patients with sinus rhythm, there may be prominent presystolic pulsations of the enlarged liver as well.
On auscultation, an opening snap (OS) of the tricuspid valve may rarely be heard approximately 0.06 s after pulmonic valve closure. The diastolic murmur of TS has many of the qualities of the diastolic murmur of MS, and because TS almost always occurs in the presence of MS, it may be missed. However, the tricuspid murmur is generally heard best along the left lower sternal border and over the xiphoid process, and is most prominent during presystole in patients with sinus rhythm. The murmur of TS is augmented during inspiration, and it is reduced during expiration and particularly during the strain phase of the Valsalva maneuver, when tricuspid transvalvular flow is reduced.
LABORATORY EXAMINATION
The electrocardiogram (ECG) features of RA enlargement (see Fig. 268-8) include tall, peaked P waves in lead II, as well as prominent, upright P waves in lead V1. The absence of ECG evidence of RV hypertrophy (RVH) in a patient with right-sided heart failure who is believed to have MS should suggest associated tricuspid valve disease. The chest x-ray in patients with combined TS and MS shows particular prominence of the RA and superior vena cava without much enlargement of the PA and with less evidence of pulmonary vascular congestion than occurs in patients with isolated MS. On echocardiographic examination, the tricuspid valve is usually thickened and domes in diastole; the transvalvular gradient can be estimated by continuous wave Doppler echocardiography. Severe TS is characterized by a valve area ≤1 cm2 or pressure half-time of ≥190 ms. The RA and inferior vena cava (IVC) are enlarged. Transthoracic echocardiography (TTE) provides additional information regarding the severity of any associated TR, mitral valve structure and function, left ventricle (LV) and RV size and function, and PA pressure. Cardiac catheterization is not routinely necessary for assessment of TS.
TRICUSPID REGURGITATION
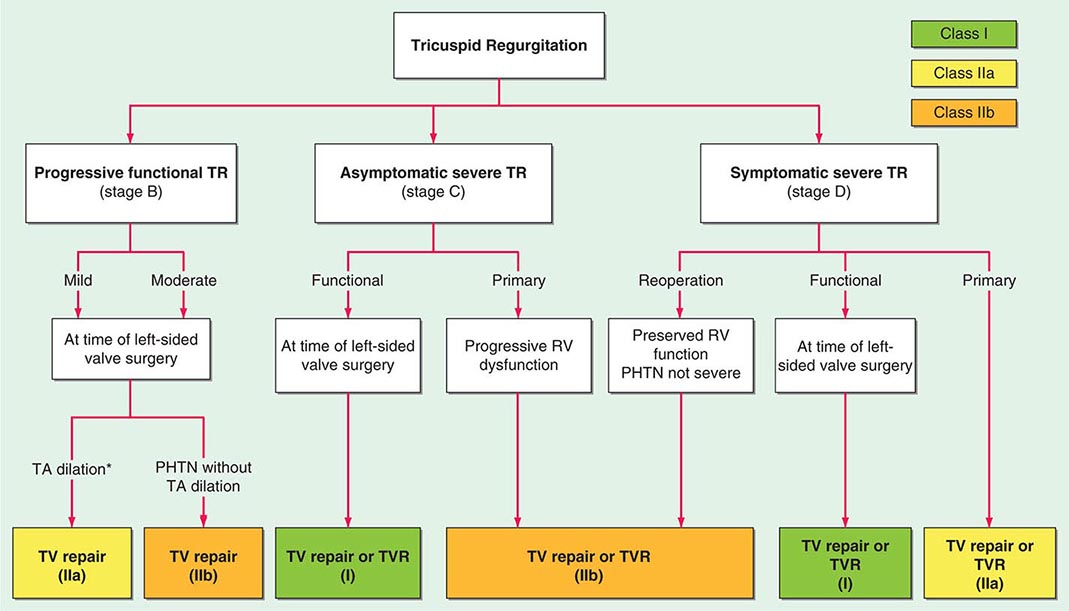
In at least 80% of cases, TR is secondary to marked dilation of the tricuspid annulus from RV enlargement due to PA hypertension (Table 285-1). Functional TR may complicate RV enlargement of any cause, however, including an inferior myocardial infarction (MI) that involves the RV. It is commonly seen in the late stages of heart failure due to rheumatic or congenital heart disease with severe PA hypertension (PA systolic pressure >55 mmHg), as well as in ischemic and idiopathic dilated cardiomyopathies. It is reversible in part if PA hypertension can be relieved. Functional TR can also develop from chronic RV apical pacing. Rheumatic fever may produce primary (organic) TR, often associated with TS. Infarction of RV papillary muscles, tricuspid valve prolapse, carcinoid heart disease, endomyocardial fibrosis, radiation, infective endocarditis, and leaflet trauma all may produce TR. Less commonly, TR results from congenitally deformed tricuspid valves, and it occurs with defects of the atrioventricular canal, as well as with Ebstein’s malformation of the tricuspid valve (Chap. 282).
PATHOPHYSIOLOGY
The incompetent tricuspid valve allows blood to flow backward from the RV into the RA, the volume of which is dependent on the driving pressure (i.e., RV systolic pressure) and the size of the regurgitant orifice. The severity and physical signs of TR can vary as a function of PA systolic pressure (in the absence of RV outflow tract stenosis), the dimension of the tricuspid valve annulus, the respiratory cycle–dependent changes in RV preload, and RA compliance. RV filling is increased during inspiration. Forward CO is reduced and does not augment with exercise. Significant degrees of TR will lead to RA enlargement and elevation of the RA and jugular venous pressures with prominent c-v waves in the pulse tracings. Progressively severe TR can lead to “ventricularization” of the RA wave form (see Fig. 267-1B). Severe TR is also characterized by RV dilation (RV volume overload) and eventual systolic dysfunction, the rate of which can be accelerated by a concomitant pressure load from PA hypertension or by myocardial fibrosis from previous injury.
SYMPTOMS
Mild or moderate degrees of TR are usually well tolerated in the absence of other hemodynamic disturbances. Because TR most often coexists with left-sided valve lesions, LV dysfunction, and/or PA hypertension, symptoms related to these lesions may dominate the clinical picture. Fatigue and exertional dyspnea owing to reduced forward CO are early symptoms of isolated, severe TR. As the disease progresses and RV function declines, patients may report cervical pulsations, abdominal fullness/bloating, diminished appetite, and muscle wasting, although with progressive weight gain and painful swelling of the lower extremities.
PHYSICAL FINDINGS
The neck veins in patients with severe TR are distended with prominent c-v waves and rapid y descents (in the absence of TS). TR is more often diagnosed by examination of the neck veins than by auscultation of the heart sounds. Other findings may include marked hepatomegaly with systolic pulsations, ascites, pleural effusions, edema, and a positive hepatojugular reflex. A prominent RV pulsation along the left parasternal region and a blowing holosystolic murmur along the lower left sternal margin, which may be intensified during inspiration (Carvallo’s sign) and reduced during expiration or the strain phase of the Valsalva maneuver, are characteristic findings. The murmur of TR may sometimes be confused with that of MR unless attention is paid to its variation during the respiratory cycle and the extent of RV enlargement is appreciated. Atrial fibrillation (AF) is usually present in the chronic phase of the disease.
LABORATORY EXAMINATION
The ECG may show changes characteristic of the lesion responsible for the TR, e.g., an inferior Q-wave MI suggestive of a prior RV MI, RVH, or a bizarre right bundle branch block type pattern with preexcitation in patients with Ebstein’s anomaly. ECG signs of RA enlargement may be present in patients with sinus rhythm; AF is frequently noted. The chest x-ray may show RA and RV enlargement, depending on the chronicity and severity of TR. TTE is usually definitive with demonstration of RA dilation and RV volume overload and prolapsing, flail, scarred, or displaced/tethered tricuspid leaflets; the diagnosis and assessment of TR can be made by color flow Doppler imaging (see Fig. 270e-8). Severe TR is accompanied by hepatic vein systolic flow reversal. Continuous wave Doppler of the TR velocity profile is useful in estimating PA systolic pressure. Accurate assessment of TR severity, PA pressures, and RV size and systolic function with TTE can be quite challenging in many patients. Real-time three-dimensional echocardiography and cardiac magnetic resonance (CMR) imaging provide alternative imaging modalities, although they are not widely available. In patients with severe TR, the CO is usually markedly reduced, and the RA pressure pulse may exhibit no x descent during early systole but a prominent c-v wave with a rapid y descent. The mean RA and RV end-diastolic pressures are often elevated. Exercise testing can be used to assess functional capacity in patients with asymptomatic severe TR. The prognostic significance of exercise-induced changes in TR severity and RV function has not been well studied.
PULMONIC STENOSIS
Pulmonic valve stenosis (PS) is essentially a congenital disorder (Table 285-1). With isolated PS, the valve is typically domed. Dysplastic pulmonic valves are seen as part of the Noonan’s syndrome (Chap. 302), which maps to chromosome 12. Much less common etiologies include carcinoid and obstructing tumors or bulky vegetations. The pulmonic valve is only very rarely affected by the rheumatic process.
PATHOPHYSIOLOGY
PS is defined hemodynamically by a systolic pressure gradient between the RV and main PA. RV hypertrophy develops as a consequence of sustained obstruction to RV outflow, and systolic ejection is prolonged. Compared with the ability of the LV to compensate for the pressure overload imposed by aortic stenosis (AS), RV dysfunction from afterload mismatch occurs earlier in the course of PS and at lower peak systolic pressures, because the RV adapts less well to this type of hemodynamic burden. With normal systolic function and CO, severe PS is defined by a peak systolic gradient across the pulmonic valve of >50 mmHg; moderate PS correlates with a peak gradient of 30–50 mmHg. PS rarely progresses in patients with peak gradients less than 30 mmHg, but may worsen in those with moderate disease due to valve thickening and calcification with age. The RA a wave elevates in relation to the higher pressures needed to fill a noncompliant, hypertrophied RV. A prominent RA v wave signifies functional TR from RV and annular dilation. The CO is maintained until late in the course of the disease.
SYMPTOMS
Patients with mild or even moderate PS are usually asymptomatic and first come to medical attention because of a heart murmur that leads to echocardiography. With severe PS, patients may report exertional dyspnea or early-onset fatigue. Anginal chest pain from RV oxygen supply-demand mismatch and syncope may occur with very severe forms of obstruction, particularly in the presence of a destabilizing trigger such as atrial fibrillation, fever, infection, or anemia.
PHYSICAL FINDINGS
The murmur of mild or moderate PS is mid-systolic in timing, crescendo-decrescendo in configuration, heard best in the left second interspace, and usually introduced by an ejection sound (click) in younger adults whose valves are still pliable. The ejection sound is the only right-sided acoustic event that decreases in intensity with inspiration. This phenomenon reflects premature opening of the pulmonic valve by the elevated RV end-diastolic (postatrial a wave) pressure. The systolic murmur increases in intensity during inspiration. With progressively severe PS, the ejection sound moves closer to the first heart sound and eventually becomes inaudible. A right-sided fourth heart sound may emerge. The systolic murmur peaks later and may persist through the aortic component of the second heart sound (A2). Pulmonic valve closure is delayed, and the pulmonic component of the second heart sound (P2) is reduced or absent. A prominent a wave, indicative of the higher atrial pressure necessary to fill the noncompliant RV, may be seen in the jugular venous pulse. A parasternal or RV lift can be felt with significant pressure overload. Signs of right heart failure, such as hepatomegaly, ascites, and edema, are uncommon but may appear very late in the disease.
LABORATORY EXAMINATION
The ECG will show right axis deviation, RVH, and RA enlargement in adult patients with severe PS. Chest x-ray findings include poststenotic dilation of the main PA in the frontal plane projection and filling of the retrosternal airspace due to RV enlargement on the lateral film. In some patients with RVH, the cardiac apex appears to be lifted off the left hemidiaphragm. The RA may also be enlarged. TTE allows definitive diagnosis and characterization in most cases, with depiction of the valve and assessment of the gradient, RV function, PA pressures (which should be low), and any associated cardiac lesions. TEE may be useful in some patients for improved delineation of the RV outflow tract (RVOT) and assessment of infundibular hypertrophy. Cardiac catheterization is not usually necessary, but if performed, pressures should be obtained from just below and above the pulmonic valve with attention to the possibility that a dynamic component to the gradient may exist. The correlation between Doppler assessment of peak instantaneous gradient and catheterization-measured peak-to-peak gradient is weak. The latter may correlate better with the Doppler mean gradient.
PULMONIC REGURGITATION
Pulmonic regurgitation (PR) may develop as a consequence of primary valve pathology, annular enlargement, or their combination; after surgical treatment of RVOT obstruction in children with such disorders as tetralogy of Fallot; or after pulmonic balloon valvotomy (Table 285-1). Carcinoid usually causes mixed pulmonic valve disease with PR and PS. Long-standing severe PA hypertension from any cause can result in dilation of the pulmonic valve ring and PR.
PATHOPHYSIOLOGY
Severe PR results in RV chamber enlargement and eccentric hypertrophy. As is the case for aortic regurgitation (AR), PR is a state of increased preload and afterload. The reverse pressure gradient from the PA to the RV, which drives the PR, progressively decreases throughout diastole and accounts for the decrescendo nature of the diastolic murmur. As RV diastolic pressure increases, the murmur becomes shorter in duration. The forward CO is preserved during the early stages of the disease, but may not increase normally with exercise and declines over time. A reduction in RV ejection fraction may be an early indicator of hemodynamic compromise. In advanced stages, there is significant enlargement of the RV and RA with marked elevation of the jugular venous pressure.
SYMPTOMS
Mild or moderate degrees of PR do not, by themselves, result in symptoms. Other problems, such as PA hypertension, may dominant the clinical picture. With progressively severe PR and RV dysfunction, fatigue, exertional dyspnea, abdominal fullness/bloating, and lower extremity swelling may be reported.
PHYSICAL FINDINGS
The physical examination hallmark of PR is a high-pitched, decrescendo diastolic murmur (Graham Steell murmur) heard along the left sternal border that can be difficult to distinguish from the more frequently appreciated murmur of aortic regurgitation. The Graham Steell murmur may become louder with inspiration and is usually associated with a loud and sometimes palpable P2 and an RV lift, as would be expected in patients with significant PA hypertension of any cause. Survivors of childhood surgery for tetralogy of Fallot or PS/pulmonary atresia may have an RV-PA conduit that is freely regurgitant because it does not contain a valve. PA pressures in these individuals are not elevated and the diastolic murmur can be misleadingly low pitched and of short duration despite significant degrees of PR and RV volume overload.
LABORATORY EXAMINATION
Depending on both the etiology and severity of PR, the ECG may show findings of RVH and RA enlargement. On chest x-ray, the RV and RA may be enlarged. Pulmonic valve morphology and function can be assessed with transthoracic Doppler echocardiography. PA pressures can be estimated from the tricuspid valve systolic jet velocity. CMR provides greater anatomic detail, particularly in patients with repaired congenital heart disease, and more precise assessment of RV volumes. Cardiac catheterization is not routinely necessary but would be performed as part of a planned transcatheter procedure.
286 | Multiple and Mixed Valvular Heart Disease |
Many acquired and congenital cardiac lesions may result in stenosis and/or regurgitation of one or more heart valves. For example, rheumatic heart disease can involve the mitral (mitral stenosis [MS], mitral regurgitation [MR], or MS and MR), aortic (aortic stenosis [AS], aortic regurgitation [AR], or AS and AR), and/or tricuspid (tricuspid stenosis [TS], tricuspid regurgitation [TR], or TS and TR) valve, alone or in combination. The common association of functional TR with significant mitral valve disease is discussed in Chap. 285. Severe mitral annular calcification can result in regurgitation (due to decreased annular shortening during systole) and mild stenosis (caused by extension of the calcification onto the leaflets resulting in restricted valve opening). Patients with severe AS may develop functional MR that may not improve after isolated aortic valve replacement (AVR). Chordal rupture has been described infrequently in patients with severe AS. Aortic valve infective endocarditis may secondarily involve the mitral apparatus either by abscess formation and contiguous spread via the intervalvular fibrosa or by “drop metastases” from the aortic leaflets onto the anterior leaflet of the mitral valve. Mediastinal radiation may result in aortic, mitral, and even tricuspid valve disease, most often with mixed stenosis and regurgitation. Carcinoid heart disease may cause mixed lesions of either or both the tricuspid and pulmonic valves. Ergotamines, and the previously used combination of fenfluramine and phentermine, can rarely result in mixed lesions of the aortic and/or mitral valve. Patients with Marfan’s syndrome may have both AR from aortic root dilation and MR due to mitral valve prolapse (MVP). Myxomatous degeneration causing prolapse of multiple valves (mitral, aortic, tricuspid) can also occur in the absence of an identifiable connective tissue disorder. Bicuspid aortic or pulmonic valve disease can result in mixed stenosis and regurgitation.
PATHOPHYSIOLOGY
In patients with multivalvular heart disease, the pathophysiologic derangements associated with the more proximal valve disease can mask the full expression of the attributes of the more distal valve lesion. For example, in patients with rheumatic mitral and aortic valve disease, the reduction in cardiac output (CO) imposed by the mitral valve disease will decrease the magnitude of the hemodynamic derangements related to the severity of the aortic valve lesion (stenotic, regurgitant, or both). Alternatively, the development of atrial fibrillation (AF) during the course of MS can lead to sudden worsening in a patient whose aortic valve disease was not previously felt to be significant. The development of reactive pulmonary vascular disease, sometimes referred to as a “secondary obstructive lesion in series,” can impose an additional challenge in these settings. As CO falls with progressive tricuspid valve disease, the severity of any associated mitral or aortic disease can be underestimated.
One of the most common examples of multivalve disease is that of functional TR in the setting of significant mitral valve disease. Functional TR occurs as a consequence of right ventricular and annular dilation; pulmonary artery (PA) hypertension is often present. The tricuspid leaflets are morphologically normal. Progressive degrees of TR lead to right ventricular volume overload and continued chamber and annular dilation. The TR is usually central in origin; reflux into the right atrium (RA) is expressed as large, systolic c-v waves in the RA pressure pulse. The height of the c-v wave is dependent on RA compliance and the volume of regurgitant flow. The RA wave form may become “ventricularized” in advanced stages of chronic, severe TR with PA hypertension. CO falls and the severity of the associated mitral valve disease may become more difficult to appreciate. Primary rheumatic tricuspid valve disease may occur with rheumatic mitral disease and cause hemodynamic changes reflective of TR, TS, or their combination. With TS, the y descent in the RA pressure pulse is prolonged.
Another example of rheumatic, multivalve disease involves the combination of mitral and aortic valve pathology, frequently characterized by MS and AR. In isolated MS, left ventricular (LV) preload and diastolic pressure are reduced as a function of the severity of inflow obstruction. With concomitant AR, however, LV filling is enhanced and diastolic pressure may rise depending on the compliance characteristics of the chamber. Because the CO falls with progressive degrees of MS, transaortic valve flows will decline, masking the potential severity of the aortic valve lesion (AR, AS, or its combination). As noted above, onset of AF in such patients can be especially deleterious.
Functional MR may complicate the course of some patients with severe AS. The mitral valve leaflets and chordae tendineae are usually normal. Incompetence is related to changes in LV geometry (remodeling) and abnormal systolic tethering of the leaflets in the context of markedly elevated LV systolic pressures. Relief of the excess afterload with surgical or transcatheter AVR often, but not always, results in reduction or elimination of the MR. Persistence of significant MR following AVR is associated with impaired functional outcomes and reduced survival. Identification of patients who would benefit from concomitant treatment of their functional MR at time of AVR is quite challenging. Most surgeons advocate for repair of moderate-to-severe or severe functional MR at time of surgical AVR.
In patients with mixed AS and AR, assessment of valve stenosis can be influenced by the magnitude of the regurgitant valve flow. Because transvalvular systolic flow velocities are augmented in patients with AR and preserved LV systolic function, the LV-aortic Doppler-derived pressure gradient and the intensity of the systolic murmur will be elevated to values higher than expected for the true systolic valve orifice size as delineated by planimetry. Uncorrected, the Gorlin formula, which relies on forward CO (systolic transvalvular flow) and the mean pressure gradient for calculation of valve area, is not accurate in the setting of mixed aortic valve disease. Similar considerations apply to patients with mixed mitral valve disease. The peak mitral valve Doppler E wave velocity (v0) is increased in the setting of severe MR because of enhanced early diastolic flow and may not accurately reflect the contribution to left atrial (LA) hypertension from any associated MS. When either AR or MR is the dominant lesion in patients with mixed aortic or mitral valve disease, respectively, the LV is dilated. When AS or MS predominates, LV chamber size will be normal or small. It can sometimes be difficult to ascertain whether stenosis or regurgitation is the dominant lesion in patients with mixed valve disease, although an integrated clinical and noninvasive assessment can usually provide clarification for purposes of patient management and follow-up.
Patients with significant AS, a nondilated LV chamber, and concentric hypertrophy will poorly tolerate the abrupt development of aortic regurgitation, as may occur, for example, with infective endocarditis or after surgical or transcatheter AVR complicated by paravalvular leakage. The noncompliant LV is not prepared to accommodate the sudden volume load, and as a result, LV diastolic pressure rises rapidly and severe heart failure develops. Indeed, paravalvular regurgitation is a significant risk factor for short- to intermediate-term death following transcatheter AVR. Conditions in which the LV may not be able to dilate in response to chronic AR (or MR) include radiation heart disease and, in some patients, the cardiomyopathy associated with obesity and diabetes. Noncompliant ventricles of small chamber size predispose to earlier onset diastolic dysfunction and heart failure in response to any further perturbation in valve function.
SYMPTOMS
Compared with patients with isolated, single-lesion valve disease, patients with multiple or mixed valve disease may develop symptoms at a relatively earlier stage in the natural history of their disease. Symptoms such as exertional dyspnea and fatigue are usually related to elevated filling pressures, reduced CO, or their combination. Palpitations may signify AF and identify mitral valve disease as an important component of the clinical presentation, even when not previously suspected. Chest pain compatible with angina could reflect left or right ventricular oxygen supply/demand mismatch on a substrate of hypertrophy and pressure/volume overload with or without superimposed coronary artery disease. Symptoms related to right heart failure (abdominal fullness/bloating, edema) are late manifestations of advanced disease.
PHYSICAL FINDINGS
Mixed disease of a single valve is most often manifested by systolic and diastolic murmurs, each with the attributes expected for the valve in question. Thus, patients with AS and AR will have characteristic mid-systolic, crescendo-decrescendo and blowing, decrescendo diastolic murmurs at the base of the heart in the second right interspace and along the left sternal edge, respectively. Many patients with significant AR have mid-systolic outflow murmurs even in the absence of valve sclerosis/stenosis, and other findings of AS must be sought. The separate murmurs of AS and AR can occasionally be difficult to distinguish from the continuous murmurs associated with either a patent ductus arteriosus (PDA) or ruptured sinus of Valsalva aneurysm. With mixed aortic valve disease, the systolic murmur should end before, and not envelope or extend through, the second heart sound (S2). The murmur associated with a PDA is heard best to the left of the upper sternum. The continuous murmur heard with a ruptured sinus of Valsalva aneurysm is often first appreciated after an episode of acute chest pain. An early ejection click, which usually defines bicuspid aortic valve disease in young adults, is often not present in patients with congenital, mixed AS and AR. As noted above, both the intensity and duration of these separate murmurs can be influenced by a reduction in CO and transvalvular flow due to coexistent mitral valve disease. In patients with isolated MS and MR, expected findings would include a blowing, holosystolic murmur and a mid-diastolic rumble (with or without an opening snap) best heard at the cardiac apex. An irregularly irregular heart rhythm in such patients would likely signify AF. Findings with TS and TR would mimic those of left-sided MS and MR, save for the expected changes in the murmurs with respiration. The murmurs of pulmonic stenosis and regurgitation behave in a fashion directionally similar to AS and AR; dynamic changes during respiration should be noted. Specific attributes of these cardiac murmurs are reviewed in Chap. 285.
LABORATORY EXAMINATION
The electrocardiogram (ECG) may show evidence of ventricular hypertrophy and/or atrial enlargement. ECG signs indicative of right-sided cardiac abnormalities in patients with left-sided valve lesions should prompt additional assessment for PA hypertension and/or right-sided valve disease. The presence of AF in patients with aortic valve disease may be a clue to the presence of previously unsuspected mitral valve disease in the appropriate context. The chest x-ray can be reviewed for evidence of cardiac chamber enlargement, valve and/or annular calcification, and any abnormalities in the appearance of the pulmonary vasculature. The latter could include enlargement of the main and proximal pulmonary arteries with PA hypertension and pulmonary venous redistribution/engorgement or Kerley B lines with increasing degrees of LA hypertension. An enlarged azygos vein in the frontal projection indicates RA hypertension. Roentgenographic findings not expected based on a single or mixed valve lesion may reflect other valve disease.
Transthoracic echocardiography (TTE) is the most commonly used imaging modality for the diagnosis and characterization of multiple and/or mixed valvular heart disease and may often demonstrate findings not clinically suspected. Transesophageal echocardiography (TEE) may sometimes be required for more accurate assessment of valve anatomy (specifically, the mitral valve) and when infective endocarditis (IE) is considered responsible for the clinical presentation. TTE findings of particular interest include those related to valve morphology and function, calcification, chamber size, ventricular wall thickness, estimated PA systolic pressure, and the dimensions of the great vessels, including the root and ascending aorta, PA, and inferior vena cava. Exercise testing (with or without echocardiography) can be useful when the degree of functional limitation reported by the patient is not adequately explained by the findings on TTE performed at rest. An integrated assessment of the clinical and TTE findings is needed to help determine the dominant valve lesion(s) and establish an appropriate plan for treatment and follow-up. Natural history is usually influenced to a relatively greater degree by the dominant lesion. Exercise testing (with or without echocardiography) can
Cardiac magnetic resonance (CMR) can be used to provide additional anatomic and physiologic information when echocardiography proves suboptimal, but is less well suited to the evaluation of valve morphology. Cardiac computed tomography (CT) has been used to assess intracardiac structures in patients with complicated IE. Coronary CT angiography provides a noninvasive alternative for the assessment of coronary artery anatomy prior to surgery.
Invasive hemodynamic evaluation with right and left heart catheterization may be required to characterize more completely the individual contributions of each lesion in patients with either multiple or mixed valvular heart disease. Measurement of PA pressures and calculation of pulmonary vascular resistance (PVR) can help inform clinical decision-making in certain patient subsets, such as those with advanced mitral and tricuspid valve disease. Attention to the accurate assessment of CO is essential. Coronary angiography (if indicated) can be performed as part of the procedure. Contrast ventriculography and great vessel angiography are performed infrequently.
287 | Cardiomyopathy and Myocarditis |
DEFINITION AND CLASSIFICATION
Cardiomyopathy is disease of the heart muscle. It is estimated that cardiomyopathy accounts for 5–10% of the heart failure in the 5–6 million patients carrying that diagnosis in the United States. This term is intended to exclude cardiac dysfunction that results from other structural heart disease, such as coronary artery disease, primary valve disease, or severe hypertension; however, in general usage, the phrase ischemic cardiomyopathy is sometimes applied to describe diffuse dysfunction attributed to multivessel coronary artery disease, and nonischemic cardiomyopathy to describe cardiomyopathy from other causes. As of 2006, cardiomyopathies are defined as “a heterogeneous group of diseases of the myocardium associated with mechanical and/or electrical dysfunction that usually (but not invariably) exhibit inappropriate ventricular hypertrophy or dilatation and are due to a variety of causes that frequently are genetic.”1
The traditional classification of cardiomyopathies into a triad of dilated, restrictive, and hypertrophic was based initially on autopsy specimens and later on echocardiographic findings. Dilated and hypertrophic cardiomyopathies can be distinguished on the basis of left ventricular wall thickness and cavity dimension; however, restrictive cardiomyopathy can have variably increased wall thickness and chamber dimensions that range from reduced to slightly increased, with prominent atrial enlargement. Restrictive cardiomyopathy is now defined more on the basis of abnormal diastolic function, which is also present but initially less prominent in dilated and hypertrophic cardiomyopathy. Restrictive cardiomyopathy can overlap in presentation, gross morphology, and etiology with both hypertrophic and dilated cardiomyopathies (Table 287-1).
PRESENTATION WITH SYMPTOMATIC CARDIOMYOPATHY |
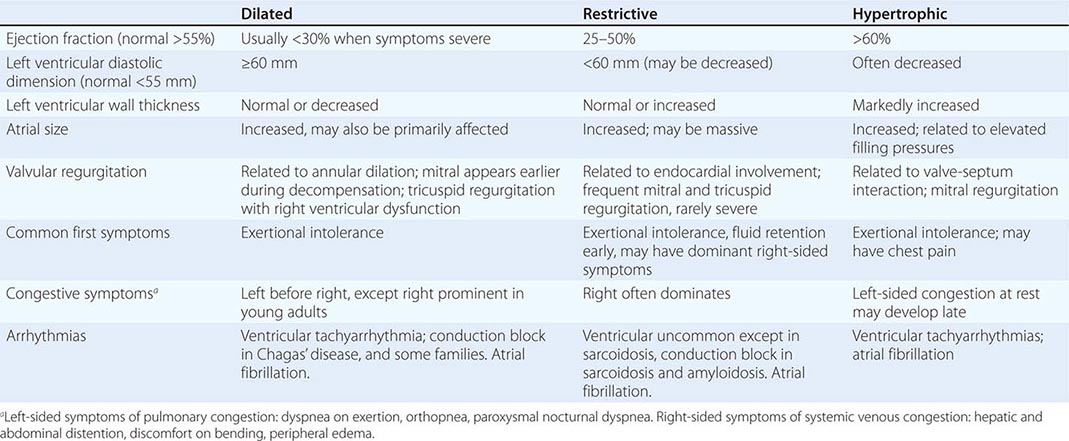
Expanding information renders this classification triad based on phenotype increasingly inadequate to define disease or therapy. Identification of more genetic determinants of cardiomyopathy has suggested a four-way classification scheme of etiology as primary (affecting primarily the heart) and secondary to other systemic disease. The primary causes are then divided into genetic, mixed genetic and acquired, and acquired; however, genetic information is often unavailable at the time of initial presentation, the phenotypic expression of a given mutation varies widely, and genetic predisposition influences the clinical phenotype of acquired cardiomyopathies, as well. Although the proposed genetic classification does not yet guide many current clinical strategies, it will likely become increasingly relevant as classification of disease moves beyond individual organ pathology to more integrated systems approaches.
GENERAL PRESENTATION
For all cardiomyopathies, the early symptoms often relate to exertional intolerance with breathlessness or fatigue, usually from inadequate cardiac reserve during exercise. These symptoms may initially go unnoticed or be attributed to other causes, commonly lung disease or age-dependent exercise limitation. As fluid retention leads to elevation of resting filling pressures, shortness of breath may occur during routine daily activity such as dressing and may manifest as dyspnea or cough when lying down at night. Although often considered the hallmark of congestion, peripheral edema may be absent despite severe fluid retention, particularly in younger patients in whom ascites and abdominal discomfort may dominate. The nonspecific term congestive heart failure describes only the resulting syndrome of fluid retention, which is common to all three types of cardiomyopathy and also to cardiac structural diseases associated with elevated filling pressures. All three types of cardiomyopathy can be associated with atrioventricular valve regurgitation, typical and atypical chest pain, atrial and ventricular tachyarrhythmias, and embolic events (Table 287-1). Initial evaluation begins with a detailed clinical history and examination, looking for clues to cardiac, extracardiac, and familial disease (Table 287-2).
INITIAL EVALUATION OF CARDIOMYOPATHY |