| USAN: | Olanzapine |
| Brand Name: | Zyprexa (Eli Lilly) |
| Molecular Weight: | 312.43 |
| FDA Approval: | 1996 |
| Drug Class: | Atypical Antipsychotic Drug |
| Indications: | Schizophrenia and Bipolar Disorder |
| Mechanism of Action: | Serotonin–Dopamine Antagonist (SDA) |
■ 1 History of Schizophrenia and Antipsychotic Drugs
Schizophrenia is a devastating mental disorder, inflicting 1.1% of the population over the age of 18, which translates to 51 million people worldwide including 2.2 million people in the United States. Schizophrenia strikes men and women at different ages; the average onset age is 18 for men and 25 for women.
Schizophrenia is characterized by positive symptoms such as delusions, hallucinations, and disorganized speech/behavior and negative symptoms including apathy, withdrawal, lack of pleasure, and impaired attention.1 While it might be relatively easy to treat positive symptoms with antipsychotics, negative symptoms are more difficult to treat. Other symptoms include depressive/anxious symptoms and aggressive symptoms such as hostility, verbal and physical abusiveness, and impulsivity. Because of schizophrenia’s complexity, it is challenging to find therapeutics that are both efficacious and safe, especially considering that the patient has to take them for a prolonged period of time.2
In ancient times, an “insane” person was often thought to be possessed by the devil, or he was being punished by God for his sins. As a consequence, beating, bleeding, starvation, hot- and cold-water shock treatment, restraint, and incarceration were widely practiced on mental patients, which only worsened their conditions. In 1927, Austrian neurologist Julius Wagner von Jauregg invented the fever shock treatment, introducing malaria in psychotics. Egaz Moniz invented the lobotomy to “introduce an organic syndrome” for the treatment of schizophrenia; both men won the Nobel Prize (for Physiology or Medicine). Today, neither malaria introduction nor lobotomy is still in use.3
Although there is still a certain stigma attached to mental illnesses, we have now amassed a tremendous amount of knowledge with regard to the genetic, biochemical, and environmental impacts on the human brain. Psychopharmacological drugs such as olanzapine (Zyprexa, 1) have significantly contributed to managing and understanding mental diseases including schizophrenia.
1.1 Typical Antipsychotics—The First Generation
Before chlorpromazine (2) became available in 1962, early treatments for schizophrenia included prolonged narcosis, known as “narcosis for psychosis.” Meanwhile, history also saw the use of excruciating treatments such as electroshock and shock introduced by fever, methiazole, and insulin. Older CNS drugs such as opiates, belladonna derivatives, bromides, barbiturates, antihistamines, and chloral hydrates (each with its unique efficacy and safety profile) have all been used to treat schizophrenia with different degrees of “success.”
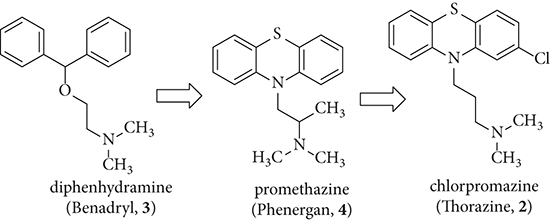
The genesis of chlorpromazine (2) can be traced back to antihistamines. In 1944, a group of scientists at Rhône-Poulenc Laboratories, led by chemist Paul Charpentier, began a program to systematically search for safer antihistamines. Their starting point was older antihistamines such as diphenhydramines (Benadryl, 3). In time, they successfully synthesized and marketed the antihistamine promethazine (Phenergan, 4). Similar to most antihistamines, promethazine (4) had side effects in the CNS that included mild antipsychotic properties. Charpentier sought to enhance these “side effects.” Structural–activity relationship (SAR) investigations led to the synthesis of RP-3277 (chlorpromazine, 2) in 1950.4 It was tested in rats, which became “indifferent.” In clinical trials, patients, too, became “disinterested” under the influence of chlorpromazine (2). In addition to the outstanding “calming” activities of RP-3277, it was later determined to have low toxicity. More importantly, chlorpromazine (2) subdued the positive symptoms (such as hallucinations and delusions) of psychotic patients.5 Chlorpromazine was introduced in 1952 in France under the trade name Largactil. The Largactil (meaning large activity) was chosen to reflect the wide range of CNS activities that chlorpromazine elicited. It was the first conventional (typical) antipsychotic discovered that was superior to opium. Chlorpromazine (2) was introduced to the US in 1954 under the trade name Thorazine. In the first eight months, the drug was administered to more than two million patients. It contributed to an 80% reduction of the resident population in mental hospitals. Thorazine (2) added a great impetus to the beginning of the psychopharmacological revolution.
Subsequently, Thorazine (2) was shown to be a potent dopamine D2 antagonist with other pharmacological properties that were thought to cause unwanted side effects. Thus, the D2-receptor antagonism of the conventional antipsychotics is thought to be responsible not only for their therapeutic effects, but also for some of their side effects. In addition, Thorazine (2) also helped our understanding of the CNS through research into the drug’s MOA.
Many “me-too” (copy-cat) drugs emerged soon after Thorazine’s (2) success.6 They included fluphenazine (Sinqualone, 5), perphenazine (Trilafon, 6), trifluoperazine (Stelazine, 7), and thioridazine (Melleril, 8). Incidentally, fluphenazine (5), along with 5-fluorouracil, was one of the first fluorine-containing drugs. Collectively known as typical antipsychotics, they all belong to the phenothiazine class. Unfortunately, they all cause a common group of side effects known as extrapyramidal symptoms (EPS), which Parkinsonian symptoms, akathisia, dyskinesia, and dystonia.


Another typical antipsychotic, haloperidol (Haldol, 9), is not a phenothiazine. Before the emergence of atypical antipsychotics, it was the gold standard for treating schizophrenia. Prepared in 1958 in Paul Janssen’s laboratories and distinctive from chlorpromazine (2) and other phenothiazines, Haldol (9) was a butyrophenone derivative. Therefore, Haldol’s chlorpromazine-like activity came as a surprise to Janssen and his colleagues. Haldol (9) was both faster and longer acting; it was potent orally as well as parentally; and more importantly, it was almost devoid of the anti-adrenergic and other autonomic effects associated with chlorpromazine (2). Haldol (9) also had a more favorable safety ratio and was surprisingly well tolerated when given chronically to laboratory animals.

Clinical trials confirmed that Haldol (9) belonged to the pharmacological family of neuroleptics. It became valuable in the treatment of agitation, delusions, and hallucinations in mental patients. Numerous chronic inpatients were able to leave the hospital and live at home thanks to Haldol (9). Until the emergence of atypical antipsychotics, Haldol remained one of the more prescribed neuroleptics 40 years after its discovery.
Haldol (9) was 50–100 times more potent than Thorazine (2), with fewer side effects—it was the most potent antipsychotic at the time of its discovery. It was developed as a more potent and selective D2 antagonist because the D2-receptor blockade in the mesolimbic pathway is believed to reduce the positive symptoms of schizophrenia. Indeed, Haldol (9) is very effective against the positive systems, however, like all other typical antipsychotics, it is ineffective in treating the negative symptoms and neurocognitive deficits of schizophrenia. In addition, administration of the drug typically causes EPS similar to other typical antipsychotics.7
With the discovery of newer atypical antipsychotics, older conventional antipsychotics are no longer used as first-line therapy, but can still be effective as a second-line or add-on treatment.
1.2 Atypical Antipsychotics—The Second Generation
As mentioned in the previous section, Haldol (9) and other typical antipsychotics are efficacious in treating positive symptoms of schizophrenia. But they are ineffective in treating the negative symptoms and neurocognitive deficits of schizophrenia. In addition, administration of typical antipsychotics often causes EPS.8
Clozapine (Clozaril, 10), the first atypical antipsychotic, was developed in 1959 by a small Swiss company, Wander AG.9 During clinical trials, Clozaril (10) showed strong sedating effects and proved efficacious for schizophrenia, but also showed some liver toxicity. Wander planned to withdraw the drug because of expected difficulty receiving regulatory approval, but clinicians who conducted trials urged Wander to provide more samples because their patients fared better on Clozaril (10) than on typical antipsychotics. For a population of patients who responded poorly to standard therapy at the time, Clozaril (10) was especially effective. Wander AG reluctantly proceeded with more trials of Clozaril and received approval to market it in a few European countries in 1971—although liver toxicity limited its widespread use. Unfortunately, the drug was eventually removed from the market in 1975 due to drug-associated agranulocytosis, a rare but potentially fatal blood disorder that resulted in lowered white-cell counts, which occurred in approximately 2–3% of patients. In Finland, eight patients died from subsequent infections. Additional side effects of Clozaril (10) therapy include sedation, weight gain, and orthostatic hypotension. Clozaril (10) was reintroduced in 1990 by Sandoz, and is now used as a second-line treatment—extensive monitoring of the patient’s blood cell count is required. Over the years Clozaril has demonstrated efficacy against treatment-resistant schizophrenia and some still consider it the gold standard treatment for refractory patients despite the inconvenience of a weekly check of white blood cell counts.
Clozaril (10) is considered the first atypical antipsychotic. Atypical antipsychotics, sometimes called serotonin–dopamine antagonists (SDAs), have reduced EPS compared to typical antipsychotics and are also believed to reduce negative, cognitive, and affective symptoms of schizophrenia more effectively. All atypical antipsychotics are potent antagonists of serotonin 5-HT2A and dopamine D2 receptors, however they also act on many other receptors including multiple serotonin receptors (5-HT1A, 5-HT1B/1D, 5-HT2C, 5-HT3, 5-HT6, 5-HT7), the noradrenergic system (α1 and α2), the cholinergic system (M1) and the histamine receptors (H1).

The second atypical antipsychotic was risperidone (Risperdal, 11) introduced by Janssen Pharmaceuticals in Belgium in 1993. With the success of Haldol (9) on the market, Janssen systematically explored its clinical utilities. They ultimately discovered Risperdal (11). Within a series of benzisoxazole derivatives, Risperdal showed a desired combination of very potent serotonin and potent dopamine antagonism. In essence, Risperdal (11) possessed the attributes of both ritanserin (a serotonin 5HT2 antagonist) and haloperidol (a dopamine antagonist), a typical antipsychotic. In schizophrenic patients, Risperdal (11) is effective against both positive and negative symptoms with reduced EPS liability and has become a first-line therapy.
The third atypical antipsychotic was Eli Lilly’s Zyprexa (1), launched in 1996. It is an antagonist against several receptors including dopamine, serotonin, histamine, adrenergic and muscarinic receptors. It was selected from a large series of chemical analogs based on behavioral tests (see below). Like many atypical antipsychotics, one of the side effects of Zyprexa is weight gain. Ironically, the weight gain side effect of some atypical anti-psychotics may be used to advantage by being prescribed off-label to patients with anorexia.
Since 2003, Zyprexa (1) has consistently had revenues of over $4 billion annually. It brought in over $5 billion, or nearly 22% of Lilly’s full-year sales, in 2011. The drug, which went generic in 2012, is the focus of this chapter.
Additional atypical antipsychotics include AstraZeneca’s quetiapine fumarate (Seroquel, 12, 1997), Pfizer’s ziprasidone (Geodon, 13, 2001), and BMS/Otsuka’s aripiprazole (Abilify, 14). One of the great advantages of ziprasidone (13) and aripiprazole (14) is that they do not have the weight gain side effect associated with other antipsychotics. Two more atypical antipsychotics—zotepine (Zoleptil, 15) and sertindole (Serlect, 16)—are available in some countries, but not in the US.



Risperdal (11), Zyprexa (1), quetiapine (12), ziprasidone (13), and aripiprazole (14) are currently considered as the first-line therapeutics for psychosis and will be highlighted in detail. Each of these drugs has a unique pharmacological and clinical profile; the clinician must therefore balance the benefit-risk factors for each patient in determining which drug to prescribe.10–12
Among the five first-line atypical antipsychotics,13,14 Risperdal (11) has high affinity for D2, 5-HT2C and α1 receptors and a very high affinity for the 5-HT2A receptor, whereas quetiapine (12) has the lowest affinity for the D2 and 5-HT2A receptors among the atypicals, and therefore relatively high doses are required for maximal efficacy. Ziprasidone (13) has high affinity for the D2 receptor, but even higher affinity for 5-HT2A and 5-HT2C receptors. Unlike other atypical antipsychotics, ziprasidone (13) also has potent 5-HT1B/1D antagonist and 5-HT1A partial agonist activity, as well as moderate SRI/NRI activity. On the other hand, aripiprazole (14) is a D2 partial agonist with an intrinsic activity of approximately 30%. Therefore, it acts as an agonist on presynaptic autoreceptors, which have a high receptor reserve, and as an antagonist on D2 post-synaptic receptors, where significant levels of endogenous dopamine exist and there is no receptor reserve.
Much remains to be discovered about the underlying pathophysiology of schizophrenia and there is still a great need for medicinal chemists to develop more efficacious and safer drugs that are devoid of clinically limiting side-effects and also address the cognitive impairment symptoms.
■ 2 Pharmacology
2.1 Mechanism of Action
After examining 17 atypical antipsychotics, Meltzer proposed the serotonin–dopamine hypothesis in 1989.15,16 Accordingly, atypical antipsychotic drugs may share common features such as relatively greater 5-HT2A receptor binding potency than D2 receptor binding potency. The difference between 5-HT2A pKi and D2 pKi is approximately greater than one. That is more than 10 times greater affinity for 5-HT2A than for D2 receptors. Of course only one factor, such as high 5-HT2A receptor binding potency or low D2 receptor binding potency, is not a sufficient condition. This pharmacological profile of atypical antipsychotic drugs has been thought to provide strong evidence for the serotonin–dopamine hypothesis that postulates a major contribution of the 5-HT2A/D2 receptor interactions to the MOA of these drugs.
As shown in Table 8.1, Zyprexa (1) is similar to all other atypical antipsychotics whose difference between 5-HT2A pKi and D2 pKi is approximately greater than one. This difference again translates to 10 times greater affinity for 5-HT2A than for D2 receptors. The only exception to the group is aripiprazole (14), whose affinity for 5-HT2A receptor is 10 times smaller than for the D2 receptor. More important, aripiprazole (14) is a partial agonist to D2 receptors (Ki = 0.74). In terms of clinical ramifications, binding to D2 receptors is associated with the drugs’ antipsychotic effects and EPS. On the other hand, binding to 5-HT2A receptors is responsible for the anti-EPS effects.17
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree



