The principle of the MIM process is illustrated in Fig. 2.3. Initially, powder and thermoplastic binder are mixed and kneaded in order to obtain the feedstock. This feedstock is heated and injected into a mold using a conventional injection molding machine. After the injection process, the binder is removed by heating, chemical extraction, or catalytic reaction, and then this is followed by sintering process to produce the final parts. The binder removal process is called the debinding process.
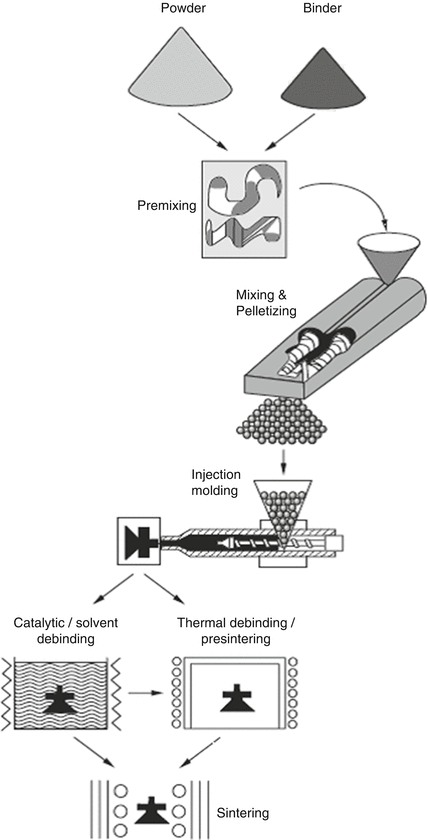

Fig. 2.3
Schematic diagram of MIM process (Reproduced with permission from Ref. [2]. Copyright 1996, Uchida Rokakuho Publishing Co., Ltd.)
The purpose of sintering is to densify the powder and to remove most of the void space left by the binders. Usually the shrinkage during sintering is in the range between 12 and 18 %; therefore, the mold is oversized to meet the required dimension. The MIM component has tolerance ranging from ±0.05 to ±0.3 %. After sintering process, the secondary operations such as grinding, machining, and drilling can be omitted. However, it depends on the accuracy of the product. For the mechanical parts, which need tighter tolerance, an approach to achieve tighter tolerance is to machine the critical surface after sintering. Hence, it is only applied for critical features. Surface roughness depends on the tools and also powder size. After sintering process, the sand blasting process can be done to reduce the surface roughness.
Another point is in the MIM process; the feedstock material can be recycled nearly 100 %. This will be beneficial for cost reduction, especially for the expensive materials. The total cost of binders, mold, debinding, and sintering process can be reduced by increasing the amount of production. In other words, the low cost of MIM process is suitable for the mass production. The summary of this section is that, with the capability of producing complex-shaped component with high properties at low cost, MIM process is expected to be one of the suitable processes to fabricate complex biomedical parts.
2.2.2 Biomedical Applications
MIM is suitable for the fabrication of small and complex-shaped parts; thus, several parts for medical applications were produced. Figure 2.4 shows the examples of biomedical components [3–7]. Those are fabricated from stainless steel, Ti alloy, and so on.


Fig. 2.4
MIM products for biomedical applications. (a) Orthodontic parts, SUS630, Ti, Al2O3; (b) surgical scalpel holder, SUS630; (c) fracture fixation plate, SUS316L; (d) heart valve, titanium; (e) an implant pump, Ti-6Al-4V; (f) dental implant, titanium
MIM is a very clean process compared to conventional manufacturing technologies. Only the possible binder residuals could represent potential candidates as toxic substances [8]. It is a good start to use exclusively nontoxic biocompatible binder components. Even if the single binder constituents may be harmless, such as paraffin or polyethylene, decomposition at high temperature could generally lead to new toxic substances. However, these substances would be trapped in pores after sintering and would cover the surface. In the first instance, they would have no contact with the body, and in the latter case, they could be removed if necessary. In the end, appropriate tests of the sintered components have to prove this.
MIM for the fabrication of medical implants is also interesting because of the possibility of generating porous components. These are beneficial with regard to tissue ingrowth. Porosity and pore diameter could be easily controlled. Figure 2.4f shows the implant that has a dense layer covered by a porous layer.
2.2.3 Titanium Alloys Fabricated by MIM
Ti and its alloys show excellent characteristics of low density, high strength, and high biocompatibility, leading to their widespread use for various industrial and biomedical applications. However, they show poor workability, so it is difficult to use them to produce parts with complicated shapes at low processing cost. Therefore, MIM process is a suitable technique for fabricating the complex-shaped parts to reduce the processing cost and material cost. Ti-6Al-4V is well known as the most widely used Ti-based alloy. Meanwhile, MIM can be used to produce various types of Ti alloys using mixed powders [8, 9].
As shown in Fig. 2.4, MIM is favored by a large number of small complex parts. Cosmetic parts had been successfully made from CP Ti powder, but for the structural applications, the oxygen level is still high especially for Ti-6Al-4V alloy. Nowadays, the oxygen content of Ti-6Al-4V products can be controlled to about 0.2 mass% with low oxygen level of starting powders, development of binders, and sintering facilities. This development promises a significant expansion of the titanium MIM marketplace into aerospace, automobiles, surgical instruments, dentistry, and so on.
2.3 Static Mechanical Properties of Ti Alloys Fabricated by MIM
In this section, the properties of injection molded Ti-6Al-4V alloy compacts using alloy powders and various mixed powders were investigated comprehensively to clarify the effect of oxygen content and relative density on the tensile properties.
On the other hand, Ti-6Al-7Nb alloy has been developed for more suitable biomaterial in place of Ti-6Al-4V alloy, because the vanadium (V) is toxic to the human body. There are few reports about MIM Ti-6Al-7Nb alloy, though the cost of starting material powder seems to be expensive because of not using a commercially available powder of gas-atomized Ti-6Al-7Nb alloy powder [10] or Al-53.8Nb pre-alloyed powder [11].
In this section, the metal injection molding process of Ti-6Al-7Nb alloy using an alloy powder and three types of mixed powders, which consisted of commercially available powders, is reviewed. The effects of powders and sintering conditions on the microstructure, relative density, and mechanical properties of injection molded compacts were mainly investigated.
2.3.1 Effect of Oxygen Content in Ti-6Al-4V Compacts
2.3.1.1 Experimental Procedures
A gas-atomized pure Ti powder (TILOP-45, Osaka Titanium Technologies Co., Ltd.), ground TiH2 powder with a different particle diameter and shape (TCH452, Toho Titanium Co., Ltd., etc.), two types of ground Al-40V alloy powders with different particle diameters (VAL-3, Nippon Denko Co., Ltd.), gas-atomized Al-40V alloy powder (60Al-40V, Daido Steel Co., Ltd.), and three types of Ti-6Al-4V alloy powders with different particle diameters and oxygen contents (TILOP64-45, Osaka Titanium Technologies Co., Ltd., etc.) were used in the experiment. The chemical compositions, particle diameter, and scanning electron microscope (SEM) images of these powders are shown in Table 2.1 and Fig. 2.5. The TiH2 powder is more cost-effective compared with gas-atomized Ti powder, which should reduce raw material costs [12]. The fine Al-40V powder shows higher oxygen content than the other Al-40V powders.

Table 2.1
Chemical composition and mean particle diameter of the powders
Chemical composition (mass%) | Particle diameter (μm) | ||||||
|---|---|---|---|---|---|---|---|
Al | V | Fe | O | C | |||
Ti | – | – | 0.044 | 0.130 | 0.008 | 24.4 | |
Ti-6Al-4V | 5.75 | 4.13 | 0.017 | 0.100 | 0.004 | 26.1 | |
TiH2 | – | – | 0.03 | 0.170 | – | 15.0 | |
Al-40 V | Fine | Bal. | 41.0 | 0.24 | 0.47 | 0.02 | 6.4 |
Coarse | Bal. | 39.7 | 0.33 | 0.20 | 0.01 | 20.0 | |
Atomized | 59.5 | Bal. | 0.13 | 0.12 | 0.02 | 26.0 | |

Fig. 2.5
SEM images of (a) titanium powder, (b) Ti-6Al-4V alloy powder, (c) titanium hydride (TiH2) powder, (d) fine Al-40V alloy powder, (e) coarse Al-40V alloy powder, and (f) atomized Al-40V powder
The pure Ti powder, TiH2 powder, and Al-40V alloy powder were weighed to obtain the chemical composition of Ti-6Al-4V and then were premixed for 3.6 ks by rotating in an argon gas-filled pot mill. The mixtures of Ti-6Al-4V alloy powder were then kneaded with binder [13] (which contained polypropylene, polymethyl methacrylate, paraffin wax, and stearic acid in a weight ratio of 30:40:29:1) using a pressure-type kneading machine (Moriyama Co., Ltd., DV1-5GHH-E) at 443 K for 8.1 ks. The binder loading was 35 vol%, and it increased with increasing TiH2 powder content because of its irregular shape and low density. The compounds obtained were crushed and screened from 2 to 8 mm diameter to prepare feedstocks for injection molding. The feedstocks were then injection molded by injection molding machine (Nissei Plastic Industrial Co., Ltd., ST20S2V) to produce tensile test specimens with a length of 75 mm, a width of 5 mm, and a thickness of 2 mm. After injection molding, solvent extraction debinding was conducted at 343 K for 21.3 ks in n-hexane [13] to partially remove the wax and polymethyl methacrylate. By this treatment, the contamination of sintered compacts by carbon derived from the binder system was reduced. Following this treatment, thermal debinding was performed from room temperature to 703 K in reduced pressure under argon gas flow. Continuous sintering was performed in high vacuum (in the order of below 10−2 Pa) at various temperatures for 3.6 to 28.8 ks, followed by furnace cooling to room temperature in high vacuum (in the order of below 10−3 Pa). The vacuum furnace (Shimadzu Mectem Inc., PVSGgr20/20) has both its heater elements and vessel made from graphite. The compacts were set on yttria or zirconia substrates in an inner vessel made from molybdenum or zirconia, and the compacts were surrounded by a spongelike Ti powder to prevent their carburization and oxidation.
The relative densities and tensile properties were measured on as-sintered compacts. The density of sintered compacts was measured by the Archimedean method with an automatic densimeter (Toyo Seiki Seisaku-Sho, Ltd., DENSIMETER H), and the relative density was calculated against the full density of Ti-6Al-4V alloy (4.42 Mg/m3). The tensile testing (in triplicate) was conducted using a universal testing instrument (Shimadzu Corp., AG-50kNIS) with an extensometer gauge length of 25 mm and crosshead speed of 8.33 × 10−5 m/s in air at room temperature. The oxygen content was determined by means of Oxygen/Nitrogen analyzer (Horiba, Ltd., EMGA-520).
2.4 Results and Discussion
Table 2.2 shows the typical properties of sintered Ti-6Al-4V alloy compacts discussed in this section. For details of these properties, refer to previous papers [12, 14–18].
Table 2.2
Characteristic properties of sintered Ti-6Al-4V alloy compacts
Powder | Sintering condition | Vessel/substrate | Relative density | Tensile strength | Elongation (%) | Oxygen content (%) |
|---|---|---|---|---|---|---|
Ti, TiH2, and coarse Al-40 V | 1,373 K X 28.8 ks | Mo/Y2O3 | 98.3 | 990 | 4.9 | 0.41 |
ZrO2/ZrO2 | 98.5 | 930 | 15.8 | 0.35 | ||
Ti and fine Al-40v | 1,473 K X 28.8 ks | Mo/ZrO2 | 98.5 | 890 | 9.6 | 0.35 |
ZrO2/ZrO2 | 97.7 | 880 | 14.5 | 0.27 | ||
Ti and coarse Al-40v | 1,373 K X 28.8 ks | Mo/Y2O3 | 98.4 | 820 | 13.7 | 0.24 |
Ti and atomized Al-40 V | 1,473 K X 14.4 ks | 96.7 | 790 | 12.3 | 0.20 | |
Ti-6Al-4V ELI | 1,423 K X 28.8 ks | 96.4 | 760 | 11.0 | 0.18 |
The relative density of the compacts using a mixture of Ti, TiH2, and Al-40V alloy powders increased with increasing TiH2 content. At the same time, the powder loading of compacts decreased with increasing TiH2 content because more binder was required to obtain enough flowability in the feedstock for injection molding. The tensile strength and oxygen content of the compacts using a mixture of Ti, TiH2, and Al-40V alloy powders increased with increasing TiH2 content. On the other hand, the elongation of the compacts using a mixture of Ti, TiH2, and Al-40V alloy powders reduced drastically with increasing TiH2 content [12].
The sintered compacts using a mixture of Ti and fine irregular Al-40V alloy powder showed the highest density, and the compacts using a mixture of Ti and coarse, irregular Al-40V alloy powder showed slightly higher density than the compacts using a mixture of Ti and spherical atomized Al-40V alloy powder. The sintered compacts using a mixture of Ti and fine irregular Al-40V alloy powder, which had the highest oxygen content compared with other Al-40V alloy powders, showed high oxygen content [14, 18]. Furthermore, the molybdenum vessel and yttria substrate could be effective to oxidation of the compacts from sintering atmosphere [15], which resulted in enough ductility, even for the prolonged sintering time at high sintering temperature to obtain high density.
The sintered compacts using Ti-6Al-4V alloy powders showed slightly lower relative density compared with the compacts using mixed powders, and their tensile properties were slightly low [16]. However, they were improved by prolonged sintering time and higher sintering temperature or using a small particle size powder [17]. The oxygen content of the sintered compacts using a Ti-6Al-4V ELI (extra-low interstitial) powder reached below 0.2 mass% as shown in Table 2.1 and Table 2.2.
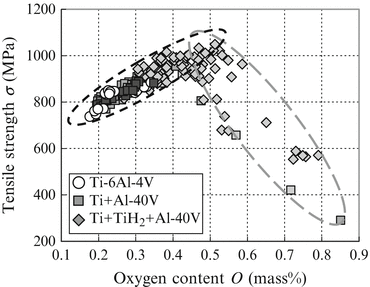
The relative density and oxygen content of the compacts in our previous studies cover a wide range from 90 to 99.8 % and 0.18 to 0.85 mass% O, respectively. As shown in Table 2.2, there is a tendency for the tensile strength of sintered compact to decrease with increasing oxygen content.
Figure 2.6 shows the effect of oxygen content on the elongation of sintered Ti-6Al-4V alloy compacts using various mixed powders or Ti-6Al-4V alloy powder. The tensile properties of the compacts with relative density below 94.5 % were excluded because of their poor properties and large fluctuation. The elongation of the compacts using any mixed powder or Ti-6Al-4V alloy powder and also any sintering conditions shows the excellent values, in excess of 10 %, when the oxygen content of the sintered compacts is below 0.35 mass%. However, the elongation of the sintered compacts is reduced dramatically if the oxygen content exceeds 0.35 mass%.
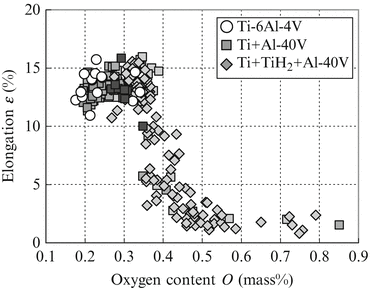

Fig. 2.6
Effect of oxygen content on the elongation of Ti-6Al-4V alloy compacts sintered under various conditions with various mixed or Ti-6Al-4V alloy powders
Figure 2.7 shows the effect of oxygen content on the elongation of sintered Ti-6Al-4V alloy compacts as a function of relative density for the oxygen content range from 0.1 to 0.35 mass%. The solid line shows the effect of oxygen content on the elongation of sintered Ti-6Al-4V alloy compacts which have relative densities from 97.5 to 98.5 %, as an approximated line. The oxygen content of sintered Ti-6AI-4V alloy compacts has little influence on the elongation when the sintered compacts have ductility greater than 10 % elongation. In addition, the relative density of sintered Ti-6Al-4V alloy compacts has very little influence on the elongation form the result of approximated line calculated for each relative density range.
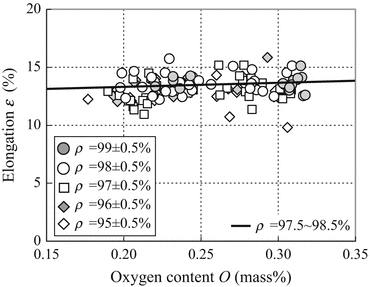

Fig. 2.7
The relationship between the oxygen content under 0.35 mass% and the elongation of Ti-6Al-4V alloy compacts sintered by various conditions with various mixed or Ti-6Al-4V alloy powders
Figure 2.8 shows the effect of oxygen content on the tensile strength of sintered Ti-6Al-4V alloy compacts using various mixtures of Ti-6Al-4V alloy powder. The tensile strength of the compacts using any mixed powder or Ti-6Al-4V alloy powder and also any sintering condition increases from approximately 800 to 1,000 MPa with increasing oxygen content below 0.5 mass%, but they are drastically reduced from the oxygen content above 0.5 mass%.