Impact of Percutaneous Image-Guided Core Biopsy on the Clinical Management of Breast Disease
Laura Liberman
INTRODUCTION
Percutaneous image-guided breast core biopsy is an accepted alternative to surgical biopsy for diagnosing breast lesions (1). Tissue-acquisition devices for percutaneous core biopsy include spring-loaded guns and automated core needles (usually 14-gauge), directional vacuum-assisted biopsy probes (14-, 11-, 9-, or 8-gauge), and intact sampling devices. Guidance methods for percutaneous biopsy include stereotaxis (2,3,4,5,6), ultrasound (7,8,9), and magnetic resonance imaging (MRI) (10,11).
Stereotactic guidance can be used for biopsy of all mammographically evident lesions but is most often used for calcifications. For stereotactic biopsy, vacuum-assisted biopsy probes (rather than automated needles) provide better retrieval and characterization of calcifications (1). Ultrasound guidance has several advantages, including lack of ionizing radiation, real-time visualization of the needle, accessibility to all parts of the breast and axilla, and multipurpose use of equipment. For ultrasound-guided biopsy, vacuum-assisted biopsy probes are fast and enable removal of larger volumes of tissue (8) but have no advantages compared to 14-gauge, automated needles in terms of missed cancers, underestimation, complications, or the need for immediate or delayed rebiopsy (9). Although ultrasound-guided biopsy is primarily used for masses, Soo et al. (12) have reported that in women with mammographically detected calcifications that are suspicious or highly suggestive of malignancy, ultrasound can identify an associated mass in 23% and can be used to guide biopsy in these cases. MRI guidance, used for biopsy of lesions detected with MRI only, requires special equipment that is safe to use in a magnetic environment. Because of the technical challenges and complex histologies encountered, vacuum-assisted biopsy devices are preferable to automated needles for MRI-guided biopsy procedures (11).
VALIDATION STUDIES
Early studies of stereotactic 14-gauge automated large core biopsy showed an 87% to 96% concordance between the results of stereotactic core biopsy and surgery (1). The best results were observed by the investigators who obtained
multiple specimens with a 14-gauge needle and a long excursion gun. In a 1993 study of ultrasound-guided 14-gauge automated large core biopsy, Parker et al. (7) reported 100% concordance between results of core biopsy and surgery in 49 lesions that went to surgery, and no carcinomas were identified at 12- to 36-month follow-up in 132 lesions after a benign finding on core needle biopsy.
multiple specimens with a 14-gauge needle and a long excursion gun. In a 1993 study of ultrasound-guided 14-gauge automated large core biopsy, Parker et al. (7) reported 100% concordance between results of core biopsy and surgery in 49 lesions that went to surgery, and no carcinomas were identified at 12- to 36-month follow-up in 132 lesions after a benign finding on core needle biopsy.
In a validation study of stereotactic 11-gauge vacuum-assisted biopsy, Pfarl et al. (6) reported false-negative findings in 3% (7 of 214) pathologically proven cancers. False-negative findings occurred in 4% (4 of 115) malignant calcifications versus 3% (3 of 99) malignant masses. The seven false-negative findings included five lesions that were highly suggestive of malignancy that yielded benign results at needle biopsy, one suspicious mass for which needle biopsy yielded benign breast tissue, and one suspicious cluster of calcifications in which no calcifications were retrieved. The false-negative rate was 10% (6 of 60) for radiologists who performed 15 or fewer previous stereotactic vacuum-assisted biopsy procedures versus 0.6% (1 of 154) for radiologists who performed more than 15 previous stereotactic vacuum-assisted biopsy procedures (ρ = 0.002). The authors concluded that stereotactic 11-gauge vacuum-assisted biopsy had a false-negative rate of 3.3% that diminished to 0.6% with experience. All false-negative findings could be identified prospectively because of failure to sample calcifications or discordance between histology and imaging findings.
INDICATIONS FOR PERCUTANEOUS CORE BIOPSY
Percutaneous core biopsy is most frequently used to evaluate nonpalpable lesions identified by screening mammography. The Breast Imaging Reporting and Data System (BI-RADS) lexicon of the American College of Radiology (13) defines assessment categories, a group of terms used to describe the level of suspicion regarding a lesion detected on a breast imaging study. Lesions are classified as BI-RADS category 0 if additional imaging is required to complete the assessment. Routine (annual) follow-up is warranted for mammograms classified as category 1 (“negative”) or 2 (“benign finding”). Short-interval follow-up (at 6 months for the ipsilateral breast, followed by both breasts at 1, 2, and 3 years after the initial mammogram) is suggested for mammographic lesions in BI-RADS category 3 (“probably benign”). Biopsy is suggested for mammographic lesions classified as category 4 (“suspicious”) or category 5 (“highly suggestive of malignancy”). After a tissue diagnosis of malignancy is made, a lesion is classified as category 6 (proven cancer) (13).
Percutaneous needle core biopsy is most often used in the assessment of category 4 lesions, most of which are benign (14,15,16). If the needle core biopsy of a category 4 lesion yields a benign diagnosis concordant with the imaging characteristics (i.e., provides a sufficient explanation for the imaging features), the woman is usually spared the need for surgery. The utility of needle core biopsy in the evaluation of category 5 lesions depends on the clinical setting. Category 5 accounts for a minority of lesions warranting biopsy, and most are malignant (14,15,16). If it is customary in the absence of needle core biopsy to perform a diagnostic surgical biopsy, followed by a second (therapeutic) surgical procedure if carcinoma is found, then performing a needle core biopsy before the therapeutic excision can spare patients with category 4 or 5 lesions a surgical procedure.
Category 3 (“probably benign”) lesions usually undergo imaging follow-up rather than biopsy. Among category 3 lesions, the frequency of carcinoma is 0.5% to 2% (17,18,19). Short-term follow-up mammography is less invasive than biopsy and is also less expensive: In an analysis of 3,184 patients with “probably benign” (category 3) lesions, Sickles and Brenner (20) found that the ratio of the cost of automated core biopsy to the cost of surveillance mammography was 8:1. Scenarios in which a tissue diagnosis may be desirable for category 3 lesions include inability to comply with follow-up mammography (e.g., a patient planning a pregnancy or traveling to an underserved area), known synchronous breast cancer, or extreme patient anxiety.
Percutaneous imaging-guided core biopsy is primarily used for nonpalpable lesions but can be helpful in the evaluation of palpable lesions, particularly if the lesion is deep, mobile, or vaguely palpable (21). Performing the biopsy under imaging guidance can help ensure that the lesion has been sampled.
ADVANTAGES OF PERCUTANEOUS BIOPSY
The patient care advantages of percutaneous core breast biopsy are numerous. Percutaneous core biopsy can spare the need for surgery in many women with benign lesions and can reduce the number of surgeries performed in women with breast cancer. Percutaneous core biopsy is less invasive and less expensive than surgical biopsy and can be performed quickly. Less tissue is removed, resulting in no deformity in the breast and less scarring on subsequent mammograms. Complications of percutaneous core biopsy are unusual, with the complication rate less than 0.2% for 14-automated core biopsy (22) and up to 3% for percutaneous 11-gauge vacuum-assisted biopsy (8,9).
Fewer Operations
An estimated 1,000,000-plus breast biopsies will be performed in the United States this year to diagnose more than 271,142 breast cancers (212,930 invasive and 58,490 in situ) (23). Among these biopsies, at least 30% (300,000) will be for nonpalpable lesions. Approximately 70% (210,000) of these 300,000 biopsies for nonpalpable lesions will be benign. If percutaneous image-guided
biopsy yields a benign diagnosis concordant with imaging characteristics, a woman is spared the need for a surgical biopsy (1).
biopsy yields a benign diagnosis concordant with imaging characteristics, a woman is spared the need for a surgical biopsy (1).
By obviating surgical biopsy in women with benign lesions, percutaneous biopsy can improve the positive predictive value of needle localization and surgical biopsy (i.e., the percentage of needle localized lesions that prove to be malignant). This effect of percutaneous needle core biopsy was demonstrated in a study by Rubin et al. (24), in which 200 patients had percutaneous breast biopsy under ultrasound (n = 167) or stereotactic (n = 33) guidance. The positive, predictive value of needle localization/surgical biopsy increased from 35%, just before needle core biopsy was introduced, to 55%, after core biopsy; for masses, the positive predictive value increased from 43% to 72%.
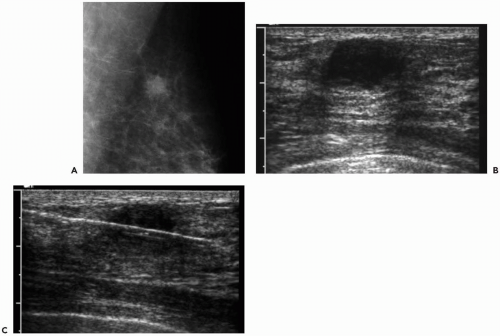
Percutaneous needle core biopsy decreases the number of surgeries necessary in women with breast cancer (Fig. 31.1). Liberman et al. (25,26) found that a single surgery was performed in 84% of women with cancer diagnosed by percutaneous biopsy, compared with 29% of women whose cancers were diagnosed by surgical biopsy. Jackman et al. (27) reported that a single surgical procedure was performed in 90% of patients whose cancers were diagnosed by core biopsy and in only 24% of patients whose cancers were diagnosed by surgical biopsy. Smith et al. (28) noted that the average number of surgeries was 1.25 in women whose cancers were diagnosed by core biopsy versus 2.01 in women with cancers diagnosed by needle localization and surgical biopsy (ρ <0.001).
In a study of 350 women who had surgery after percutaneous diagnosis of nonpalpable breast cancer, Liberman et al. (29) found that one operation was performed in 283 of 350 (81%) women, including 106 of 111 (96%) women who had mastectomy and 177 of 239 (74%) women who had breast conservation. In women who required two or more surgeries after percutaneous diagnosis of breast cancer, the most common reasons for additional surgeries included tumor at or close to the margins of resection, ductal carcinoma in situ (DCIS) underestimation (i.e., percutaneous biopsy yielded DCIS but subsequent surgery showed invasive cancer), and tumor in sentinel nodes. In multivariate analysis, one operation was significantly more likely if underestimation were absent or if mastectomy were performed; for women who had breast conservation, one operation was significantly more likely if underestimation were absent or if a mammographic mass were present.
For women with multiple breast lesions, percutaneous biopsy of two or more areas can help determine whether carcinoma is present at a single site or multiple sites. This information may influence surgical treatment: The presence of cancer at two sites in the same quadrant (multifocal disease) may indicate the need for wider excision, and
the presence of cancer in two separate quadrants (multicentric disease) generally warrants mastectomy (30). Percutaneous biopsy can also decrease the number of surgeries in women with new mammographically detected lesions after breast conservation (31). If percutaneous biopsy yields a benign diagnosis concordant with the imaging characteristics, surgery may be avoided, and if percutaneous biopsy reveals recurrent carcinoma in the treated breast, the patient may proceed with definitive surgery, often mastectomy. Percutaneous biopsy is also helpful in women with locally advanced or metastatic breast cancer; samples can be analyzed for the presence of receptors for estrogen, progesterone, and HER-2/neu, aiding in the selection of chemotherapy.
the presence of cancer in two separate quadrants (multicentric disease) generally warrants mastectomy (30). Percutaneous biopsy can also decrease the number of surgeries in women with new mammographically detected lesions after breast conservation (31). If percutaneous biopsy yields a benign diagnosis concordant with the imaging characteristics, surgery may be avoided, and if percutaneous biopsy reveals recurrent carcinoma in the treated breast, the patient may proceed with definitive surgery, often mastectomy. Percutaneous biopsy is also helpful in women with locally advanced or metastatic breast cancer; samples can be analyzed for the presence of receptors for estrogen, progesterone, and HER-2/neu, aiding in the selection of chemotherapy.
Lower Cost
Substantial savings can result from the use of needle core biopsy rather than surgical biopsy for the evaluation of BI-RADS category 4 or 5 lesions. Several studies in the 1990s addressed the impact of stereotactic automated core biopsy on the cost of diagnosis. In a theoretical model, Lindfors and Rosenquist (32) found that the marginal cost per year of life saved by screening was reduced a maximum of 23% with the use of automated needle core biopsy instead of surgery. In clinical practice, Liberman et al. (33) found that stereotactic 14-gauge automated core biopsy spared a surgical procedure in 77% of women, resulting in a decrease in the cost of diagnosis of more than 50%. Based on these data, the estimated annual national savings from stereotactic 14-gauge automated core biopsy approached $200 million. Lee et al. (34) found that stereotactic, 14-gauge automated core biopsy spared a surgical procedure in 81% of patients. Cost savings (based on Medicare estimates) were observed in all groups but varied with mammographic features. Savings were least for calcifications and highly suspicious lesions and greatest for masses and indeterminate lesions.
Cost savings for ultrasound-guided automated core biopsy exceed those of stereotactic automated core biopsy. In a study of 151 women who had ultrasound-guided core biopsy of nonpalpable masses, 128 (85%) were spared a surgical procedure (35). Using Medicare figures, ultrasound-guided core biopsy resulted in a 56% decrease in the cost of diagnosis compared to surgical biopsy. If stereotactic rather than ultrasound-guided core biopsy had been used, the cost savings compared to surgical biopsy would have been 39%. This study suggests that both stereotactic core biopsy and ultrasound-guided core biopsy are less expensive than surgery, but that the savings are greater if the biopsy is performed under ultrasound guidance.
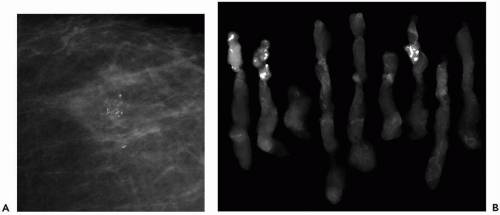
Vacuum-assisted biopsy probes are more expensive than automated core needles but can still result in substantial cost savings. Liberman and Sama (36) reviewed 200 consecutive, solitary, nonpalpable lesions that had stereotactic, 11-gauge, vacuum-assisted biopsy. Of these 200 lesions, 106 (53%) were not amenable to 14-gauge, automated core biopsy due to small size, superficial location, or inadequate breast thickness. Stereotactic 11-gauge vacuum assisted biopsy spared a surgical procedure in 151 (76%) of 200 lesions, including 112 (73%) of 154 calcific lesions (Fig. 31.2) and 39 (85%) of 46 masses. Stereotactic, 11-gauge, vacuum-assisted biopsy decreased the cost of diagnosis by 20% compared with the cost of surgical biopsy. These data suggest that although savings per case are modest, 11-gauge, vacuum-assisted biopsy expands the spectrum of lesions amenable to stereotactic biopsy, increasing cost savings in the population.