Other classes exhibit antiemetic properties. 5-HT3 antagonists such as ondansetron and granisetron can prevent postoperative vomiting, radiation therapy–induced symptoms, and cancer chemotherapy–induced emesis, but also are used for other causes of emesis with limited evidence for efficacy. Tricyclic antidepressant agents provide symptomatic benefit in patients with chronic idiopathic nausea and functional vomiting as well as in long-standing diabetic patients with nausea and vomiting. Other antidepressants such as mirtazapine and olanzapine also may exhibit antiemetic effects.
GASTROINTESTINAL MOTOR STIMULANTS
Drugs that stimulate gastric emptying are used for gastroparesis (Table 54-2). Metoclopramide, a combined 5-HT4 agonist and D2 antagonist, is effective in gastroparesis, but antidopaminergic side effects, such as dystonias and mood and sleep disturbances, limit use in ~25% of cases. Erythromycin increases gastroduodenal motility by action on receptors for motilin, an endogenous stimulant of fasting motor activity. Intravenous erythromycin is useful for inpatients with refractory gastroparesis, but oral forms have some utility. Domperidone, a D2 antagonist not available in the United States, exhibits prokinetic and antiemetic effects but does not cross into most brain regions; thus, anxiety and dystonic reactions are rare. The main side effects of domperidone relate to induction of hyperprolactinemia via effects on pituitary regions served by a porous blood-brain barrier.
Refractory motility disorders pose significant challenges. Intestinal pseudoobstruction may respond to the somatostatin analogue octreotide, which induces propagative small-intestinal motor complexes. Acetylcholinesterase inhibitors such as pyridostigmine are also observed to benefit some patients with small-bowel dysmotility. Pyloric injections of botulinum toxin are reported in uncontrolled studies to reduce gastroparesis symptoms, but small controlled trials observe benefits no greater than sham treatments. Surgical pyloroplasty has improved symptoms in case series. Placing a feeding jejunostomy reduces hospitalizations and improves overall health in some patients with drug-refractory gastroparesis. Postvagotomy gastroparesis may improve with near-total gastric resection; similar operations are now being tried for other gastroparesis etiologies. Implanted gastric electrical stimulators may reduce symptoms, enhance nutrition, improve quality of life, and decrease health care expenditures in medication-refractory gastroparesis, but small controlled trials do not report convincing benefits.
SAFETY CONSIDERATIONS
Safety concerns about selected antiemetics have been emphasized. Centrally acting antidopaminergics, especially metoclopramide, can cause irreversible movement disorders such as tardive dyskinesia, particularly in older patients. This complication should be carefully explained and documented in the medical record. Some agents with antiemetic properties including domperidone, erythromycin, tricyclics, and 5-HT3 antagonists can induce dangerous cardiac rhythm disturbances, especially in those with QTc interval prolongation on electrocardiography (ECG). Surveillance ECG testing has been advocated for some of these agents.
SELECTED CLINICAL SETTINGS
Some cancer chemotherapies are intensely emetogenic (Chap. 103e). Combining a 5-HT3 antagonist, an NK1 antagonist, and a glucocorticoid provides significant control of both acute and delayed vomiting after highly emetogenic chemotherapy. Unlike other drugs in the same class, the 5-HT3 antagonist palonosetron exhibits efficacy at preventing delayed chemotherapy-induced vomiting. Benzodiazepines such as lorazepam can reduce anticipatory nausea and vomiting. Miscellaneous therapies with benefit in chemotherapy-induced emesis include cannabinoids, olanzapine, and alternative therapies like ginger. Most antiemetic regimens produce greater reductions in vomiting than in nausea.
Clinicians should exercise caution in managing pregnant patients with nausea. Studies of the teratogenic effects of antiemetic agents provide conflicting results. Few controlled trials have been performed in nausea of pregnancy. Antihistamines such as meclizine and doxylamine, antidopaminergics such as prochlorperazine, and antiserotonergics such as ondansetron demonstrate limited efficacy. Some obstetricians offer alternative therapies such as pyridoxine, acupressure, or ginger.
Managing cyclic vomiting syndrome is a challenge. Prophylaxis with tricyclic antidepressants, cyproheptadine, or β-adrenoceptor antagonists can reduce the severity and frequency of attacks. Intravenous 5-HT3 antagonists combined with the sedating effects of a benzodiazepine like lorazepam are a mainstay of treatment of acute flares. Small studies report benefits with antimigraine agents, including the 5-HT1 agonist sumatriptan, as well as selected anticonvulsants such as zonisamide and levetiracetam.
INDIGESTION
MECHANISMS
The most common causes of indigestion are gastroesophageal reflux and functional dyspepsia. Other cases are a consequence of organic illness.
Gastroesophageal Reflux Gastroesophageal reflux results from many physiologic defects. Reduced lower esophageal sphincter (LES) tone contributes to reflux in scleroderma and pregnancy and may be a factor in some patients without systemic illness. Others exhibit frequent transient LES relaxations (TLESRs) that cause bathing of the esophagus by acid or nonacidic fluid. Overeating and aerophagia override the barrier function of the LES, whereas reductions in esophageal body motility or salivary secretion prolong fluid exposure. Increased intragastric pressure promotes gastroesophageal reflux in obesity. The role of hiatal hernias is controversial—most reflux patients have hiatal hernias, but most with hiatal hernias do not have excess heartburn.
Gastric Motor Dysfunction Disturbed gastric motility may contribute to gastroesophageal reflux in up to one-third of cases. Delayed gastric emptying is also found in ~30% of functional dyspeptics. Conversely, some dyspeptics exhibit rapid gastric emptying. The relation of these defects to symptom induction is uncertain; studies show poor correlation between symptom severity and degrees of motor dysfunction. Impaired gastric fundus relaxation after eating (i.e., accommodation) may underlie selected dyspeptic symptoms like bloating, nausea, and early satiety in ~40% of patients.
Visceral Afferent Hypersensitivity Disturbed gastric sensation is another pathogenic factor in functional dyspepsia. Visceral hypersensitivity was first reported in IBS with demonstration of heightened perception of rectal balloon inflation without changes in compliance. Similarly, ~35% of dyspeptic patients note discomfort with fundic distention to lower pressures than healthy controls. Others with dyspepsia exhibit hypersensitivity to chemical stimulation with capsaicin or with acid or lipid exposure in the duodenum. Some individuals with functional heartburn without increased acid or nonacid reflux are believed to have heightened perception of normal esophageal pH and volume.
Other Factors Helicobacter pylori has a clear etiologic role in peptic ulcer disease, but ulcers cause a minority of dyspepsia cases. H. pylori is a minor factor in the genesis of functional dyspepsia. Functional dyspepsia is associated with chronic fatigue, produces reduced physical and mental well-being, and is exacerbated by stress. Anxiety, depression, and somatization may have contributing roles in some cases. Functional MRI studies show increased activation of several brain regions, emphasizing contributions from central nervous system factors. Analgesics cause dyspepsia, whereas nitrates, calcium channel blockers, theophylline, and progesterone promote gastroesophageal reflux. Other stimuli that induce reflux include ethanol, tobacco, and caffeine via LES relaxation. Genetic factors may promote development of reflux and dyspepsia.
DIFFERENTIAL DIAGNOSIS
Gastroesophageal Reflux Disease Gastroesophageal reflux disease (GERD) is prevalent. Heartburn is reported once monthly by 40% of Americans and daily by 7–10%. Most cases of heartburn occur because of excess acid reflux, but reflux of nonacidic fluid produces similar symptoms. Alkaline reflux esophagitis produces GERD-like symptoms most often in patients who have had surgery for peptic ulcer disease. Ten percent of patients with heartburn exhibit normal esophageal acid exposure and no increase in nonacidic reflux (functional heartburn).
Functional Dyspepsia Nearly 25% of the populace has dyspepsia at least six times yearly, but only 10–20% present to clinicians. Functional dyspepsia, the cause of symptoms in >60% of dyspeptic patients, is defined as ≥3 months of bothersome postprandial fullness, early satiety, or epigastric pain or burning with symptom onset at least 6 months before diagnosis in the absence of organic cause. Functional dyspepsia is subdivided into postprandial distress syndrome, characterized by meal-induced fullness, early satiety, and discomfort, and epigastric pain syndrome, which presents with epigastric burning pain unrelated to meals. Most cases follow a benign course, but some with H. pylori infection or on nonsteroidal anti-inflammatory drugs (NSAIDs) develop ulcers. As with idiopathic gastroparesis, some cases of functional dyspepsia result from prior infection.
Ulcer Disease In most GERD patients, there is no destruction of the esophagus. However, 5% develop esophageal ulcers, and some form strictures. Symptoms cannot distinguish nonerosive from erosive or ulcerative esophagitis. A minority of cases of dyspepsia stem from gastric or duodenal ulcers. The most common causes of ulcer disease are H. pylori infection and use of NSAIDs. Other rare causes of gastroduodenal ulcers include Crohn’s disease (Chap. 351) and Zollinger-Ellison syndrome (Chap. 348), resulting from gastrin overproduction by an endocrine tumor.
Malignancy Dyspeptic patients often seek care because of fear of cancer, but few cases result from malignancy. Esophageal squamous cell carcinoma occurs most often with long-standing tobacco or ethanol intake. Other risks include prior caustic ingestion, achalasia, and the hereditary disorder tylosis. Esophageal adenocarcinoma usually complicates prolonged acid reflux. Eight to 20% of GERD patients exhibit esophageal intestinal metaplasia, termed Barrett’s metaplasia, a condition that predisposes to esophageal adenocarcinoma (Chap. 109). Gastric malignancies include adenocarcinoma, which is prevalent in certain Asian societies, and lymphoma.
Other Causes Opportunistic fungal or viral esophageal infections may produce heartburn but more often cause odynophagia. Other causes of esophageal inflammation include eosinophilic esophagitis and pill esophagitis. Biliary colic is in the differential diagnosis of unexplained upper abdominal pain, but most patients with biliary colic report discrete acute episodes of right upper quadrant or epigastric pain rather than the chronic burning, discomfort, and fullness of dyspepsia. Twenty percent of patients with gastroparesis report a predominance of pain or discomfort rather than nausea and vomiting. Intestinal lactase deficiency as a cause of gas, bloating, and discomfort occurs in 15–25% of whites of northern European descent but is more common in blacks and Asians. Intolerance of other carbohydrates (e.g., fructose, sorbitol) produces similar symptoms. Small-intestinal bacterial overgrowth may cause dyspepsia, often associated with bowel dysfunction, distention, and malabsorption. Eosinophilic infiltration of the duodenal mucosa is described in some dyspeptics, particularly with postprandial distress syndrome. Celiac disease, pancreatic disease (chronic pancreatitis, malignancy), hepatocellular carcinoma, Ménétrier’s disease, infiltrative diseases (sarcoidosis, eosinophilic gastroenteritis), mesenteric ischemia, thyroid and parathyroid disease, and abdominal wall strain cause dyspepsia. Gluten sensitivity in the absence of celiac disease is reported to evoke unexplained upper abdominal symptoms. Extraperitoneal etiologies of indigestion include congestive heart failure and tuberculosis.
55 | Diarrhea and Constipation |
Diarrhea and constipation are exceedingly common and, together, exact an enormous toll in terms of mortality, morbidity, social inconvenience, loss of work productivity, and consumption of medical resources. Worldwide, >1 billion individuals suffer one or more episodes of acute diarrhea each year. Among the 100 million persons affected annually by acute diarrhea in the United States, nearly half must restrict activities, 10% consult physicians, ~250,000 require hospitalization, and ~5000 die (primarily the elderly). The annual economic burden to society may exceed $20 billion. Acute infectious diarrhea remains one of the most common causes of mortality in developing countries, particularly among impoverished infants, accounting for 1.8 million deaths per year. Recurrent, acute diarrhea in children in tropical countries results in environmental enteropathy with long-term impacts on physical and intellectual development.
Constipation, by contrast, is rarely associated with mortality and is exceedingly common in developed countries, leading to frequent self-medication and, in a third of those, to medical consultation. Population statistics on chronic diarrhea and constipation are more uncertain, perhaps due to variable definitions and reporting, but the frequency of these conditions is also high. United States population surveys put prevalence rates for chronic diarrhea at 2–7% and for chronic constipation at 12–19%, with women being affected twice as often as men. Diarrhea and constipation are among the most common patient complaints presenting to internists and primary care physicians, and they account for nearly 50% of referrals to gastroenterologists.
Although diarrhea and constipation may present as mere nuisance symptoms at one extreme, they can be severe or life-threatening at the other. Even mild symptoms may signal a serious underlying gastrointestinal lesion, such as colorectal cancer, or systemic disorder, such as thyroid disease. Given the heterogeneous causes and potential severity of these common complaints, it is imperative for clinicians to appreciate the pathophysiology, etiologic classification, diagnostic strategies, and principles of management of diarrhea and constipation, so that rational and cost-effective care can be delivered.
NORMAL PHYSIOLOGY
While the primary function of the small intestine is the digestion and assimilation of nutrients from food, the small intestine and colon together perform important functions that regulate the secretion and absorption of water and electrolytes, the storage and subsequent transport of intraluminal contents aborally, and the salvage of some nutrients that are not absorbed in the small intestine after bacterial metabolism of carbohydrate allows salvage of short-chain fatty acids. The main motor functions are summarized in Table 55-1. Alterations in fluid and electrolyte handling contribute significantly to diarrhea. Alterations in motor and sensory functions of the colon result in highly prevalent syndromes such as irritable bowel syndrome (IBS), chronic diarrhea, and chronic constipation.
NORMAL GASTROINTESTINAL MOTILITY: FUNCTIONS AT DIFFERENT ANATOMIC LEVELS |
Abbreviation: MMC, migrating motor complex.
NEURAL CONTROL
The small intestine and colon have intrinsic and extrinsic innervation. The intrinsic innervation, also called the enteric nervous system, comprises myenteric, submucosal, and mucosal neuronal layers. The function of these layers is modulated by interneurons through the actions of neurotransmitter amines or peptides, including acetylcholine, vasoactive intestinal peptide (VIP), opioids, norepinephrine, serotonin, adenosine triphosphate (ATP), and nitric oxide (NO). The myenteric plexus regulates smooth-muscle function through intermediary pacemaker-like cells called the interstitial cells of Cajal, and the submucosal plexus affects secretion, absorption, and mucosal blood flow. The enteric nervous system receives input from the extrinsic nerves, but it is capable of independent control of these functions.
The extrinsic innervations of the small intestine and colon are part of the autonomic nervous system and also modulate motor and secretory functions. The parasympathetic nerves convey visceral sensory pathways from and excitatory pathways to the small intestine and colon. Parasympathetic fibers via the vagus nerve reach the small intestine and proximal colon along the branches of the superior mesenteric artery. The distal colon is supplied by sacral parasympathetic nerves (S2–4) via the pelvic plexus; these fibers course through the wall of the colon as ascending intracolonic fibers as far as, and in some instances including, the proximal colon. The chief excitatory neurotransmitters controlling motor function are acetylcholine and the tachykinins, such as substance P. The sympathetic nerve supply modulates motor functions and reaches the small intestine and colon alongside their arterial vessels. Sympathetic input to the gut is generally excitatory to sphincters and inhibitory to non-sphincteric muscle. Visceral afferents convey sensation from the gut to the central nervous system (CNS); initially, they course along sympathetic fibers, but as they approach the spinal cord they separate, have cell bodies in the dorsal root ganglion, and enter the dorsal horn of the spinal cord. Afferent signals are conveyed to the brain along the lateral spinothalamic tract and the nociceptive dorsal column pathway and are then projected beyond the thalamus and brainstem to the insula and cerebral cortex to be perceived. Other afferent fibers synapse in the prevertebral ganglia and reflexly modulate intestinal motility, blood flow, and secretion.
INTESTINAL FLUID ABSORPTION AND SECRETION
On an average day, 9 L of fluid enter the gastrointestinal (GI) tract, ~1 L of residual fluid reaches the colon, and the stool excretion of fluid constitutes about 0.2 L/d. The colon has a large capacitance and functional reserve and may recover up to four times its usual volume of 0.8 L/d, provided the rate of flow permits reabsorption to occur. Thus, the colon can partially compensate for excess fluid delivery to the colon that may result from intestinal absorptive or secretory disorders.
In the small intestine and colon, sodium absorption is predominantly electrogenic (i.e., it can be measured as an ionic current across the membrane because there is not an equivalent loss of a cation from the cell), and uptake takes place at the apical membrane; it is compensated for by the export functions of the basolateral sodium pump. There are several active transport proteins at the apical membrane, especially in the small intestine, whereby sodium ion entry is coupled to monosaccharides (e.g., glucose through the transporter SGLT1, or fructose through GLUT-5). Glucose then exits the basal membrane through a specific transport protein, GLUT-5, creating a glucose concentration gradient between the lumen and the intercellular space, drawing water and electrolytes passively from the lumen. A variety of neural and nonneural mediators regulate colonic fluid and electrolyte balance, including cholinergic, adrenergic, and serotonergic mediators. Angiotensin and aldosterone also influence colonic absorption, reflecting the common embryologic development of the distal colonic epithelium and the renal tubules.
SMALL-INTESTINAL MOTILITY
During the fasting period, the motility of the small intestine is characterized by a cyclical event called the migrating motor complex (MMC), which serves to clear nondigestible residue from the small intestine (the intestinal “housekeeper”). This organized, propagated series of contractions lasts, on average, 4 min, occurs every 60–90 min, and usually involves the entire small intestine. After food ingestion, the small intestine produces irregular, mixing contractions of relatively low amplitude, except in the distal ileum where more powerful contractions occur intermittently and empty the ileum by bolus transfers.
ILEOCOLONIC STORAGE AND SALVAGE
The distal ileum acts as a reservoir, emptying intermittently by bolus movements. This action allows time for salvage of fluids, electrolytes, and nutrients. Segmentation by haustra compartmentalizes the colon and facilitates mixing, retention of residue, and formation of solid stools. There is increased appreciation of the intimate interaction between the colonic function and the luminal ecology. The resident microorganisms, predominantly anaerobic bacteria, in the colon are necessary for the digestion of unabsorbed carbohydrates that reach the colon even in health, thereby providing a vital source of nutrients to the mucosa. Normal colonic flora also keeps pathogens at bay by a variety of mechanisms. In health, the ascending and transverse regions of colon function as reservoirs (average transit time, 15 h), and the descending colon acts as a conduit (average transit time, 3 h). The colon is efficient at conserving sodium and water, a function that is particularly important in sodium-depleted patients in whom the small intestine alone is unable to maintain sodium balance. Diarrhea or constipation may result from alteration in the reservoir function of the proximal colon or the propulsive function of the left colon. Constipation may also result from disturbances of the rectal or sigmoid reservoir, typically as a result of dysfunction of the pelvic floor, the anal sphincters, the coordination of defecation, or dehydration.
COLONIC MOTILITY AND TONE
The small intestinal MMC only rarely continues into the colon. However, short duration or phasic contractions mix colonic contents, and high-amplitude (>75 mmHg) propagated contractions (HAPCs) are sometimes associated with mass movements through the colon and normally occur approximately five times per day, usually on awakening in the morning and postprandially. Increased frequency of HAPCs may result in diarrhea or urgency. The predominant phasic contractions in the colon are irregular and non-propagated and serve a “mixing” function.
Colonic tone refers to the background contractility upon which phasic contractile activity (typically contractions lasting <15 s) is superimposed. It is an important cofactor in the colon’s capacitance (volume accommodation) and sensation.
COLONIC MOTILITY AFTER MEAL INGESTION
After meal ingestion, colonic phasic and tonic contractility increase for a period of ~2 h. The initial phase (~10 min) is mediated by the vagus nerve in response to mechanical distention of the stomach. The subsequent response of the colon requires caloric stimulation (e.g., intake of at least 500 kcal) and is mediated, at least in part, by hormones (e.g., gastrin and serotonin).
DEFECATION
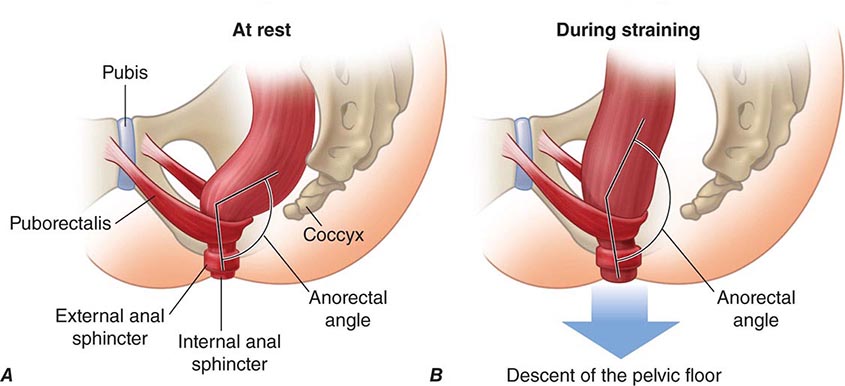
Tonic contraction of the puborectalis muscle, which forms a sling around the rectoanal junction, is important to maintain continence; during defecation, sacral parasympathetic nerves relax this muscle, facilitating the straightening of the rectoanal angle (Fig. 55-1). Distention of the rectum results in transient relaxation of the internal anal sphincter via intrinsic and reflex sympathetic innervation. As sigmoid and rectal contractions, as well as straining (Valsalva maneuver), which increases intraabdominal pressure, increase the pressure within the rectum, the rectosigmoid angle opens by >15°. Voluntary relaxation of the external anal sphincter (striated muscle innervated by the pudendal nerve) in response to the sensation produced by distention permits the evacuation of feces. Defecation can also be delayed voluntarily by contraction of the external anal sphincter.
FIGURE 55-1 Sagittal view of the anorectum (A) at rest and (B) during straining to defecate. Continence is maintained by normal rectal sensation and tonic contraction of the internal anal sphincter and the puborectalis muscle, which wraps around the anorectum, maintaining an anorectal angle between 80° and 110°. During defecation, the pelvic floor muscles (including the puborectalis) relax, allowing the anorectal angle to straighten by at least 15°, and the perineum descends by 1–3.5 cm. The external anal sphincter also relaxes and reduces pressure on the anal canal. (Reproduced with permission from A Lembo, M Camilleri: N Engl J Med 349:1360, 2003.)
DIARRHEA
DEFINITION
Diarrhea is loosely defined as passage of abnormally liquid or unformed stools at an increased frequency. For adults on a typical Western diet, stool weight >200 g/d can generally be considered diarrheal. Diarrhea may be further defined as acute if <2 weeks, persistent if 2–4 weeks, and chronic if >4 weeks in duration.
Two common conditions, usually associated with the passage of stool totaling <200 g/d, must be distinguished from diarrhea, because diagnostic and therapeutic algorithms differ. Pseudodiarrhea, or the frequent passage of small volumes of stool, is often associated with rectal urgency, tenesmus, or a feeling of incomplete evacuation, and accompanies IBS or proctitis. Fecal incontinence is the involuntary discharge of rectal contents and is most often caused by neuromuscular disorders or structural anorectal problems. Diarrhea and urgency, especially if severe, may aggravate or cause incontinence. Pseudodiarrhea and fecal incontinence occur at prevalence rates comparable to or higher than that of chronic diarrhea and should always be considered in patients complaining of “diarrhea.” Overflow diarrhea may occur in nursing home patients due to fecal impaction that is readily detectable by rectal examination. A careful history and physical examination generally allow these conditions to be discriminated from true diarrhea.
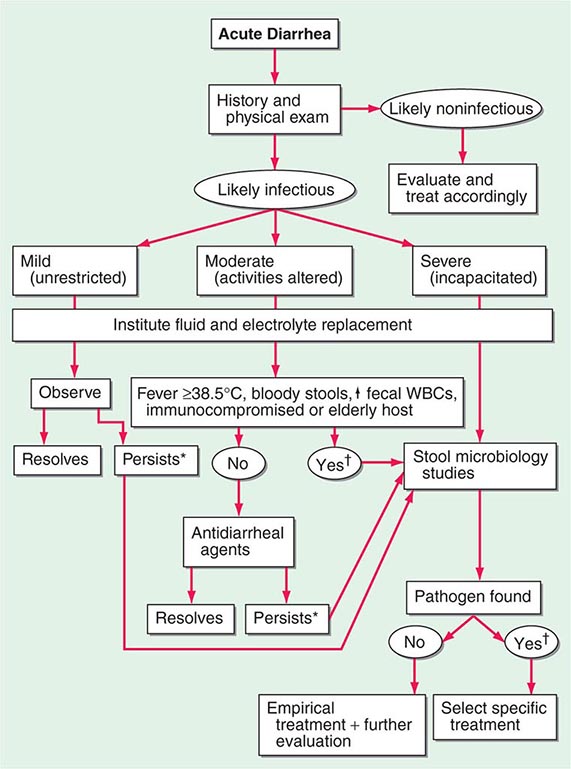
ACUTE DIARRHEA
More than 90% of cases of acute diarrhea are caused by infectious agents; these cases are often accompanied by vomiting, fever, and abdominal pain. The remaining 10% or so are caused by medications, toxic ingestions, ischemia, food indiscretions, and other conditions.
Infectious Agents Most infectious diarrheas are acquired by fecal-oral transmission or, more commonly, via ingestion of food or water contaminated with pathogens from human or animal feces. In the immunocompetent person, the resident fecal microflora, containing >500 taxonomically distinct species, are rarely the source of diarrhea and may actually play a role in suppressing the growth of ingested pathogens. Disturbances of flora by antibiotics can lead to diarrhea by reducing the digestive function or by allowing the overgrowth of pathogens, such as Clostridium difficile (Chap. 161). Acute infection or injury occurs when the ingested agent overwhelms or bypasses the host’s mucosal immune and nonimmune (gastric acid, digestive enzymes, mucus secretion, peristalsis, and suppressive resident flora) defenses. Established clinical associations with specific enteropathogens may offer diagnostic clues.
In the United States, five high-risk groups are recognized:
1. Travelers. Nearly 40% of tourists to endemic regions of Latin America, Africa, and Asia develop so-called traveler’s diarrhea, most commonly due to enterotoxigenic or enteroaggregative Escherichia coli as well as to Campylobacter, Shigella, Aeromonas, norovirus, Coronavirus, and Salmonella. Visitors to Russia (especially St. Petersburg) may have increased risk of Giardia-associated diarrhea; visitors to Nepal may acquire Cyclospora. Campers, backpackers, and swimmers in wilderness areas may become infected with Giardia. Cruise ships may be affected by outbreaks of gastroenteritis caused by agents such as norovirus.
2. Consumers of certain foods. Diarrhea closely following food consumption at a picnic, banquet, or restaurant may suggest infection with Salmonella, Campylobacter, or Shigella from chicken; enterohemorrhagic E. coli (O157:H7) from undercooked hamburger; Bacillus cereus from fried rice or other reheated food; Staphylococcus aureus or Salmonella from mayonnaise or creams; Salmonella from eggs; Listeria from uncooked foods or soft cheeses; and Vibrio species, Salmonella, or acute hepatitis A from seafood, especially if raw. State departments of public health issue communications regarding food-related illnesses, which may have originated domestically or been imported, but ultimately cause epidemics in the United States (e.g., the Cyclospora epidemic of 2013 in midwestern states that resulted from bagged salads).
3. Immunodeficient persons. Individuals at risk for diarrhea include those with either primary immunodeficiency (e.g., IgA deficiency, common variable hypogammaglobulinemia, chronic granulomatous disease) or the much more common secondary immunodeficiency states (e.g., AIDS, senescence, pharmacologic suppression). Common enteric pathogens often cause a more severe and protracted diarrheal illness, and, particularly in persons with AIDS, opportunistic infections, such as by Mycobacterium species, certain viruses (cytomegalovirus, adenovirus, and herpes simplex), and protozoa (Cryptosporidium, Isospora belli, Microsporida, and Blastocystis hominis) may also play a role (Chap. 226). In patients with AIDS, agents transmitted venereally per rectum (e.g., Neisseria gonorrhoeae, Treponema pallidum, Chlamydia) may contribute to proctocolitis. Persons with hemochromatosis are especially prone to invasive, even fatal, enteric infections with Vibrio species and Yersinia infections and should avoid raw fish.
4. Daycare attendees and their family members. Infections with Shigella, Giardia, Cryptosporidium, rotavirus, and other agents are very common and should be considered.
5. Institutionalized persons. Infectious diarrhea is one of the most frequent categories of nosocomial infections in many hospitals and long-term care facilities; the causes are a variety of microorganisms but most commonly C. difficile. C. difficile can affect those with no history of antibiotic use and may be acquired in the community.
The pathophysiology underlying acute diarrhea by infectious agents produces specific clinical features that may also be helpful in diagnosis (Table 55-2). Profuse, watery diarrhea secondary to small-bowel hypersecretion occurs with ingestion of preformed bacterial toxins, enterotoxin-producing bacteria, and enteroadherent pathogens. Diarrhea associated with marked vomiting and minimal or no fever may occur abruptly within a few hours after ingestion of the former two types; vomiting is usually less, abdominal cramping or bloating is greater, and fever is higher with the latter. Cytotoxin-producing and invasive microorganisms all cause high fever and abdominal pain. Invasive bacteria and Entamoeba histolytica often cause bloody diarrhea (referred to as dysentery). Yersinia invades the terminal ileal and proximal colon mucosa and may cause especially severe abdominal pain with tenderness mimicking acute appendicitis.
ASSOCIATION BETWEEN PATHOBIOLOGY OF CAUSATIVE AGENTS AND CLINICAL FEATURES IN ACUTE INFECTIOUS DIARRHEA |
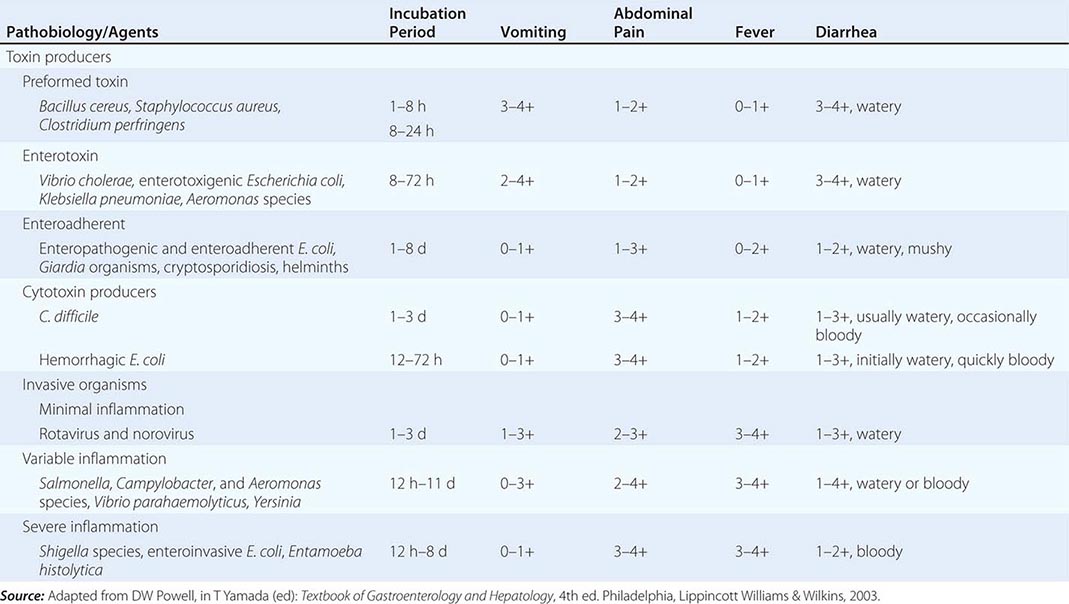
Finally, infectious diarrhea may be associated with systemic manifestations. Reactive arthritis (formerly known as Reiter’s syndrome), arthritis, urethritis, and conjunctivitis may accompany or follow infections by Salmonella, Campylobacter, Shigella, and Yersinia. Yersiniosis may also lead to an autoimmune-type thyroiditis, pericarditis, and glomerulonephritis. Both enterohemorrhagic E. coli (O157:H7) and Shigella can lead to the hemolytic-uremic syndrome with an attendant high mortality rate. The syndrome of postinfectious IBS has now been recognized as a complication of infectious diarrhea. Similarly, acute gastroenteritis may precede the diagnosis of celiac disease or Crohn’s disease. Acute diarrhea can also be a major symptom of several systemic infections including viral hepatitis, listeriosis, legionellosis, and toxic shock syndrome.
Other Causes Side effects from medications are probably the most common noninfectious causes of acute diarrhea, and etiology may be suggested by a temporal association between use and symptom onset. Although innumerable medications may produce diarrhea, some of the more frequently incriminated include antibiotics, cardiac antidysrhythmics, antihypertensives, nonsteroidal anti-inflammatory drugs (NSAIDs), certain antidepressants, chemotherapeutic agents, bronchodilators, antacids, and laxatives. Occlusive or nonocclusive ischemic colitis typically occurs in persons >50 years; often presents as acute lower abdominal pain preceding watery, then bloody diarrhea; and generally results in acute inflammatory changes in the sigmoid or left colon while sparing the rectum. Acute diarrhea may accompany colonic diverticulitis and graft-versus-host disease. Acute diarrhea, often associated with systemic compromise, can follow ingestion of toxins including organophosphate insecticides; amanita and other mushrooms; arsenic; and preformed environmental toxins in seafood, such as ciguatera and scombroid. Acute anaphylaxis to food ingestion can have a similar presentation. Conditions causing chronic diarrhea can also be confused with acute diarrhea early in their course. This confusion may occur with inflammatory bowel disease (IBD) and some of the other inflammatory chronic diarrheas that may have an abrupt rather than insidious onset and exhibit features that mimic infection.
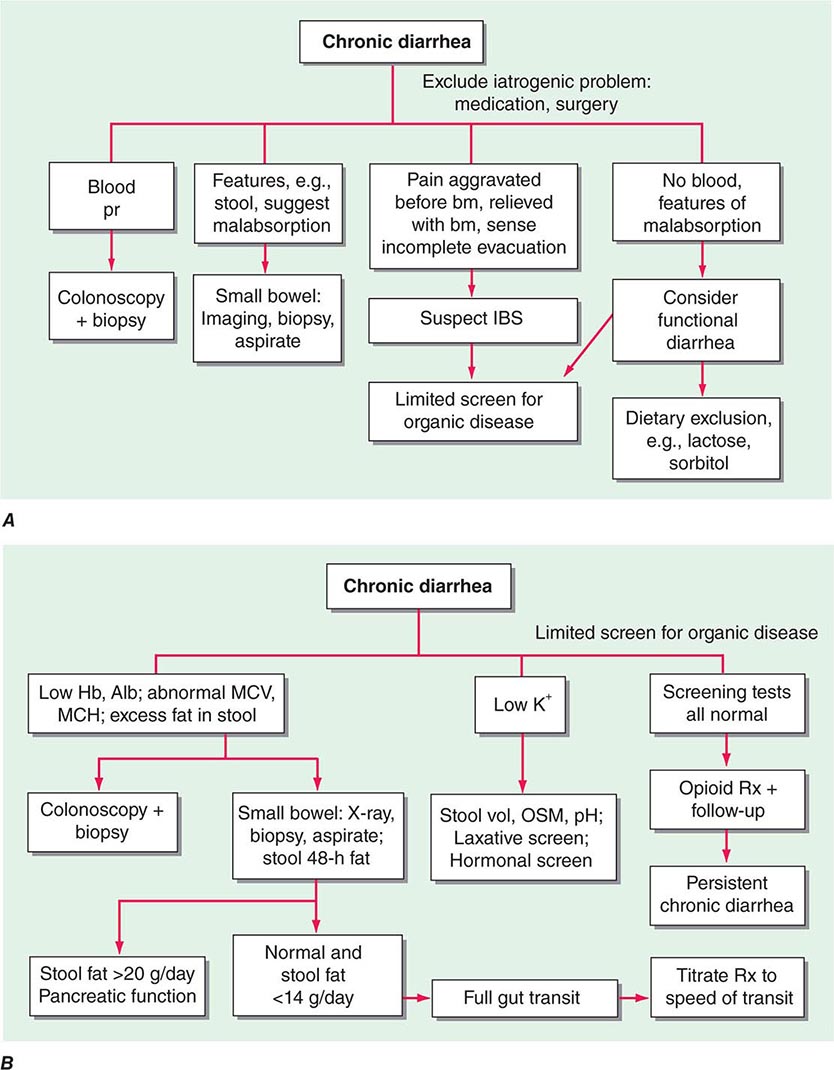
CHRONIC DIARRHEA
Diarrhea lasting >4 weeks warrants evaluation to exclude serious underlying pathology. In contrast to acute diarrhea, most of the causes of chronic diarrhea are noninfectious. The classification of chronic diarrhea by pathophysiologic mechanism facilitates a rational approach to management, although many diseases cause diarrhea by more than one mechanism (Table 55-3).
MAJOR CAUSES OF CHRONIC DIARRHEA ACCORDING TO PREDOMINANT PATHOPHYSIOLOGIC MECHANISM |
Abbreviation: FODMAP, fermentable oligosaccharides, disaccharides, monosaccharides, and polyols.
Secretory Causes Secretory diarrheas are due to derangements in fluid and electrolyte transport across the enterocolonic mucosa. They are characterized clinically by watery, large-volume fecal outputs that are typically painless and persist with fasting. Because there is no malabsorbed solute, stool osmolality is accounted for by normal endogenous electrolytes with no fecal osmotic gap.
MEDICATIONS Side effects from regular ingestion of drugs and toxins are the most common secretory causes of chronic diarrhea. Hundreds of prescription and over-the-counter medications (see earlier section, “Acute Diarrhea, Other Causes”) may produce diarrhea. Surreptitious or habitual use of stimulant laxatives (e.g., senna, cascara, bisacodyl, ricinoleic acid [castor oil]) must also be considered. Chronic ethanol consumption may cause a secretory-type diarrhea due to enterocyte injury with impaired sodium and water absorption as well as rapid transit and other alterations. Inadvertent ingestion of certain environmental toxins (e.g., arsenic) may lead to chronic rather than acute forms of diarrhea. Certain bacterial infections may occasionally persist and be associated with a secretory-type diarrhea.
BOWEL RESECTION, MUCOSAL DISEASE, OR ENTEROCOLIC FISTULA These conditions may result in a secretory-type diarrhea because of inadequate surface for reabsorption of secreted fluids and electrolytes. Unlike other secretory diarrheas, this subset of conditions tends to worsen with eating. With disease (e.g., Crohn’s ileitis) or resection of <100 cm of terminal ileum, dihydroxy bile acids may escape absorption and stimulate colonic secretion (cholerheic diarrhea). This mechanism may contribute to so-called idiopathic secretory diarrhea or bile acid diarrhea (BAD), in which bile acids are functionally malabsorbed from a normal-appearing terminal ileum. This idiopathic bile acid malabsorption (BAM) may account for an average of 40% of unexplained chronic diarrhea. Reduced negative feedback regulation of bile acid synthesis in hepatocytes by fibroblast growth factor 19 (FGF-19) produced by ileal enterocytes results in a degree of bile-acid synthesis that exceeds the normal capacity for ileal reabsorption, producing BAD. An alternative cause of BAD is a genetic variation in the receptor proteins (β-klotho and fibroblast growth factor 4) on the hepatocyte that normally mediate the effect of FGF-19. Dysfunction of these proteins prevents FGF-19 inhibition of hepatocyte bile acid synthesis.
Partial bowel obstruction, ostomy stricture, or fecal impaction may paradoxically lead to increased fecal output due to fluid hypersecretion.
HORMONES Although uncommon, the classic examples of secretory diarrhea are those mediated by hormones. Metastatic gastrointestinal carcinoid tumors or, rarely, primary bronchial carcinoids may produce watery diarrhea alone or as part of the carcinoid syndrome that comprises episodic flushing, wheezing, dyspnea, and right-sided valvular heart disease. Diarrhea is due to the release into the circulation of potent intestinal secretagogues including serotonin, histamine, prostaglandins, and various kinins. Pellagra-like skin lesions may rarely occur as the result of serotonin overproduction with niacin depletion. Gastrinoma, one of the most common neuroendocrine tumors, most typically presents with refractory peptic ulcers, but diarrhea occurs in up to one-third of cases and may be the only clinical manifestation in 10%. While other secretagogues released with gastrin may play a role, the diarrhea most often results from fat maldigestion owing to pancreatic enzyme inactivation by low intraduodenal pH. The watery diarrhea hypokalemia achlorhydria syndrome, also called pancreatic cholera, is due to a non-β cell pancreatic adenoma, referred to as a VIPoma, that secretes VIP and a host of other peptide hormones including pancreatic polypeptide, secretin, gastrin, gastrin-inhibitory polypeptide (also called glucose-dependent insulinotropic peptide), neurotensin, calcitonin, and prostaglandins. The secretory diarrhea is often massive with stool volumes >3 L/d; daily volumes as high as 20 L have been reported. Life-threatening dehydration; neuromuscular dysfunction from associated hypokalemia, hypomagnesemia, or hypercalcemia; flushing; and hyperglycemia may accompany a VIPoma. Medullary carcinoma of the thyroid may present with watery diarrhea caused by calcitonin, other secretory peptides, or prostaglandins. Prominent diarrhea is often associated with metastatic disease and poor prognosis. Systemic mastocytosis, which may be associated with the skin lesion urticaria pigmentosa, may cause diarrhea that is either secretory and mediated by histamine or inflammatory due to intestinal infiltration by mast cells. Large colorectal villous adenomas may rarely be associated with a secretory diarrhea that may cause hypokalemia, can be inhibited by NSAIDs, and are apparently mediated by prostaglandins.
CONGENITAL DEFECTS IN ION ABSORPTION Rarely, defects in specific carriers associated with ion absorption cause watery diarrhea from birth. These disorders include defective Cl–/HCO3– exchange (congenital chloridorrhea) with alkalosis (which results from a mutated DRA [down-regulated in adenoma] gene) and defective Na+/H+ exchange (congenital sodium diarrhea), which results from a mutation in the NHE3 (sodium-hydrogen exchanger) gene and results in acidosis.
Some hormone deficiencies may be associated with watery diarrhea, such as occurs with adrenocortical insufficiency (Addison’s disease) that may be accompanied by skin hyperpigmentation.
Osmotic Causes Osmotic diarrhea occurs when ingested, poorly absorbable, osmotically active solutes draw enough fluid into the lumen to exceed the reabsorptive capacity of the colon. Fecal water output increases in proportion to such a solute load. Osmotic diarrhea characteristically ceases with fasting or with discontinuation of the causative agent.
OSMOTIC LAXATIVES Ingestion of magnesium-containing antacids, health supplements, or laxatives may induce osmotic diarrhea typified by a stool osmotic gap (>50 mosmol/L): serum osmolarity (typically 290 mosmol/kg) – (2 × [fecal sodium + potassium concentration]). Measurement of fecal osmolarity is no longer recommended because, even when measured immediately after evacuation, it may be erroneous because carbohydrates are metabolized by colonic bacteria, causing an increase in osmolarity.
CARBOHYDRATE MALABSORPTION Carbohydrate malabsorption due to acquired or congenital defects in brush-border disaccharidases and other enzymes leads to osmotic diarrhea with a low pH. One of the most common causes of chronic diarrhea in adults is lactase deficiency, which affects three-fourths of nonwhites worldwide and 5–30% of persons in the United States; the total lactose load at any one time influences the symptoms experienced. Most patients learn to avoid milk products without requiring treatment with enzyme supplements. Some sugars, such as sorbitol, lactulose, or fructose, are frequently malabsorbed, and diarrhea ensues with ingestion of medications, gum, or candies sweetened with these poorly or incompletely absorbed sugars.
WHEAT AND FODMAP INTOLERANCE Chronic diarrhea, bloating, and abdominal pain are recognized as symptoms of nonceliac gluten intolerance (which is associated with impaired intestinal or colonic barrier function) and intolerance of fermentable oligosaccharides, disaccharides, monosaccharides, and polyols (FODMAPs). The latter’s effects represent the interaction between the GI microbiome and the nutrients.
Steatorrheal Causes Fat malabsorption may lead to greasy, foul-smelling, difficult-to-flush diarrhea often associated with weight loss and nutritional deficiencies due to concomitant malabsorption of amino acids and vitamins. Increased fecal output is caused by the osmotic effects of fatty acids, especially after bacterial hydroxylation, and, to a lesser extent, by the neutral fat. Quantitatively, steatorrhea is defined as stool fat exceeding the normal 7 g/d; rapid-transit diarrhea may result in fecal fat up to 14 g/d; daily fecal fat averages 15–25 g with small-intestinal diseases and is often >32 g with pancreatic exocrine insufficiency. Intraluminal maldigestion, mucosal malabsorption, or lymphatic obstruction may produce steatorrhea.
INTRALUMINAL MALDIGESTION This condition most commonly results from pancreatic exocrine insufficiency, which occurs when >90% of pancreatic secretory function is lost. Chronic pancreatitis, usually a sequel of ethanol abuse, most frequently causes pancreatic insufficiency. Other causes include cystic fibrosis; pancreatic duct obstruction; and, rarely, somatostatinoma. Bacterial overgrowth in the small intestine may deconjugate bile acids and alter micelle formation, impairing fat digestion; it occurs with stasis from a blind-loop, small-bowel diverticulum or dysmotility and is especially likely in the elderly. Finally, cirrhosis or biliary obstruction may lead to mild steatorrhea due to deficient intraluminal bile acid concentration.
MUCOSAL MALABSORPTION Mucosal malabsorption occurs from a variety of enteropathies, but it most commonly occurs from celiac disease. This gluten-sensitive enteropathy affects all ages and is characterized by villous atrophy and crypt hyperplasia in the proximal small bowel and can present with fatty diarrhea associated with multiple nutritional deficiencies of varying severity. Celiac disease is much more frequent than previously thought; it affects ~1% of the population, frequently presents without steatorrhea, can mimic IBS, and has many other GI and extraintestinal manifestations. Tropical sprue may produce a similar histologic and clinical syndrome but occurs in residents of or travelers to tropical climates; abrupt onset and response to antibiotics suggest an infectious etiology. Whipple’s disease, due to the bacillus Tropheryma whipplei and histiocytic infiltration of the small-bowel mucosa, is a less common cause of steatorrhea that most typically occurs in young or middle-aged men; it is frequently associated with arthralgias, fever, lymphadenopathy, and extreme fatigue, and it may affect the CNS and endocardium. A similar clinical and histologic picture results from Mycobacterium avium-intracellulare infection in patients with AIDS. Abetalipoproteinemia is a rare defect of chylomicron formation and fat malabsorption in children, associated with acanthocytic erythrocytes, ataxia, and retinitis pigmentosa. Several other conditions may cause mucosal malabsorption including infections, especially with protozoa such as Giardia; numerous medications (e.g., olmesartan, mycophenolate mofetil, colchicine, cholestyramine, neomycin); amyloidosis; and chronic ischemia.
POSTMUCOSAL LYMPHATIC OBSTRUCTION The pathophysiology of this condition, which is due to the rare congenital intestinal lymphangiectasia or to acquired lymphatic obstruction secondary to trauma, tumor, cardiac disease or infection, leads to the unique constellation of fat malabsorption with enteric losses of protein (often causing edema) and lymphocytopenia. Carbohydrate and amino acid absorption are preserved.
Inflammatory Causes Inflammatory diarrheas are generally accompanied by pain, fever, bleeding, or other manifestations of inflammation. The mechanism of diarrhea may not only be exudation but, depending on lesion site, may include fat malabsorption, disrupted fluid/electrolyte absorption, and hypersecretion or hypermotility from release of cytokines and other inflammatory mediators. The unifying feature on stool analysis is the presence of leukocytes or leukocyte-derived proteins such as calprotectin. With severe inflammation, exudative protein loss can lead to anasarca (generalized edema). Any middle-aged or older person with chronic inflammatory-type diarrhea, especially with blood, should be carefully evaluated to exclude a colorectal tumor.
IDIOPATHIC INFLAMMATORY BOWEL DISEASE The illnesses in this category, which include Crohn’s disease and chronic ulcerative colitis, are among the most common organic causes of chronic diarrhea in adults and range in severity from mild to fulminant and life-threatening. They may be associated with uveitis, polyarthralgias, cholestatic liver disease (primary sclerosing cholangitis), and skin lesions (erythema nodosum, pyoderma gangrenosum). Microscopic colitis, including both lymphocytic and collagenous colitis, is an increasingly recognized cause of chronic watery diarrhea, especially in middle-aged women and those on NSAIDs, statins, proton pump inhibitors (PPIs), and selective serotonin reuptake inhibitors (SSRIs); biopsy of a normal-appearing colon is required for histologic diagnosis. It may coexist with symptoms suggesting IBS or with celiac sprue or drug-induced enteropathy. It typically responds well to anti-inflammatory drugs (e.g., bismuth), to the opioid agonist loperamide, or to budesonide.
PRIMARY OR SECONDARY FORMS OF IMMUNODEFICIENCY Immunodeficiency may lead to prolonged infectious diarrhea. With selective IgA deficiency or common variable hypogammaglobulinemia, diarrhea is particularly prevalent and often the result of giardiasis, bacterial overgrowth, or sprue.
EOSINOPHILIC GASTROENTERITIS Eosinophil infiltration of the mucosa, muscularis, or serosa at any level of the GI tract may cause diarrhea, pain, vomiting, or ascites. Affected patients often have an atopic history, Charcot-Leyden crystals due to extruded eosinophil contents may be seen on microscopic inspection of stool, and peripheral eosinophilia is present in 50–75% of patients. While hypersensitivity to certain foods occurs in adults, true food allergy causing chronic diarrhea is rare.
OTHER CAUSES Chronic inflammatory diarrhea may be caused by radiation enterocolitis, chronic graft-versus-host disease, Behçet’s syndrome, and Cronkhite-Canada syndrome, among others.
Dysmotility Causes Rapid transit may accompany many diarrheas as a secondary or contributing phenomenon, but primary dysmotility is an unusual etiology of true diarrhea. Stool features often suggest a secretory diarrhea, but mild steatorrhea of up to 14 g of fat per day can be produced by maldigestion from rapid transit alone. Hyperthyroidism, carcinoid syndrome, and certain drugs (e.g., prostaglandins, prokinetic agents) may produce hypermotility with resultant diarrhea. Primary visceral neuromyopathies or idiopathic acquired intestinal pseudoobstruction may lead to stasis with secondary bacterial overgrowth causing diarrhea. Diabetic diarrhea, often accompanied by peripheral and generalized autonomic neuropathies, may occur in part because of intestinal dysmotility.
The exceedingly common IBS (10% point prevalence, 1–2% per year incidence) is characterized by disturbed intestinal and colonic motor and sensory responses to various stimuli. Symptoms of stool frequency typically cease at night, alternate with periods of constipation, are accompanied by abdominal pain relieved with defecation, and rarely result in weight loss.
Factitial Causes Factitial diarrhea accounts for up to 15% of unexplained diarrheas referred to tertiary care centers. Either as a form of Munchausen syndrome (deception or self-injury for secondary gain) or eating disorders, some patients covertly self-administer laxatives alone or in combination with other medications (e.g., diuretics) or surreptitiously add water or urine to stool sent for analysis. Such patients are typically women, often with histories of psychiatric illness, and disproportionately from careers in health care. Hypotension and hypokalemia are common co-presenting features. The evaluation of such patients may be difficult: contamination of the stool with water or urine is suggested by very low or high stool osmolarity, respectively. Such patients often deny this possibility when confronted, but they do benefit from psychiatric counseling when they acknowledge their behavior.