General Concepts, Sex Cord/Gonadal Stromal Tumors
Steven S. Shen, MD, PhD
Jae Y. Ro, MD, PhD
TERMINOLOGY
Definitions
Neoplasms that have features of Leydig (interstitial) cells, Sertoli cells, granulosa cells, or rarely, theca cells
Sex cord stromal tumor (SCST) may be of mixed classifiable sex cord stromal cells (mixed SCST) or unclassifiable sex cord stromal cell (unclassified SCST)
Cases of mixed germ cell and SCST reported (gonadoblastoma and unclassified type)
EPIDEMIOLOGY
Incidence
4-6% of adult testicular neoplasms
30% of testicular tumors in infants and children
Unlike germ cell tumors, there is no racial difference in frequency
ETIOLOGY/PATHOGENESIS
Histogenesis
Poorly understood in general
May relate to disruption of hypothalamic-pituitarytesticular axis and hormonal disturbance
No definitive association with cryptorchidism
Specific types of sex cord stromal tumor may be associated with genetic syndromes; e.g., large cell calcifying Sertoli cell tumor in Peutz-Jeghers syndrome and testicular feminization syndrome for Sertoli cell tumors
Although testicular granulosa cell or theca cell tumors have been reported, no granulosa or theca cells are present in normal testis
CLINICAL IMPLICATIONS
Presentation
Painless mass (rarely painful)
Asymptomatic or hormone-related symptoms
Infants with Leydig cell tumor usually present with isosexual pseudoprecocity
Some types may cause gynecomastia or impotence
Treatment
Surgical approaches
Orchiectomy is curative; staging work-up required
Testis sparing resection possible
Prognosis
Approximately 10% of adult-type SCSTs are malignant and may metastasize
Metastasis is the only reliable criterion for malignancy; histologic factors alone may not predict malignancy
Features that may be associated with disease progression include
Nuclear pleomorphism, frequent and abnormal mitosis (> 4/10 high-power fields), necrosis
Infiltrative growth, large size (> 5 cm), extension to paratesticular tissue, and vascular invasion
Tumors occurring in infants and children are almost always benign with rare exceptions
Clinical Presentation
Painless testicular swelling or mass
Symptomatology of hormonal disturbance or genetic syndrome
MACROSCOPIC FINDINGS
General Features
Size
Range from microscopic to several cm (malignant forms usually larger, > 5 cm)
Specimen Handling
Total Resection
Procure cord margin before cutting into testis
Small tumors may be entirely embedded
Submit at least 1 section/cm tumor
Sections to include: Tumor with adjacent parenchyma
Sections to include: Rete testis, epididymis, and spermatic cord
At least 2 sections of grossly normal parenchyma
Subtotal Resection
Ink resection margin 1st
Take perpendicular sections of tumor with margin
Submit entire tumor, if appropriate
Sections to include: Normal parenchyma
MICROSCOPIC FINDINGS
Normal Histology
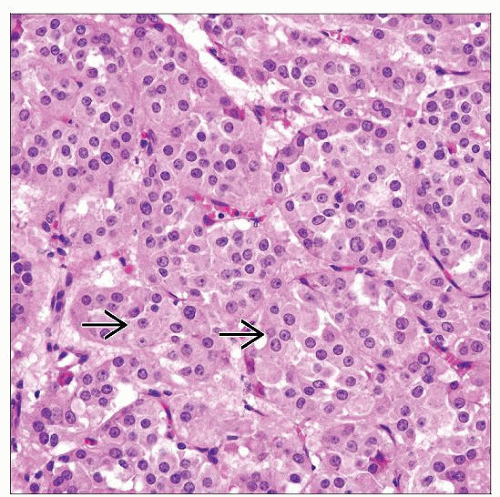
Interstitial (Leydig) cells
Present in interstitium as single cells or in clusters
They may be also present in tunica albuginea, rete testis, epididymis, and spermatic cord (ectopic Leydig cells)
Leydig cells have uniform, round nuclei, prominent nucleoli, and abundant eosinophilic cytoplasm
Intracytoplasmic lipofuscin pigment may be seen, particularly in older men
Reinke crystalloids (better demonstrated with trichrome stains)
Immunoreactive with vimentin, inhibin, and calretinin, but not cytokeratin (may be focally and weakly positive)
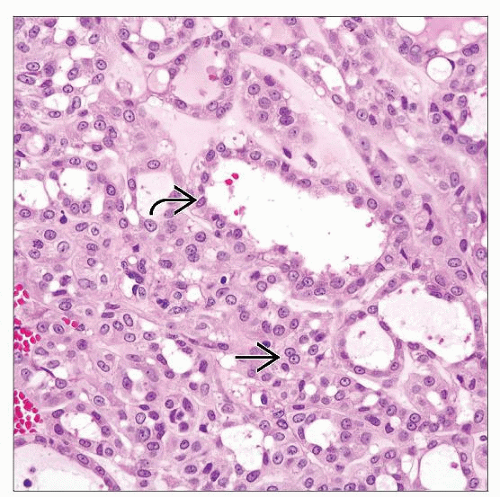
Seminiferous tubules and Sertoli cells
Sertoli cells are located within seminiferous tubules and comprise approximately 10-15% of cells within tubules (germ cell:Sertoli cell ratio is ~ 13:1)
Located 1 or 2 cells away from basement membrane of tubules
Pyramidal shaped-cells with round to ovoid nuclei, finely granular chromatin, often prominent nucleoli
Cytoplasm is eosinophilic and granular with fine vacuoles
Immunoreactive with vimentin, Cam5.2, CK19, inhibin, and calretinin
Granulosa cells
Probably represent precursor Sertoli cells in fetal seminiferous tubules
These cells are cuboidal or columnar in shape and rest on tubular basement membrane
Not found in normal adult testis
Undifferentiated sex cord stromal cells
Present early in fetal gonadal development
Primitive cells with potential for elaboration of steroid hormones
Not found in normal adult testis
General Features
World Health Organization (WHO) Histologic Classification of sex cord stromal tumors of testis
Leydig cell tumor (LCT)
Malignant LCT
Sertoli cell tumor (SCT)
SCT, lipid-rich variant
Sclerosing SCT
Large cell calcifying SCT
Malignant SCT
Granulosa cell tumor (GCT)
Adult type GCT
Juvenile GCT
Tumor of thecoma/fibroma group
Thecoma
Fibroma
Sex cord/gonadal stromal tumor (SCST), mixed/unclassified type
Incompletely differentiated SCST
SCSTs, mixed forms
Malignant SCSTs
Tumors containing both germ cell and sex cord/gonadal stromal elements
Gonadoblastoma
Germ cell sex cord/gonadal stromal tumors, unclassified
Cytologic Features
Depends on tumor types
Predominant Pattern
Solid or nested pattern
Usually seen in LCTs, GCTs, unclassified SCST, fibroma-thecoma
Pseudoglandular, trabecular, or tubular
Usually seen in SCTs, may be seen in LCTs, GCTs
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree