Chapter 30 Supplement One Health
Interdependence of People, Other Species, and the Planet
Applications of One Health to Millennium Development Goals
The One Health approach can provide integrated and collaborative solutions to a number of important global health and sustainability challenges. As an example of its widespread applicability, we highlight the Millennium Development Goals (MDGs, www.un.org/millenniumgoals), one of the most globally accepted development metric paradigms. The United Nations spearheaded the MDGs to address poverty, education, equity, mortality, sustainability, and health. The eight targets for the MDGs address wide-reaching and complex global issues, and as such, they require multisectoral, integrated approaches. Adopting a One Health approach could help in achieving the MDGs by strengthening cross-sectoral collaboration and approaching problems with a preventive focus.1–3 In fact, six of the eight MDGs could benefit from a strategic application of the One Health approach (Table S30-1).
Table S30-1 Millennium Development Goals as Defined by the United Nations
| Millennium Development Goals | Potential Benefits of One Health Approach |
|---|---|
| Goal 1: Eradicate extreme poverty and hunger.* | Improved crop agriculture and livestock production; better understanding of how climate change will affect food security. |
| Goal 2: Achieve universal primary education. | Indirect relevance. |
| Goal 3: Promote gender equality and empower women. | Indirect relevance. |
| Goal 4: Reduce child mortality.* | Reduce diarrheal infections, one of the biggest killers of children, by improving water quality and food safety. |
| Goal 5: Improve maternal health.* | Improve water quality and food safety; reduce use of biomass for fuel and promote use of alternative, cleaner stoves. |
| Goal 6: Combat HIV/AIDS, malaria, and other diseases.* | Understand the environmental and behavioral drivers of disease emergence; approaches to vector control; and relevance of animal reservoirs of disease. |
| Goal 7: Ensure environmental sustainability.* | Reduce the rate of environmental degradation; incorporate more efficient, less costly, and less environmentally damaging agricultural and industrial methods; recognize the importance of addressing climate change. |
| Goal 8: Develop a global partnership for development.* | Integrate health, environmental stewardship, energy, trade, business, and public infrastructure systems to improve health. |
*These goals could benefit from applications of a One Health approach.
ONE HEALTH CASE STUDY 1 Deforestation, Intensive Livestock Production, and Nipah Virus Emergence4
An outbreak of a novel paramyxovirus, the Nipah virus, struck Malaysia in late September of 1998. Although the virus is native to fruit bats (Pteropodidae family),5 unusually close contact between bats and swine during 1998 allowed the virus to jump species. Those in contact with infected swine quickly became ill, and the virus rapidly spread across peninsular Malaysia and into Singapore through the transport of infected pigs (Fig. S30-1). By the time the outbreak was contained in May 1999, 105 individuals had died, most of whom were directly involved with swine farming. Additionally, more than 1.1 million pigs had been slaughtered at a cost of $97 million, effectively devastating Malaysia’s swine industry.
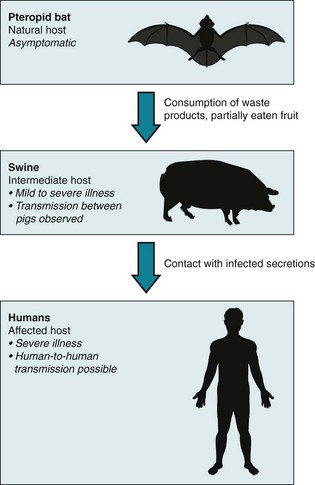
Figure S30-1 The Nipah virus transmission cycle in Malaysia, 1998-99.
(Modified from Nadimpalli M, Akoroda U, Williams JT: Nipah virus in Malaysia, 1998-99: a One Health perspective. In Barrett MA, Sackey-Harris M, Stroud C, editors. Applications of the One Health approach to current health and sustainability challenges: an educational resource, vol 1, Durham, NC, Duke University, University of North Carolina, North Carolina State University [In press].)
Retrospectively, it was determined that ineffective control measures and poor disease surveillance greatly exacerbated the spread and severity of disease. The novel exposure of humans to Nipah virus was caused by a unique combination of environmental, animal, and human factors: deforestation, forest fires, and a drought in 1998 are thought to have forced fruit bats to concentrate in fruit orchards in northern Malaysia.6,7 The proximity of these orchards to pig nurseries allowed for the spillover of Nipah virus from bats to pigs,8 and unprotected physical contact between pigs and pig farmers allowed the virus to rapidly infect humans. Unfortunately, the disconnect among Malaysia’s human, animal, and environmental health entities made recognizing Nipah virus as the causative agent of the outbreak particularly complicated for the Malaysian government. A One Health approach involving interdisciplinary collaborations among these entities could have resulted in more rapid identification of the outbreak and implementation of more suitable control measures, saving lives.
Fruit bats are native to several countries in or proximal to Southeast Asia, including Indonesia, Madagascar, India, Bangladesh, China, Thailand, Cambodia, Papua New Guinea, and Australia. Bats in all these countries have tested seropositive for either Nipah virus or Hendra virus, a closely related paramyxovirus also capable of infecting humans.9 There is potential for overlap in distribution of Hendra and Nipah viruses and for pteropid bats to act as vectors for long-distance transmission to humans or animals.10 Although the 1998-99 Nipah virus outbreak affected only Malaysia and Singapore, future outbreaks of Nipah virus could occur in any country within the geographic range of these bats. Pteropid bats migrate in response to available food sources, and their movements do not recognize national boundaries. Human outbreaks have recently occurred in both India and Bangladesh, for example, from the consumption of contaminated fruit and fruit products.9 A One Health approach could be invaluable to any country dealing with or hoping to prevent an outbreak of Nipah virus in the future.
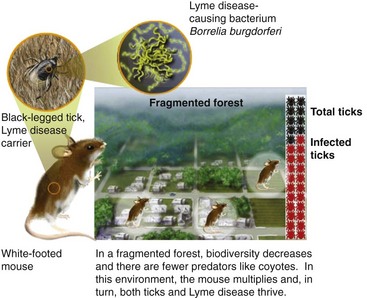
ONE HEALTH CASE STUDY 2 Biodiversity Loss, Land Use, and Lyme Disease11
Animal-Human-Ecosystem Dynamics
Lyme disease is transmitted to humans and domesticated animals by certain species of ticks from wildlife. Although the geographic range of the bacterium causing Lyme disease has expanded and contracted for millennia with environmental change (i.e., interglacials) in Eurasia and North America, the Lyme epidemic in the United States was likely caused by human-induced changes in land use. One example includes farmland reverted to woodland as the result of changing economics and policy in agriculture. This caused increased deer and tick populations and a culture of outdoor recreational activity in these woodlands, which enabled greater interaction of people with infected ticks. As development increased forest fragmentation, exposure of humans to infected ticks also increased (Fig. S30-2). Evolution of B. burgdorferi for different reservoir hosts has resulted in genetic variants that cause different disease entities in humans.11a
ONE HEALTH CASE STUDY 3 Rift Valley Fever at the Interface of Humans, Domestic Animals, and the Environment11
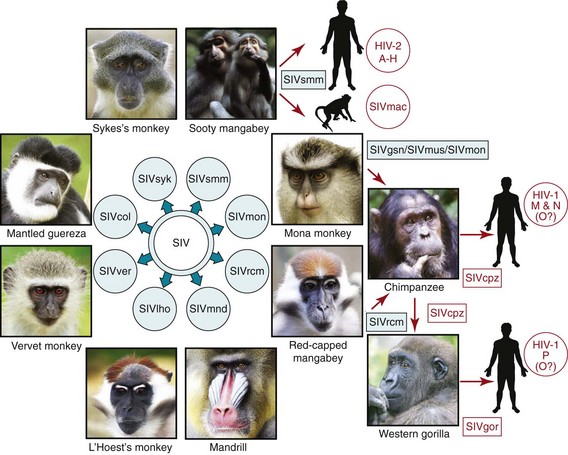
ONE HEALTH CASE STUDY 4 Origins of Human Immunodeficiency Virus
Two types of human immunodeficiency virus (HIV) can infect humans: HIV-1, which causes the majority of HIV infections worldwide, and HIV-2, which is largely geographically confined to West Africa.12,13 HIV-1 has infected more than 60 million people worldwide and has resulted in more than 25 million deaths.14 Both types have been traced back to simian immunodeficiency viruses (SIVs) endemic in more than 26 different species of nonhuman primates15 (Fig. S30-3). The pandemic strain of HIV-1 (group M) is most closely related to SIV documented in chimpanzees and originated from one distinct cross-species transmission event. HIV-2 is most closely related to SIV from wild sooty mangabeys. Based on banked human blood, tissue samples and estimates of viral mutation rates, scientists have calculated that the HIV-1 (group M) jump from chimpanzees to humans occurred in central Africa during the late 19th or early 20th century, a time of rapid urbanization and social change in the region.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree