Chapter 5 Gene Therapy and Emerging Molecular Therapies
| Abbreviations | |
|---|---|
| ADA | Adenosine deaminase |
| cDNA | Complementary DNA |
| CF | Cystic fibrosis |
| CFTR | Cystic fibrosis transmembrane conductance regulator |
| DNA | Deoxyribonucleic acid |
| FDA | Food and Drug Administration |
| HIV | Human immunodeficiency virus |
| HS-TK | Herpes simplex virus thymidine kinase |
| mRNA | Messenger RNA |
| RISC | RNA-induced silencing complex |
| RNA | Ribonucleic acid |
| RNAi | RNA interference |
| siRNA | Short interfering RNA |
Advances in understanding molecular mechanisms of disease and manipulating genetic material, protein receptors, and antibodies provide new approaches for treatment of human diseases. Emerging therapeutic reagents include not only novel antibody and protein therapies, but also nucleic acid-based molecules, including complete genes, deoxyribonucleic acid (DNA), complementary DNA (cDNA), ribonucleic acid (RNA), and oligonucleotides. Together with cell therapies and targeted approaches for protein and nucleic acid delivery, these reagents and strategies for administration are conceptualized within the broad borders of gene therapy and the emerging field of molecular medicine. Gene therapy was originally conceived as a treatment for monogenic (Mendelian) disease by complementation of a mutant gene with a normal (wild type) gene. However, gene therapy also includes treatment of acquired human disease by delivery of DNA encoding a therapeutic protein, or introducing a fragment of nucleic acid to interrupt the messenger RNA (mRNA) of a pathogenic protein. A key component of the use of nucleic acids is the spectrum of strategies used to focus therapies on specific organs, deliver genes to specific cells, or both. Targeting is achieved through genetically engineered viruses, receptor-ligand interactions, and antibodies. Therefore these gene-based reagents and the vehicles used to deliver them represent a novel form of “gene as drug.” This chapter presents a discussion of these molecular therapies.
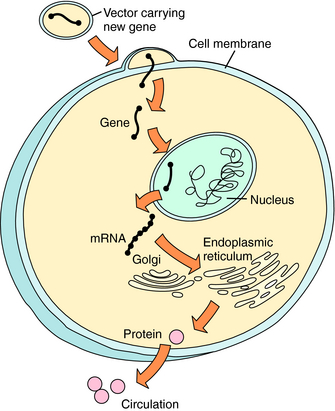
Basic concepts of gene transfer and gene expression are similar, regardless of the vehicle used to carry genetic material to the target cell (Fig. 5-1). After administration of the DNA-vector, the vehicle carrying DNA enters the cell by passing through the cell membrane or by active uptake via a specific receptor. The DNA is taken into the nucleus, where it can be processed. The host cell supplies enzymes necessary for transcription of the DNA into messenger RNA (mRNA) and translation of the mRNA into protein within the cytoplasm. The protein functions intracellularly or extracellularly to replace a hereditary deficient or defective protein, or to provide a therapeutic function. The protein may: (1) function intracellularly, such as adenosine deaminase (ADA) used to correct the mutation in lymphocytes responsible for one form of the severe combined immunodeficiency disease (SCID) syndrome; (2) replace a cell membrane protein, such as the cystic fibrosis (CF) transmembrane conductance regulator chloride channel (CFTR) mutant in CF; or (3) introduce a secreted protein, such as factor VIII, which is deficient in hemophilia. Successful gene transfer (also called transfection) and expression are evaluated by measuring RNA, protein, and, importantly, function in the target cell. In contrast to traditional pharmacological approaches, the goal of this therapy is alteration of the genotype of the cell, rather than alteration of the functional phenotype only.
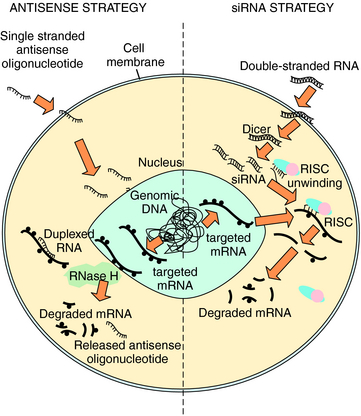
Nucleic acids can also be delivered into the cell to interrupt specific mRNA translation and subsequent protein production. The two general classes of nucleic acids, using independent mechanisms to silence genes through binding and triggering destruction of targeted mRNA, are antisense and RNA interference (RNAi) (Fig. 5-2). Antisense oligonucleotides are 12 to 28 nucleotide, single-strand sequences that are chemically modified to enhance their t1/2. Binding of these oligonucleotides to a complementary mRNA sequence results in cleavage of the targeted mRNA by endogenous RNaseH, an endoribonuclease that specifically recognizes RNA-DNA heteroduplexes. The cleaved mRNA and oligonucleotide are degraded, and protein translation is reduced. However, a high abundance of oligonucleotides is required for efficient antisense silencing. Larger oligonucleotides can also be engineered as a ribozyme to bind and directly cleave mRNA, and then repeat this process without self degradation. More than 50 different antisense oligonucleotides and ribozymes are in clinical trials, primarily directed toward oncogenic genes and infectious virus RNAs.
A recently developed mRNA silencing system is RNAi that takes advantage of endogenous RNA regulatory pathways (see Fig. 5-2). Large, double-stranded RNA sequences designed to target endogenous mRNA enter the cell and are cut into short interfering RNA (siRNA) by the dicer enzyme. Alternatively, synthetic siRNA may be delivered to the cell directly. In either case, the double-strand siRNA molecules are bound by a group of proteins called the RNA-induced silencing complex (RISC). The RISC proteins activate unwinding the siRNA to single-strand RNA that binds a specific mRNA molecule. The RISC cuts the mRNA in the region paired with the antisense siRNA sequence, and the cleaved mRNA is degraded to prevent protein translation. Clinical trials are in development using RNAi.
PRINCIPLES OF CLINICAL GENE THERAPY
Prerequisites for Human Gene Therapy
Application of gene transfer principles to gene therapy requires several critical considerations (Box 5-1). First, a candidate disease for gene therapy must be selected. Typically, this is a disease not successfully treated by currently available therapies. Second, the genetic basis of the disease must be determined by identifying the gene that encodes for a mutant protein or by locating the mutant gene using classical genetic studies. Third, the pathophysiology of the disease must be known so that the cellular site of normal and abnormal gene expression can be ascertained to target the therapy. A corollary is that the magnitude and duration of exogenous gene expression likely to ameliorate the disease should be estimated. Fourth, tools for detection of gene expression must be in hand, including methods for detection of RNA, protein, and protein function. Fifth, preclinical in vitro and in vivo systems for testing efficacy of gene transfer must be developed. This usually mandates that an animal model of disease be available. With few exceptions, these steps are developed in the laboratory before a strategy for clinical gene therapy is considered. Finally, pharmaceutical-grade nucleic acid or virus vector must be produced free of biological and chemical contaminants.