Immune System
ORGANIZATION OF THE IMMUNE SYSTEM
Like any security force, the components and mechanisms of the immune system are organized in an efficient—almost military-like—manner. They are not just ready on a moment’s notice; they are continually patrolling the body for foreign or internal enemies and shoring up the various lines of defense to fend off a possible attack. Before we begin studying the specifics of immunity, we will spend a moment mapping out the overall defensive strategy of the immune system.
First, it is important to recognize that cells, viruses, and other particles have unique molecules and groups of molecules on their surfaces that can be used to identify them. These molecular markers visible to the immune system are called antigens. This is similar to military operations in which enemy aircraft, vehicles, or soldiers can be identified by their distinctive insignia that are different from the insignia seen on “our side.” Likewise, our own cells have unique cell markers embedded in our plasma membranes that identify each of our cells as self—that is, belonging to us as an individual. And foreign cells or particles have nonself molecules that serve as recognition markers for our immune system. The ability of our immune system to attack abnormal or foreign cells but spare our own normal cells is called self-tolerance.
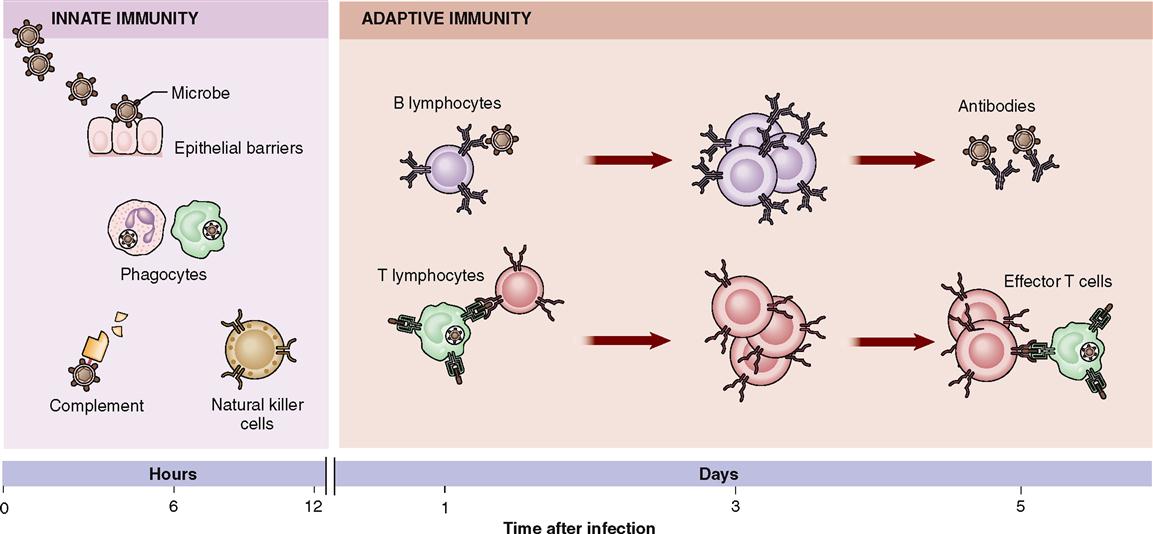
In human society, any good security force employs numerous and varied strategies to guard its territory and take action if necessary. So, too, does the body’s “society of cells” employ a system that uses many different kinds of mechanisms to ensure the integrity and survival of the internal environment. All of these defense mechanisms can be categorized into one of two major categories of immune mechanisms: innate immunity and adaptive immunity.
Innate immunity is called such because it is “in place” before a person is exposed to a particular harmful particle or condition. The word innate refers to something that is already present naturally at birth. Because it includes mechanisms that resist a wide variety of threatening agents or conditions, innate immunity is also called nonspecific immunity. The term nonspecific implies that these immune mechanisms do not act on only one or two specific invaders but rather provide a more general defense by simply acting against a wide variety of particles recognized as nonself.
Adaptive immunity, on the other hand, involves mechanisms that recognize specific threatening agents and then adapt, or respond, by targeting their activity against these agents—and these agents only. Because it targets only specific harmful particles, adaptive immunity is also called specific immunity. Adaptive immune mechanisms often take some time to recognize their targets and react with sufficient force to overcome the threat, at least on their first exposure to a specific kind of threatening agent. Innate mechanisms, because they are already in place, have the advantage of being able to meet an enemy as soon as it presents itself. As we discuss examples of each major type of immunity, you will come to understand how each type works and appreciate the distinction between them. You will also come to appreciate the value in having two complementary strategies for defending the body.
As in any body system, the work of the immune system is done by cells or substances made by cells. The primary types of cells involved in innate immunity are these: epithelial barrier cells, phagocytic cells (neutrophils, macrophages), and aptly named natural killer (NK) cells. The primary types of cells involved in adaptive immunity are two types of lymphocytes called T cells and B cells.
Cytokines, which are chemicals released from cells to trigger or regulate innate and adaptive immune responses, also participate in innate immunity. Examples of cytokines include interleukins (ILs), leukotrienes, and interferons (IFNs)—all of which are described later in this chapter. Other chemicals, in addition to cytokines, play a regulatory role in immunity—these include complements, other enzymes, and the amine histamine.
Awesome indeed is the army of cells and molecules that make up the immune system. Over one trillion lymphocytes, for example, and 100 million trillion (1020) plasma protein molecules (antibodies) are a few of the many agents that help your body resist damage and disease. Figure 24-1 and Table 24-1 summarize some of the essential characteristics of innate and adaptive immunity that we discuss in this chapter.
TABLE 24-1
| INNATE IMMUNITY | ADAPTIVE IMMUNITY | ||
| Synonyms | Frequently used alternate terminology | Nonspecific immunity, native immunity, genetic immunity | Specific immunity, acquired immunity |
| Characteristics | |||
| Specificity | Unique antigens produce unique responses of the immune system | Not specific—recognizes variety of different groups of foreign cells or particles | Specific—recognizes specific antigens on specific cells or particles |
| Speed of reaction | Reaction time of the immune responses | Rapid: immediate up to several hours | Slower: several hours to several days |
| Memory | Enhanced responses to repeated exposures to the same antigen | None | Yes |
| Does not react to self | Prevents injury to the individual’s own cells* | Yes | Yes |
| Components | |||
| Barriers | Prevent entry of harmful particles | Skin, mucosa, antimicrobial chemicals | Lymphocytes in epithelia; antibodies released at epithelial surfaces |
| Blood proteins | Circulate throughout body, providing wide area of protection | Complement, interferon (IFN), others | Antibodies |
| Cells | Types of leukocytes involved in immunity | Phagocytes (macrophages, neutrophils), natural killer (NK) cells | Lymphocytes (B cells and T cells) |
*Assumes healthy function. Anti-self immunity (autoimmunity) is a characteristic of many disorders.
INNATE IMMUNITY
The general, innate defensive mechanisms of the body are many and varied (Table 24-2). Only the major types of innate immune mechanisms are listed here; many other examples appear in other chapters throughout this book. You will probably recognize examples in this chapter that you have encountered already in previous chapters. Phagocytes are a good example. They are often referred to by different names when identified in specific body areas. For example, phagocytic cells in the skin were identified as dendritic cells (DCs) of the epidermis (Langerhans cells; Chapter 7, p. 173).
TABLE 24-2
| MECHANISM | DESCRIPTION |
| Species Resistance | Genetic characteristics of the human species protect the body from certain pathogens |
| Mechanical and Chemical Barriers | Physical impediments to the entry of foreign cells or substances |
| Skin and mucosa | Forms a continuous wall that separates the internal environment from the external environment, preventing the entry of pathogens |
| Secretions | Secretions such as sebum, mucus, acids, and enzymes chemically inhibit the activity of pathogens |
| Inflammation | The inflammatory response isolates the pathogens and stimulates the speedy arrival of large numbers of immune cells |
| Fever | Fever may enhance immune reactions and inhibit pathogens |
| Phagocytosis | Ingestion and destruction of pathogens by phagocytic cells |
| Neutrophils | Granular leukocytes that are usually the first phagocytic cell to arrive at the scene of an inflammatory response |
| Macrophages | Monocytes that have enlarged to become giant phagocytic cells capable of consuming many pathogens; often called by other, more specific names when found in specific tissues of the body |
| Natural Killer (NK) Cells | Group of lymphocytes that kill many different types of cancer cells and virus-infected cells |
| Interferon | Protein produced by cells after they become infected by a virus; inhibits the spread or further development of a viral infection |
| Complement | Group of plasma proteins (inactive enzymes) that produce a cascade of chemical reactions that ultimately causes lysis (rupture) of a foreign cell; the complement cascade can be triggered by adaptive or innate immune mechanisms |
| Toll-like Receptors (TLRs) | Membrane receptors that recognize nonspecific patterns in microbial molecules (not human molecules) and trigger a variety of innate immune responses (many of those listed in this table) |
Species Resistance
Species resistance refers to a phenomenon in which the genetic characteristics common to a particular kind of organism, or species, provide defense against certain pathogens (disease-causing agents). The human species (Homo sapiens), for example, is resistant to many life-threatening infections and infestations that often spread easily among plants and other animals. For example, humans do not have to worry about getting Dutch elm disease, a fungal infection that nearly eradicated the American elm tree, or becoming infected with canine viral distemper, a virus to which young dogs are susceptible. Usually, species resistance in humans results from the fact that our internal environment is not suitable for certain pathogens. We may also have resistance because a particular microbe may not be biochemically compatible with the various molecules on our cell membranes that the microbes would need to gain entry into a host cell.
Mechanical and Chemical Barriers
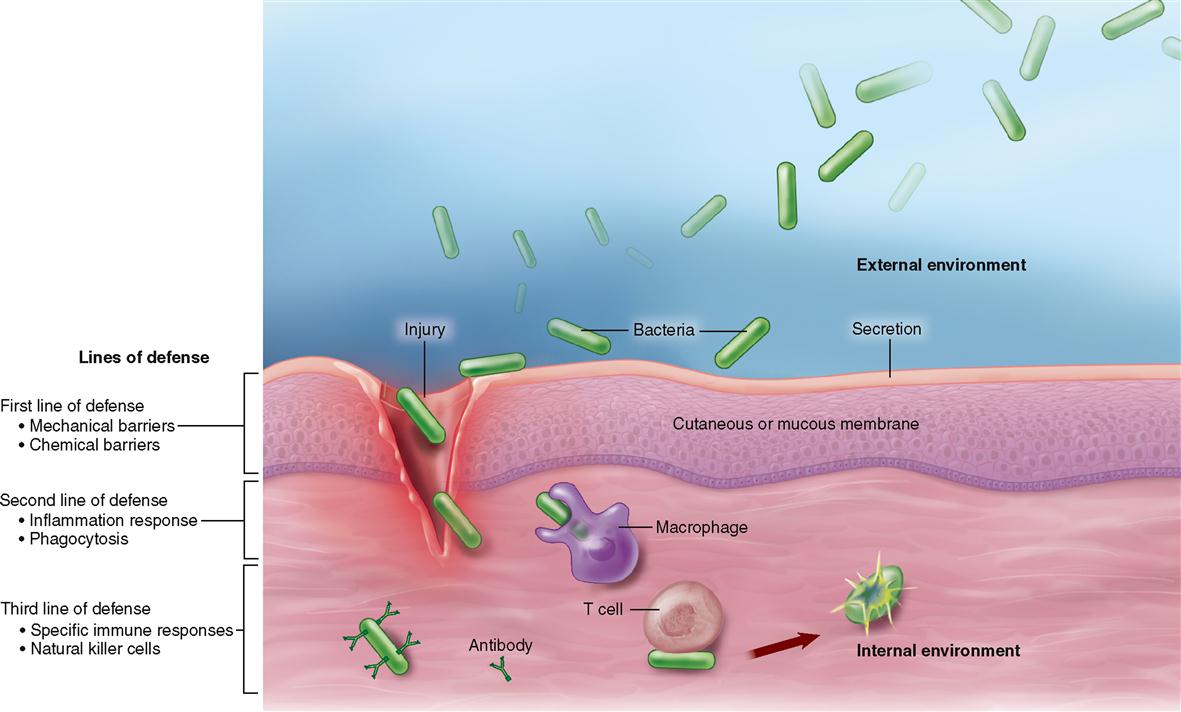
The internal environment of the human body is protected by a continuous mechanical barrier formed by the cutaneous membrane (skin) and mucous membranes (see Figure 6-39, p. 155). Often called the first line of defense, these membranes provide several layers of densely packed cells and other materials—forming a sort of “castle wall”—that protects the internal environment from invasion by foreign cells (Figure 24-2).
Besides forming a protective wall, the skin and mucous membranes operate various additional immune mechanisms. For example, substances such as sebum (which contains pathogen-inhibiting agents), mucus (in which pathogens become stuck and are then swept away), enzymes (which may hydrolyze pathogens), and hydrochloric acid in gastric mucosa (which may destroy pathogens) also may be present to act as innate defense mechanisms. These chemical barriers act as a sort of moat around the castle wall formed by the membranes.
The epithelial barriers of the body are essentially innate, nonspecific defenses. However, the protective epithelial membranes also have adaptive (specific) defenses that reinforce them. The combined innate and adaptive immune functions of protective mucous membranes are discussed later in the chapter in a separate boxed essay (see Box 24-8 on p. 771).
Inflammation and Fever
THE INFLAMMATORY RESPONSE
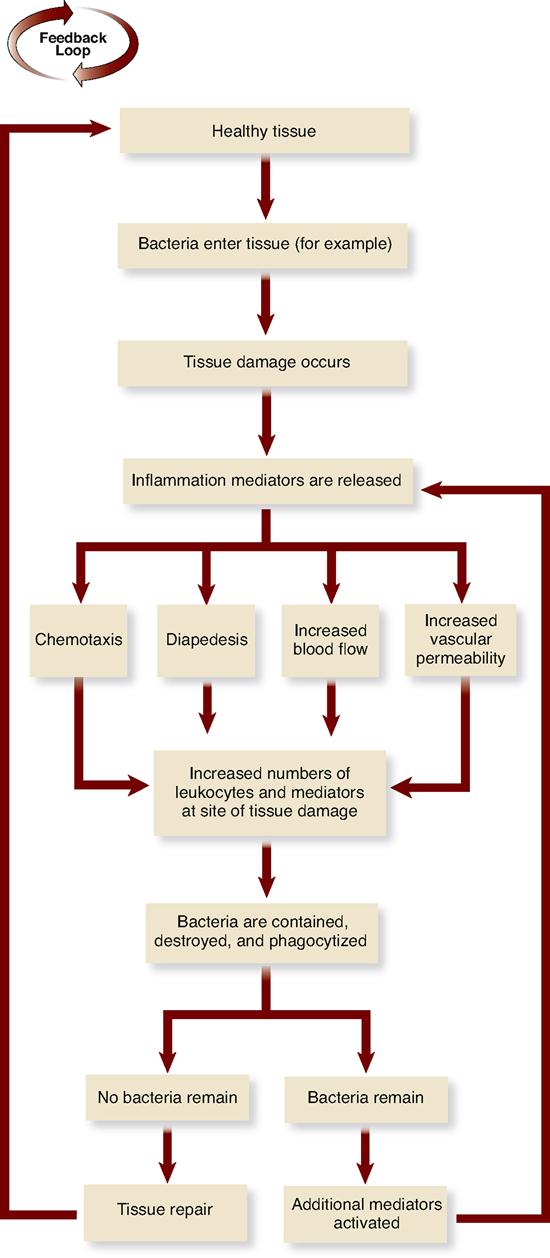
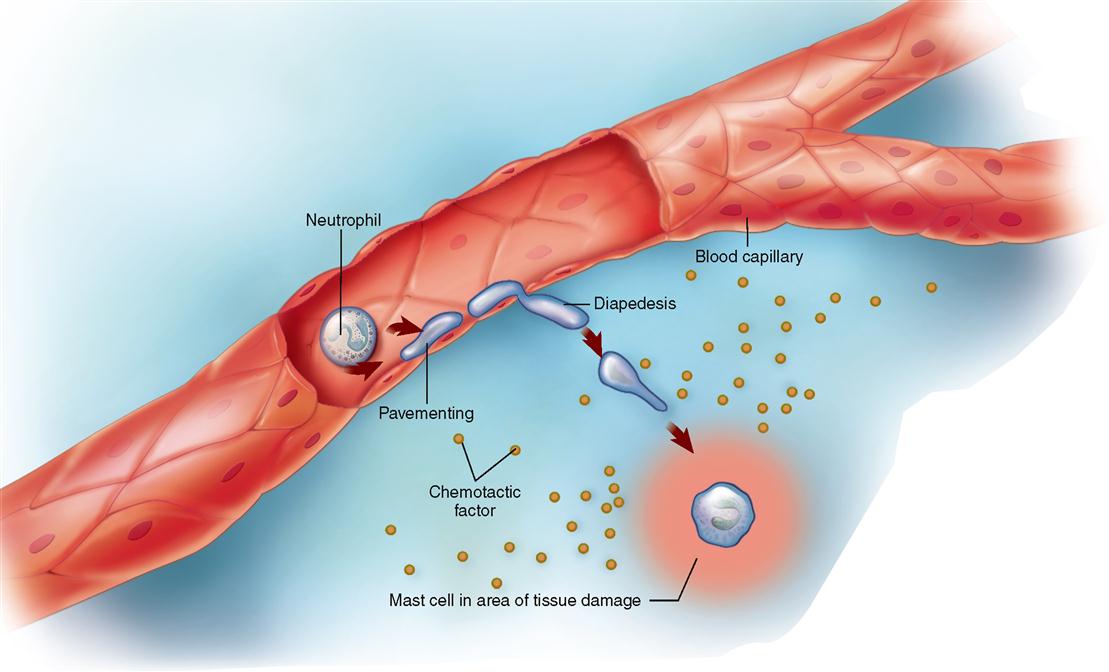
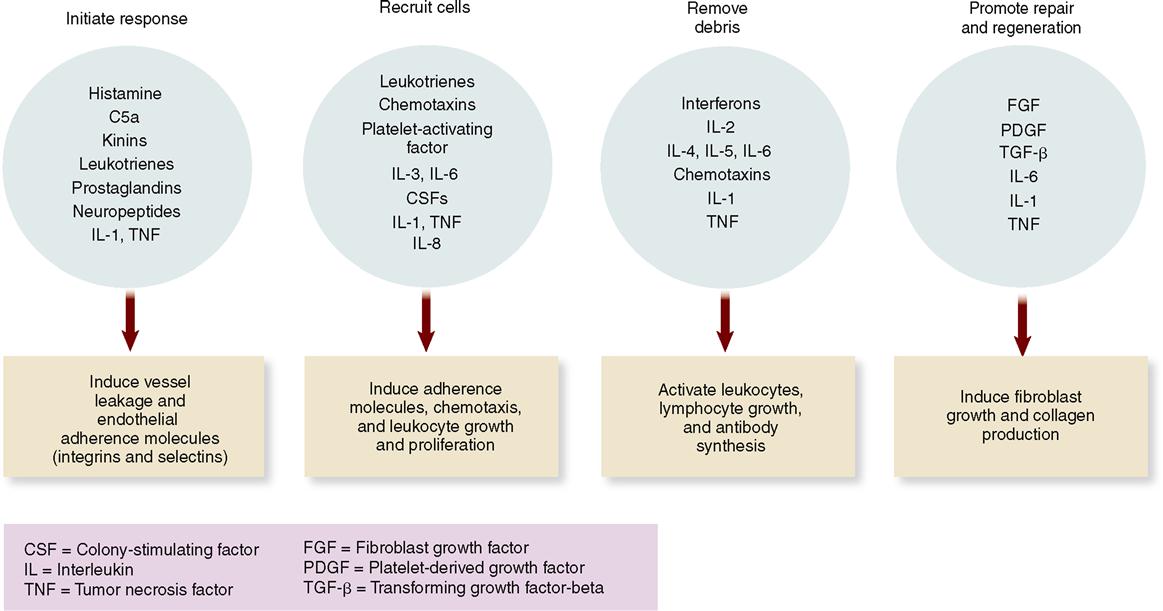
If bacteria or other invaders break through the chemical and mechanical barriers formed by the membranes and their secretions, the body has a second line of defense at the ready: the inflammatory response (see Figure 24-2). The inflammatory response has already been discussed in some detail in Chapter 6 (see Box 6-3 on p. 146 and A&P Connect: Inflammation online). For the purpose of a quick review, recall that tissue damage elicits a host of responses that counteract the injury and promote a return to normal. An example of how local inflammation works is illustrated in Figure 24-3. In the example, bacteria cause tissue damage that, in turn, triggers the release of various inflammation mediators from cells such as the mast cells found in connective tissues (Figure 24-4). These inflammation mediators include histamine, kinins, prostaglandins, leukotrienes, interleukins (ILs), and related compounds. Many of these mediators are chemotactic factors, that is, substances that attract white blood cells to the area in a process called chemotaxis. As Figure 24-5 shows, chemotaxis is the process by which a cell navigates toward the source of the chemotactic factor (chemotaxin) by way of detecting and then moving toward higher concentrations of the factor.
Additionally, many of the factors released from tissue cells and phagocytes, such as the peptide fragment called C5a from complement, produce the mechanisms that cause characteristic signs of inflammation: heat, redness, pain, and swelling (Figure 24-6). These signs result from increased blood flow and vascular permeability in the affected region, which help phagocytic white blood cells reach the general area and then enter the affected tissue. Also, some inflammation mediators trigger fibroblasts to grow and produce more collagen fibers to promote repair and regeneration.
FEVER
Besides local inflammation, systemic inflammation may occur when the inflammation mediators trigger responses that occur on a body-wide basis. A body-wide inflammatory response may be manifested by a fever—a state of abnormally high body temperature. For example, bacterial infections that spread widely throughout the body may produce systemic inflammatory response syndrome (SIRS). SIRS involves an abnormally high neutrophil (phagocytic white blood cell [WBC]) count and fever. Viral infections, tumors, allergies, and other abnormal conditions also can cause fevers.
Recall from Box 1-2 on p. 21 that fevers result from a “reset” of the body’s thermostat in the hypothalamus, which temporarily increases the set point or target temperature to a higher-than-normal value. The body then shivers or we cover ourselves and otherwise seek heat until the new set point temperature—a fever—is reached.
As we have already learned in earlier chapters, pyrogen molecules trigger the fever response by promoting production of prostaglandins (PGs), which then reset the body’s thermostat in the hypothalamus. Aspirin and other cyclooxygenase (COX) inhibitors reduce the activity of the COX enzymes (COX-1 and COX-2) that produces these prostaglandins—thus having a fever-reducing effect (see Figure 18-13 on p. 557). Pyrogens can be released from damaged cells (endogenous pyrogens) or could be introduced from outside the body (exogenous pyrogens).
The elevated temperature of a fever may facilitate some immune reactions and may also inhibit the reproduction of some microbial pathogens. However, immunologists still debate the role of fever in protecting the body.
Phagocytosis
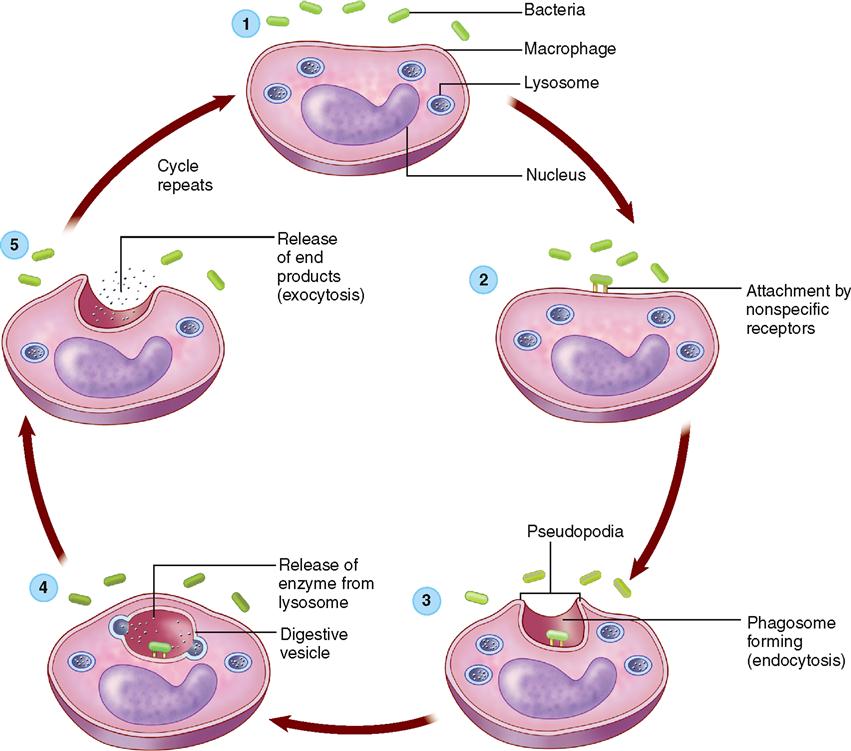
A major component of the body’s second line of defense is the mechanism of phagocytosis—the ingestion and destruction of microorganisms or other small particles. There are many types of phagocytes, that is, cells capable of phagocytosis, in the body. As Figure 24-7 shows, when phagocytes approach a microorganism, they extend footlike projections (pseudopods) toward it. Soon the pseudopods encircle the organism and form a complete sac, called a phagosome, around it. The phagosome then moves into the interior of the cell, where a lysosome fuses with it. The contents of the lysosome, chiefly digestive enzymes and hydrogen peroxide, drain into the phagosome and destroy the microorganisms within it.
Because phagocytosis defends us against various kinds of agents, it is classified as an innate defense. However, phagocytes also “cross over” to play an important role in adaptive immunity as well. After digesting the offending particle, a phagocyte will often process the proteins and display bits of the protein—peptides—on the surface of the phagocyte. These peptides are then recognized by cells of the adaptive immune system as antigens, thus possibly triggering an adaptive immune response. Cells that perform this function are called antigen-presenting cells (APCs).
You learned in Chapter 20 that the most numerous type of phagocyte is the neutrophil, a granular, neutral-staining type of WBC. After being released at a site of inflammation or tissue damage, chemotactic factors diffuse into adjoining capillaries. Once in the bloodstream, they cause neutrophils and other phagocytes to adhere to the vessel’s endothelial lining in a process called pavementing (see Figure 24-5). Numbers of these adherent phagocytes pass between the endothelial cells that form the capillary wall, dissolve the underlying basement membrane, and then exit through the vessel wall in the inflamed area. The movement of phagocytes from blood vessel to inflammation site is called diapedesis. Phagocytes have a very short life span, and thus dead cells tend to “pile up” at the inflammation site—forming most of the white substance called pus.
Another common type of phagocyte is the macrophage (meaning “large eater”). Macrophages are phagocytic monocytes (nongranular WBCs) that have grown to several times their original size after migrating out of the bloodstream. Macrophages are important APCs.
Yet another important type of phagocyte is the dendritic cell (DC) found in many tissues of the body that are in contact with the external environment, such as the skin and mucous membranes (Figure 24-8). This type of phagocytic APC is called dendritic because of its many branches (dendr- branch). Dendritic cells are also sometimes called stellate (“star shaped”) cells.
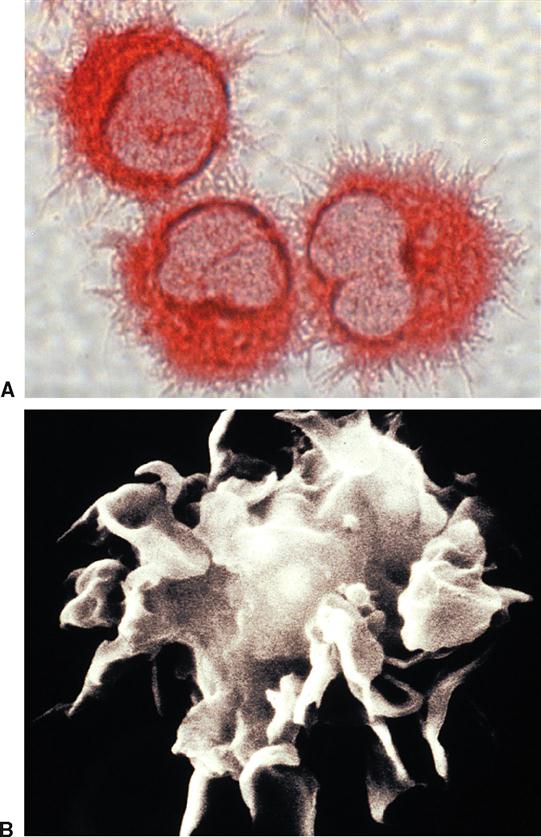
Phagocytic APCs of various types are present in many areas of the body, even on the outside surface of some mucous membranes (e.g., in the respiratory tract). Phagocyte types are often known by specific names that designate their location (Table 24-3). The importance of phagocytes to our overall defense of the body is made clear by the fact that 10% to 15% (by number) of all cells in any organ of the body are phagocytic cells!
TABLE 24-3
Examples of Phagocyte Locations
| PHAGOCYTES | LOCATION |
| Circulating phagocytes | Bloodstream |
| Osteoclasts; bone marrow (fixed) phagocytes | Bone and bone marrow |
| Microglia | Central nervous system |
| Histiocytes | Connective tissues |
| Epidermal dendritic cells | Epidermis |
| Kupffer cells | Liver |
| Alveolar macrophages (dust cells); dendritic cells | Lung |
| Fixed and free lymphoid macrophages; dendritic cells | Lymph nodes |
| Pleural macrophages; peritoneal macrophages | Serous fluids |
| Splenic macrophages | Spleen |
Natural Killer Cells
Besides phagocytes, the body has another important set of cells that provides innate defense of the body. These are the natural killer (NK) cells. NK cells are a group of lymphocytes that kill many types of tumor cells and cells infected by different kinds of viruses. As a group they are produced in the red bone marrow and constitute about 15% of the total lymphocyte cell numbers. NK cells are neither T cells nor B cells, as described later under the topic of adaptive immunity. Because they have such a broad action and do not have to be activated by a specific foreign antigen to become active, they are usually included among the innate immune strategies.
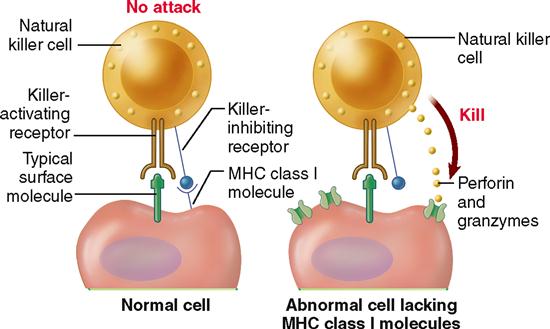
NK cells recognize abnormal cells by using two different recognition receptors: a killer-activating receptor and a killer-inhibiting receptor (Figure 24-9). The killer-activating receptor binds to any of several common surface molecules found in cells. Thus the NK cell can bind to any cell of the body as well as any foreign cells. However, if the killer-inhibiting receptor happens to bind to an MHC (major histocompatibility complex) protein also, then the killing action is stopped. Box 24-1 explains that MHCs are surface proteins on all normal cells and are unique to each individual person. Thus only abnormal and foreign cells fail to bind to the killer-inhibitor centers—and therefore are killed by the NK cell.