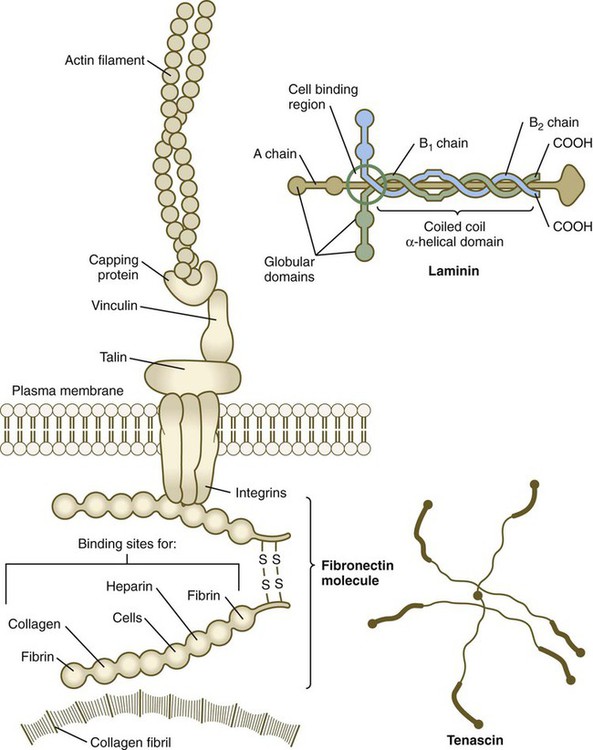
The neural crest, whose existence has been recognized for more than a century, forms an exceptionally wide range of cell types and structures, including several types of nerves and glia, connective tissue, bones, and pigment cells. Its importance and prominence are such that the neural crest has often been called the fourth germ layer of the body. Not until adequate methods of marking neural crest cells became available—first with isotopic labels and subsequently with stable biological markers, monoclonal antibodies, intracellular dyes, and genetic markers—did the neural crest become one of the most widely studied components of the vertebrate embryo. Most studies on the neural crest have been conducted on the avian embryo because of its accessibility and the availability of specific markers (see Fig. 9.31). More recently, emphasis has shifted to studies on the mouse, especially for dissecting molecular controls, but it appears that most of the information on the biology of the neural crest derived from birds can be applied to mammalian embryos. Some important syndromes and malformations are based on abnormalities of the neural crest. Some of these syndromes are presented in Clinical Correlation 12.1, at the end of the chapter. According to the most recent data, the earliest stages of neural crest induction may occur as early as gastrulation, but according to the classical model, the neural crest arises as the result of inductive actions by the adjacent non-neural ectoderm and possibly nearby mesoderm on the neural plate (Fig. 12.1). The ectodermal inductive signals are bone morphogenetic proteins (BMPs) and Wnts. Fibroblast growth factor-8 (FGF-8) from mesoderm plays a role in neural crest induction in amphibians, and it seems to be involved in mammals as well. The role of BMPs is complex and relates to a concentration gradient along the ectodermal layer as neurulation proceeds. The highest concentrations of BMP are seen in the lateral ectoderm, and cells exposed to these concentrations remain ectodermal. Cells within the neural plate are exposed to the lowest concentrations of BMP because of the local inhibitory actions of noggin and chordin (see Fig. 5.8C), and, by default, they remain neural. Cells at the border of the neural plate are exposed to intermediate levels of BMP, and, in this environment, they are induced to form neural crest precursor cells. Within the neural tube, neural crest precursor cells are epithelial and are tightly adherent to other neuroepithelial cells through a variety of intercellular connections. Prominent among them are the cadherins. Among the new transcription factors upregulated in induced neural crest precursor cells are snail-1 and snail-2 (formerly called slug) and Foxd-3, which are instrumental in allowing the neural crest cells to break free from the neural epithelium and then migrate away as mesenchymal cells.* Under the influence of snail-1 and snail-2, the profile of cadherins produced by the neural crest precursors changes from type I cadherins (e.g., N-cadherin and E-cadherin), which are strongly adhesive, to type II cadherins, which are less adhesive. After leaving the neuroepithelium, the neural crest cells first encounter a relatively cell-free environment rich in extracellular matrix molecules (Fig. 12.2). In this environment, the cells undergo extensive migrations along several well-defined pathways. These migrations are determined by intrinsic properties of the neural crest cells and features of the external environment encountered by the migrating cells. Neural crest migration is influenced by a variety of molecules residing in the extracellular matrix. Although the presence of a basal lamina can inhibit their emigration from the neural tube, neural crest cells often prefer to migrate along basal laminae, such as those of the surface ectoderm or neural tube, after they have left the neural tube. Components of the extracellular matrix permissive for migration include molecules found in basal laminae, such as fibronectin, laminin, and type IV collagen (Fig. 12.3). Attachment to and migration over these substrate molecules are mediated by the family of attachment proteins called integrins. Other molecules, such as chondroitin sulfate proteoglycans, are not good substrates for neural crest cells and inhibit their migration. Neural crest cells emigrate from the neural tube or neural folds in streams, with each cell in contact with neighbors through filopodial contacts. During their migratory phase, neural crest cells are exquisitely sensitive to guidance molecules, most of which are inhibitory. Among the most important of these guidance molecules are the ligand/receptor pairs Robo/Slit, Neuropilin/Semaphorin and Ephrin/Eph (see Table 11.1). Much less is known about attractive influences on neural crest cell migration. During migration, neural crest cells extend protrusions that both test the environment and are part of the propulsive mechanism. If an inhibitory influence is encountered, the protrusions collapse through signals derived from a planar cell polarity pathway (see p. 87). This mechanism acts as a brake when the cells encounter an inhibitory environment, but it is also involved in their forward propulsion. In a migrating stream of neural crest cells, contact with the cells behind also results in the pulling of protrusions at the trailing edge of the cells, thus resulting in a net forward motion of the leading cells. Specific examples of the environmental control of neural crest cell migrations are given later in this chapter. Much remains to be learned about what causes neural crest cells to stop migrating, but often they stop migrating in areas where repulsive signals are low. Neural crest cells ultimately differentiate into an astonishing array of adult structures (Table 12.1). What controls their differentiation is one of the principal questions of neural crest biology. Two opposing hypotheses have been proposed. According to one, all neural crest cells are equal in developmental potential, and their ultimate differentiation is entirely determined by the environment through which they migrate and into which they finally settle. The other hypothesis suggests that premigratory crest cells are already programmed for different developmental fates, and that certain stem cells are favored, whereas others are inhibited from further development during migration. More recent research indicates that the real answer can be found somewhere between these two positions. Increasing evidence suggests that among migrating neural crest cells is a mix of cells whose fate has been predetermined within the neural tube and cells whose ultimate phenotype depends on environmental influences. Table 12.1 Major Derivatives of the Neural Crest A more striking example is the conversion of cells of the periocular neural crest mesenchyme, which in birds would normally form cartilage, into neurons if they are associated with embryonic hindgut tissue in vitro. Many of the regional influences on the differentiation of local populations of neural crest cells are now recognized to be interactions between the migrating neural crest cells and specific tissues that they encounter during migration. Examples of tissue interactions that promote the differentiation of specific neural crest derivatives are given in Table 12.2. Table 12.2 Environmental Factors Promoting Differentiation of Neural Crest Cells Many neural crest cells are bipotential, depending on signals from their local environment for cues to their final differentiation. Cultured heart cells secrete a protein that converts postmitotic sympathetic neurons from an adrenergic (norepinephrine transmitter) phenotype to a cholinergic (acetylcholine-secreting) phenotype (see Fig. 11.22). During normal development, the sympathetic neurons that innervate sweat glands are catecholaminergic until their axons actually contact the sweat glands. At that point, they become cholinergic. The neural crest of the trunk extends from the level of the sixth somite to the most caudal somites. Three pathways of migration are commonly described (Fig. 12.4). These pathways occur in different sequences and are subject to different controls. The first neural crest cells to leave the neural tube migrate around and between the somites, which are still in an epithelial configuration. Their migratory path follows the intersomitic blood vessels, and the cells rapidly reach the region of the dorsal aorta (see Fig. 12.4, pathway 1). It may be that at this early stage no other pathway is available to these migrating cells. These cells constitute the sympathoadrenal lineage.
Neural Crest
Developmental History of the Neural Crest
Origin, Induction, and Specification
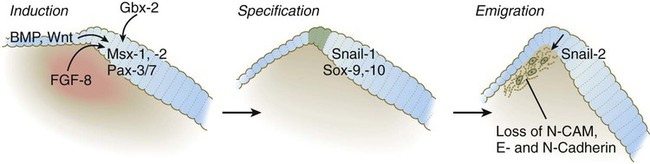
BMP, bone morphogenetic protein; FGF-8, fibroblast growth factor-8; N-CAM, neural cell adhesion molecule.
Epitheliomesenchymal Transformation and Emigration from the Neural Tube
Neural Crest Cell Migration
Differentiation of Neural Crest Cells
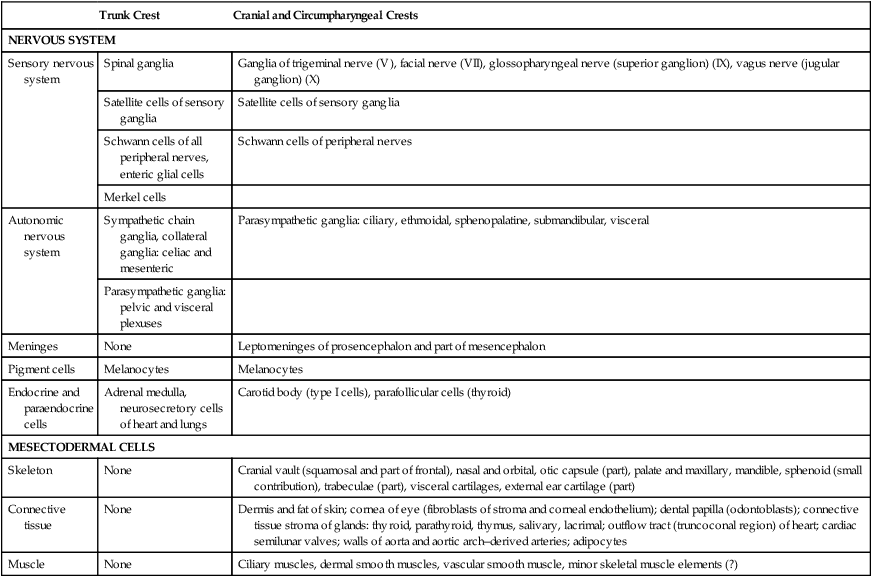
Trunk Crest
Cranial and Circumpharyngeal Crests
NERVOUS SYSTEM
Sensory nervous system
Spinal ganglia
Ganglia of trigeminal nerve (V), facial nerve (VII), glossopharyngeal nerve (superior ganglion) (IX), vagus nerve (jugular ganglion) (X)
Satellite cells of sensory ganglia
Satellite cells of sensory ganglia
Schwann cells of all peripheral nerves, enteric glial cells
Schwann cells of peripheral nerves
Merkel cells
Autonomic nervous system
Sympathetic chain ganglia, collateral ganglia: celiac and mesenteric
Parasympathetic ganglia: ciliary, ethmoidal, sphenopalatine, submandibular, visceral
Parasympathetic ganglia: pelvic and visceral plexuses
Meninges
None
Leptomeninges of prosencephalon and part of mesencephalon
Pigment cells
Melanocytes
Melanocytes
Endocrine and paraendocrine cells
Adrenal medulla, neurosecretory cells of heart and lungs
Carotid body (type I cells), parafollicular cells (thyroid)
MESECTODERMAL CELLS
Skeleton
None
Cranial vault (squamosal and part of frontal), nasal and orbital, otic capsule (part), palate and maxillary, mandible, sphenoid (small contribution), trabeculae (part), visceral cartilages, external ear cartilage (part)
Connective tissue
None
Dermis and fat of skin; cornea of eye (fibroblasts of stroma and corneal endothelium); dental papilla (odontoblasts); connective tissue stroma of glands: thyroid, parathyroid, thymus, salivary, lacrimal; outflow tract (truncoconal region) of heart; cardiac semilunar valves; walls of aorta and aortic arch–derived arteries; adipocytes
Muscle
None
Ciliary muscles, dermal smooth muscles, vascular smooth muscle, minor skeletal muscle elements (?)
Neural Crest Derivative
Interacting Structure
Bones of cranial vault
Brain
Bones of base of skull
Notochord, brain
Pharyngeal arch cartilages
Pharyngeal endoderm
Meckel’s cartilage
Cranial ectoderm
Maxillary bone
Maxillary ectoderm
Mandible
Mandibular ectoderm
Palate
Palatal ectoderm
Otic capsule
Otic vesicle
Dentin of teeth
Oral ectoderm
Glandular stroma: thyroid, parathyroid, thymus, salivary
Local epithelium
Adrenal medullary chromaffin cells
Glucocorticoids secreted by adrenal cortex
Enteric neurons
Gut wall
Sympathetic neurons
Spinal cord, notochord, somites
Sensory neurons
Peripheral target tissue
Pigment cells
Extracellular matrix along pathway of migration
Major Divisions of the Neural Crest
Trunk Neural Crest
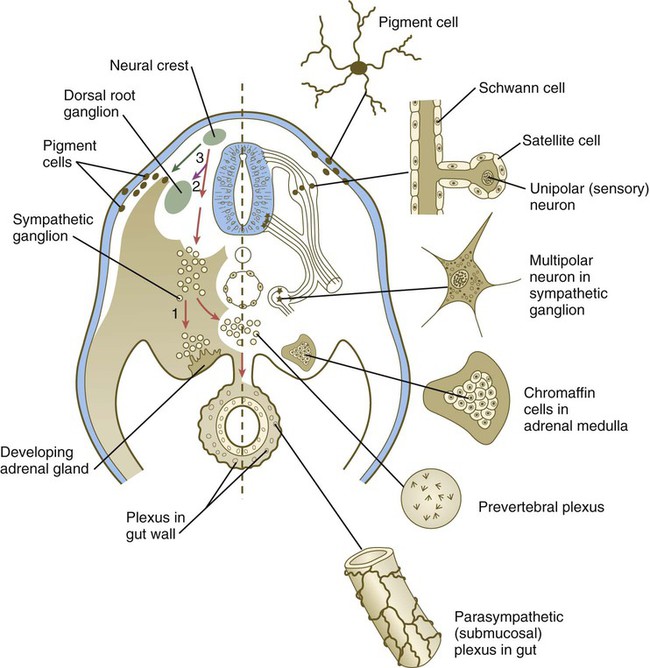
Left, Pathways in the early embryo. The first emigrating cells (pathway 1) follow the ventral (sympathoadrenal) pathway (red arrows). The second wave of emigrating cells (pathway 2) follows the ventrolateral pathway indicated by the purple arrow. The last cells to leave the neural tube (pathway 3) follow the dorsolateral pathway (green arrow) as they go on to differentiate into pigment cells.![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Neural Crest