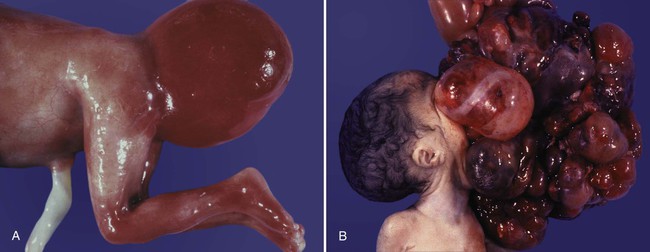
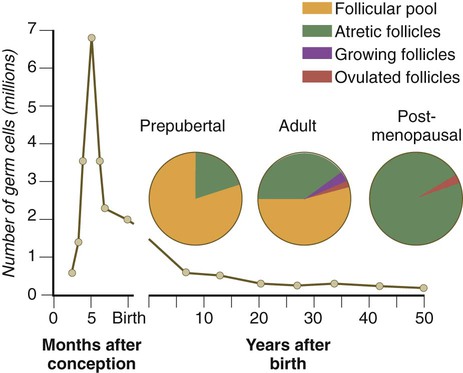
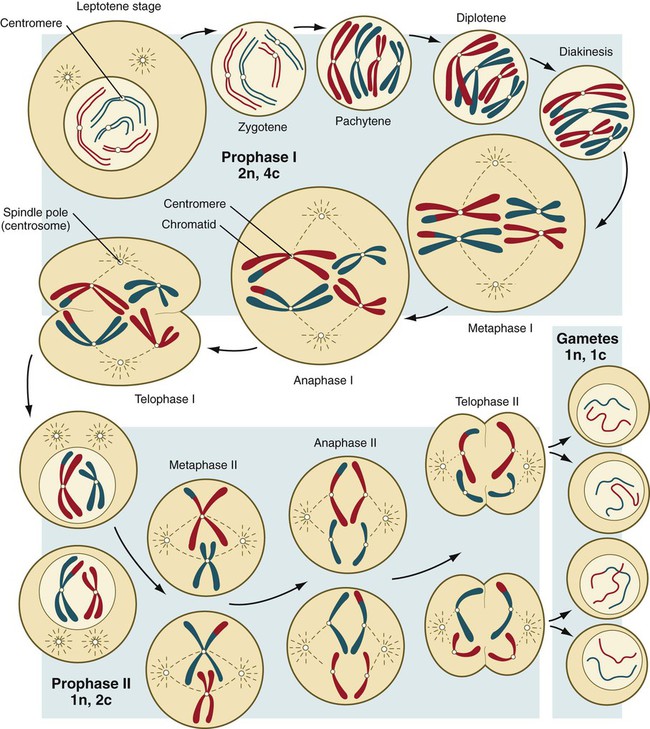
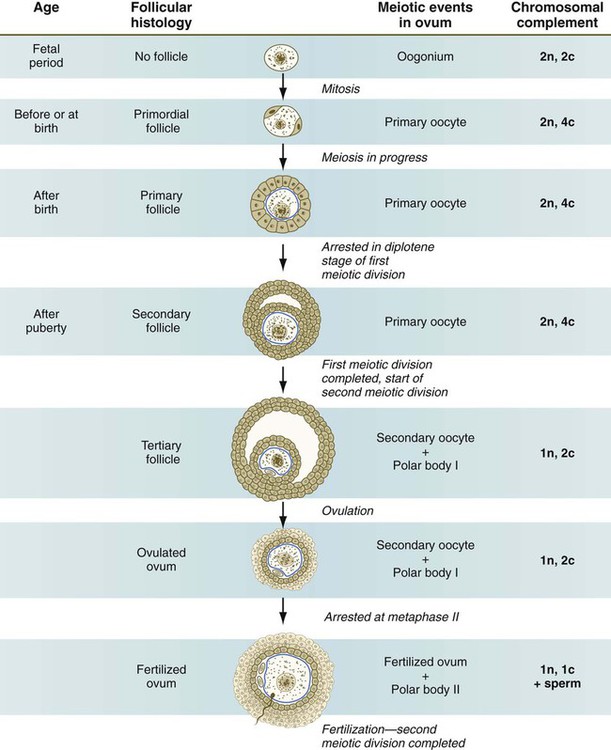
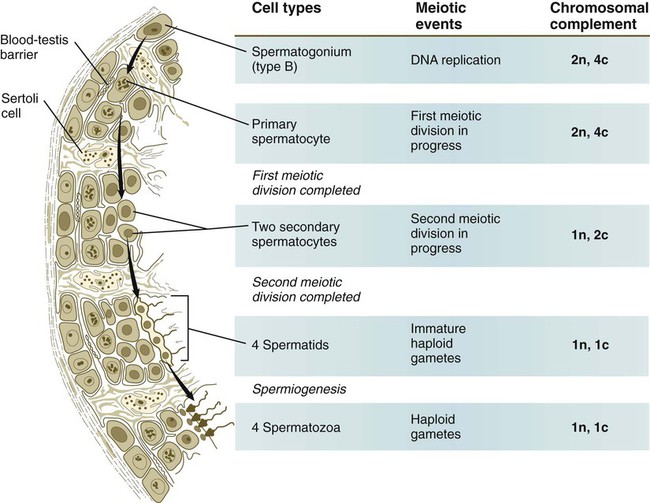
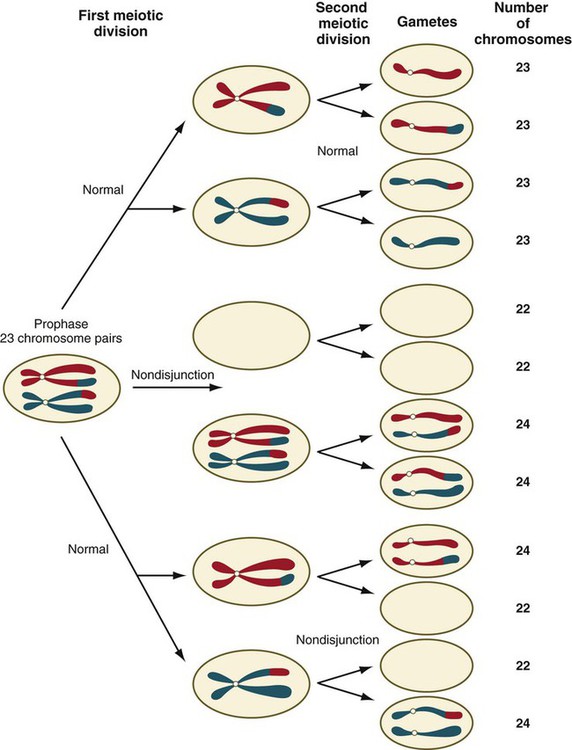
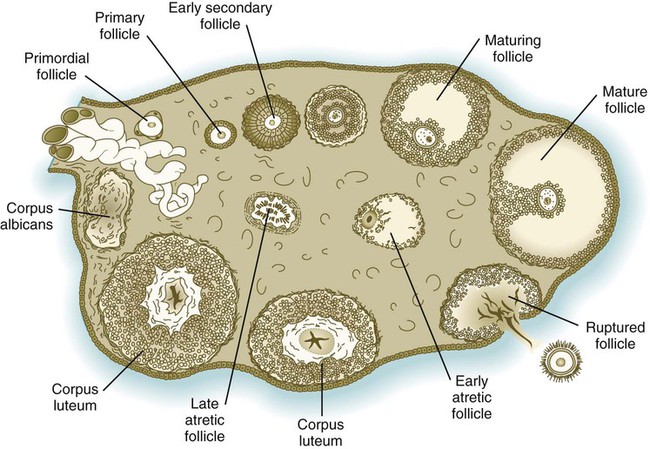
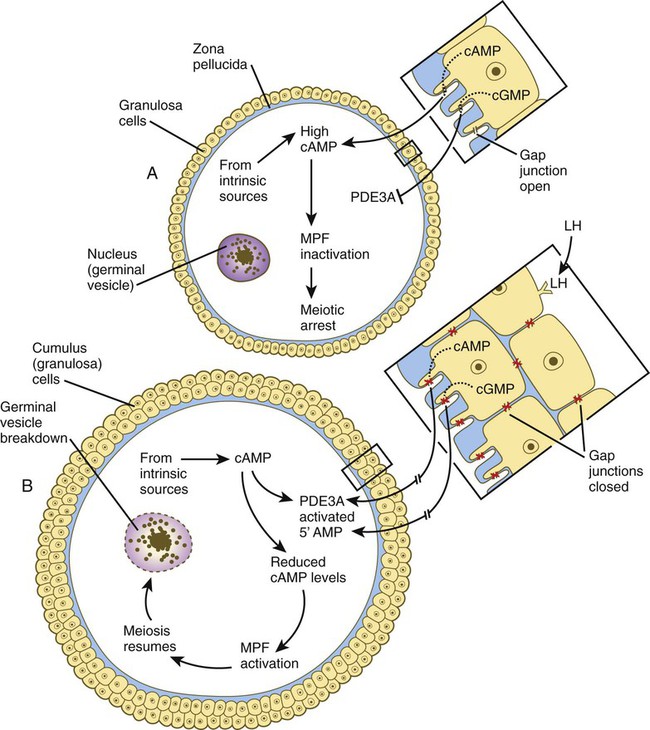
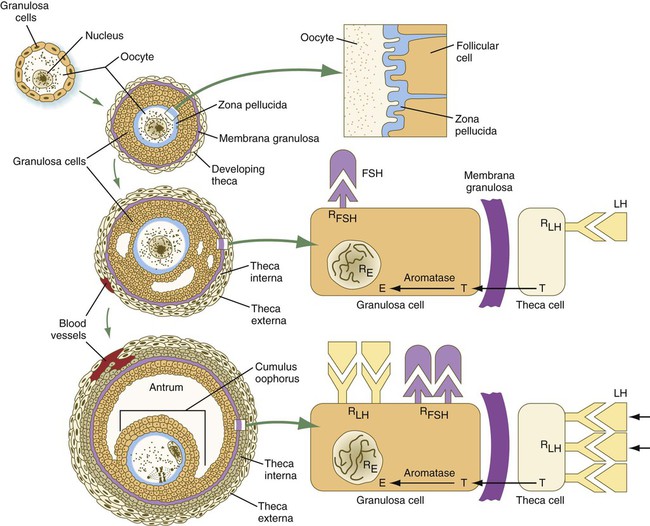
Chapter 1 Primordial germ cells, the earliest recognizable precursors of gametes, arise outside the gonads and migrate into the gonads during early embryonic development. Human primordial germ cells first become readily recognizable at 24 days after fertilization in the endodermal layer of the yolk sac (Fig. 1.1A) by their large size and high content of the enzyme alkaline phosphatase. In the mouse, their origin has been traced even earlier in development (see p. 390). Germ cells exit from the yolk sac into the hindgut epithelium and then migrate* through the dorsal mesentery until they reach the primordia of the gonads (Fig. 1.1B). In the mouse, an estimated 100 cells leave the yolk sac, and through mitotic multiplication (6 to 7 rounds of cell division), about 4000 primordial germ cells enter the primitive gonads. Misdirected primordial germ cells that lodge in extragonadal sites usually die, but if such cells survive, they may develop into teratomas. Teratomas are bizarre growths that contain scrambled mixtures of highly differentiated tissues, such as skin, hair, cartilage, and even teeth (Fig. 1.2). They are found in the mediastinum, the sacrococcygeal region, and the oral region. After they arrive in the gonads, the primordial germ cells begin a phase of rapid mitotic proliferation. In a mitotic division, each germ cell produces two diploid progeny that are genetically equal. Through several series of mitotic divisions, the number of primordial germ cells increases exponentially from hundreds to millions. The pattern of mitotic proliferation differs markedly between male and female germ cells. Oogonia, as mitotically active germ cells in the female are called, go through a period of intense mitotic activity in the embryonic ovary from the second through the fifth month of pregnancy in the human. During this period, the population of germ cells increases from only a few thousand to nearly 7 million (Fig. 1.3). This number represents the maximum number of germ cells that is ever found in the ovaries. Shortly thereafter, numerous oogonia undergo a natural degeneration called atresia. Atresia of germ cells is a continuing feature of the histological landscape of the human ovary until menopause. Meiosis involves two sets of divisions (Fig. 1.4). Before the first meiotic division, deoxyribonucleic acid (DNA) replication has already occurred, so at the beginning of meiosis, the cell is 2n, 4c. (In this designation, n is the species number of chromosomes, and c is the amount of DNA in a single set [n] of chromosomes.) The cell contains the normal number (2n) of chromosomes, but as a result of replication, its DNA content (4c) is double the normal amount (2c). In the first meiotic division, often called the reductional division, a prolonged prophase (see Fig. 1.4) leads to the pairing of homologous chromosomes and frequent crossing-over, resulting in the exchange of segments between members of the paired chromosomes. Crossing-over even occurs in the sex chromosomes. This takes place in a small region of homology between the X and Y chromosomes. Crossing-over is not a purely random process. Rather, it occurs at sites along the chromosomes known as hot spots. Their location is based on configurations of proteins that organize the chromosomes early in meiosis. One such protein is cohesin, which helps to hold sister chromatids together during division. Hypermethylation of histone proteins in the chromatin indicates specific sites where the DNA strands break and are later repaired after crossing-over is completed. Another protein, condensin, is important in compaction of the chromosomes, which is necessary for both mitotic and meiotic divisions to occur. Meiosis in the human female is a very leisurely process. As the primary oocytes enter the diplotene stage of the first meiotic division in the early months after birth, the first of two blocks in the meiotic process occurs (Fig. 1.5). The suspended diplotene phase of meiosis is the period when the primary oocyte prepares for the needs of the embryo. In oocytes of amphibians and other lower vertebrates, which must develop outside the mother’s body and often in a hostile environment, it is highly advantageous for the early stages of development to occur very rapidly so that the stage of independent locomotion and feeding is attained as soon as possible. These conditions necessitate a strategy of storing up the materials needed for early development well in advance of ovulation and fertilization because normal synthetic processes would not be rapid enough to produce the materials required for the rapidly cleaving embryo. In such species, yolk is accumulated, the genes for producing ribosomal ribonucleic acid (rRNA) are amplified, and many types of RNA molecules are synthesized and stored in an inactive form for later use. RNA synthesis in the amphibian oocyte occurs on the lampbrush chromosomes, which are characterized by many prominent loops of spread-out DNA on which messenger RNA (mRNA) molecules are synthesized. The amplified genes for producing rRNA are manifested by the presence of 600 to 1000 nucleoli within the nucleus. Primary oocytes also prepare for fertilization by producing several thousand cortical granules, which are of great importance during the fertilization process (see Chapter 2). Because cortical granules play an important role in preventing the entry of excess spermatozoa during fertilization in human eggs (see p. 31), the formation of cortical granules (mainly from the Golgi apparatus) continues to be one of the functions of the diplotene stage that is preserved in humans. Roughly 4500 cortical granules are produced in the mouse oocyte. A higher number is likely in the human oocyte. With the completion of the first meiotic division shortly before ovulation, two unequal cellular progeny result. One is a large cell, called the secondary oocyte. The other is a small cell called the first polar body (see Fig. 1.5). The secondary oocytes begin the second meiotic division, but again the meiotic process is arrested, this time at metaphase. The stimulus for the release from this meiotic block is fertilization by a spermatozoon. Unfertilized secondary oocytes fail to complete the second meiotic division. The second meiotic division is also unequal; one of the daughter cells is relegated to becoming a second polar body. The first polar body may also divide during the second meiotic division. Formation of both the first and second polar bodies involves highly asymmetric cell divisions. To a large extent, this is accomplished by displacement of the mitotic spindle apparatus toward the periphery of the oocyte through the actions of the cytoskeletal protein actin (see Fig. 2.7). Meiosis in the male does not begin until after puberty. In contrast to the primary oocytes in the female, not all spermatogonia enter meiosis at the same time. Large numbers of spermatogonia remain in the mitotic cycle throughout much of the reproductive lifetime of males. When the progeny of a spermatogonium have entered the meiotic cycle as primary spermatocytes, they spend several weeks passing through the first meiotic division (Fig. 1.6). The result of the first meiotic division is the formation of two secondary spermatocytes, which immediately enter the second meiotic division. About 8 hours later, the second meiotic division is completed, and four haploid (1n, 1c) spermatids remain as progeny of the single primary spermatocyte. The total length of human spermatogenesis is 64 days. Disturbances that can occur during meiosis and result in chromosomal aberrations are discussed in Clinical Correlation 1.1 and Figure 1.7. In the embryo, oogonia are naked, but after meiosis begins, cells from the ovary partially surround the primary oocytes to form primordial follicles (see Fig. 1.5). By birth, the primary oocytes are invested with a complete layer of follicular cells, and the complex of primary oocyte and the follicular (granulosa) cells is called a primary follicle (Fig. 1.8). Both the oocyte and the surrounding follicular cells develop prominent microvilli and gap junctions that connect the two cell types. The meiotic arrest at the diplotene stage of the first meiotic division is the result of a complex set of interactions between the oocyte and its surrounding layer of follicular (granulosa) cells. The principal factor in maintaining meiotic arrest is a high concentration of cyclic adenosine monophosphate (cAMP) in the cytoplasm of the oocyte (Fig. 1.9). This is accomplished by both the intrinsic production of cAMP by the oocyte and the production of cAMP by the follicular cells and its transport into the oocyte through gap junctions connecting the follicular cells to the oocyte. In addition, the follicular cells produce and transport into the oocyte cyclic guanosine monophosphate (cGMP), which inactivates phosphodiesterase 3A (PDE3A), an enzyme that converts cAMP to 5′AMP. The high cAMP within the oocyte inactivates maturation promoting factor (MPF), which at a later time functions to lead the oocyte out of meiotic arrest and to complete the first meiotic division. As the primary follicle takes shape, a prominent, translucent, noncellular membrane called the zona pellucida forms between the primary oocyte and its enveloping follicular cells (Fig. 1.10). The microvillous connections between the oocyte and follicular cells are maintained through the zona pellucida. In rodents, the components of the zona pellucida (four glycoproteins and glycosaminoglycans) are synthesized almost entirely by the egg, but in other mammals, follicular cells also contribute materials to the zona. The zona pellucida contains sperm receptors and other components that are important in fertilization and early postfertilization development. (The functions of these molecules are discussed more fully in Chapter 2.) Early development of the follicle occurs without the significant influence of hormones, but as puberty approaches, continued follicular maturation requires the action of the pituitary gonadotropic hormone follicle-stimulating hormone (FSH) on the granulosa cells, which have by this time developed FSH receptors on their surfaces (see Fig. 1.10). In addition, the oocyte itself exerts a significant influence on follicular growth. After blood-borne FSH is bound to the FSH receptors, the stimulated granulosa cells produce small amounts of estrogens. The most obvious indication of the further development of some of the follicles is the formation of an antrum, a cavity filled with a fluid called liquor folliculi. Initially formed by secretions of the follicular cells, the antral fluid is later formed mostly as a transudate from the capillaries on the outer side of the membrana granulosa. Formation of the antrum divides the follicular cells into two groups. The cells immediately surrounding the oocyte are called cumulus cells, and the cells between the antrum and the membrana granulosa become the mural granulosa cells. Factors secreted by the oocyte confer different properties on the cumulus cells from the mural granulosa cells. In the absence of a direct stimulus from the oocyte, the granulosa cells follow a default pathway and begin to assemble hormone receptors on their surface (see Fig. 1.10). In contrast, the cumulus cells do not express hormone receptors, but under the influence of the oocyte, they undergo changes that facilitate the release of the ovum at the time of ovulation. Enlargement of the follicle results largely from the proliferation of granulosa cells. The direct stimulus for granulosa cell proliferation is a locally produced signaling protein, activin, a member of the transforming growth factor-β family of signaling molecules (see Table 4.1). The local action of activin is enhanced by the actions of FSH. Responding to the stimulus of pituitary hormones, secondary follicles produce significant amounts of steroid hormones. The cells of the theca interna possess receptors for luteinizing hormone (LH), also secreted by the anterior pituitary (see Fig. 1.15). The theca interna cells produce androgens (e.g., testosterone), which pass through the membrana granulosa to the granulosa cells. The influence of FSH induces the granulosa cells to synthesize the enzyme (aromatase), which converts the theca-derived androgens into estrogens (mainly 17β-estradiol). Not only does the estradiol leave the follicle to exert important effects on other parts of the body, but also it stimulates the formation of LH receptors on the granulosa cells. Through this mechanism, the follicular cells are able to respond to the large LH surge that immediately precedes ovulation (see Fig. 1.16). Under multiple hormonal influences, the follicle enlarges rapidly (Fig. 1.11; see Fig. 1.10) and presses against the surface of the ovary. At this point, it is called a tertiary (graafian) follicle. About 10 to 12 hours before ovulation, meiosis resumes. The resumption of meiosis in response to the LH surge is initiated by the cumulus (granulosa) cells, because the oocyte itself does not possess LH receptors. Responding to LH, the cumulus cells shut down their gap junctions (see Fig. 1.9B
Getting Ready for Pregnancy
Gametogenesis
Phase 1: Origin and Migration of Germ Cells
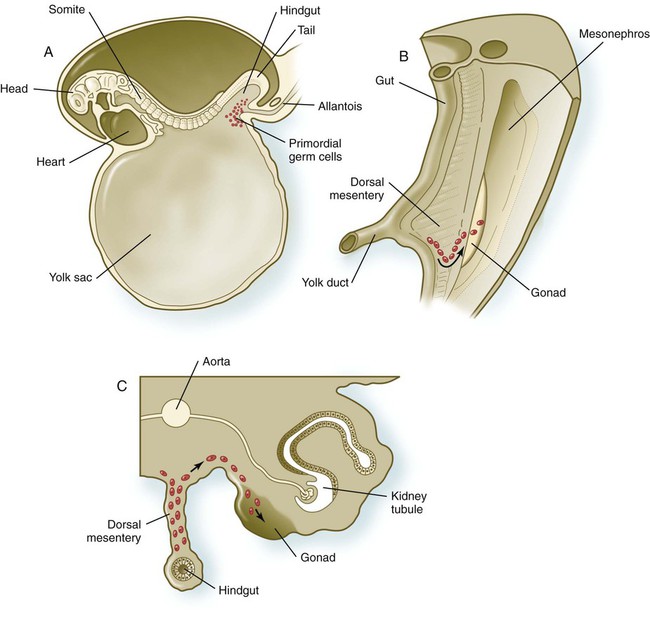
A, Location of primordial germ cells in the 16-somite human embryo (midsagittal view). B, Pathway of migration (arrow) through the dorsal mesentery. C, Cross section showing the pathway of migration (arrows) through the dorsal mesentery and into the gonad.
Phase 2: Increase in the Number of Germ Cells by Mitosis
Phase 3: Reduction in Chromosomal Number by Meiosis
Stages of Meiosis
Meiosis in Females
Meiosis in Males
Phase 4: Final Structural and Functional Maturation of Eggs and Sperm
Oogenesis
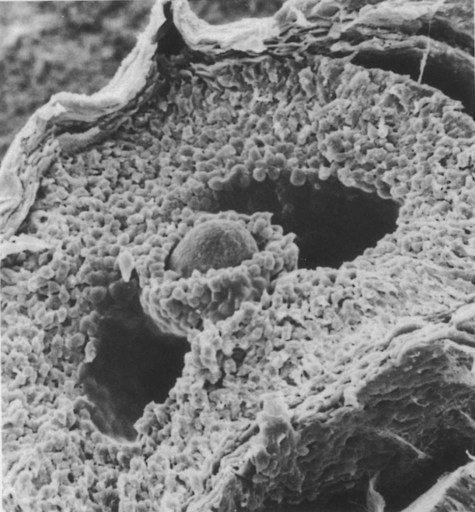
The spherical oocyte (center) is surrounded by smaller cells of the corona radiata, which projects into the antrum. (×840.) (Courtesy of P. Bagavandoss, Ann Arbor, Mich.)
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Getting Ready for Pregnancy