CHAPTER 9 Neuroanatomy of the Spinal Cord
The vertebral column and its adjacent musculature are discussed in detail in the previous chapters. Because of the intimate anatomic and functional relationship between the vertebral column and spinal cord (which is protected by the vertebral column), knowledge of both is equally important. Chapter 3 describes the meninges, external surface, and vasculature of the spinal cord. In addition, it provides a cursory description of the cord’s internal organization. The purpose of this chapter is to elaborate on this internal organization by discussing the neurons, which form the circuitry of the spinal cord, and the ascending and descending tracts, which provide a connection among the spinal cord, the peripheral nerves, and the higher centers of the central nervous system. This information forms the basis of important neuroanatomic concepts that are necessary for an understanding of clinical neuroscience. The knowledge of these concepts is imperative for diagnosing pathologic conditions of the cord, some of which may be caused by vertebral column dysfunction. Examples demonstrating the application of these neuroanatomic principles to pathologic conditions are presented at the end of the chapter.
PERIPHERAL NERVOUS SYSTEM
Peripheral Receptors
Receptors may be divided into two types, rapidly adapting and slowly adapting. A slowly adapting receptor, such as a Merkel’s disc, responds continuously to a sustained stimulus, whereas a rapidly adapting receptor does not. A rapidly adapting receptor, such as a pacinian corpuscle, responds to any dynamic change in the receptor. For example, if the pressure onto a pacinian corpuscle is continuously increased, the corpuscle will continue to produce action potentials until the stimulus strength becomes constant. A stimulus furnishes a receptor with four basic characteristics: modality (type of stimulus), intensity (strength of stimulus), duration (perceived time that the stimulus is present), and location (where on the body the stimulus is being perceived). When a receptor is adequately stimulated, a generator potential occurs across its membrane and may lead to an action potential. The action potential propagates along the sensory neuron into the CNS. The CNS then is able to combine the four characteristics of the stimulus into a perceived sensation.
The sensory neurons of the peripheral nervous system (PNS) are pseudounipolar neurons. Their cell bodies are located in the dorsal root ganglia. The peripheral process, which may be myelinated or unmyelinated, is the part of the fiber continuous with the receptor. It is the sensory component of a peripheral nerve. The other part of the fiber, the central process, enters the CNS. A bundle of central processes form a dorsal root. The peripheral processes are classified according to their conduction velocity, and conduction velocity is related to the axon diameter. Fibers with large diameters conduct the fastest. Based on the relationship between velocity and diameter, cutaneous fibers are classified alphabetically as A-beta, A-delta, and C fibers. Similarly, afferents from muscle tissue usually are classified numerically from heavily myelinated to unmyelinated as I, II, III, and IV. Type I also has subgroups of Ia and Ib. Afferents from visceral interoceptors often are classified as A-delta and C fibers. Motor (efferent) fibers also are classified according to the alphabetic listing. Large somatic motor neurons correspond to the A-alpha and A-gamma group, and autonomic efferent fibers correspond to the B and C groups. Table 9-1 summarizes the classifications of the afferent and efferent fibers.
Peripheral receptors can be classified by their morphology, location, and the type of stimulus to which they respond. Morphologically, receptors may be encapsulated by connective tissue and nonneural cells, or they may simply be nonencapsulated, bare arborizing endings. Receptors classified by their location of distribution are called exteroceptors, proprioceptors, or interoceptors. Exteroceptors are superficial and located in the skin. Modalities such as nociception (pain), temperature, and touch (and the submodalities of pressure and vibration) are conveyed by these receptors. Proprioceptors are located in the muscles, tendons, and joints of the body and provide information concerning limb position, while the limbs are either stationary (static) or moving (dynamic or kinesthetic) (Williams et al., 1995; Kiernan, 1998; Pearson and Gordon, 2000a; Nolte, 2002). Interoceptors are located in the viscera, glands, and vessels and convey poorly localized information from such systems as the digestive and urinary. Examples of the types of information conveyed by interoceptors include distention or fullness and ischemic pain.
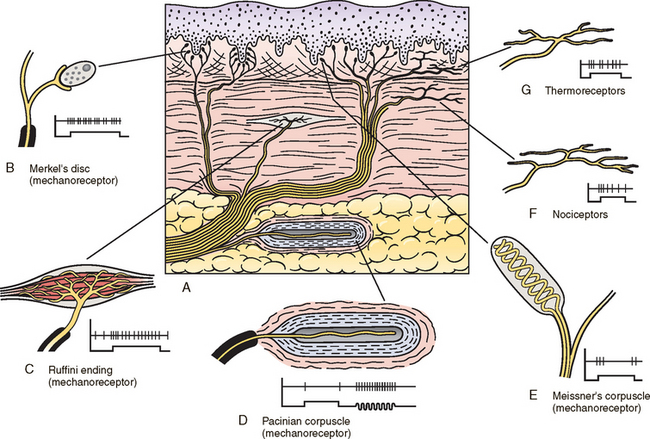
Cutaneous Receptors.
Cutaneous receptors (exteroceptors) include mechanoreceptors, thermoreceptors, and nociceptors and subserve such modalities as touch, pressure, vibration, temperature, and nociception (pain) (Fig. 9-1). Mechanoreceptors include the nonencapsulated Merkel’s discs, nonencapsulated endings surrounding hair follicles (peritrichial), and encapsulated endings such as Ruffini endings, pacinian corpuscles, and Meissner’s corpuscles. The fibers supplying these receptors are primarily A-beta. Thermoreceptors are nonencapsulated, free nerve endings that occupy areas approximately 1 mm in diameter. Cold thermoreceptors respond in the range of 5° C (41° F) to 40° C (104° F) relative to the normal skin temperature of 34° C (93.2° F) and fire most frequently at 25° C (77° F). Warm thermoreceptors are stimulated in the temperature range 29° C (84.2° F) to 45° C (113° F) and are most active at 45° C (Gardner, Martin, and Jessell, 2000). Cold receptors are supplied by A-delta or C fibers, but warm receptors are supplied by C fibers alone.
An understanding of nociceptors is helpful in the comprehension of pain of spinal origin. Nociceptors are free nerve endings, and they respond to stimuli that may threaten or actually damage adjacent tissue cells. The damage to cells causes the release of chemical mediators that may sensitize (e.g., prostaglandins, leukotrienes, substance P) previously unresponsive free nerve endings by lowering their threshold for activation or activate (e.g., histamine, bradykinin, potassium, serotonin) the free nerve endings. In some instances, activated free nerve endings release substance P and calcitonin gene-related peptide into the surrounding area, causing vasodilation, extravasation of fluid, and release of histamine from tissue cells. These changes in turn lead to lowering the threshold for activation of more nociceptors. The inflammatory process that results from the activity of these nociceptors is termed neurogenic inflammation (Basbaum and Jessell, 2000). Three types of nociceptors appear to exist: (a) mechanical, which are stimulated by mechanical damage such as by a sharp object; (b) thermal, which are stimulated by temperatures higher than 45° C (113° F) or lower than 20° C (68° F); and (c) polymodal, which respond to damaging mechanical, thermal, or chemical stimuli. Mechanical and thermal nociceptors send their information via A-delta fibers, whereas polymodal receptors use C fibers. The three types of nociceptors often are activated simultaneously and thus work together. For example, if a person stubs a toe, initially there is sharp (fast) pain resulting from stimulating mechanical nociceptors followed by an achy prolonged pain resulting from stimulating polymodal nociceptors that use slower C fibers.
The cutaneous fibers of these receptors form overlapping horizontal plexuses in the dermis and subcutaneous layers of the skin. The density and variety of receptors vary in different regions. For example, in hairy skin the peritrichial endings are most common, but Merkel’s discs and free nerve endings are also present. In glabrous (hairless) skin, free nerve endings are present, as are Merkel’s discs and Meissner’s corpuscles. The latter two receptors have small receptive fields and help to discriminate the spatial relationship of stimuli. This ability to discriminate is well developed on the fingertips. In fact, Meissner’s corpuscles have been located only in primate animals (Kiernan, 1998). The subcutaneous tissues of both types of skin are provided with pacinian corpuscles and Ruffini endings, both of which have large receptive fields and therefore are less discriminatory (Gardner, Martin, and Jessell, 2000).
Muscle, Tendon, and Joint Receptors.
The majority of the receptors located in muscles, tendons, and joints are involved with the sense of proprioception. The receptors are classified as proprioceptors based on their location. They are classified as mechanoreceptors based on the type of stimulus to which they respond. These receptors convey proprioception, the term used to describe the sensory information that contributes to the sense of movement and position of one’s own limbs and body without visual input. The mechanoreceptors involved with providing proprioception (excluding the vestibular system of the inner ear) are the joint receptors, neuromuscular spindles, and Golgi tendon organs (neurotendinous spindles). They function in the coordination and control of movements and the maintenance of upright posture by monitoring both the stationary position and movement (kinesthesia) of body parts, and then relaying that information into the CNS. This information, often called joint position sense, may be perceived consciously. In addition, cutaneous mechanoreceptors also may function in the overall assessment of proprioception. For example, they may aid other proprioceptors in the task of discriminating different thicknesses of objects held between the thumb and finger and in the manipulation of objects of different shapes and sizes (McCloskey, 1994; Gardner, Martin, and Jessell, 2000).
Joint receptors are located in the superficial and deep layers of the joint capsules and in the ligaments. Four types of receptors exist, classified as I, II, III, and IV. The first three types are encapsulated mechanoreceptors that have a proprioceptive function. The fourth type consists of unmyelinated free nerve endings. The receptors classified as I, II, or III provide information about such activities as the direction, velocity, and initiation of joint movements. They do this by responding to tension applied to the connective tissue surrounding them. The group IV free nerve endings, which mediate nociception and normally are silent, respond to potentially injurious mechanical or inflammatory processes (Wyke, 1985).
Golgi tendon organs (GTOs), or neurotendinous spindles, respond to tension that is applied to a tendon. They consist of tendon collagen fibers surrounded by connective tissue, with group Ib afferent fibers twisted among the collagen fibers in such a way that they may become “squeezed” under the appropriate amount of tension. On stimulation, the group Ib afferent fiber stimulates a Ib interneuron. This interneuron then inhibits the alpha motor neuron that supplies the skeletal muscle associated with that stimulated GTO. This action is opposite to that of a stimulated neuromuscular spindle, which produces excitation of the alpha motor neuron supplying the skeletal muscle associated with that spindle. The GTOs and spindles not only function at the spinal level, but also send input to higher centers (see Ascending Tracts). The section Spinal Motor Neurons and Motor Coordination describes the functional relationship between muscle spindles and GTOs.
Visceral Receptors.
These receptors also may be classified as interoceptors because of their location. They include mechanoreceptors that respond to movement or distention of the viscera. These are found in locations such as the mesentery, connective tissue enclosing the organs, and along blood vessels. Nociceptors are found in the viscera, as well. These are capable of responding to noxious mechanical, thermal, and chemical stimuli (Willis Jr. and Coggeshall, 1991). Chapter 10 discusses the relationship of these receptors and their afferent fibers to somatic and autonomic efferents.
Peripheral Nerves
Thirty-one pairs of spinal nerves exist, and each is formed by the convergence of a dorsal root and a ventral root usually within the intervertebral foramen (IVF). Just distal to this union, each spinal nerve divides into a dorsal ramus (posterior primary division, PPD) and a ventral ramus (anterior primary division, APD) (see Fig. 3-3). The dorsal rami of spinal nerves innervate the zygapophysial joints, skin over the back, and deepest muscles of the neck and back. The ventral rami innervate the extremities and the ventrolateral aspect of the trunk. Successive thoracic ventral rami retain a clear segmental distribution along the thoracic region. However, the back of the head, the anterior and lateral neck, shoulder, and upper and lower extremities are innervated by plexuses. Each plexus is formed by a regrouping of adjacent ventral rami. The plexuses are called the cervical, brachial, and lumbosacral plexuses, and each is briefly described here.
The cervical plexus is formed by ventral rami of the C1 through C4 cervical nerves. It supplies cutaneous innervation to the dorsolateral part of the head, neck, and shoulder. Motor fibers in this plexus course to the deep cervical muscles, hyoid muscles, diaphragm, and sternocleidomastoid and trapezius muscles (see Chapter 5).
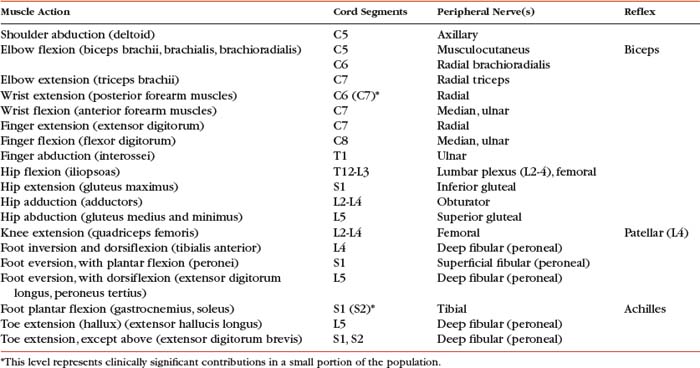
The brachial plexus is formed by ventral rami of the C5 through T1 spinal nerves (often with a contribution from C4 and T2). This plexus supplies the upper extremity. Subsequent to the mixing of the ventral rami in the plexus, numerous branches are formed, including five large terminal branches: the axillary, musculocutaneous, radial, ulnar, and median nerves. The axillary nerve (C5 to C6) supplies cutaneous branches to the deltoid region and muscular branches to the deltoid and teres minor muscles. The musculocutaneous nerve (C5 to C7) is sensory to the anterolateral and posterolateral aspect of the forearm. It supplies motor innervation to the flexors of the arm, which includes the biceps brachii. The radial nerve (C5 to C8, possibly T1) has an extensive area of distribution in both the arm and forearm. Its cutaneous branches innervate the posterior aspect of the arm and forearm. The superficial radial nerve supplies the skin of the lateral half of the dorsum of the hand and the first three and a half digits, excluding the nails. The radial nerve also innervates the extensor muscles of the upper extremity. The ulnar nerve (C8 and T1, sometimes C7) courses through the arm to supply structures in the forearm and hand. Its motor distribution includes one and a half forearm flexors (ulnar side) and intrinsic hand muscles, including the hypothenar and all interossei muscles and some thenar muscles. Its cutaneous distribution is present only in the hand and encompasses the ulnar half of the hand, including the fifth and medial one half of the fourth digits. The median nerve (C6 to C8 and T1, sometimes C5) supplies motor fibers to the forearm flexors (excluding those with ulnar innervation) and some intrinsic hand muscles, including most of the thenar muscles. As with the ulnar nerve, its sensory area is only in the hand and includes the lateral palmar surface and first three and a half digits, including the nails (Williams et al., 1995) (Figs. 9-2 and 9-3 and Table 9-2). (See Chapter 5 for a full description of the brachial plexus and its proximal branches.)
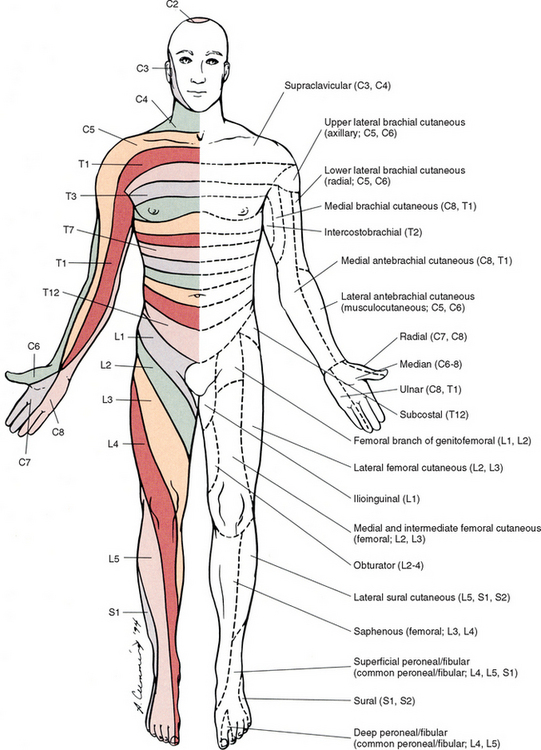
FIG. 9-2 Anterior view of the body showing its cutaneous innervation. Left, Dermatomal pattern, which may vary according to different authors. This dermatomal mapping is based on studies by JG Keegan and FV Garrett (1948). Right, Areas of cutaneous peripheral nerve distributions. Note the similarity of cord segment origins between the two sides.
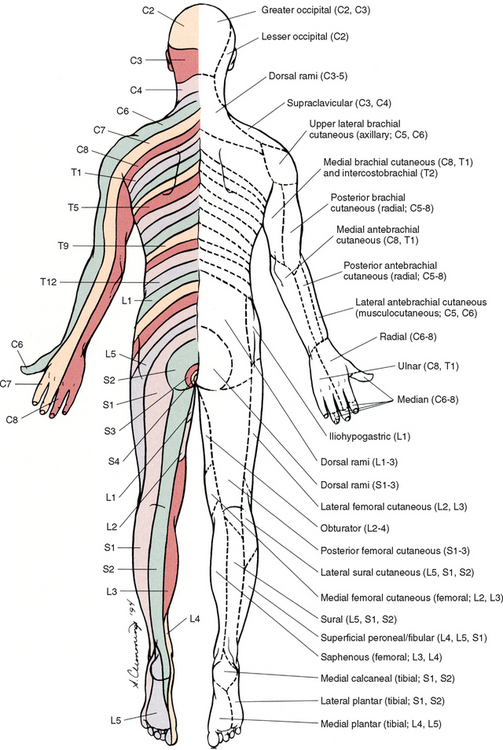
FIG. 9-3 Posterior view of the body showing its cutaneous innervation. Left, Dermatomal pattern, which may vary according to different authors. This dermatomal mapping is based on studies by JG Keegan and FV Garrett (1948). Right, Areas of cutaneous peripheral nerve distributions. Note the similarity of cord segment origins between the two sides.
Table 9-2 Muscles Supplied by Terminal Branches of the Brachial Plexus and the Lumbosacral Plexus
| Muscle(s) Supplied | Peripheral Nerve | Cord Segments |
|---|---|---|
| Deltoid, teres minor | Axillary | C5,C6 |
| Anterior arm | Musculocutaneous | C5-7 |
| Extensors of upper extremity | Radial | C5-8 (T1)* |
| Flexor carpi ulnaris, flexor digitorum profundus (medial half), hypothenar eminence, interossei, adductor pollicis, lumbricals (3rd and 4th) | Ulnar | (C7)*, C8-T1 |
| Anterior forearm (except above), thenar eminence (except above), lumbricals (1st and 2nd) | Median | (C5)*, C6-T1 |
| Medial thigh | Obturator | L2-4 |
| Anterior thigh | Femoral | L2-4 (mostly L3 and L4) |
| Posterior thigh | Sciatic | L4-S3 |
| Common fibular (peroneal) | L4-S2 | |
| Lateral leg | Superficial | (mostly S1) |
| Anterior leg | Deep | |
| Posterior leg | Tibial | L4-S3 (mostly S1) |
| Gluteus maximus | Inferior gluteal | L5-S2 (mostly S1) |
| Gluteus medius and minimus | Superior gluteal | L4-S1 (mostly L5) |
* This level represents clinically significant contributions in a small portion of the population.
The lumbosacral plexus is the third plexus, which is composed of ventral rami L2 through S2 (with contributions from L1 and S3). The major branches of this network are the femoral, obturator, gluteal, sciatic, common fibular (peroneal) (and its branches), and tibial nerves. Cutaneous nerves with large areas of distribution include, but are not limited to, the lateral femoral cutaneous, saphenous, posterior femoral cutaneous, and sural nerves. (Chapters 7 and 8 describe the lumbar plexus and its smaller branches [iliohypogastric and ilioinguinal] and the sacral plexus and its branches.)
The obturator nerve (L2 to L4) supplies motor branches to the adductor muscles of the thigh and gracilis muscle. Also it is cutaneous to the inner thigh. The femoral nerve (L2 to L4) sends motor branches to the anterior thigh muscles (e.g., quadriceps), which extend the leg. Cutaneous innervation by the femoral nerve supplies the anterior and anteromedial thigh and, via the saphenous nerve (L3 and L4), the medial leg and foot. The thigh’s lateral side is innervated by cutaneous branches of the lateral femoral cutaneous nerve (L2 and L3). The sciatic nerve (L4 to S3) is the body’s largest nerve. This nerve actually is composed of two parts (tibial and common fibular) but usually is ensheathed to form one nerve in the posterior thigh. Motor branches in the posterior thigh innervate the hamstring muscles (biceps femoris, semitendinosus, semimembranosus), which flex the leg. The cutaneous innervation of the posterior thigh is furnished by the posterior femoral cutaneous nerve (S1 to S3). The sciatic nerve divides into the common fibular (peroneal) and tibial nerves at varying levels proximal to the knee.
The common fibular (peroneal) nerve (L4, L5, S1, and S2) courses laterally around the neck of the fibula and divides into two major branches: superficial fibular (peroneal) and deep fibular (peroneal). The superficial fibular (peroneal) nerve supplies muscular branches to the fibularis (peronei) muscles, which are responsible for eversion of the foot, and cutaneous branches to the distal and anterolateral third of the leg and dorsum of the foot (excluding the first digital interspace). The deep fibular (peroneal) nerve sends motor fibers to the anterior leg muscles, which provide dorsiflexion of the foot and extension of the toes. Cutaneous branches supply the skin between the first two toes. The other branch of the sciatic nerve is the tibial nerve (L4, L5, S1, S2, and S3). This nerve provides motor innervation to posterior leg muscles (including the gastrocnemius muscle), which are responsible for plantar flexion of the foot. A branch of the tibial nerve and contributing fibers from the common fibular (peroneal) nerve form the sural nerve. This supplies sensory innervation to the posterior and lateral surfaces of the leg. The tibial nerve divides into medial and lateral plantar nerves at the region of the medial malleolus. These nerves supply motor and sensory innervation to the plantar aspect of the foot (see Figs. 9-2 and 9-3 and Table 9-2).
The inferior and superior gluteal nerves innervate muscles moving the hip joint. The inferior gluteal nerve (L5, S1, and S2) is responsible for the motor innervation of the strongest hip extensor, the gluteus maximus. The superior gluteal nerve (L4, L5, and S1) innervates the gluteus medius and minimus muscles and the tensor fascia latae muscle, which are responsible for hip abduction (see Table 9-2).
Realize the differences between peripheral nerve and dermatomal patterns (and remember there is much variation in dermatomal maps) (see Figs. 9-2 and 9-3). Know both the segmental and peripheral innervation of major skeletal muscles. This knowledge of peripheral innervation of muscles and skin is imperative, because a neurologic examination includes the assessment of a patient’s motor functions (reflexes and muscle strength; Table 9-3) and sensory functions. The information gained from this assessment is useful for distinguishing if the lesion is in the CNS or PNS and subsequently for determining the specific location of the lesion along one of these two systems.
INTERNAL ORGANIZATION OF THE SPINAL CORD
Gray Matter
The neurons that compose the gray matter are subdivided into four groups: motor neurons, the axons of which leave the spinal cord and innervate the effector tissues (skeletal, smooth, and cardiac muscles, and glands); tract neurons, the axons of which ascend in the white matter to higher centers; interneurons, which have short processes; and propriospinal neurons, the axons of which provide communication between cord segments. Propriospinal neurons are classified as long, intermediate, or short. Long propriospinal neurons extend the length of the cord in the ventral funiculus and ventral part of the lateral funiculus (see White Matter). Descending propriospinal fibers (axons) course bilaterally and the ascending axons project mostly contralaterally. Short propriospinal neurons extend six to eight segments in the ipsilateral lateral funiculus, and intermediate propriospinal neurons course primarily ipsilaterally more than eight segments but less than the cord’s entire length. Most of the propriospinal fibers are located immediately adjacent to the gray matter in the fasciculus proprius, but some travel more laterally in the funiculi. The axons of medial propriospinal neurons are long and include extensive branches. For example, some extend the length of the cord to coordinate the movements of neck and pelvic axial musculature for postural corrections. Laterally placed propriospinal fibers influence neurons innervating more distal muscles, communicate with a smaller number of segments, and branch less extensively. Because the distal musculature is less connected to other muscle groups, these muscles function more independently. This independence permits more diverse somatic motor activities (Williams et al., 1995; Ghez and Krakauer, 2000). Through their communication with neurons in other cord segments, propriospinal neurons are involved not only with the coordination of somatic motor activity, but also with the autonomic innervation of sweat glands, smooth muscle of the vasculature, and viscera such as the bladder and bowel (Williams et al., 1995).
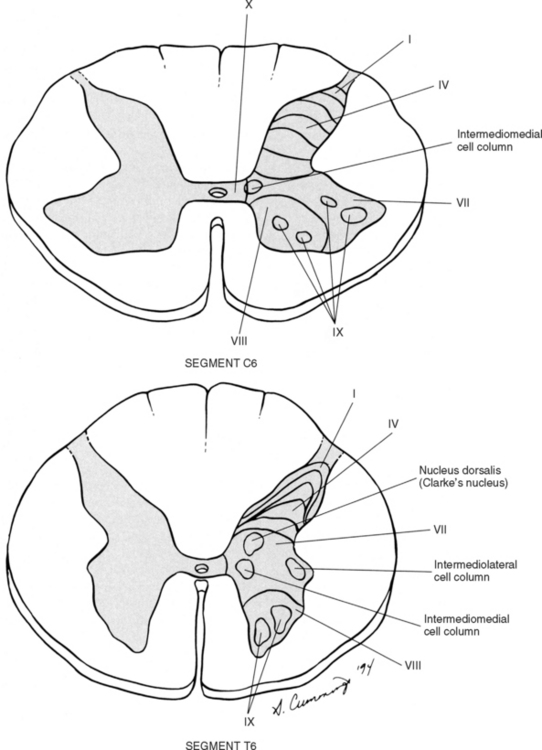
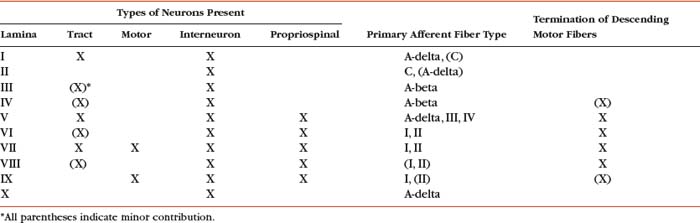
In the early 1950s, Rexed (1952) studied feline spinal cords and proposed that the organization of the gray matter formed 10 layers, or laminae. He described lamina I as being located at the tip of the dorsal horn, followed sequentially into the ventral horn by laminae II through IX. Lamina X formed the connecting crossbar of the gray matter, that is, the gray commissure. This organization has been accepted for the human spinal cord as well (Fig. 9-4). Each lamina includes at least one of the four general types of neurons: motor, tract, interneuron, or propriospinal. Each lamina also may be the site of the termination of primary afferents, descending tracts, propriospinal neurons, and interneurons of neighboring laminae. The laminae may vary in size throughout regions of the spinal cord and even may be absent in some regions. Also within each lamina, neurons may be organized into smaller groups, called nuclei or cell columns, based on commonalities such as cell morphology and function. The following is a brief description of each of these laminae.
Laminae I through VI (Dorsal Horn).
The dorsal horn consists of laminae I through VI. Laminae I through IV form the head, lamina V the neck, and lamina VI the base of the dorsal horn. Laminae I and II are collectively known as the superficial dorsal horn and are heavily involved with the processing of nociception. The majority of A-delta and C fibers terminate here. Lamina I is also known as the marginal zone of Waldeyer. Most of the primary afferent input into lamina I originates from cutaneous nociceptors and thermoreceptors via A-delta fibers. Additional input is conveyed by C fibers (from nociceptors, thermoreceptors, and histamine-sensitive receptors conveying the sensation of “itch” [Schmelz et al., 1997; Andrew and Craig, 2001; Craig, Zhang, and Blomqvist, 2002]) and a small group of thinly myelinated muscle, joint, and visceral afferent fibers (Willis and Coggeshall, 1991; Williams et al., 1995). Many neurons within lamina I are considered to be nociceptive specific, whereas others are classified as wide-dynamic-range neurons that respond to both noxious and innocuous stimuli. Thermoreceptive-specific neurons also terminate in lamina I (Han, Zhang, and Craig, 1998) along with numerous interneurons. Tract neurons originate in lamina I as well, and provide the major output for the superficial dorsal horn.
Lamina II is known as the substantia gelatinosa of Rolando. The many processes, the presence of small neurons, and the absence of myelinated axons gives this layer a gelatinous appearance on close inspection. The primary afferent input into lamina II enters by C afferent fibers from cutaneous nociceptors, thermoreceptors, and mechanoreceptors. A few A-delta fibers also terminate here. The neurons of lamina II are almost entirely interneurons (both excitatory and inhibitory), the dendrites of which arborize within the lamina and also project into other laminae. Some interneurons respond to noxious stimuli, whereas others respond to both noxious and innocuous stimuli (Basbaum and Jessell, 2000).
Lamina V forms the neck of the dorsal horn. Primary afferent input comes via A-delta fibers from cutaneous mechanical nociceptors and group III and IV muscle and joint afferents, and nociceptive visceral afferents (Willis and Coggeshall, 1991; Basbaum and Jessell, 2000). Additional input to this lamina (such as input from C fibers) is most likely received via the dendritic projections located within more dorsal laminae. Many neurons within this lamina are wide-dynamic-range tract neurons that are the site of viscerosomatic convergence for visceral referred pain (Benarroch et al., 1999), whereas others are interneurons and propriospinal neurons.
In summary, the first six laminae that comprise the dorsal horn are the major receiving areas for sensory information. Pain and temperature input appears to terminate primarily in superficial layers; mechanical types of stimuli terminate in the middle region; and proprioceptive input ends in the base of the dorsal horn near the motor regions. Each primary afferent fiber has many collateral branches that synapse in the circuitry of more than one lamina and feed into more than one ascending pathway. For example, one Ia afferent fiber from a neuromuscular spindle may have 500 or more branches terminating within the gray matter of the cord (Nolte, 2002). Many of the laminae communicate with each other via profuse dendritic branching and the axonal projections of their interneurons. This provides a mechanism by which incoming sensory signals may be processed and modified (modulated) before ascending to higher centers. This modulation occurs at the level of the synapse between presynaptic and postsynaptic neurons and involves not only the release of neurotransmitters and neuromodulators but also the different receptors with which these chemicals bind. This is exemplified by studies of the nociceptive interneurons in lamina II that show that numerous chemicals can modulate synaptic transmission.
Dorsal horn interneurons.
Interneurons in the dorsal horn play a key role in modifying sensory information and ongoing movements, as well as controlling all reflexive movements. The extensive branching of the afferent inputs to the spinal cord results in simultaneous activation of many types of interneurons. The activity of these interneurons and their subsequent synapses on cells in the ventral horn (motor systems), ascending tracts (sensory perception of stimuli), descending tracts (sensitivity of motor and sensory pools), and other interneurons in the spinal cord controls the overall sensitivity and general responsiveness of all neurons in the spinal cord. In addition to the input from afferent fibers, the spinal interneurons also receive input from the descending systems, ventral horn alpha motor neurons, local (at the level) interneurons, short propriospinal interneurons (from one or two levels above or below), and long propriospinal interneurons (ascending and descending between all levels of the cord). This vast array of interconnections among and between the various interneurons also contributes to the general responsiveness of the spinal cord to both afferent (sensory or reflexive) and efferent (motor or voluntary) stimuli. The interconnections between the interneurons are mutually inhibitory, meaning that only one set of pathways can be active at any time. When one particular response pathway and its corresponding interneurons are activated, all other interneuronal pathways typically are inhibited. This allows the spinal cord to respond selectively to any input, either afferent or efferent, without inappropriately activating other antagonistic pathways that might hinder the appropriate response. In addition, interneurons can modulate or alter the normal output of higher-order neurons. One example of this is related to the gate control theory of pain (see Fig. 11-6), in which nonnociceptive afferents reduce the output of second-order pain fibers via inhibitory interneurons in the dorsal horn. Therefore interneurons are the key building blocks of all spinal reflexes, and can control or modulate most afferent and efferent information at the level of the spinal cord (see the Spinal Motor Neurons and Motor Coordination section later in this chapter).
Relationship between the dorsal horn and trigeminal nerve.
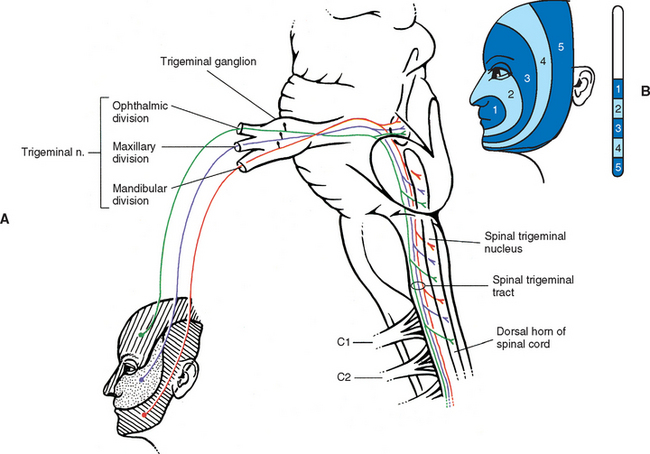
The dorsal horn of the upper three or four cervical cord segments has an interesting relationship with the trigeminal nerve. The trigeminal nerve (cranial nerve V) provides sensory innervation to the skin of the face and other structures, including the paranasal sinuses, cornea, temporomandibular joint, and oral and nasal mucosae. Cranial nerve V (CN V) also supplies motor fibers to the muscles of mastication and several other small muscles of the head. The afferent fibers of CN V enter the brain stem at the level of the pons and synapse in a nuclear column that extends from the midbrain through the pons and medulla and into the upper cervical cord segments. The portion of the nuclear column located in the medulla and upper cervical cord segments is known as the spinal trigeminal nucleus (Fig. 9-5). Afferent fibers conveying pain and temperature (and some touch) that enter the brain stem within CN V (and also in the facial and glossopharyngeal nerves) descend as the spinal trigeminal tract and synapse in this nucleus. The most inferior portion of the spinal trigeminal nucleus is the nucleus caudalis. This nuclear region continues from the caudal medulla into the upper three or four cervical cord segments and blends with laminae I through IV, and possibly V and VI, of the dorsal horn of those segments (Carpenter, 1991; Williams et al., 1995). This area of gray matter that is the site of convergence of trigeminal and cervical afferents is also called the trigeminocervical (Bogduk, 1992; Biondi, 2001) or trigeminospinal nucleus. The descending spinal trigeminal tract fibers also continue into the dorsolateral tract of Lissauer in the upper cervical segments. Afferent fibers conveying similar information and traveling in dorsal roots of the upper three or four cervical nerves also synapse in these same laminae. In fact, some of these cervical dorsal root afferents may ascend into the rostral medulla and synapse in the spinal trigeminal nucleus (Abrahams, 1989).
The relationship between the dorsal horn and trigeminal system is clinically significant (Pollman, Keidel, and Pfaffenrath, 1997; Biondi, 2000, 2001; Bogduk, 2001). The upper three cervical spinal nerves innervate the muscles (including the trapezius and sternocleidomastoid muscles), ligaments, and joints in the region of the upper three cervical vertebral segments, C2-3 intervertebral discs, vertebral and internal carotid arteries, and dura mater of the upper spinal cord and posterior cranial fossa of the skull. Second-order neurons in the dorsal horn, which project to the brain, receive afferents from pain generators in these structures and potentially from pain generators innervated by lower cervical nerves whose central processes have ascended in the dorsolateral tract of Lissauer. In addition, these second-order neurons receive input from pain generators innervated by CN V primary afferents. When the brain receives this nociceptive information from these spinal pain generators, it can misinterpret the input as coming from the more familiar source, resulting in the perception that the origin is in the territory of CN V. Thus this region of convergence becomes the anatomic basis for pain referral from the neck to regions innervated by CN V (typically the forehead or orbital region), and vice versa (sometimes called the trigemino-cervical reflex (Lance, 1989; Pollmann, Keidel, and Pfaffenrath, 1997; Browne et al., 1998; Sessle, 1998; Milanov and Bogdanova, 2003). Head pain or headache that is the result of stimulation of pain generators located in the cervical spine is called cervicogenic headache.
Laminae VII through X.
Lamina VII composes most of the intermediate region of the gray matter and also extends into the ventral horn (see Fig. 9-4). The shape of lamina VII varies in different regions. For example, in the T1 to L2 or L3 segments, lamina VII includes the lateral horn. Most primary afferent input into this lamina (excluding the lateral horn) and the remaining ventral horn concerns proprioception, although some neurons receive noxious stimuli via polysynaptic connections (Basbaum and Jessell, 2000). The lateral part of lamina VII (excluding the lateral horn) is involved with the regulation of posture and movement. These neurons, including tract neurons, have numerous ascending (tract neurons) and descending connections with the cerebellum and midbrain. The medial part of lamina VII includes interneurons and propriospinal neurons that connect adjacent laminae and cord segments for reflexes concerned with movement and autonomic activities. In the ventral part of the lamina, inhibitory interneurons, such as Renshaw cells and Ia inhibitory interneurons, have been identified (Williams et al., 1995). Clearly defined nuclei and cell columns also are within lamina VII. One of these is the nucleus dorsalis of Clarke (nucleus thoracicus). This oval nucleus consists of tract neurons and is located in the medial part of lamina VII in segments C8 through L3. It is best defined in the T10 to L2 segments (Carpenter and Sutin, 1983). The axons of these neurons ascend ipsilaterally in the spinal cord white matter to the cerebellum as the dorsal spinocerebellar tract (see Ascending Tracts). A cluster of four nuclei that is associated with the innervation of autonomic effectors also is located in lamina VII (see Chapter 10). The largest of the four is the intermediolateral cell column, which is located in the lateral horn in cord segments T1 to L2 or L3 and consists of autonomic motor neuron cell bodies. The axons of these motor neurons are preganglionic sympathetic fibers that exit in the ventral root and synapse in autonomic sympathetic ganglia. They are involved with the innervation of smooth and cardiac muscles and glands. The sacral autonomic nucleus is found in cord segments S2 to S4, and although there is no lateral horn at that level, this nucleus is located in a similar position to that of the intermediolateral cell column. The sacral autonomic nucleus contains cell bodies, the axons of which form preganglionic parasympathetic fibers. These fibers exit via the S2 to S4 ventral roots, synapse in autonomic ganglia, and innervate the smooth muscle and glands of the pelvic and lower abdominal regions. An additional group of neurons forms the intermediomedial nucleus of lamina VII. This is found in the medial aspect of this lamina and is considered by some authors to be the termination site of visceral afferent fibers (Carpenter and Sutin, 1983). Chapter 10 provides a thorough description of the autonomic nervous system. From this description, it is apparent that lamina VII is composed of all four neuron types: interneurons, tract, propriospinal, and motor neurons.
Lamina IX is found in the ventral horn and consists of well-defined medial and lateral longitudinal nuclear columns, and a central nuclear group of motor neurons (anterior horn cells) (see Fig. 9-4) (Williams et al., 1995). The position and presence of the groups vary at different spinal regions. The medial group (subdivided into dorsal and ventral parts) is found in all cord segments and provides the axons that innervate the axial musculature. The lateral group (subdivided into dorsal, ventral, and retrodorsal groups) is responsible for innervating the muscles of the extremities. These clusters of neurons are organized so that the more lateral the motor neurons, the more distal the muscles they innervate. Also, the dorsal motor neuronal groups innervate the flexor muscles, and the ventral motor neuronal groups innervate the extensor muscles. In addition, one specific group of ventrolateral neurons located in the S1 and S2 segments forms Onuf’s nucleus. The axons from this nucleus innervate the perineal musculature (Williams et al., 1995). The addition of these lateral motor neuronal groups in cord segments supplying the extremity muscles creates a distinctive lateral enlargement in the ventral horn.
The third nuclear group of lamina IX is the central group. This group includes three nuclei located in specific cord segments. One of these is the lumbosacral nucleus, which is located in the L2-S1 cord segments. The projections from this nucleus are unknown (Williams et al., 1995). Another nuclear column forms the phrenic nucleus, which is found in the C3 to C5 segments. The axons of these segments, and in particular C4, form the phrenic nerve, which provides innervation to the diaphragm. Based on cadaveric studies, Routal and Pal (1999) suggest that the phrenic nucleus is not part of a central column but instead actually is a subdivision of the medial column. The diaphragm develops as an axial muscle and therefore the phrenic nucleus is located near the other motor neurons that innervate the axial musculature (i.e., the medial motor column). The location of the nucleus in the C3-5 segments and the location of the nucleus’ efferent fibers also may have clinical significance. As exemplified by two case studies, severe cases of cervical spondylotic myelopathy could traumatize the phrenic nucleus or its efferent fibers, resulting in phrenic paresis (Parke and Whalen, 2001). The other nucleus is the accessory nucleus, which extends from the lower medulla into the C5 cord segment (Routal and Pal, 1999). Axons from this nucleus form the spinal root of the spinal accessory nerve (CN XI). They ascend in the vertebral canal dorsal to the denticulate ligament and travel through the foramen magnum to enter the cranial cavity and briefly join the cranial root of CN XI. Subsequently this nerve exits the cranial cavity, and the spinal root fibers branch away to innervate the sternocleidomastoid and trapezius muscles. This nucleus may be somatotopically organized in a craniocaudal direction and subdivided into two parts, with the upper part of the nucleus projecting to the sternocleidomastoid muscle, and the lower part projecting to the trapezius muscle (Routal and Pal, 1999).
Each of these three major groups of lamina IX includes alpha motor neurons, which project to extrafusal skeletal muscle fibers and are subdivided into tonic (to slow muscle fibers) and phasic (to clusters of fast muscle fibers) neurons; beta motor neurons, which course to extrafusal and intrafusal fibers; and gamma motor neurons, which innervate the contractile portion of the neuromuscular spindles located within skeletal muscles and are subdivided into static and dynamic types (Williams et al., 1995; FitzGerald and Folan-Curran, 2002). The alpha and gamma motor neurons are tightly packed into pools responsible for the innervation of a particular skeletal muscle. Both types of motor neurons receive excitatory and inhibitory input from neighboring interneurons that have formed local reflex circuits, propriospinal neurons, and some descending tract fibers. These connections modulate the activity of the motor neurons. Alpha motor neurons also receive the primary afferent fibers from neuromuscular spindles that form the sensory arc of the stretch (myotatic) reflex (see Spinal Motor Neurons and Motor Coordination). The cell bodies of the alpha motor neurons, which are large and range from 30 to 70 µm in size, receive a tremendous amount of synaptic input. An estimated 10, 000 excitatory boutons of descending fibers from the brain and propriospinal neurons may synapse on the extensive dendritic tree of a typical alpha motor neuron, and some 5000 inhibitory propriospinal boutons synapse on the cell bodies of alpha motor neurons (FitzGerald and Folan-Curran, 2002). These connections indicate the extensive level of neuronal integration occurring at this cell. Gamma motor neurons are smaller (10 to 30 µm) and have a lower threshold to stimuli than alpha motor neurons (Davidoff and Hackman, 1991; Williams et al., 1995). In addition to the motor neurons, lamina IX includes some interneurons and propriospinal neurons (Carpenter and Sutin, 1983; Williams et al., 1995; Kiernan, 1998).
The last lamina to be mentioned is lamina X. This region forms the commissural area between the two halves of the gray matter and surrounds the central canal. It consists of interneurons and some decussating axons. Lamina X receives input from A-delta fibers transmitting information from mechanical nociceptors and group C visceral afferents (Willis and Coggeshall, 1991).
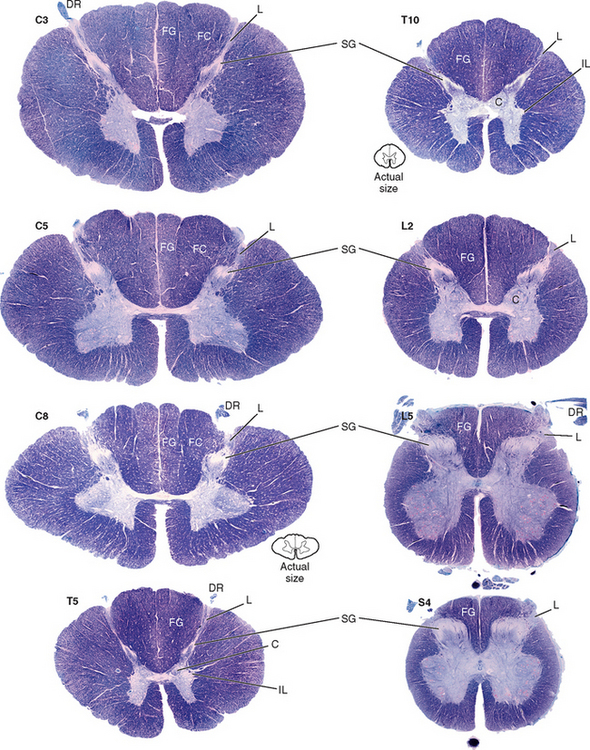
In summary, the gray matter is divided into 10 laminae, each of which may vary in size and shape within the different cord segments (Fig. 9-6). This variation results in a notable difference in the overall amount and shape of gray matter present in the various regions of the spinal cord. Also, there is a difference in the appearance and amount of white matter throughout the cord because of the presence or absence of certain tracts at different levels. For example, the amount of white matter is greater in the cervical cord than in other areas because all ascending and descending tracts to and from the brain traverse this region (Fig. 9-6). Table 9-4 summarizes the types of neuron cell bodies found in each lamina, the primary afferent fiber types terminating in each lamina, and the laminae in which descending (motor) fibers from higher centers synapse.
Dorsal Root Entry Zone
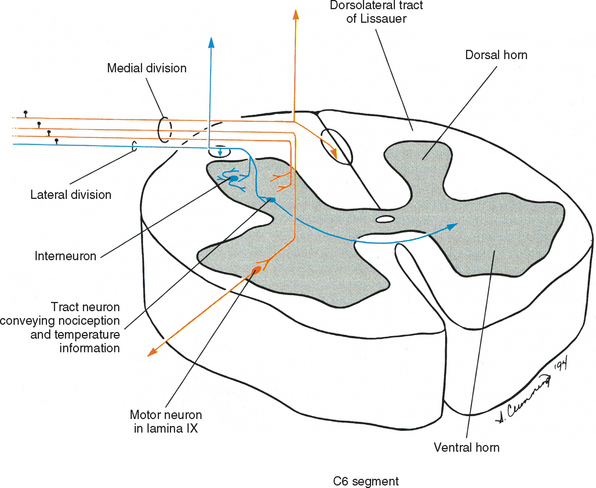
As the rootlet fibers enter the dorsal root entry zone, they become arranged into lateral and medial divisions (Fig. 9-7). The lateral division contains thinly myelinated and unmyelinated fibers, which include the nociceptive (pain) and temperature, or A-delta and C fibers. These fibers first enter an area of white matter located at the tip of the dorsal horn called the dorsolateral tract of Lissauer and then continue into the gray matter of the cord. The dorsolateral tract of Lissauer is found in all cord segments, but it is most developed in the upper cervical segments. Collateral branches of the entering fibers are given off within the dorsolateral tract of Lissauer, some of which ascend or descend a few segments before also synapsing in the gray matter. It has been suggested that visceral afferent fibers may give off collateral branches that span as many as five segments (Chandler, Zhang, and Foreman, 1996; Jänig, 1996). In addition to the ascending and descending branches, the dorsolateral tract of Lissauer also contains fibers from the substantia gelatinosa (lamina II) that interconnect with laminae II of other levels. Other propriospinal fibers also have been identified in the dorsolateral tract (Williams et al., 1995). At all levels, the lateral division primary afferent fibers may synapse on tract neurons in the dorsal horn and intermediate gray, interneurons in the dorsal horn, and interneurons involved with somatic and visceral reflex responses.
When the primary afferents enter the dorsal horn, they synapse in the gray matter and release neurotransmitters. These neurotransmitters include excitatory amino acids and neuropeptides. Many primary afferents appear to release glutamate (or aspartate or both) as their primary neurotransmitter. Glutamate has been localized in peripheral nerves, dorsal root ganglia, dorsal roots, and the dorsal horn. Excitatory neuropeptides found in the presynaptic terminals of small-diameter primary afferent fibers include substance P and calcitonin gene-related peptide (CGRP). Substance P is synthesized in the cell bodies found in the dorsal root ganglia and is transported (via fast axonal transport) to the terminals of small-diameter nociceptive fibers in the dorsal horn. The fibers’ terminals are prevalent in laminae I and II. CGRP has been identified in the cell bodies found in the dorsal root ganglia (often co-localized along with substance P), in group A-delta and C dorsal root fibers, Lissauer’s tract, and the presynaptic terminals of afferent fibers that synapse in dorsal horn laminae. Additional excitatory neuropeptides released by small-diameter afferent fibers, which are less well known, include somatostatin, cholecystokinin, thyrotropin-releasing hormone, and vasoactive intestinal polypeptide (Willis and Coggeshall, 1991).
White Matter
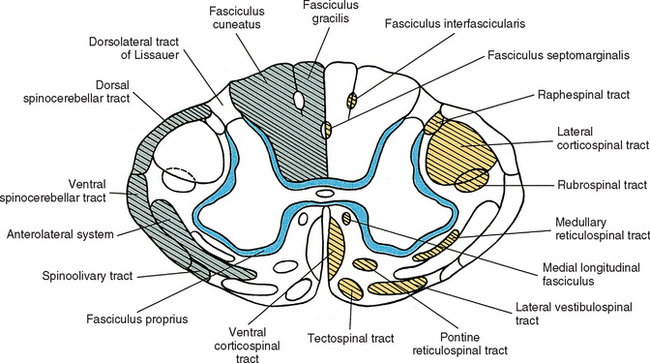
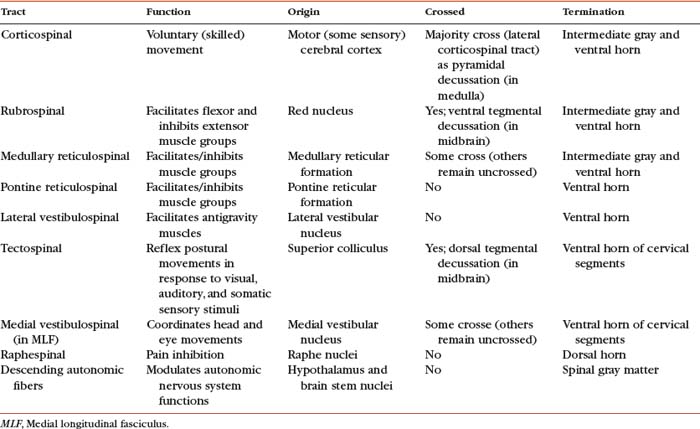
The white matter of the spinal cord is seen in cross section to be a distinct region located peripheral to the gray matter. It contains myelinated and unmyelinated axons, glial cells, and capillaries. The white matter consists of three regions: a dorsal funiculus (column) between the dorsal horns, a lateral funiculus (column) between each dorsal horn and ventral horn, and a ventral funiculus (column) between the ventral horns. Tracts, or fasciculi, are present within each of these regions (Fig. 9-8). These may be ascending tracts, which convey sensory information to higher centers, or descending tracts, which originate in higher centers and send descending signals to the cord. These descending signals are involved primarily with some type of motor information. Other axons are confined to the spinal cord and interconnect cord segments at various levels. These form the fasciculus proprius, which is an area of white matter located immediately adjacent to the gray matter. It consists of descending and ascending axons of propriospinal neurons. Concerning the ascending and descending tracts associated with higher centers, each tract contains axons that have a common origin, destination, and function. During a neurologic examination, the integrity of certain tracts is tested. Therefore familiarity of the location of these tracts aids the clinician in localizing lesions within the CNS.
Ascending Tracts.
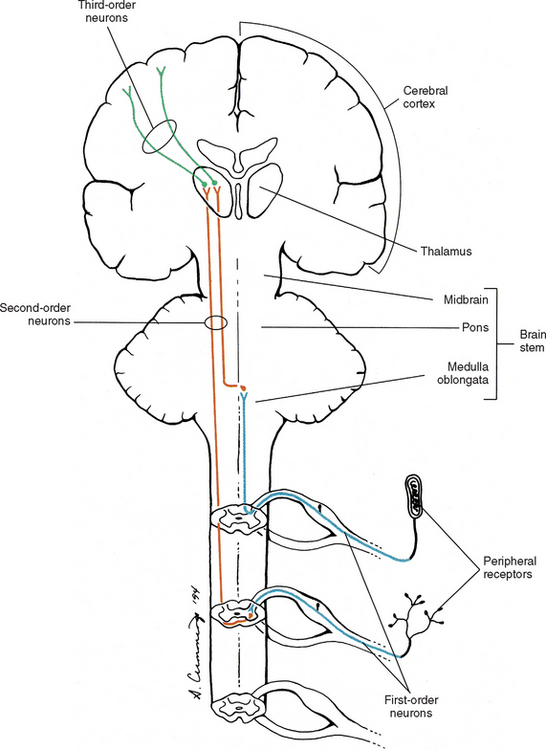
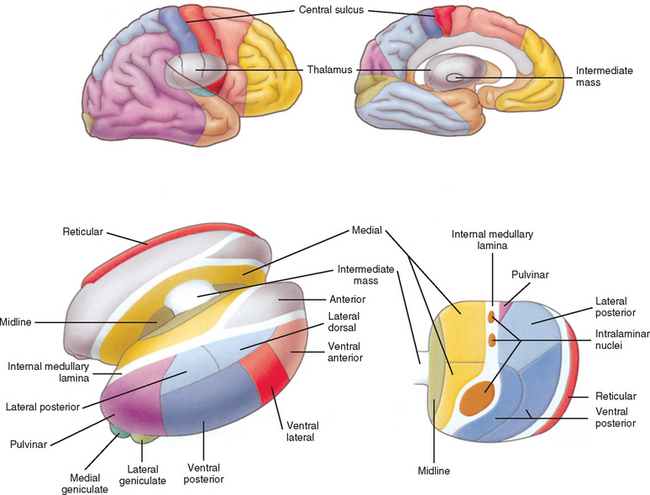
In general, an ascending tract can be classified into one of three groups based on the three different functions that are provided by the somatosensory system. One group contains tracts that transmit information concerning the type, location, and intensity of a stimulus. These are considered to be direct or discriminatory tracts. A second group consists of tracts that transmit information about the initiation of reflexes that may be concerned with arousal, affective, and motivational responses to a stimulus. The third group of tracts includes those that transmit information concerning the unconscious monitoring and control of motor activity, such as posture and movement (Benarroch et al., 1999). Ascending tracts convey information that has originated from a stimulated peripheral receptor located in the skin, muscles, tendons, joints, or viscera. When a receptor is stimulated, it transmits an action potential via the peripheral and central processes of sensory (afferent) neurons to the CNS. The sensory fibers that convey the action potential from the peripheral receptors are sometimes called first-order neurons, because they are the first neuron in a chain of neurons that proceeds to a higher center such as the cerebral cortex for conscious awareness of the stimulus, or the cerebellar cortex for regulation of motor patterns (Fig. 9-9). On entering the cord, numerous first-order neurons synapse on neurons in the gray matter of the spinal cord, whereas others, after contributing collateral branches to the gray matter, ascend and synapse in nuclear gray matter in the caudal medulla of the brain stem. The next neuron of the chain that leaves the gray matter of the cord or medulla to ascend to higher centers is known as the second-order neuron. Along with many others, this neuron makes up a specific tract. If sensory information is perceived consciously, the second-order neuron decussates and subsequently synapses with a third-order neuron in the thalamus, which is located in the diencephalon of the brain. The thalamus is an oval-shaped area of gray matter that consists of numerous nuclear subgroups (Fig. 9-10). All sensory information traveling to the cerebral cortex (except olfaction) synapses in one of these thalamic nuclei. From here, the third-order thalamic neuron fibers course in a thick bundle of axons called the internal capsule (which also includes efferent fibers from the cerebral cortex) and terminate in the cortex, thus completing the chain. The thalamus is not simply a relay station through which sensory information is routed to reach its cortical destination. In fact, there is a crude awareness of pain and temperature sensations at this level. In reality, the thalamus functions more like a complex processing center, or gatekeeper. Through the processing within a thalamic nucleus, and in conjunction with excitatory and inhibitory input from other centers (e.g., brain stem nuclei, reticular formation, and cerebral cortex), the thalamus is able to modulate its sensory output to the cerebral cortex based on the immediate needs of the individual (Amaral, 2000).
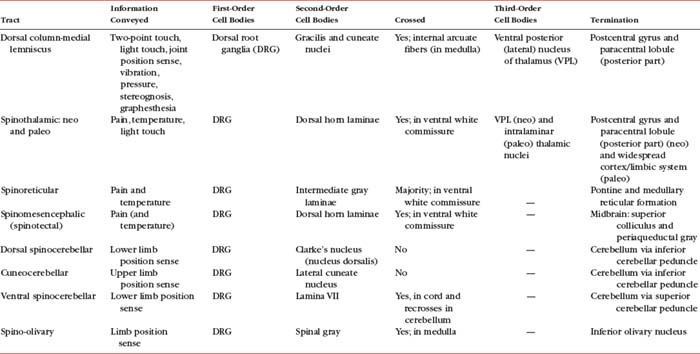
Certain important characteristics should be identified when considering the tracts of the CNS. These include the direction of the tract (i.e., ascending or descending, which usually is indicated by the name), the specific type of information the tract is conveying, if the tract crosses, and the location of crossing. This information is summarized in Tables 9-5 and 9-6. The ascending tracts are discussed first, beginning with the most clinically and anatomically relevant. These major tracts are well defined, and much information has been gathered about them. Secondary tracts are then discussed, about which limited information is available. They appear to supplement the major tracts by conveying similar types of information.
Dorsal column-medial lemniscal system.
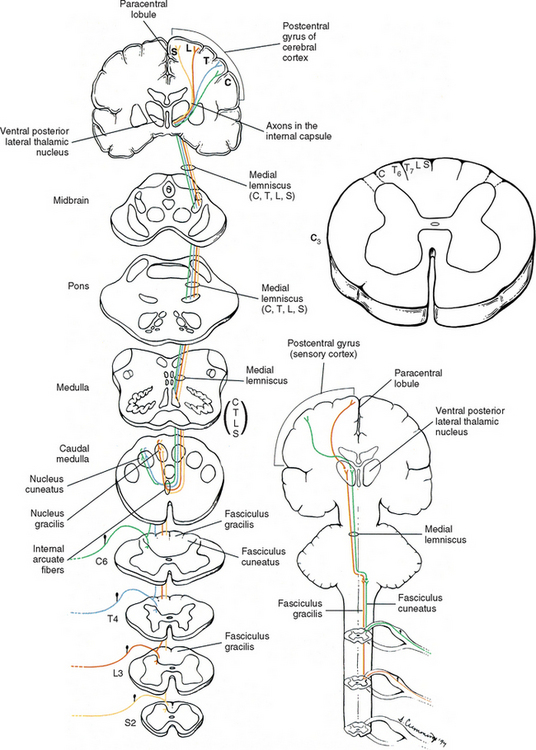
The first system of ascending fibers to be discussed is the dorsal column-medial lemniscus (DC-ML). Dorsal column refers to the first-order fibers located ipsilaterally (in reference to the side of fiber entry) in the dorsal white column of the spinal cord. Medial lemniscus refers to the second-order fibers located contralaterally in the brain stem. The DC-ML system conveys discriminatory (two-point) touch, some light (crude) touch, pressure, vibration, and joint position sense (conscious proprioception). This input provides temporal and spatial discriminatory qualities that allow for the conscious awareness of body part position at rest and during movements, and the conscious appreciation of the intensity and localization of touch, pressure, and vibration.
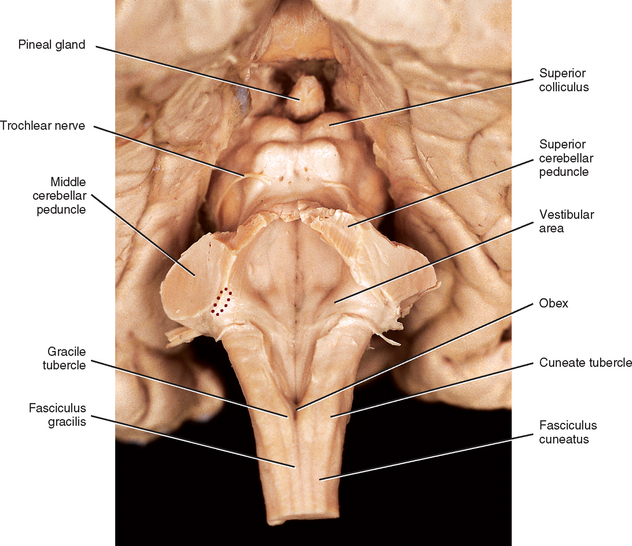
The peripheral receptors, which are mechanoreceptors (e.g., neuromuscular spindles, joint receptors, GTOs), have been discussed. The afferent fibers of these mechanoreceptors are large diameter; therefore they are located in the medial division of the dorsal root entry zone and subsequently enter the cord just medial to the dorsolateral tract of Lissauer. As these first-order neurons enter, they bifurcate into long ascending and short descending branches. The descending branches descend as the fasciculus interfascicularis in the upper half of the cord and as the fasciculus septomarginalis in the lower cord segments (see Fig. 9-8). These synapse in spinal gray matter and are involved in mediating reflex responses. The longer fibers contribute collateral branches to the spinal gray matter to participate in intersegmental reflexes and ascend ipsilaterally in the dorsal (white) column of the cord and continue into the medulla of the brain stem (Fig. 9-11). The first synapse occurs here in the nuclei gracilis and cuneatus, which are deep to the tubercles of the same name (Fig. 9-12). As the first-order neurons enter the dorsal white column, each neuron comes to lie more laterally to the fibers that entered more inferiorly. For example, information entering via a lumbar nerve ascends in the dorsal column in axons located lateral to the axons conveying information entering via a sacral nerve. In a cross section of the C3 spinal cord, first-order neurons conveying information (specific for the DC-ML system) from areas of the body innervated by sacral nerves are found most medial, followed in a lateral sequence by axons conveying information from areas innervated by lumbar, thoracic, and cervical nerves (see Fig. 9-11).
At the midthoracic level of the cord and above, the dorsal column is divided by the dorsal intermediate sulcus into a medial fasciculus gracilis and lateral fasciculus cuneatus. The dorsal intermediate sulcus extends ventrally from the cord’s periphery to approximately one half of the way into the dorsal column. This sulcus acts as a mechanical barrier preventing medial migration of the cuneate fibers (Smith and Deacon, 1984). Thus the fasciculus gracilis, which is found in all cord segments, includes axons of middle and lower thoracic, lumbar, and sacral nerves and therefore generally conveys information from the ipsilateral lower extremity. Smith and Deacon (1984) investigated the dorsal columns of human spinal cords and found that the orientation of the fasciculus gracilis fibers varied. In the most caudal part of the fasciculus, the fibers were oriented parallel to the medial side of the dorsal horn, whereas the upper lumbar and lower thoracic fibers were parallel to the dorsal median septum. However, the fibers in the fasciculus gracilis located in the remaining cord segments were oriented obliquely in a ventromedial-to-dorsolateral fashion. The authors also found that some overlapping of fibers occurred within the fasciculus gracilis but little if any occurred between the fasciculus gracilis and fasciculus cuneatus.
The fasciculus cuneatus includes axons of upper and middle thoracic and cervical nerves and, in general, conveys information from the ipsilateral upper extremity (see Fig. 9-11).
In addition to the mediolateral arrangement of fibers in the dorsal column, the type of modality is organized during the fibers’ ascent such that input from hair receptors is superficial, whereas tactile and vibratory information ascends via deeper fibers (Williams et al., 1995). Smith and Deacon (1984) demonstrated that the cross-sectional shape of each fasciculus (both gracilis and cuneatus) is different in each of the upper thoracic and cervical segments, and thus characteristic of that particular segment. The authors also believe that the fasciculi gracilis and cuneatus should be regarded as separate anatomic entities.
As mentioned, the first-order neurons of the fasciculi gracilis and cuneatus synapse with second-order neurons in the nuclei of the same name in the caudal medulla. The axons of the second-order neurons decussate (cross) in the caudal medulla (in a region called the sensory or lemniscal decussation); as they do so, they form a bundle of fibers known as the internal arcuate fibers (see Fig. 9-11). These second-order neurons then ascend through the brain stem as a fiber bundle known as the medial lemniscus. The lemniscal fibers are organized in the medulla such that information originating from the lower extremity and transmitted to the spinal cord in lumbar and sacral nerves is conveyed by fibers that are ventral to fibers conveying information originating from the upper extremity and transmitted to the cord in (primarily) cervical nerves. In the pons the fibers shift so that the lower extremity information is conveyed by fibers located lateral to the fibers conveying information from the upper extremity. In the midbrain of the brain stem, the lower extremity fibers become located dorsolateral to the upper extremity fibers. Clinically, it is imperative to recognize that the decussation of fibers occurs in the medulla. A unilateral lesion (e.g., trauma, vascular insufficiency, tumor) in the medial lemniscus of the brain stem produces contralateral deficits (e.g., loss of vibration, loss of joint position sense), whereas a lesion in the dorsal column of the spinal cord produces ipsilateral deficits.
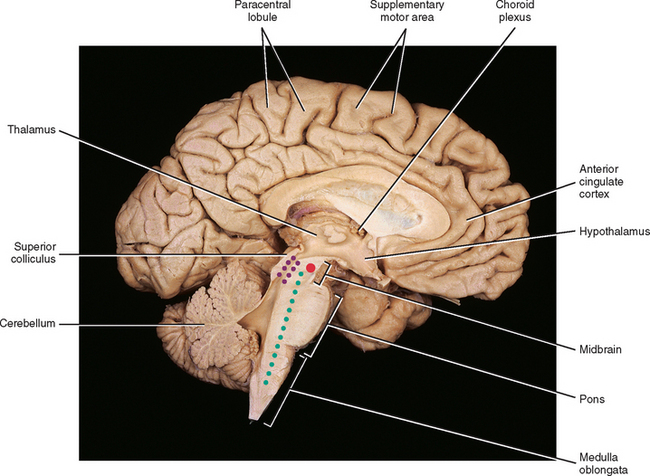
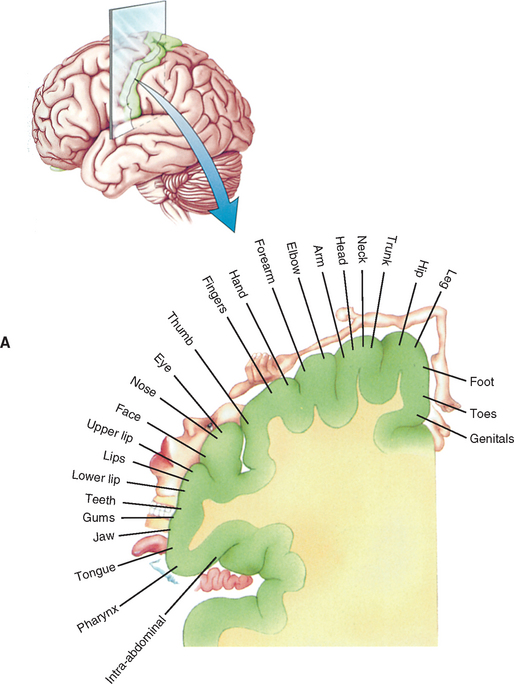
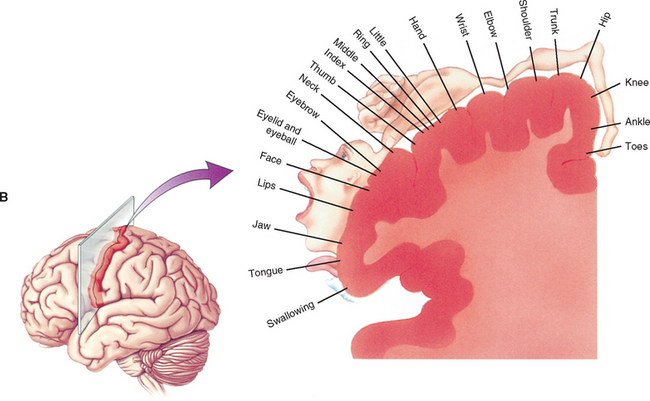
The medial lemniscus ascends to the ventral posterior (lateral part) nucleus of the thalamus and synapses on third-order neurons (see Figs. 9-10, 9-11, and 9-14). The axons of the third-order neurons travel in the internal capsule (a mass of axons going to and coming from the cerebral cortex) by way of the corona radiata to the primary sensory area of the cerebral cortex, which is located in the postcentral gyrus and paracentral lobule (posterior part) of the parietal lobe (Figs. 9-13 and 9-14; see also Fig. 9-11). From here, neurons project to adjacent cortical areas for further higher-level processing. This includes the integration of the information that is useful for the execution of movements and the same for the appreciation of the significance of the sensory input. For example, through the sense of touch and without visual input, one is able to recognize an object placed in the hand (stereognosis) or identify numbers or letters traced on the skin (graphesthesia). The DC-ML system maintains the spatial relationships of all parts of the body throughout its course in the CNS and allows the surface and underlying body structures to be mapped onto the primary sensory area of the cerebral cortex. This arrangement is called somatotopic organization. This organization is illustrated by a mapping of the entire body surface on the somatosensory cortex that depicts the location and amount of cortex dedicated to the processing of sensory information from a particular part of the body. The map, called the homunculus, represents the density of the innervation, rather than the size, of that particular body part (Fig. 9-15).
Anterolateral system.
The remaining tracts discussed in this chapter follow a basic plan in which first-order neurons terminate in the spinal cord gray matter. One group of tracts conveys nociception (pain) and temperature and some light touch. The tracts of this group ascend in the anterolateral quadrant of the spinal cord white matter and are collectively called the anterolateral system (ALS). This system consists of the spinothalamic, spinoreticular, and spinomesencephalic tracts (Young, 1986; Willis and Coggeshall, 1991; Basbaum and Jessell, 2000; Nolte, 2002). Nociception is conveyed by fast-conducting A-delta fibers (6 to 30 m/s) and slow-conducting C fibers (0.5 to 2 m/s) and is divided into two main types of pain. The A-delta fibers convey nociceptive information that is precisely localized and is perceived as being sharp, acute, or pinpricklike pain. It is perceived approximately 0.1 seconds after the stimulus has occurred and acts to alert the individual to potential tissue damage. It is typically confined to the skin. Slow-conducting C fibers convey nociceptive information that is described as burning, aching, or throbbing pain and is felt 1 second or later after the stimulus has occurred. This pain is appreciated secondarily to the fast pain, lasts much longer, and is poorly localized. It can occur from damage in any tissue (Snell, 2001). Temperature sense is also conveyed by A-delta and C fibers.
The first-order neurons of all of the ALS tracts enter the cord via the lateral division of the dorsal root entry zone and pass into the dorsolateral tract of Lissauer. Here, collateral branches ascend and descend, as well as enter and then synapse, in spinal cord laminae (Fig. 9-16). The spinothalamic tract originates primarily in laminae I, IV to VI, and even from laminae VII and VIII (Young, 1986; Hodge and Apkarian, 1990; Noback, Strominger, and Demarest, 1991; Willis and Coggeshall, 1991; Williams et al., 1995; Willis and Westlund, 1997). The second-order fibers decussate in the cord’s ventral white commissure within one or two segments of entry, although recent studies suggest that the fibers cross transversely rather than diagonally (Nathan, Smith, and Deacon, 2001
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree