FIGURE 44-3 Acute epiglottitis. In this lateral soft tissue radiograph of the neck, the arrow indicates the enlarged edematous epiglottis (the “thumbprint sign”).
INFECTIONS OF DEEP NECK STRUCTURES
Deep neck infections are usually extensions of infection from other primary sites, most often within the pharynx or oral cavity. Many of these infections are life threatening but are difficult to detect at early stages, when they may be more easily managed. Three of the most clinically relevant spaces in the neck are the submandibular (and sublingual) space, the lateral pharyngeal (or parapharyngeal) space, and the retropharyngeal space. These spaces communicate with one another and with other important structures in the head, neck, and thorax, providing pathogens with easy access to areas that include the mediastinum, carotid sheath, skull base, and meninges. Once infection reaches these sensitive areas, mortality rates can be as high as 20–50%.
Infection of the submandibular and/or sublingual space typically originates from an infected or recently extracted lower tooth. The result is the severe, life-threatening infection referred to as Ludwig’s angina (see “Oral Infections,” above). Infection of the lateral pharyngeal (or parapharyngeal) space is most often a complication of common infections of the oral cavity and upper respiratory tract, including tonsillitis, peritonsillar abscess, pharyngitis, mastoiditis, and periodontal infection. This space, situated deep in the lateral wall of the pharynx, contains a number of sensitive structures, including the carotid artery, internal jugular vein, cervical sympathetic chain, and portions of cranial nerves IX through XII; at its distal end, it opens into the posterior mediastinum. Involvement of this space with infection can therefore be rapidly fatal. Examination may reveal some tonsillar displacement, trismus, and neck rigidity, but swelling of the lateral pharyngeal wall can easily be missed. The diagnosis can be confirmed by CT. Treatment consists of airway management, operative drainage of fluid collections, and at least 10 days of IV therapy with an antibiotic active against streptococci and oral anaerobes (e.g., ampicillin/sulbactam). A particularly severe form of this infection involving the components of the carotid sheath (postanginal septicemia, Lemierre’s disease) is described above (see “Oral Infections”). Infection of the retropharyngeal space also can be extremely dangerous, as this space runs posterior to the pharynx from the skull base to the superior mediastinum. Infections in this space are more common among children <5 years old because of the presence of several small retropharyngeal lymph nodes that typically atrophy by age 4 years. Infection is usually a consequence of extension from another site of infection—most commonly, acute pharyngitis. Other sources include otitis media, tonsillitis, dental infections, Ludwig’s angina, and anterior extension of vertebral osteomyelitis. Retropharyngeal space infection also can follow penetrating trauma to the posterior pharynx (e.g., from an endoscopic procedure). Infections are commonly polymicrobial, involving a mixture of aerobes and anaerobes; group A β-hemolytic streptococci and S. aureus are the most common pathogens. M. tuberculosis was a common cause in the past but now is rarely involved in the United States.
Patients with retropharyngeal abscess typically present with sore throat, fever, dysphagia, and neck pain and are often drooling because of difficulty and pain with swallowing. Examination may reveal tender cervical adenopathy, neck swelling, and diffuse erythema and edema of the posterior pharynx as well as a bulge in the posterior pharyngeal wall that may not be obvious on routine inspection. A soft tissue mass is usually demonstrable by lateral neck radiography or CT. Because of the risk of airway obstruction, treatment begins with securing of the airway, followed by a combination of surgical drainage and IV antibiotic administration. Initial empirical therapy should cover streptococci, oral anaerobes, and S. aureus; ampicillin/sulbactam, clindamycin plus ceftriaxone, or meropenem is usually effective. Complications result primarily from extension to other areas (e.g., rupture into the posterior pharynx may lead to aspiration pneumonia and empyema). Extension may also occur to the lateral pharyngeal space and mediastinum, resulting in mediastinitis and pericarditis, or into nearby major blood vessels. All these events are associated with a high mortality rate.
45 | Oral Manifestations of Disease |
As primary care physicians and consultants, internists are often asked to evaluate patients with disease of the oral soft tissues, teeth, and pharynx. Knowledge of the oral milieu and its unique structures is necessary to guide preventive services and recognize oral manifestations of local or systemic disease (Chap. 46e). Furthermore, internists frequently collaborate with dentists in the care of patients who have a variety of medical conditions that affect oral health or who undergo dental procedures that increase their risk of medical complications.
DISEASES OF THE TEETH AND PERIODONTAL STRUCTURES
Tooth formation begins during the sixth week of embryonic life and continues through 17 years of age. Teeth start to develop in utero and continue to develop until after the tooth erupts. Normally, all 20 deciduous teeth have erupted by age 3 and have been shed by age 13. Permanent teeth, eventually totaling 32, begin to erupt by age 6 and have completely erupted by age 14, though third molars (“wisdom teeth”) may erupt later.
The erupted tooth consists of the visible crown covered with enamel and the root submerged below the gum line and covered with bonelike cementum. Dentin, a material that is denser than bone and exquisitely sensitive to pain, forms the majority of the tooth substance, surrounding a core of myxomatous pulp containing the vascular and nerve supply. The tooth is held firmly in the alveolar socket by the periodontium, supporting structures that consist of the gingivae, alveolar bone, cementum, and periodontal ligament. The periodontal ligament tenaciously binds the tooth’s cementum to the alveolar bone. Above this ligament is a collar of attached gingiva just below the crown. A few millimeters of unattached or free gingiva (1–3 mm) overlap the base of the crown, forming a shallow sulcus along the gum-tooth margin.
Dental Caries, Pulpal and Periapical Disease, and Complications Dental caries usually begin asymptomatically as a destructive infectious process of the enamel. Bacteria—principally Streptococcus mutans—colonize the organic buffering biofilm (plaque) on the tooth surface. If not removed by brushing or by the natural cleansing and antibacterial action of saliva, bacterial acids can demineralize the enamel. Fissures and pits on the occlusal surfaces are the most frequent sites of early decay. Surfaces between the teeth, adjacent to tooth restorations and exposed roots, are also vulnerable, particularly as individuals age. Over time, dental caries extend to the underlying dentin, leading to cavitation of the enamel. Without management, the caries will penetrate to the tooth pulp, producing acute pulpitis. At this stage, when the pulp infection is limited, the tooth may become sensitive to percussion and to hot or cold, and pain resolves immediately when the irritating stimulus is removed. Should the infection spread throughout the pulp, irreversible pulpitis occurs, leading to pulp necrosis. At this later stage, pain can be severe and has a sharp or throbbing visceral quality that may be worse when the patient lies down. Once pulp necrosis is complete, pain may be constant or intermittent, but cold sensitivity is lost.
Treatment of caries involves removal of the softened and infected hard tissue and restoration of the tooth structure with silver amalgam, glass ionomer, composite resin, or gold. Once irreversible pulpitis occurs, root canal therapy becomes necessary; removal of the contents of the pulp chamber and root canals is followed by thorough cleaning and filling with an inert material. Alternatively, the tooth may be extracted.
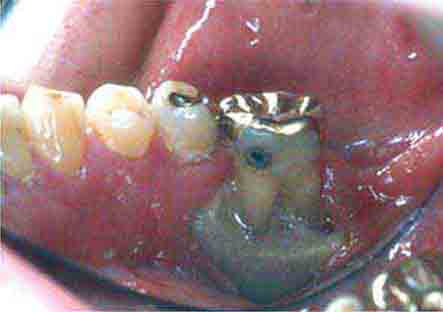
Pulpal infection leads to periapical abscess formation, which can produce pain on chewing. If the infection is mild and chronic, a periapical granuloma or eventually a periapical cyst forms, either of which produces radiolucency at the root apex. When unchecked, a periapical abscess can erode into the alveolar bone, producing osteomyelitis; penetrate and drain through the gingivae, producing a parulis (gumboil); or track along deep fascial planes, producing virulent cellulitis (Ludwig’s angina) involving the submandibular space and floor of the mouth (Chap. 201). Elderly patients, patients with diabetes mellitus, and patients taking glucocorticoids may experience little or no pain or fever as these complications develop.
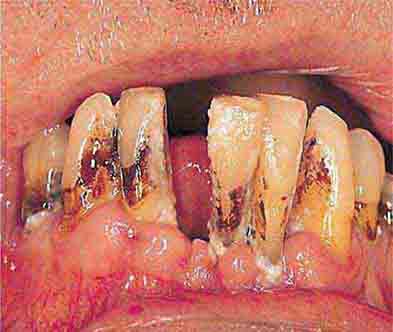
Periodontal Disease Periodontal disease and dental caries are the primary causes of tooth loss. Like dental caries, chronic infection of the gingiva and anchoring structures of the tooth begins with formation of bacterial plaque. The process begins at the gum line. Plaque and calculus (calcified plaque) are preventable by appropriate daily oral hygiene, including periodic professional cleaning. Left undisturbed, chronic inflammation can ensue and produce hyperemia of the free and attached gingivae (gingivitis), which then typically bleed with brushing. If this issue is ignored, severe periodontitis can develop, leading to deepening of the physiologic sulcus and destruction of the periodontal ligament. Gingival pockets develop around the teeth. As the periodontium (including the supporting bone) is destroyed, the teeth loosen. A role for chronic inflammation due to chronic periodontal disease in promoting coronary heart disease and stroke has been proposed. Epidemiologic studies have demonstrated a moderate but significant association between chronic periodontal inflammation and atherogenesis, though a causal role remains unproven.
Acute and aggressive forms of periodontal disease are less common than the chronic forms described above. However, if the host is stressed or exposed to a new pathogen, rapidly progressive and destructive disease of the periodontal tissue can occur. A virulent example is acute necrotizing ulcerative gingivitis. Stress and poor oral hygiene are risk factors. The presentation includes sudden gingival inflammation, ulceration, bleeding, interdental gingival necrosis, and fetid halitosis. Localized juvenile periodontitis, which is seen in adolescents, is particularly destructive and appears to be associated with impaired neutrophil chemotaxis. AIDS-related periodontitis resembles acute necrotizing ulcerative gingivitis in some patients and a more destructive form of adult chronic periodontitis in others. It may also produce a gangrene-like destructive process of the oral soft tissues and bone that resembles noma, an infectious condition seen in severely malnourished children in developing nations.
Prevention of Tooth Decay and Periodontal Infection Despite the reduced prevalences of dental caries and periodontal disease in the United States (due in large part to water fluoridation and improved dental care, respectively), both diseases constitute a major public health problem worldwide, particularly in certain groups. The internist should promote preventive dental care and hygiene as part of health maintenance. Populations at high risk for dental caries and periodontal disease include those with hyposalivation and/or xerostomia, diabetics, alcoholics, tobacco users, persons with Down syndrome, and those with gingival hyperplasia. Furthermore, patients lacking access to dental care (e.g., as a result of low socioeconomic status) and patients with a reduced ability to provide self-care (e.g., individuals with disabilities, nursing home residents, and persons with dementia or upper-extremity disability) suffer at a disproportionate rate. It is important to provide counseling regarding regular dental hygiene and professional cleaning, use of fluoride-containing toothpaste, professional fluoride treatments, and (for patients with limited dexterity) use of electric toothbrushes and also to instruct persons caring for those who are not capable of self-care. Cost, fear of dental care, and differences in language and culture create barriers that prevent some people from seeking preventive dental services.
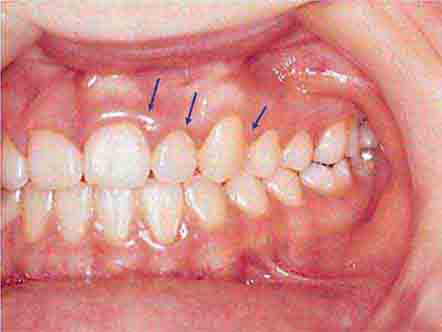
Developmental and Systemic Disease Affecting the Teeth and Periodontium In addition to posing cosmetic issues, malocclusion, the most common developmental oral problem, can interfere with mastication unless corrected through orthodontic and surgical techniques. Impacted third molars are common and can become infected or erupt into an insufficient space. Acquired prognathism due to acromegaly may also lead to malocclusion, as may deformity of the maxilla and mandible due to Paget’s disease of the bone. Delayed tooth eruption, a receding chin, and a protruding tongue are occasional features of cretinism and hypopituitarism. Congenital syphilis produces tapering, notched (Hutchinson’s) incisors and finely nodular (mulberry) molar crowns. Enamel hypoplasia results in crown defects ranging from pits to deep fissures of primary or permanent teeth. Intrauterine infection (syphilis, rubella), vitamin deficiency (A, C, or D), disorders of calcium metabolism (malabsorption, vitamin D–resistant rickets, hypoparathyroidism), prematurity, high fever, and rare inherited defects (amelogenesis imperfecta) are all causes. Tetracycline, given in sufficiently high doses during the first 8 years of life, may produce enamel hypoplasia and discoloration. Exposure to endogenous pigments can discolor developing teeth; etiologies include erythroblastosis fetalis (green or bluish-black), congenital liver disease (green or yellow-brown), and porphyria (red or brown that fluoresces with ultraviolet light). Mottled enamel occurs if excessive fluoride is ingested during development. Worn enamel is seen with age, bruxism, or excessive acid exposure (e.g., chronic gastric reflux or bulimia). Celiac disease is associated with nonspecific enamel defects in children but not in adults.
Total or partial tooth loss resulting from periodontitis is seen with cyclic neutropenia, Papillon-Lefévre syndrome, Chédiak-Higashi syndrome, and leukemia. Rapid focal tooth loosening is most often due to infection, but rarer causes include Langerhans cell histiocytosis, Ewing’s sarcoma, osteosarcoma, and Burkitt’s lymphoma. Early loss of primary teeth is a feature of hypophosphatasia, a rare congenital error of metabolism.
Pregnancy may produce gingivitis and localized pyogenic granulomas. Severe periodontal disease occurs in uncontrolled diabetes mellitus. Gingival hyperplasia may be caused by phenytoin, calcium channel blockers (e.g., nifedipine), and cyclosporine, though excellent daily oral care can prevent or reduce its occurrence. Idiopathic familial gingival fibromatosis and several syndrome-related disorders cause similar conditions. Discontinuation of the medication may reverse the drug-induced form, though surgery may be needed to control both of the latter entities. Linear gingival erythema is variably seen in patients with advanced HIV infection and probably represents immune deficiency and decreased neutrophil activity. Diffuse or focal gingival swelling may be a feature of early or late acute myelomonocytic leukemia as well as of other lymphoproliferative disorders. A rare but pathognomonic sign of granulomatosis with polyangiitis is a red-purplish, granular gingivitis (strawberry gums).
DISEASES OF THE ORAL MUCOSA
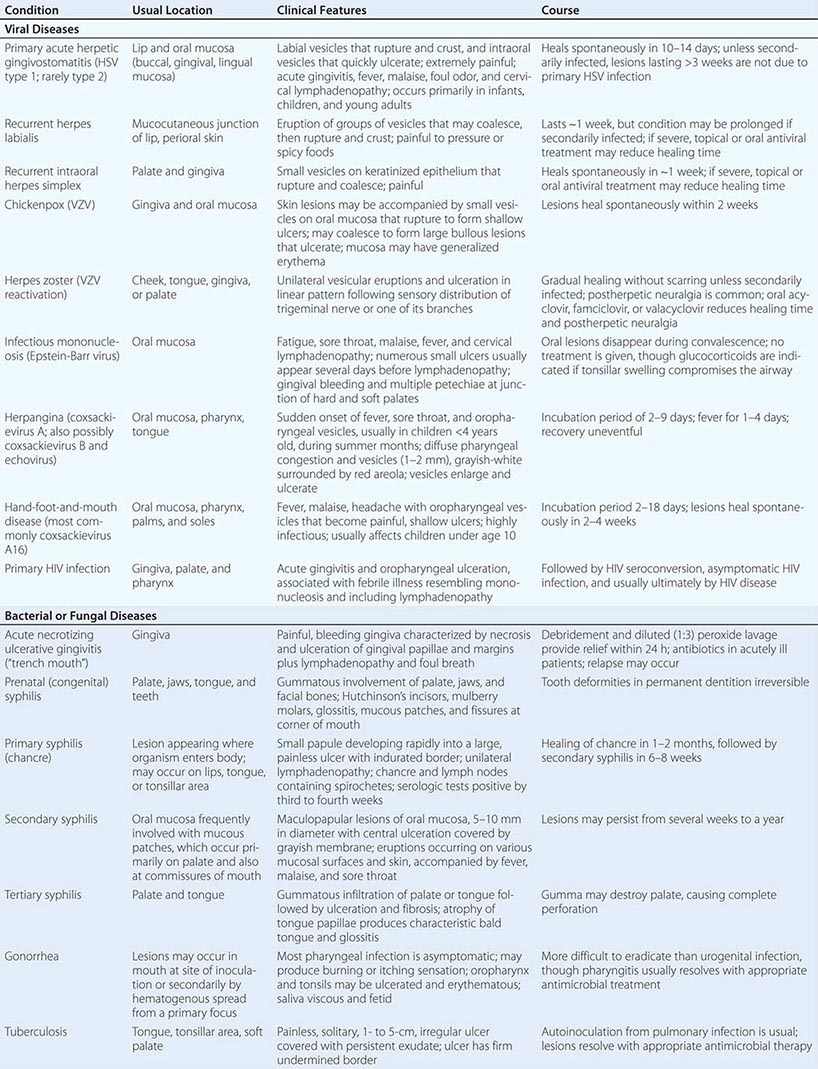
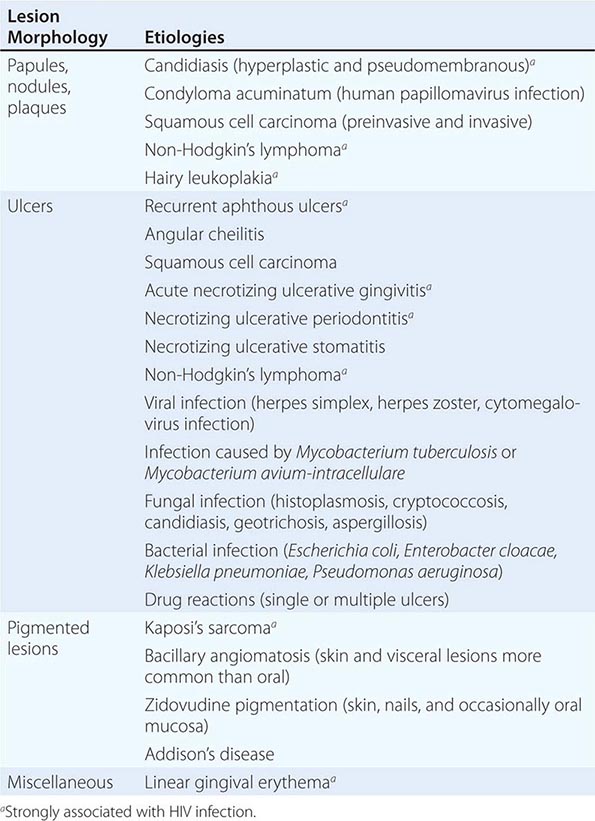
Infections Most oral mucosal diseases involve microorganisms (Table 45-1).
VESICULAR, BULLOUS, OR ULCERATIVE LESIONS OF THE ORAL MUCOSA |
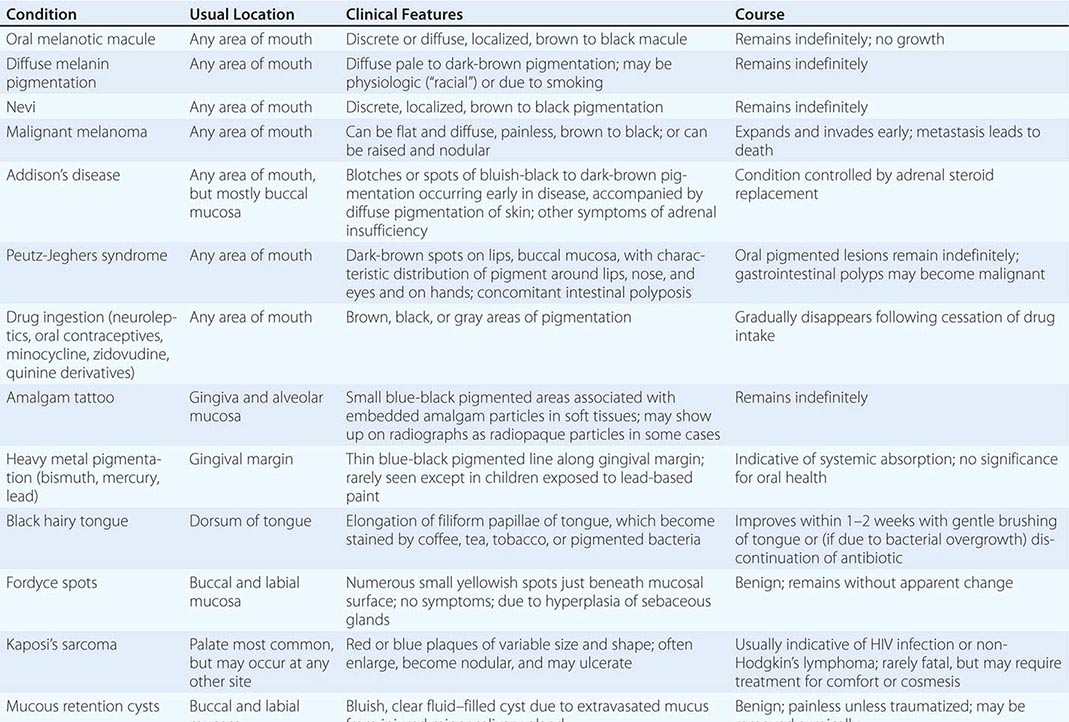
Pigmented Lesions See Table 45-2.
PIGMENTED LESIONS OF THE ORAL MUCOSA |
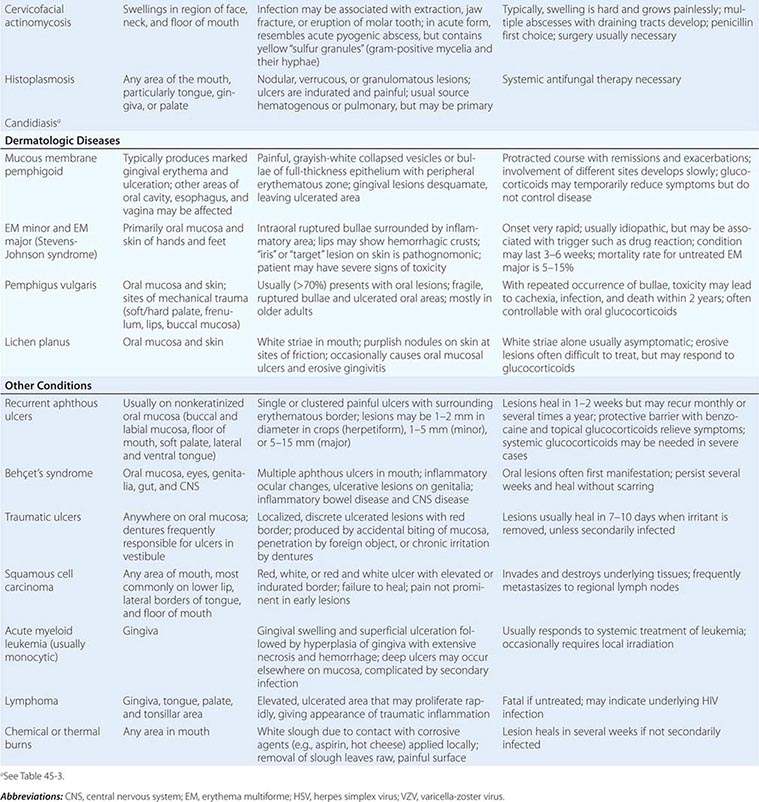
Dermatologic Diseases See Tables 45-1, 45-2, and 45-3 and Chaps. 70–74.
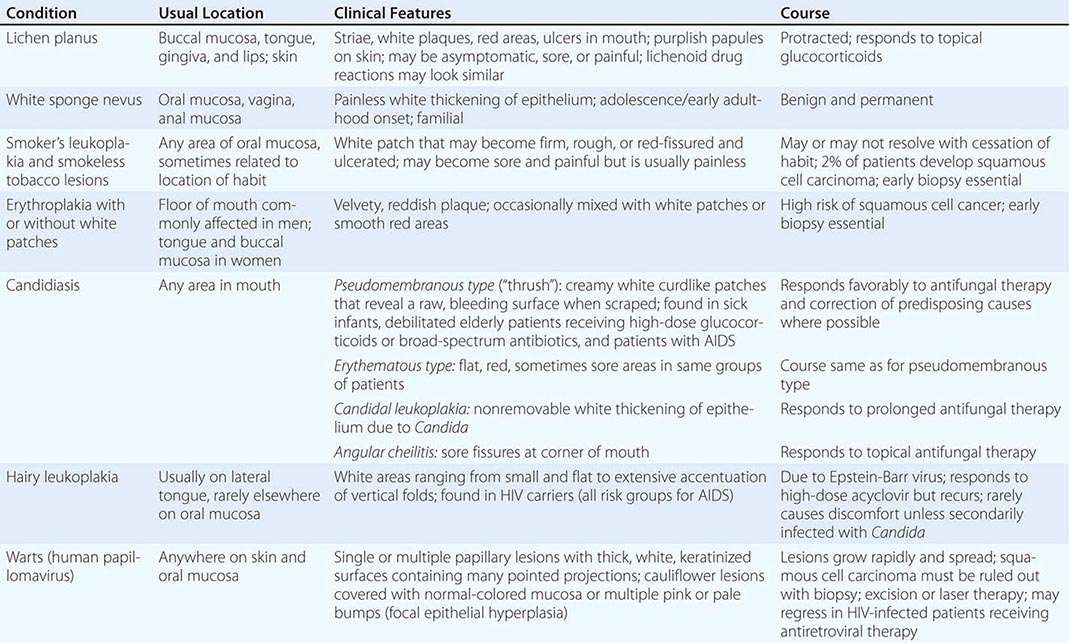
WHITE LESIONS OF ORAL MUCOSA |
Diseases of the Tongue See Table 45-4.
ALTERATIONS OF THE TONGUE |

HIV Disease and AIDS See Tables 45-1, 45-2, 45-3, and 45-5; Chap. 226; and Fig. 218-3.
ORAL LESIONS ASSOCIATED WITH HIV INFECTION |
Ulcers Ulceration is the most common oral mucosal lesion. Although there are many causes, the host and the pattern of lesions, including the presence of organ system features, narrow the differential diagnosis (Table 45-1). Most acute ulcers are painful and self-limited. Recurrent aphthous ulcers and herpes simplex account for the majority. Persistent and deep aphthous ulcers can be idiopathic or can accompany HIV/AIDS. Aphthous lesions are often the presenting symptom in Behçet’s syndrome (Chap. 387). Similar-appearing, though less painful, lesions may occur in reactive arthritis, and aphthous ulcers are occasionally present during phases of discoid or systemic lupus erythematosus (Chap. 382). Aphthous-like ulcers are seen in Crohn’s disease (Chap. 351), but, unlike the common aphthous variety, they may exhibit granulomatous inflammation on histologic examination. Recurrent aphthae are more prevalent in patients with celiac disease and have been reported to remit with elimination of gluten.
Of major concern are chronic, relatively painless ulcers and mixed red/white patches (erythroplakia and leukoplakia) of >2 weeks’ duration. Squamous cell carcinoma and premalignant dysplasia should be considered early and a diagnostic biopsy performed. This awareness and this procedure are critically important because early-stage malignancy is vastly more treatable than late-stage disease. High-risk sites include the lower lip, floor of the mouth, ventral and lateral tongue, and soft palate–tonsillar pillar complex. Significant risk factors for oral cancer in Western countries include sun exposure (lower lip), tobacco and alcohol use, and human papillomavirus infection. In India and some other Asian countries, smokeless tobacco mixed with betel nut, slaked lime, and spices is a common cause of oral cancer. Rarer causes of chronic oral ulcer, such as tuberculosis, fungal infection, granulomatosis with polyangiitis, and midline granuloma may look identical to carcinoma. Making the correct diagnosis depends on recognizing other clinical features and performing a biopsy of the lesion. The syphilitic chancre is typically painless and therefore easily missed. Regional lymphadenopathy is invariably present. The syphilitic etiology is confirmed with appropriate bacterial and serologic tests.
Disorders of mucosal fragility often produce painful oral ulcers that fail to heal within 2 weeks. Mucous membrane pemphigoid and pemphigus vulgaris are the major acquired disorders. While their clinical features are often distinctive, a biopsy or immunohistochemical examination should be performed to diagnose these entities and to distinguish them from lichen planus and drug reactions.
Hematologic and Nutritional Disease Internists are more likely to encounter patients with acquired, rather than congenital, bleeding disorders. Bleeding should stop 15 min after minor trauma and within an hour after tooth extraction if local pressure is applied. More prolonged bleeding, if not due to continued injury or rupture of a large vessel, should lead to investigation for a clotting abnormality. In addition to bleeding, petechiae and ecchymoses are prone to occur at the vibrating line between the soft and hard palates in patients with platelet dysfunction or thrombocytopenia.
All forms of leukemia, but particularly acute myelomonocytic leukemia, can produce gingival bleeding, ulcers, and gingival enlargement. Oral ulcers are a feature of agranulocytosis, and ulcers and mucositis are often severe complications of chemotherapy and radiation therapy for hematologic and other malignancies. Plummer-Vinson syndrome (iron deficiency, angular stomatitis, glossitis, and dysphagia) raises the risk of oral squamous cell cancer and esophageal cancer at the postcricoidal tissue web. Atrophic papillae and a red, burning tongue may occur with pernicious anemia. Deficiencies in B-group vitamins produce many of these same symptoms as well as oral ulceration and cheilosis. Consequences of scurvy include swollen, bleeding gums; ulcers; and loosening of the teeth.
NONDENTAL CAUSES OF ORAL PAIN
Most, but not all, oral pain emanates from inflamed or injured tooth pulp or periodontal tissues. Nonodontogenic causes are often overlooked. In most instances, toothache is predictable and proportional to the stimulus applied, and an identifiable condition (e.g., caries, abscess) is found. Local anesthesia eliminates pain originating from dental or periodontal structures, but not referred pains. The most common nondental source of pain is myofascial pain referred from muscles of mastication, which become tender and ache with increased use. Many sufferers exhibit bruxism (grinding of the teeth) secondary to stress and anxiety. Temporomandibular joint disorder is closely related. It affects both sexes, with a higher prevalence among women. Features include pain, limited mandibular movement, and temporomandibular joint sounds. The etiologies are complex; malocclusion does not play the primary role once attributed to it. Osteoarthritis is a common cause of masticatory pain. Anti-inflammatory medication, jaw rest, soft foods, and heat provide relief. The temporomandibular joint is involved in 50% of patients with rheumatoid arthritis, and its involvement is usually a late feature of severe disease. Bilateral preauricular pain, particularly in the morning, limits range of motion.
Migrainous neuralgia may be localized to the mouth. Episodes of pain and remission without an identifiable cause and a lack of relief with local anesthesia are important clues. Trigeminal neuralgia (tic douloureux) can involve the entire branch or part of the mandibular or maxillary branch of the fifth cranial nerve and can produce pain in one or a few teeth. Pain may occur spontaneously or may be triggered by touching the lip or gingiva, brushing the teeth, or chewing. Glossopharyngeal neuralgia produces similar acute neuropathic symptoms in the distribution of the ninth cranial nerve. Swallowing, sneezing, coughing, or pressure on the tragus of the ear triggers pain that is felt in the base of the tongue, pharynx, and soft palate and may be referred to the temporomandibular joint. Neuritis involving the maxillary and mandibular divisions of the trigeminal nerve (e.g., maxillary sinusitis, neuroma, and leukemic infiltrate) is distinguished from ordinary toothache by the neuropathic quality of the pain. Occasionally, phantom pain follows tooth extraction. Pain and hyperalgesia behind the ear and on the side of the face in the day or so before facial weakness develops often constitute the earliest symptom of Bell’s palsy. Likewise, similar symptoms may precede visible lesions of herpes zoster infecting the seventh nerve (Ramsey-Hunt syndrome) or trigeminal nerve. Postherpetic neuralgia may follow either condition. Coronary ischemia may produce pain exclusively in the face and jaw; as in typical angina pectoris, this pain is usually reproducible with increased myocardial demand. Aching in several upper molar or premolar teeth that is unrelieved by anesthetizing the teeth may point to maxillary sinusitis.
Giant cell arteritis is notorious for producing headache, but it may also produce facial pain or sore throat without headache. Jaw and tongue claudication with chewing or talking is relatively common. Tongue infarction is rare. Patients with subacute thyroiditis often experience pain referred to the face or jaw before the tenderness of the thyroid gland and transient hyperthyroidism are appreciated.
“Burning mouth syndrome” (glossodynia) occurs in the absence of an identifiable cause (e.g., vitamin B12 deficiency, iron deficiency, diabetes mellitus, low-grade Candida infection, food sensitivity, or subtle xerostomia) and predominantly affects postmenopausal women. The etiology may be neuropathic. Clonazepam, α-lipoic acid, and cognitive behavioral therapy have benefited some patients. Some cases associated with an angiotensin-converting enzyme inhibitor have remitted when treatment with the drug was discontinued.
DISEASES OF THE SALIVARY GLANDS
Saliva is essential to oral health. Its absence leads to dental caries, periodontal disease, and difficulties in wearing dental prostheses, masticating, and speaking. Its major components, water and mucin, serve as a cleansing solvent and lubricating fluid. In addition, saliva contains antimicrobial factors (e.g., lysozyme, lactoperoxidase, secretory IgA), epidermal growth factor, minerals, and buffering systems. The major salivary glands secrete intermittently in response to autonomic stimulation, which is high during a meal but low otherwise. Hundreds of minor glands in the lips and cheeks secrete mucus continuously throughout the day and night. Consequently, oral function becomes impaired when salivary function is reduced. The sensation of a dry mouth (xerostomia) is perceived when salivary flow is reduced by 50%. The most common etiology is medication, especially drugs with anticholinergic properties but also alpha and beta blockers, calcium channel blockers, and diuretics. Other causes include Sjögren’s syndrome, chronic parotitis, salivary duct obstruction, diabetes mellitus, HIV/AIDS, and radiation therapy that includes the salivary glands in the field (e.g., for Hodgkin’s disease and for head and neck cancer). Management involves the elimination or limitation of drying medications, preventive dental care, and supplementation with oral liquid or salivary substitutes. Sugarless mints or chewing gum may stimulate salivary secretion if dysfunction is mild. When sufficient exocrine tissue remains, pilocarpine or cevimeline has been shown to increase secretions. Commercial saliva substitutes or gels relieve dryness. Fluoride supplementation is critical to prevent caries.
Sialolithiasis presents most often as painful swelling but in some instances as only swelling or only pain. Conservative therapy consists of local heat, massage, and hydration. Promotion of salivary secretion with mints or lemon drops may flush out small stones. Antibiotic treatment is necessary when bacterial infection in suspected. In adults, acute bacterial parotitis is typically unilateral and most commonly affects postoperative, dehydrated, and debilitated patients. Staphylococcus aureus (including methicillin-resistant strains) and anaerobic bacteria are the most common pathogens. Chronic bacterial sialadenitis results from lowered salivary secretion and recurrent bacterial infection. When suspected bacterial infection is not responsive to therapy, the differential diagnosis should be expanded to include benign and malignant neoplasms, lymphoproliferative disorders, Sjögren’s syndrome, sarcoidosis, tuberculosis, lymphadenitis, actinomycosis, and granulomatosis with polyangiitis. Bilateral nontender parotid enlargement occurs with diabetes mellitus, cirrhosis, bulimia, HIV/AIDS, and drugs (e.g., iodide, propylthiouracil).
Pleomorphic adenoma comprises two-thirds of all salivary neoplasms. The parotid is the principal salivary gland affected, and the tumor presents as a firm, slow-growing mass. Although this tumor is benign, its recurrence is common if resection is incomplete. Malignant tumors such as mucoepidermoid carcinoma, adenoid cystic carcinoma, and adenocarcinoma tend to grow relatively fast, depending upon grade. They may ulcerate and invade nerves, producing numbness and facial paralysis. Surgical resection is the primary treatment. Radiation therapy (particularly neutron-beam therapy) is used when surgery is not feasible and as post-resection for certain histologic types with a high risk of recurrence. Malignant salivary gland tumors have a 5-year survival rate of ~68%.
DENTAL CARE FOR MEDICALLY COMPLEX PATIENTS
Routine dental care (e.g., uncomplicated extraction, scaling and cleaning, tooth restoration, and root canal) is remarkably safe. The most common concerns regarding care of dental patients with medical disease are excessive bleeding for patients taking anticoagulants, infection of the heart valves and prosthetic devices from hematogenous seeding by the oral flora, and cardiovascular complications resulting from vasopressors used with local anesthetics during dental treatment. Experience confirms that the risk of any of these complications is very low.
Patients undergoing tooth extraction or alveolar and gingival surgery rarely experience uncontrolled bleeding when warfarin anticoagulation is maintained within the therapeutic range currently recommended for prevention of venous thrombosis, atrial fibrillation, or mechanical heart valve. Embolic complications and death, however, have been reported during subtherapeutic anticoagulation. Therapeutic anticoagulation should be confirmed before and continued through the procedure. Likewise, low-dose aspirin (e.g., 81–325 mg) can safely be continued. For patients taking aspirin and another antiplatelet medication (e.g., clopidogrel), the decision to continue the second antiplatelet medication should be based on individual consideration of the risks of thrombosis and bleeding.
Patients at risk for bacterial endocarditis (Chap. 155) should maintain optimal oral hygiene, including flossing, and have regular professional cleanings. Currently, guidelines recommend that prophylactic antibiotics be restricted to those patients at high risk for bacterial endocarditis who undergo dental and oral procedures involving significant manipulation of gingival or periapical tissue or penetration of the oral mucosa. If unexpected bleeding occurs, antibiotics given within 2 h after the procedure provide effective prophylaxis.
Hematogenous bacterial seeding from oral infection can undoubtedly produce late prosthetic-joint infection and therefore requires removal of the infected tissue (e.g., drainage, extraction, root canal) and appropriate antibiotic therapy. However, evidence that late prosthetic-joint infection follows routine dental procedures is lacking. For this reason, antibiotic prophylaxis is not recommended before dental surgery for patients with orthopedic pins, screws, and plates. Antibiotic prophylaxis is recommended for patients within the first 2 years after joint replacement who have inflammatory arthropathies, immunosuppression, type 1 diabetes mellitus, previous prosthetic-joint infection, hemophilia, or malnourishment.
Concern often arises regarding the use of vasoconstrictors to treat patients with hypertension and heart disease. Vasoconstrictors enhance the depth and duration of local anesthesia, thus reducing the anesthetic dose and potential toxicity. If intravascular injection is avoided, 2% lidocaine with 1:100,000 epinephrine (limited to a total of 0.036 mg of epinephrine) can be used safely in patients with controlled hypertension and stable coronary heart disease, arrhythmia, or congestive heart failure. Precautions should be taken with patients taking tricyclic antidepressants and nonselective beta blockers because these drugs may potentiate the effect of epinephrine.
Elective dental treatments should be postponed for at least 1 month and preferably for 6 months after myocardial infarction, after which the risk of reinfarction is low provided the patient is medically stable (e.g., stable rhythm, stable angina, and no heart failure). Patients who have suffered a stroke should have elective dental care deferred for 6 months. In both situations, effective stress reduction requires good pain control, including the use of the minimal amount of vasoconstrictor necessary to provide good hemostasis and local anesthesia.
Bisphosphonate therapy is associated with osteonecrosis of the jaw. However, the risk with oral bisphosphonate therapy is very low. Most patients affected have received high-dose aminobisphosphonate therapy for multiple myeloma or metastatic breast cancer and have undergone tooth extraction or dental surgery. Intraoral lesions, of which two-thirds are painful, appear as exposed yellow-white hard bone involving the mandible or maxilla. Screening tests for determining risk of osteonecrosis are unreliable. Patients slated for aminobisphosphonate therapy should receive preventive dental care that reduces the risk of infection and the need for future dentoalveolar surgery.
HALITOSIS
Halitosis typically emanates from the oral cavity or nasal passages. Volatile sulfur compounds resulting from bacterial decay of food and cellular debris account for the malodor. Periodontal disease, caries, acute forms of gingivitis, poorly fitting dentures, oral abscess, and tongue coating are common causes. Treatment includes correcting poor hygiene, treating infection, and tongue brushing. Hyposalivation can produce and exacerbate halitosis. Pockets of decay in the tonsillar crypts, esophageal diverticulum, esophageal stasis (e.g., achalasia, stricture), sinusitis, and lung abscess account for some instances. A few systemic diseases produce distinctive odors: renal failure (ammoniacal), hepatic (fishy), and ketoacidosis (fruity). Helicobacter pylori gastritis can also produce ammoniacal breath. If a patient presents because of concern about halitosis but no odor is detectable, then pseudohalitosis or halitophobia must be considered.
AGING AND ORAL HEALTH
While tooth loss and dental disease are not normal consequences of aging, a complex array of structural and functional changes that occur with age can affect oral health. Subtle changes in tooth structure (e.g., diminished pulp space and volume, sclerosis of dentinal tubules, and altered proportions of nerve and vascular pulp content) result in the elimination or diminution of pain sensitivity and a reduction in the reparative capacity of the teeth. In addition, age-associated fatty replacement of salivary acini may reduce physiologic reserve, thus increasing the risk of hyposalivation. In healthy older adults, there is minimal, if any, reduction in salivary flow.
Poor oral hygiene often results when general health fails or when patients lose manual dexterity and upper-extremity flexibility. This situation is particularly common among frail older adults and nursing home residents and must be emphasized because regular oral cleaning and dental care reduce the incidence of pneumonia and oral disease as well as the mortality risk in this population. Other risks for dental decay include limited lifetime fluoride exposure. Without assiduous care, decay can become quite advanced yet remain asymptomatic. Consequently, much of a tooth—or the entire tooth—can be destroyed before the patient is aware of the process.
Periodontal disease, a leading cause of tooth loss, is indicated by loss of alveolar bone height. More than 90% of the U.S. population has some degree of periodontal disease by age 50. Healthy adults who have not had significant alveolar bone loss by the sixth decade of life do not typically experience significant worsening with advancing age.
Complete edentulousness with advanced age, though less common than in previous decades, still affects <50% of the U.S. population ≥85 years of age. Speech, mastication, and facial contours are dramatically affected. Edentulousness may also exacerbate obstructive sleep apnea, particularly in asymptomatic individuals who wear dentures. Dentures can improve verbal articulation and restore diminished facial contours. Mastication can also be restored; however, patients expecting dentures to facilitate oral intake are often disappointed. Accommodation to dentures requires a period of adjustment. Pain can result from friction or traumatic lesions produced by loose dentures. Poor fit and poor oral hygiene may permit the development of candidiasis. This fungal infection may be either asymptomatic or painful and is suggested by erythematous smooth or granular tissue conforming to an area covered by the appliance. Individuals with dentures and no natural teeth need regular (annual) professional oral examinations.
46e | Atlas of Oral Manifestations of Disease |
The health status of the oral cavity is linked to cardiovascular disease, diabetes, and other systemic illnesses. Thus, examining the oral cavity for signs of disease is a key part of the physical exam. This chapter presents numerous outstanding clinical photographs illustrating many of the conditions discussed in Chap. 45, Oral Manifestations of Disease. Conditions affecting the teeth, periodontal tissues, and oral mucosa are all represented.
FIGURE 46e-1 Gingival overgrowth secondary to calcium channel blocker use.
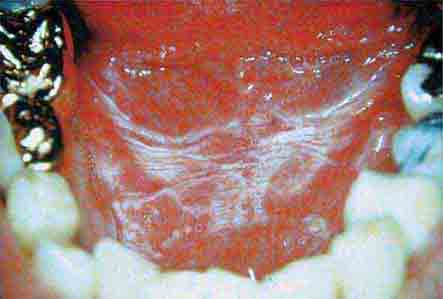
FIGURE 46e-2 Oral lichen planus.
FIGURE 46e-3 Erosive lichen planus.
FIGURE 46e-4 Stevens-Johnson syndrome—reaction to nevirapine.
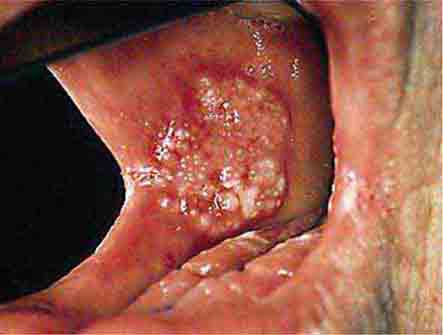
FIGURE 46e-5 Erythematosus candidiasis under a denture (i.e., the patient should be treated for this fungal infection).
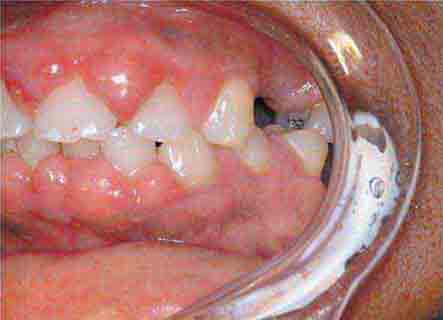
FIGURE 46e-6 Severe periodontitis.
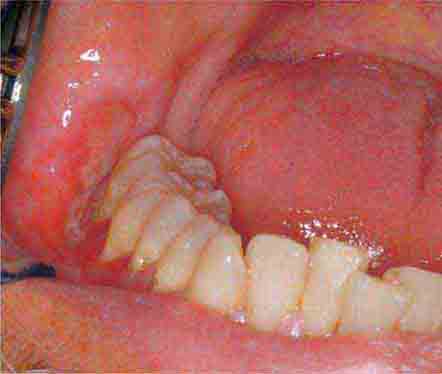
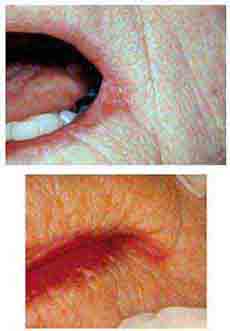
FIGURE 46e-7 Angular cheilitis.
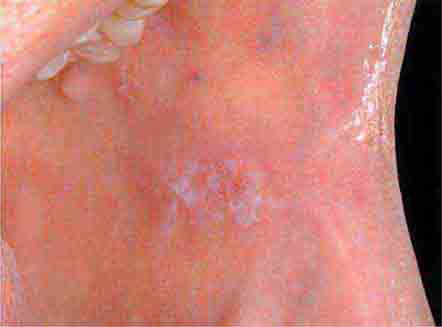
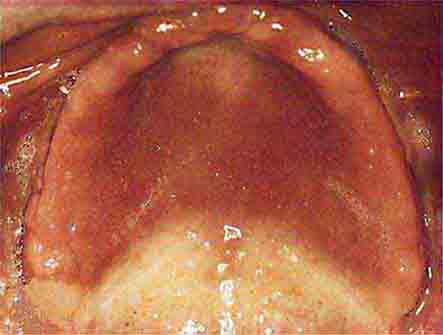
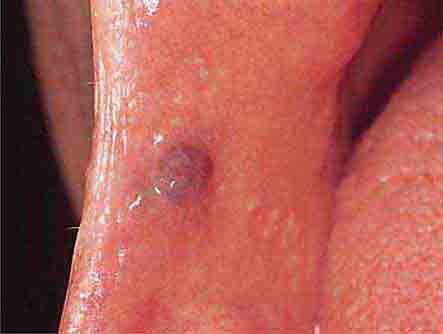
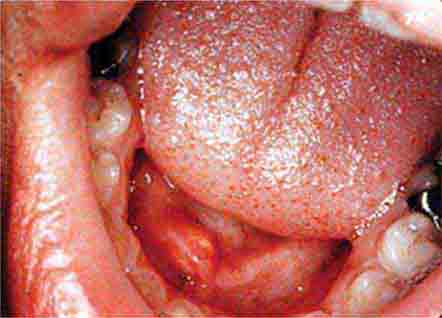
FIGURE 46e-8 Sublingual leukoplakia.
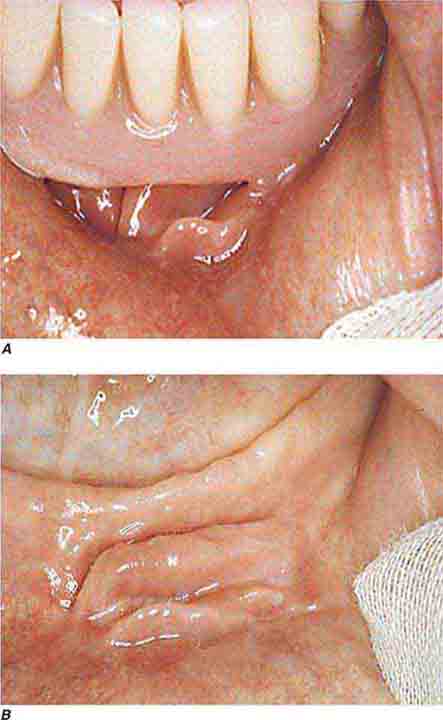
FIGURE 46e-9 A. Epulis (gingival hypertrophy) under denture. B. Epulis fissuratum.
FIGURE 46e-10 Traumatic lesion inside of cheek.
FIGURE 46e-11 Oral leukoplakia, subtype homogenous leukoplakia.
FIGURE 46e-12 Oral carcinoma.
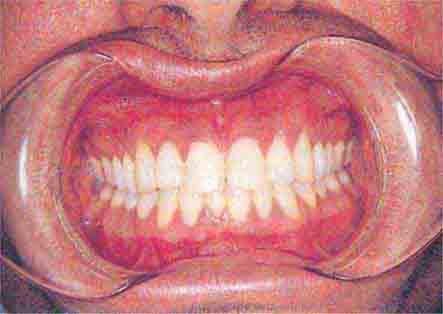
FIGURE 46e-13 Healthy mouth.
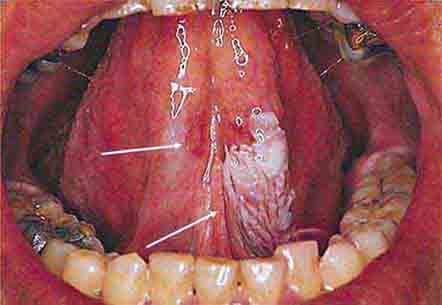
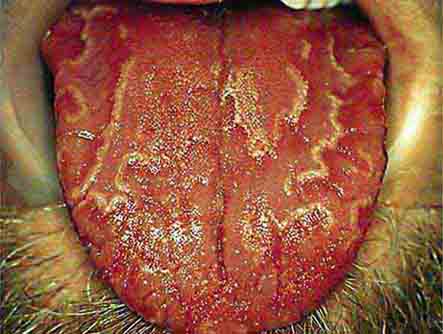
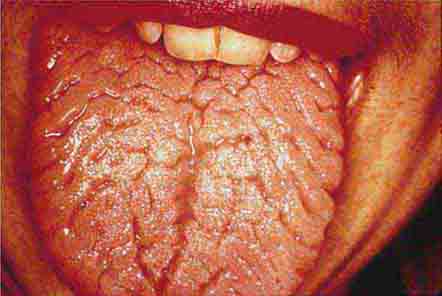
FIGURE 46e-14 Geographic tongue.
FIGURE 46e-15 Moderate gingivitis.
FIGURE 46e-16 Gingival recession.
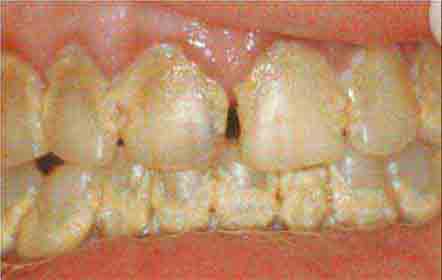
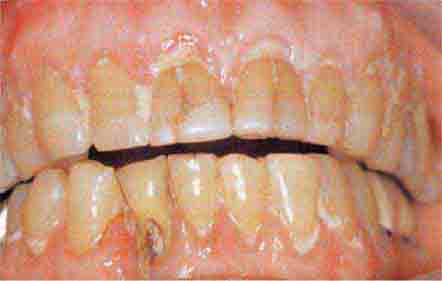
FIGURE 46e-17 Heavy calculus and gingival inflammation.
FIGURE 46e-18 Severe gingival inflammation and heavy calculus.
FIGURE 46e-19 Root cavity in presence of severe periodontal disease.
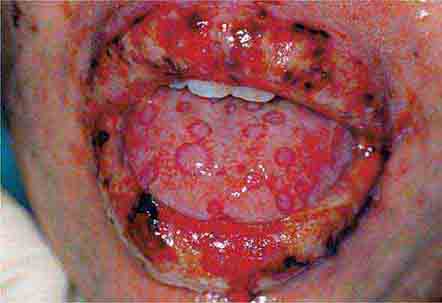
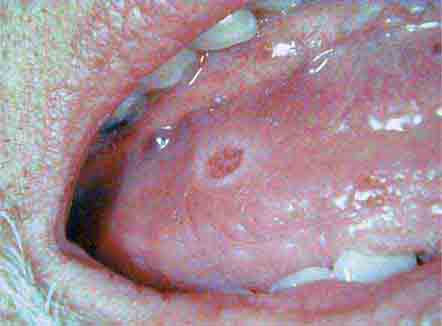
FIGURE 46e-20 Ulcer on lateral border of tongue—potential carcinoma.
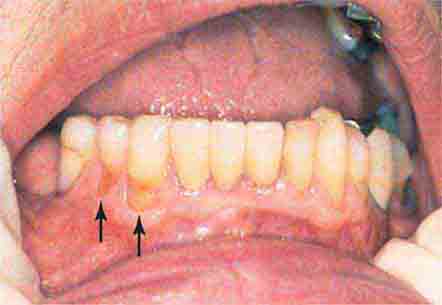
FIGURE 46e-21 Osteonecrosis.
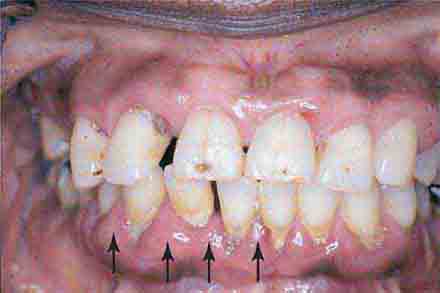
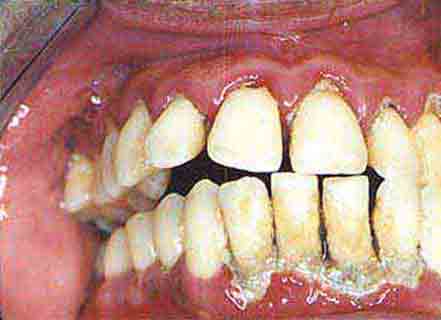
FIGURE 46e-22 Severe periodontal disease, missing tooth, very mobile teeth.
FIGURE 46e-23 Salivary stone.
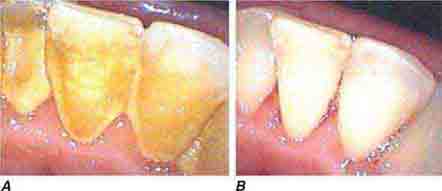
FIGURE 46e-24 A. Calculus. B. Teeth cleaned.
FIGURE 46e-25 Traumatic ulcer.
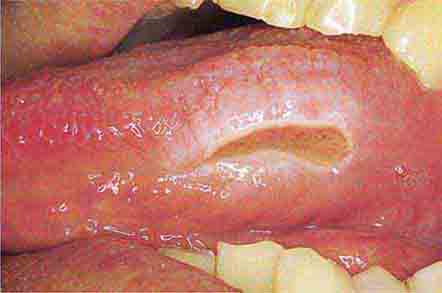
FIGURE 46e-26 Fissured tongue.
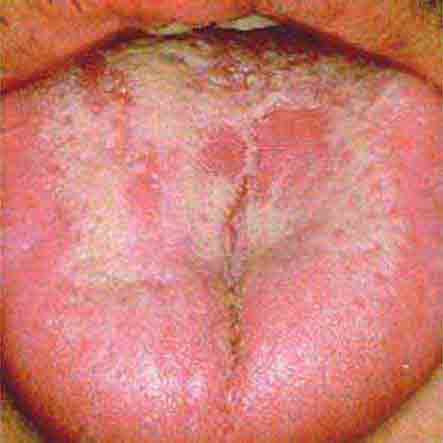
FIGURE 46e-27 White coated tongue—likely candidiasis.
ACKNOWLEDGMENT
Dr. Jane Atkinson was a co-author of this chapter in the 17th edition. Some of the materials have been carried over into this edition.
SECTION 5 | ALTERATIONS IN CIRCULATORY AND RESPIRATORY FUNCTIONS |
47e | Dyspnea |
DYSPNEA
The American Thoracic Society defines dyspnea as a “subjective experience of breathing discomfort that consists of qualitatively distinct sensations that vary in intensity. The experience derives from interactions among multiple physiological, psychological, social, and environmental factors and may induce secondary physiological and behavioral responses.” Dyspnea, a symptom, can be perceived only by the person experiencing it and must be distinguished from the signs of increased work of breathing.
MECHANISMS OF DYSPNEA
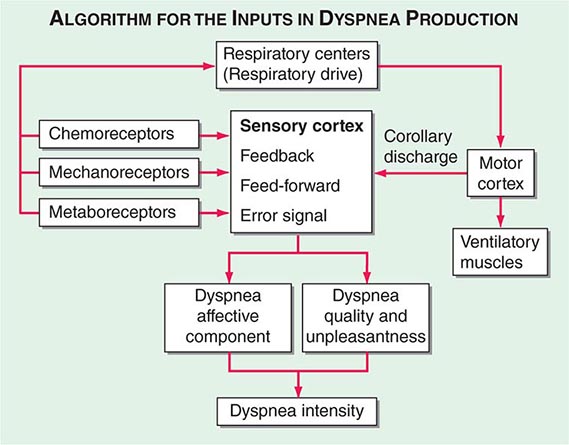
Respiratory sensations are the consequence of interactions between the efferent, or outgoing, motor output from the brain to the ventilatory muscles (feed-forward) and the afferent, or incoming, sensory input from receptors throughout the body (feedback) as well as the integrative processing of this information that we infer must be occurring in the brain (Fig. 47e-1). In contrast to painful sensations, which can often be attributed to the stimulation of a single nerve ending, dyspnea sensations are more commonly viewed as holistic, more akin to hunger or thirst. A given disease state may lead to dyspnea by one or more mechanisms, some of which may be operative under some circumstances (e.g., exercise) but not others (e.g., a change in position).
FIGURE 47e-1 Hypothetical model for integration of sensory inputs in the production of dyspnea. Afferent information from the receptors throughout the respiratory system projects directly to the sensory cortex to contribute to primary qualitative sensory experiences and to provide feedback on the action of the ventilatory pump. Afferents also project to the areas of the brain responsible for control of ventilation. The motor cortex, responding to input from the control centers, sends neural messages to the ventilatory muscles and a corollary discharge to the sensory cortex (feed-forward with respect to the instructions sent to the muscles). If the feed-forward and feedback messages do not match, an error signal is generated and the intensity of dyspnea increases. An increasing body of data supports the contribution of affective inputs to the ultimate perception of unpleasant respiratory sensations. (Adapted from MA Gillette, RM Schwartzstein, in SH Ahmedzai, MF Muer [eds]. Supportive Care in Respiratory Disease. Oxford, UK, Oxford University Press, 2005.)
Motor Efferents Disorders of the ventilatory pump—most commonly, increased airway resistance or stiffness (decreased compliance) of the respiratory system—are associated with increased work of breathing or the sense of an increased effort to breathe. When the muscles are weak or fatigued, greater effort is required, even though the mechanics of the system are normal. The increased neural output from the motor cortex is sensed via a corollary discharge, a neural signal that is sent to the sensory cortex at the same time that motor output is directed to the ventilatory muscles.
Sensory Afferents Chemoreceptors in the carotid bodies and medulla are activated by hypoxemia, acute hypercapnia, and acidemia. Stimulation of these receptors and of others that lead to an increase in ventilation produce a sensation of “air hunger.” Mechanoreceptors in the lungs, when stimulated by bronchospasm, lead to a sensation of chest tightness. J-receptors, which are sensitive to interstitial edema, and pulmonary vascular receptors, which are activated by acute changes in pulmonary artery pressure, appear to contribute to air hunger. Hyperinflation is associated with the sensation of increased work of breathing, an inability to get a deep breath, or an unsatisfying breath. Metaboreceptors, which are located in skeletal muscle, are believed to be activated by changes in the local biochemical milieu of the tissue active during exercise and, when stimulated, contribute to breathing discomfort.
Integration: Efferent-Reafferent Mismatch A discrepancy or mismatch between the feed-forward message to the ventilatory muscles and the feedback from receptors that monitor the response of the ventilatory pump increases the intensity of dyspnea. This mismatch is particularly important when there is a mechanical derangement of the ventilatory pump, as in asthma or chronic obstructive pulmonary disease (COPD).
Contribution of Emotional or Affective Factors to Dyspnea Acute anxiety or fear may increase the severity of dyspnea either by altering the interpretation of sensory data or by leading to patterns of breathing that heighten physiologic abnormalities in the respiratory system. In patients with expiratory flow limitation, for example, the increased respiratory rate that accompanies acute anxiety leads to hyperinflation, increased work and effort of breathing, and the sense of an unsatisfying breath.
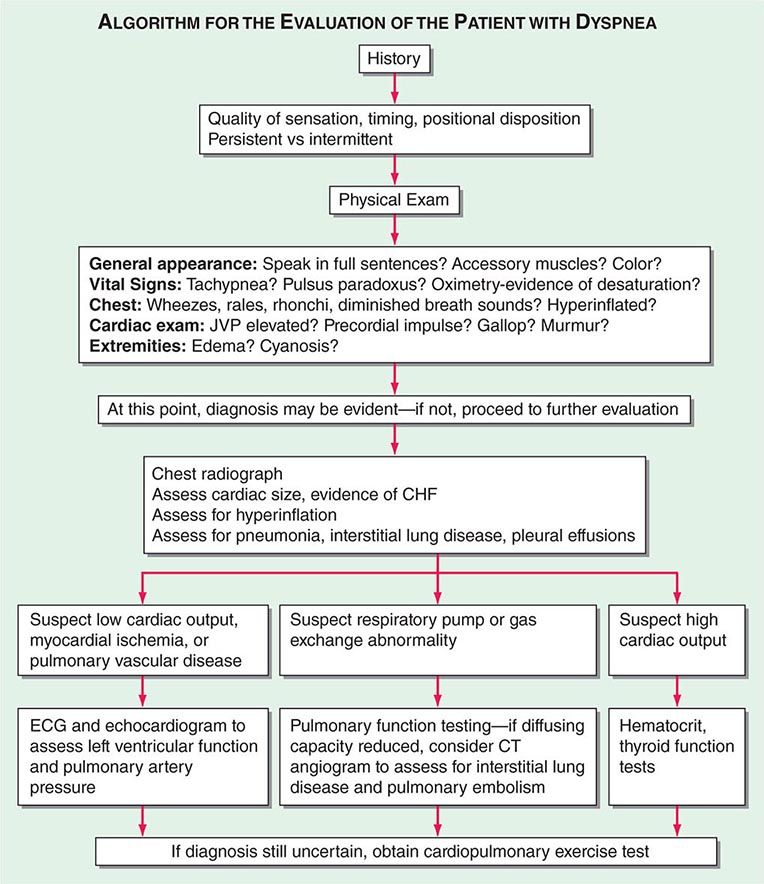
ASSESSING DYSPNEA
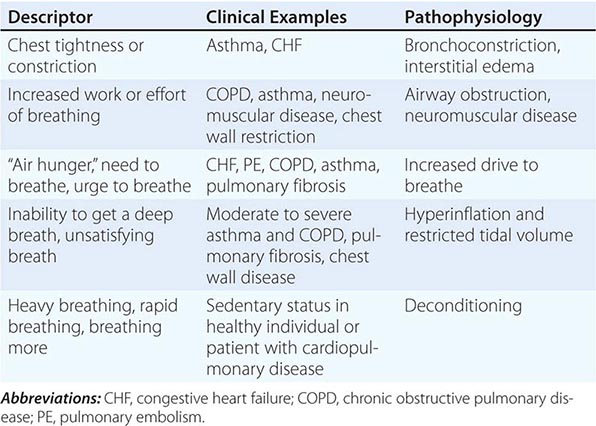
Quality of Sensation Like pain assessment, dyspnea assessment begins with a determination of the quality of the patient’s discomfort (Table 47e-1). Dyspnea questionnaires or lists of phrases commonly used by patients assist those who have difficulty describing their breathing sensations.
ASSOCIATION OF QUALITATIVE DESCRIPTORS, CLINICAL CHARACTERISTICS, AND PATHOPHYSIOLOGIC MECHANISMS OF SHORTNESS OF BREATH |
Sensory Intensity A modified Borg scale or visual analogue scale can be utilized to measure dyspnea at rest, immediately following exercise, or on recall of a reproducible physical task, such as climbing the stairs at home. An alternative approach is to gain a sense of the patient’s disability by inquiring about what activities are possible. These methods indirectly assess dyspnea and may be affected by nonrespiratory factors, such as leg arthritis or weakness. The Baseline Dyspnea Index and the Chronic Respiratory Disease Questionnaire are commonly used tools for this purpose.
Affective Dimension For a sensation to be reported as a symptom, it must be perceived as unpleasant and interpreted as abnormal. Laboratory studies have demonstrated that air hunger evokes a stronger affective response than does increased effort or work of breathing. Some therapies for dyspnea, such as pulmonary rehabilitation, may reduce breathing discomfort, in part, by altering this dimension.
DIFFERENTIAL DIAGNOSIS
Dyspnea most often results from deviations from normal function in the cardiovascular and respiratory systems. These deviations produce breathlessness as a consequence of increased drive to breathe; increased effort or work of breathing; and/or stimulation of receptors in the heart, lungs, or vascular system. Most diseases of the respiratory system are associated with alterations in the mechanical properties of the lungs and/or chest wall, and some stimulate pulmonary receptors. In contrast, disorders of the cardiovascular system more commonly lead to dyspnea by causing gas-exchange abnormalities or stimulating pulmonary and/or vascular receptors (Table 47e-2).
MECHANISMS OF DYSPNEA IN COMMON DISEASES |
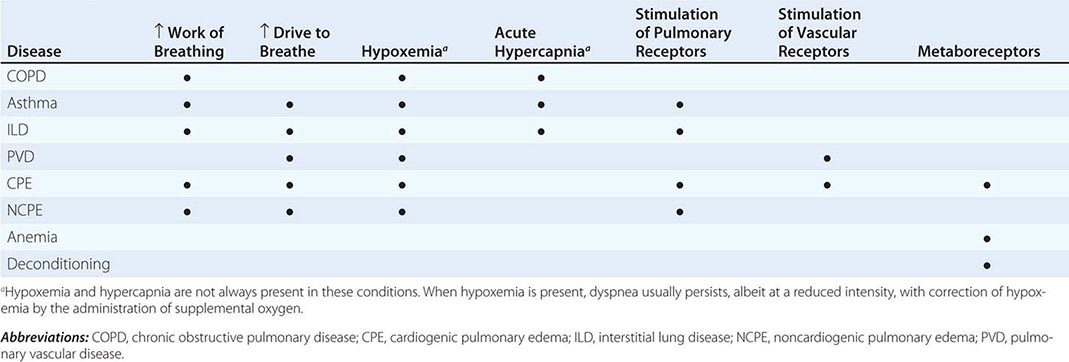
Respiratory System Dyspnea • DISEASES OF THE AIRWAYS Asthma and COPD, the most common obstructive lung diseases, are characterized by expiratory airflow obstruction, which typically leads to dynamic hyperinflation of the lungs and chest wall. Patients with moderate to severe disease have both increased resistive and elastic loads (a term that relates to the stiffness of the system) on the ventilatory muscles and experience increased work of breathing. Patients with acute bronchoconstriction also report a sense of tightness, which can exist even when lung function is still within the normal range. These patients are commonly tachypneic; this condition leads to hyperinflation and reduced respiratory system compliance and also limits tidal volume. Both the chest tightness and the tachypnea are probably due to stimulation of pulmonary receptors. Both asthma and COPD may lead to hypoxemia and hypercapnia from ventilation-perfusion (![]() /Q) mismatch (and diffusion limitation during exercise with emphysema); hypoxemia is much more common than hypercapnia as a consequence of the different ways in which oxygen and carbon dioxide bind to hemoglobin.
/Q) mismatch (and diffusion limitation during exercise with emphysema); hypoxemia is much more common than hypercapnia as a consequence of the different ways in which oxygen and carbon dioxide bind to hemoglobin.
DISEASES OF THE CHEST WALL Conditions that stiffen the chest wall, such as kyphoscoliosis, or that weaken ventilatory muscles, such as myasthenia gravis or the Guillain-Barré syndrome, are also associated with an increased effort to breathe. Large pleural effusions may contribute to dyspnea, both by increasing the work of breathing and by stimulating pulmonary receptors if there is associated atelectasis.
DISEASES OF THE LUNG PARENCHYMA Interstitial lung diseases, which may arise from infections, occupational exposures, or autoimmune disorders, are associated with increased stiffness (decreased compliance) of the lungs and increased work of breathing. In addition, ![]() /Q mismatch and the destruction and/or thickening of the alveolar-capillary interface may lead to hypoxemia and an increased drive to breathe. Stimulation of pulmonary receptors may further enhance the hyperventilation characteristic of mild to moderate interstitial disease.
/Q mismatch and the destruction and/or thickening of the alveolar-capillary interface may lead to hypoxemia and an increased drive to breathe. Stimulation of pulmonary receptors may further enhance the hyperventilation characteristic of mild to moderate interstitial disease.
Cardiovascular System Dyspnea • DISEASES OF THE LEFT HEART Diseases of the myocardium resulting from coronary artery disease and nonischemic cardiomyopathies cause a greater left-ventricular end-diastolic volume and an elevation of the left-ventricular end-diastolic as well as pulmonary capillary pressures. These elevated pressures lead to interstitial edema and stimulation of pulmonary receptors, thereby causing dyspnea; hypoxemia due to ![]() /Q mismatch may also contribute to breathlessness. Diastolic dysfunction, characterized by a very stiff left ventricle, may lead to severe dyspnea with relatively mild degrees of physical activity, particularly if it is associated with mitral regurgitation.
/Q mismatch may also contribute to breathlessness. Diastolic dysfunction, characterized by a very stiff left ventricle, may lead to severe dyspnea with relatively mild degrees of physical activity, particularly if it is associated with mitral regurgitation.
DISEASES OF THE PULMONARY VASCULATURE Pulmonary thromboembolic disease and primary diseases of the pulmonary circulation (primary pulmonary hypertension, pulmonary vasculitis) cause dyspnea via increased pulmonary-artery pressure and stimulation of pulmonary receptors. Hyperventilation is common, and hypoxemia may be present. However, in most cases, use of supplemental oxygen has only a minimal impact on the severity of dyspnea and hyperventilation.
DISEASES OF THE PERICARDIUM Constrictive pericarditis and cardiac tamponade are both associated with increased intracardiac and pulmonary vascular pressures, which are the likely cause of dyspnea in these conditions. To the extent that cardiac output is limited (at rest or with exercise) metaboreceptors may be stimulated if cardiac output is compromised to the degree that lactic acidosis develops; chemoreceptors will also be activated.
Dyspnea with Normal Respiratory and Cardiovascular Systems Mild to moderate anemia is associated with breathing discomfort during exercise. This symptom is thought to be related to stimulation of metaboreceptors; oxygen saturation is normal in patients with anemia. The breathlessness associated with obesity is probably due to multiple mechanisms, including high cardiac output and impaired ventilatory pump function (decreased compliance of the chest wall). Cardiovascular deconditioning (poor fitness) is characterized by the early development of anaerobic metabolism and the stimulation of chemoreceptors and metaboreceptors. Dyspnea that is medically unexplained has been associated with increased sensitivity to the unpleasantness of acute hypercapnia.
PULMONARY EDEMA
MECHANISMS OF FLUID ACCUMULATION
The extent to which fluid accumulates in the interstitium of the lung depends on the balance of hydrostatic and oncotic forces within the pulmonary capillaries and in the surrounding tissue. Hydrostatic pressure favors movement of fluid from the capillary into the interstitium. The oncotic pressure, which is determined by the protein concentration in the blood, favors movement of fluid into the vessel. Levels of albumin, the primary protein in the plasma, may be low in conditions such as cirrhosis and nephrotic syndrome. While hypoalbuminemia favors movement of fluid into the tissue for any given hydrostatic pressure in the capillary, it is usually not sufficient by itself to cause interstitial edema. In a healthy individual, the tight junctions of the capillary endothelium are impermeable to proteins, and the lymphatics in the tissue carry away the small amounts of protein that may leak out; together, these factors result in an oncotic force that maintains fluid in the capillary. Disruption of the endothelial barrier, however, allows protein to escape the capillary bed and enhances the movement of fluid into the tissue of the lung.
CARDIOGENIC PULMONARY EDEMA
(See also Chap. 326) Cardiac abnormalities that lead to an increase in pulmonary venous pressure shift the balance of forces between the capillary and the interstitium. Hydrostatic pressure is increased and fluid exits the capillary at an increased rate, resulting in interstitial and, in more severe cases, alveolar edema. The development of pleural effusions may further compromise respiratory system function and contribute to breathing discomfort.
Early signs of pulmonary edema include exertional dyspnea and orthopnea. Chest radiographs show peribronchial thickening, prominent vascular markings in the upper lung zones, and Kerley B lines. As the pulmonary edema worsens, alveoli fill with fluid; the chest radiograph shows patchy alveolar filling, typically in a perihilar distribution, which then progresses to diffuse alveolar infiltrates. Increasing airway edema is associated with rhonchi and wheezes.
NONCARDIOGENIC PULMONARY EDEMA
In noncardiogenic pulmonary edema, lung water increases due to damage of the pulmonary capillary lining with consequent leakage of proteins and other macromolecules into the tissue; fluid follows the protein as oncotic forces are shifted from the vessel to the surrounding lung tissue. This process is associated with dysfunction of the surfactant lining the alveoli, increased surface forces, and a propensity for the alveoli to collapse at low lung volumes. Physiologically, noncardiogenic pulmonary edema is characterized by intrapulmonary shunt with hypoxemia and decreased pulmonary compliance leading to lower functional residual capacity. On pathologic examination, hyaline membranes are evident in the alveoli, and inflammation leading to pulmonary fibrosis may be seen. Clinically, the picture ranges from mild dyspnea to respiratory failure. Auscultation of the lungs may be relatively normal despite chest radiographs that show diffuse alveolar infiltrates. CT scans demonstrate that the distribution of alveolar edema is more heterogeneous than was once thought. Although normal intracardiac pressures are considered by many to be part of the definition of noncardiogenic pulmonary edema, the pathology of the process, as described above, is distinctly different, and a combination of cardiogenic and noncardiogenic pulmonary edema is observed in some patients.
It is useful to categorize the causes of noncardiogenic pulmonary edema in terms of whether the injury to the lung is likely to result from direct, indirect, or pulmonary vascular causes (Table 47e-3). Direct injuries are mediated via the airways (e.g., aspiration) or as the consequence of blunt chest trauma. Indirect injury is the consequence of mediators that reach the lung via the bloodstream. The third category includes conditions that may result from acute changes in pulmonary vascular pressures, possibly due to sudden autonomic discharge (in the case of neurogenic and high-altitude pulmonary edema) or sudden swings of pleural pressure as well as transient damage to the pulmonary capillaries (in the case of reexpansion pulmonary edema).
COMMON CAUSES OF NONCARDIOGENIC PULMONARY EDEMA |
DISTINGUISHING CARDIOGENIC FROM NONCARDIOGENIC PULMONARY EDEMA
The history is essential for assessing the likelihood of underlying cardiac disease as well as for identification of one of the conditions associated with noncardiogenic pulmonary edema. The physical examination in cardiogenic pulmonary edema is notable for evidence of increased intracardiac pressures (S3 gallop, elevated jugular venous pulse, peripheral edema) and rales and/or wheezes on auscultation of the chest. In contrast, the physical examination in noncardiogenic pulmonary edema is dominated by the findings of the precipitating condition; pulmonary findings may be relatively normal in the early stages. The chest radiograph in cardiogenic pulmonary edema typically shows an enlarged cardiac silhouette, vascular redistribution, interstitial thickening, and perihilar alveolar infiltrates; pleural effusions are common. In noncardiogenic pulmonary edema, heart size is normal, alveolar infiltrates are distributed more uniformly throughout the lungs, and pleural effusions are uncommon. Finally, the hypoxemia of cardiogenic pulmonary edema is due largely to ![]() /Q to mismatch and responds to the administration of supplemental oxygen. In contrast, hypoxemia in noncardiogenic pulmonary edema is due primarily to intrapulmonary shunting and typically persists despite high concentrations of inhaled oxygen.
/Q to mismatch and responds to the administration of supplemental oxygen. In contrast, hypoxemia in noncardiogenic pulmonary edema is due primarily to intrapulmonary shunting and typically persists despite high concentrations of inhaled oxygen.
48 | Cough and Hemoptysis |
COUGH
Cough performs an essential protective function for human airways and lungs. Without an effective cough reflex, we are at risk for retained airway secretions and aspirated material predisposing to infection, atelectasis, and respiratory compromise. At the other extreme, excessive coughing can be exhausting; can be complicated by emesis, syncope, muscular pain, or rib fractures; and can aggravate abdominal or inguinal hernias and urinary incontinence. Cough is often a clue to the presence of respiratory disease. In many instances, cough is an expected and accepted manifestation of disease, as in acute respiratory tract infection. However, persistent cough in the absence of other respiratory symptoms commonly causes patients to seek medical attention.
COUGH MECHANISM
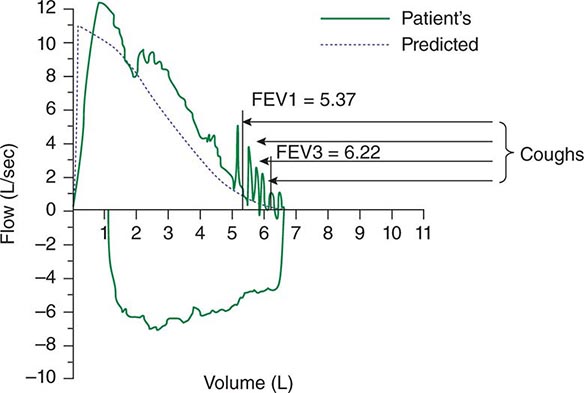
Spontaneous cough is triggered by stimulation of sensory nerve endings that are thought to be primarily rapidly adapting receptors and C fibers. Both chemical (e.g., capsaicin) and mechanical (e.g., particulates in air pollution) stimuli may initiate the cough reflex. A cationic ion channel—the type 1 vanilloid receptor—found on rapidly adapting receptors and C fibers is the receptor for capsaicin, and its expression is increased in patients with chronic cough. Afferent nerve endings richly innervate the pharynx, larynx, and airways to the level of the terminal bronchioles and extend into the lung parenchyma. They may also be located in the external auditory meatus (the auricular branch of the vagus nerve, or the Arnold nerve) and in the esophagus. Sensory signals travel via the vagus and superior laryngeal nerves to a region of the brainstem in the nucleus tractus solitarius vaguely identified as the “cough center.” The cough reflex involves a highly orchestrated series of involuntary muscular actions, with the potential for input from cortical pathways as well. The vocal cords adduct, leading to transient upper-airway occlusion. Expiratory muscles contract, generating positive intrathoracic pressures as high as 300 mmHg. With sudden release of the laryngeal contraction, rapid expiratory flows are generated, exceeding the normal “envelope” of maximal expiratory flow seen on the flow-volume curve (Fig. 48-1). Bronchial smooth-muscle contraction together with dynamic compression of airways narrows airway lumens and maximizes the velocity of exhalation. The kinetic energy available to dislodge mucus from the inside of airway walls is directly proportional to the square of the velocity of expiratory airflow. A deep breath preceding a cough optimizes the function of the expiratory muscles; a series of repetitive coughs at successively lower lung volumes sweeps the point of maximal expiratory velocity progressively further into the lung periphery.
FIGURE 48-1 Flow-volume curve shows spikes of high expiratory flow achieved with cough. FEV1, forced expiratory volume in 1 s.
IMPAIRED COUGH
Weak or ineffective cough compromises the ability to clear lower respiratory tract infections, predisposing to more serious infections and their sequelae. Weakness, paralysis, or pain of the expiratory (abdominal and intercostal) muscles is foremost on the list of causes of impaired cough (Table 48-1). Cough strength is generally assessed qualitatively; peak expiratory flow or maximal expiratory pressure at the mouth can be used as a surrogate marker for cough strength. A variety of assistive devices and techniques have been developed to improve cough strength, running the gamut from simple (splinting of the abdominal muscles with a tightly-held pillow to reduce postoperative pain while coughing) to complex (a mechanical cough-assist device supplied via face mask or tracheal tube that applies a cycle of positive pressure followed rapidly by negative pressure). Cough may fail to clear secretions despite a preserved ability to generate normal expiratory velocities; such failure may be due to either abnormal airway secretions (e.g., bronchiectasis due to cystic fibrosis) or structural abnormalities of the airways (e.g., tracheomalacia with expiratory collapse during cough).
CAUSES OF IMPAIRED COUGH |
SYMPTOMATIC COUGH
The cough of chronic bronchitis in long-term cigarette smokers rarely leads the patient to seek medical advice. It lasts for only seconds to a few minutes, is productive of benign-appearing mucoid sputum, and generally does not cause discomfort. Cough may occur in the context of other respiratory symptoms that together point to a diagnosis; for example, cough accompanied by wheezing, shortness of breath, and chest tightness after exposure to a cat or other sources of allergens suggests asthma. At times, however, cough is the dominant or sole symptom of disease, and it may be of sufficient duration and severity that relief is sought. The duration of cough is a clue to its etiology. Acute cough (<3 weeks) is most commonly due to a respiratory tract infection, aspiration, or inhalation of noxious chemicals or smoke. Subacute cough (3–8 weeks in duration) is a common residuum of tracheobronchitis, as in pertussis or “postviral tussive syndrome.” Chronic cough (>8 weeks) may be caused by a wide variety of cardiopulmonary diseases, including those of inflammatory, infectious, neoplastic, and cardiovascular etiologies. When initial assessment with chest examination and radiography is normal, cough-variant asthma, gastroesophageal reflux, nasopharyngeal drainage, and medications (angiotensin-converting enzyme [ACE] inhibitors) are the most common causes of chronic cough.
ASSESSMENT OF CHRONIC COUGH
Details as to the sound, the time of occurrence during the day, and the pattern of coughing infrequently provide useful etiologic clues. Regardless of cause, cough often worsens upon first lying down at night, with talking, or with the hyperpnea of exercise; it frequently improves with sleep. An exception may involve the cough that occurs only with certain allergic exposures or exercise in cold air, as in asthma. Useful historical questions include what circumstances surround the onset of cough, what makes the cough better or worse, and whether or not the cough produces sputum.
The physical examination seeks clues suggesting the presence of cardiopulmonary disease, including findings such as wheezing or crackles on chest examination. Examination of the auditory canals and tympanic membranes (for irritation of the latter resulting in stimulation of Arnold’s nerve), the nasal passageways (for rhinitis or polyps), and the nails (for clubbing) may also provide etiologic clues. Because cough can be a manifestation of a systemic disease such as sarcoidosis or vasculitis, a thorough general examination is equally important.
In virtually all instances, evaluation of chronic cough merits a chest radiograph. The list of diseases that can cause persistent cough without other symptoms and without detectable abnormalities on physical examination is long. It includes serious illnesses such as sarcoidosis or Hodgkin’s disease in young adults, lung cancer in older patients, and (worldwide) pulmonary tuberculosis. An abnormal chest film prompts an evaluation aimed at explaining the cough. In a patient with chronic productive cough, examination of expectorated sputum is warranted. Purulent-appearing sputum should be sent for routine bacterial culture and, in certain circumstances, mycobacterial culture as well. Cytologic examination of mucoid sputum may be useful to assess for malignancy and to distinguish neutrophilic from eosinophilic bronchitis. Expectoration of blood—whether streaks of blood, blood mixed with airway secretions, or pure blood—deserves a special approach to assessment and management (see “Hemoptysis,” below).
CHRONIC COUGH WITH A NORMAL CHEST RADIOGRAPH
It is commonly held that (alone or in combination) the use of an ACE inhibitor; postnasal drainage; gastroesophageal reflux; and asthma account for more than 90% of cases of chronic cough with a normal or noncontributory chest radiograph. However, clinical experience does not support this contention, and strict adherence to this concept discourages the search for alternative explanations by both clinicians and researchers. ACE inhibitor–induced cough occurs in 5–30% of patients taking these agents and is not dose dependent. ACE metabolizes bradykinin and other tachykinins, such as substance P. The mechanism of ACE inhibitor–associated cough may involve sensitization of sensory nerve endings due to accumulation of bradykinin. In support of this hypothesis, polymorphisms in the neurokinin-2 receptor gene are associated with ACE inhibitor–induced cough. Any patient with chronic unexplained cough who is taking an ACE inhibitor should have a trial period off the medication, regardless of the timing of the onset of cough relative to the initiation of ACE inhibitor therapy. In most instances, a safe alternative is available; angiotensin-receptor blockers do not cause cough. Failure to observe a decrease in cough after 1 month off medication argues strongly against this etiology. Postnasal drainage of any etiology can cause cough as a response to stimulation of sensory receptors of the cough-reflex pathway in the hypopharynx or aspiration of draining secretions into the trachea. Clues suggesting this etiology include postnasal drip, frequent throat clearing, and sneezing and rhinorrhea. On speculum examination of the nose, excess mucoid or purulent secretions, inflamed and edematous nasal mucosa, and/or polyps may be seen; in addition, secretions or a cobblestoned appearance of the mucosa along the posterior pharyngeal wall may be noted. Unfortunately, there is no means by which to quantitate postnasal drainage. In many instances, this diagnosis must rely on subjective information provided by the patient. This assessment must also be counterbalanced by the fact that many people who have chronic postnasal drainage do not experience cough.
Linking gastroesophageal reflux to chronic cough poses similar challenges. It is thought that reflux of gastric contents into the lower esophagus may trigger cough via reflex pathways initiated in the esophageal mucosa. Reflux to the level of the pharynx (laryngopharyngeal reflux), with consequent aspiration of gastric contents, causes a chemical bronchitis and possibly pneumonitis that can elicit cough for days afterward. Retrosternal burning after meals or on recumbency, frequent eructation, hoarseness, and throat pain may be indicative of gastroesophageal reflux. Nevertheless, reflux may also elicit minimal or no symptoms. Glottic inflammation detected on laryngoscopy may be a manifestation of recurrent reflux to the level of the throat, but it is a nonspecific finding. Quantification of the frequency and level of reflux requires a somewhat invasive procedure to measure esophageal pH directly (either nasopharyngeal placement of a catheter with a pH probe into the esophagus for 24 h or endoscopic placement of a radiotransmitter capsule into the esophagus). The precise interpretation of test results that permits an etiologic linking of reflux events and cough remains debated. Again, assigning the cause of cough to gastroesophageal reflux must be weighed against the observation that many people with symptomatic reflux do not experience chronic cough.
Cough alone as a manifestation of asthma is common among children but not among adults. Cough due to asthma in the absence of wheezing, shortness of breath, and chest tightness is referred to as “cough-variant asthma.” A history suggestive of cough-variant asthma ties the onset of cough to exposure to typical triggers for asthma and the resolution of cough to discontinuation of exposure. Objective testing can establish the diagnosis of asthma (airflow obstruction on spirometry that varies over time or reverses in response to a bronchodilator) or exclude it with certainty (a negative response to a bronchoprovocation challenge—e.g., with methacholine). In a patient capable of taking reliable measurements, home expiratory peak flow monitoring can be a cost-effective method to support or discount a diagnosis of asthma.
Chronic eosinophilic bronchitis causes chronic cough with a normal chest radiograph. This condition is characterized by sputum eosinophilia in excess of 3% without airflow obstruction or bronchial hyperresponsiveness and is successfully treated with inhaled glucocorticoids.
Treatment of chronic cough in a patient with a normal chest radiograph is often empirical and is targeted at the most likely cause(s) of cough as determined by history, physical examination, and possibly pulmonary-function testing. Therapy for postnasal drainage depends on the presumed etiology (infection, allergy, or vasomotor rhinitis) and may include systemic antihistamines; antibiotics; nasal saline irrigation; and nasal pump sprays with glucocorticoids, antihistamines, or anticholinergics. Antacids, histamine type 2 (H2) receptor antagonists, and proton-pump inhibitors are used to neutralize or decrease the production of gastric acid in gastroesophageal reflux disease; dietary changes, elevation of the head and torso during sleep, and medications to improve gastric emptying are additional therapeutic measures. Cough-variant asthma typically responds well to inhaled glucocorticoids and intermittent use of inhaled β-agonist bronchodilators.
Patients who fail to respond to treatment targeting the common causes of chronic cough or who have had these causes excluded by appropriate diagnostic testing should undergo chest CT. Diseases causing cough that may be missed on chest x-ray include tumors, early interstitial lung disease, bronchiectasis, and atypical mycobacterial pulmonary infection. On the other hand, patients with chronic cough who have normal findings on chest examination, lung function testing, oxygenation assessment, and chest CT can be reassured as to the absence of serious pulmonary pathology.
SYMPTOM-BASED TREATMENT OF COUGH
Chronic idiopathic cough, also called cough hypersensitivity syndrome, is distressingly common. It is often experienced as a tickle or sensitivity in the throat, occurs more often in women, and is typically “dry” or at most productive of scant amounts of mucoid sputum. It can be exhausting, interfere with work, and cause social embarrassment. Once serious underlying cardiopulmonary pathology has been excluded, an attempt at cough suppression is appropriate. Most effective are narcotic cough suppressants, such as codeine or hydrocodone, which are thought to act in the “cough center” in the brainstem. The tendency of narcotic cough suppressants to cause drowsiness and constipation and their potential for addictive dependence limit their appeal for long-term use. Dextromethorphan is an over-the-counter, centrally acting cough suppressant with fewer side effects and less efficacy than the narcotic cough suppressants. Dextromethorphan is thought to have a different site of action than narcotic cough suppressants and can be used in combination with them if necessary. Benzonatate is thought to inhibit neural activity of sensory nerves in the cough-reflex pathway. It is generally free of side effects; however, its effectiveness in suppressing cough is variable and unpredictable. Case series have reported benefit from off-label use of gabapentin or amitriptyline for chronic idiopathic cough. Novel cough suppressants without the limitations of currently available agents are greatly needed. Approaches that are being explored include the development of neurokinin receptor antagonists, type 1 vanilloid receptor antagonists, and novel opioid and opioid-like receptor agonists.
HEMOPTYSIS
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree