Figure 47-1. Worldwide geographic variability in gastric cancer incidence. (From Ferlay J, Soerjomataram I, Ervik M, et al. GLOBOCAN 2012 v1.0, Cancer Incidence and Mortality Worldwide: IARC CancerBase No. 11 [Internet]. Lyon, France: International Agency for Research on Cancer; 2013. Available from: http://globocan.iarc.fr, accessed on August 1, 2015.)
1 Adenomatous polyps also tend to be asymptomatic, but in contrast to hyperplastic polyps, adenomatous polyps carry a distinct risk for the development of malignancy.10 Mucosal atypia is frequent, and mitotic figures are more common than in hyperplastic polyps. Dysplasia and carcinoma in situ have developed in adenomatous polyps observed over time. The risk for the development of carcinoma has been estimated at 10% to 20% and is greatest for polyps more than 2 cm in diameter. The presence of multiple adenomatous polyps increases the risk of cancer. The presence of an adenomatous polyp is also a marker indicating a diffusely increased risk for the development of cancer in the remainder of the gastric mucosa.
Endoscopic removal is indicated for pedunculated lesions and is sufficient if the polyp is completely removed and shows no evidence of invasive cancer on histologic examination. Operative excision is recommended for sessile lesions larger than 2 cm, for polyps with biopsy-proved invasive carcinoma, and for polyps complicated by pain or bleeding. After removal, repeat endoscopic surveillance is indicated to rule out recurrence at the site of previous excision, evaluate for new or missed polyps or an early carcinoma, and confirm eradication of H. pylori, if applicable.
Gastritis
The incidence of both gastric cancer and atrophic gastritis increases with age. Chronic gastritis is frequently associated with intestinal metaplasia and mucosal dysplasia, and these histologic features are often observed in mucosa adjacent to gastric cancer. Gastritis is frequently progressive and severe in the gastric mucosa of patients with cancer.
Gastric malignancy seems to be increased in patients with chronic gastritis associated with pernicious anemia, although the risk appears to have been overstated in the past. This disease, characterized by fundic mucosal atrophy, loss of parietal and chief cells, hypochlorhydria, and hypergastrinemia, is present in 3% of people older than 60 years. For people in whom pernicious anemia has been active for more than 5 years, the risk of gastric cancer is twice that of age-matched control subjects. Patients with pernicious anemia also have an increased risk of gastric carcinoid development. This increased risk warrants aggressive investigation of new symptoms in patients with long-standing pernicious anemia, but it is not high enough to justify repeated endoscopic surveillance.
Intestinal metaplasia, the presence of intestinal glands within the gastric mucosa, is also commonly associated with both gastritis and gastric cancer. The evolution from metaplasia to dysplasia to carcinoma to invasive cancer has been demonstrated in other organs and in adenocarcinoma arising in the gastroesophageal junction. However, no direct evidence has been provided for this progression in gastric cancer.
Helicobacter Pylori
2 As outlined above, infection with H. pylori has been unequivocally associated with chronic inflammatory conditions in the stomach, and this association has stimulated interest in the role of chronic infection by this organism in gastric carcinogenesis. Childhood acquisition of H. pylori infection is frequent in areas of high gastric cancer incidence, and high rates of infection have been identified in patients with premalignant lesions and invasive cancer. Infection with H. pylori is associated with an increased risk of adenocarcinoma of both major histologic types (intestinal and diffuse) and of both the body and the antrum of the stomach.11 In contrast, H. pylori infection is not a risk factor for cancers of the gastroesophageal junction, which are frequently associated with mucosal abnormalities of Barrett esophagus and which seem to follow the metaplasia to dysplasia to carcinoma pattern of development.
The mechanism of carcinogenesis related to H. pylori infection is incompletely understood but is thought to be related to the chronic inflammation caused by the organism.12 However, only ∼1% of patients chronically infected with H. pylori will develop the gastric cancer phenotype, which consists of corpus-predominant gastritis, multifocal atrophic gastritis, high gastrin levels, hypo/achlorhydria, and low pepsinogen I/II ratio. The majority of subjects infected with H. pylori will develop the simple gastritis phenotype which is not associated with any significant clinical outcome, or the duodenal ulcer phenotype (10% to 15% of infected subjects) which consists of antral-predominant gastritis and high gastrin and acid secretion and actually provides protection from developing gastric cancer.13 Of note, there is variable distribution of these three phenotypes geographically, with particular prevalence of the gastric cancer phenotype in certain parts of Asia where gastric cancer is common.14 Bacterial virulence factors, environmental exposures, and host genetic factors also clearly play an important role in the pathogenesis of gastric carcinogenesis following infection-related gastritis.12,13,15 Eradication of H. pylori may not prevent the development of gastric cancer once premalignant lesions have already developed.16
Previous Gastric Surgery
A number of uncontrolled reports have suggested that gastric cancer is more likely to develop in people who have undergone previous partial gastrectomy, with patients who have undergone a gastrojejunal (Billroth II) anastomosis at apparently higher risk for carcinogenesis than those reconstructed with a gastroduodenal anastomosis (Billroth I).17,18 The so-called gastric remnant cancer is a true clinical entity, although the risk for development of this gastric neoplasm appears to have been overestimated. Several large, prospective studies with long-term follow-up indicate that the relative risk is not increased for up to 15 years after gastric resection, likely due to surgical removal of mucosa at risk for development of gastric cancer, followed by modest increases in cancer risk (three times the control value) observed only after 25 years.19–22
The cellular mechanisms that contribute to the development of neoplasia in the remnant stomach are unknown. Decreased luminal pH, bacterial overgrowth with increased production of N-nitroso carcinogens, and reflux of bile acids into the stomach have been postulated to promote cancer development, but remains unproved. Vagotomy, often performed in conjunction with gastric surgery for benign disease, does not appear to promote cancer development. A population-based study from Sweden of 7,198 vagotomized patients followed for up to 18 years did not report increased risk.20
A recent study explored genetic alterations in gastric remnant cancer and found that the microsatellite instability high (MSI-H) phenotype was much more common (43%) in gastric remnant cancers than in sporadic gastric cancers (6%), and that this incidence was much higher in patients who had undergone a Billroth II anastomosis (67%) as compared to those who had undergone a Billroth I anastomosis (11%). The MSI-H phenotype in these tumors was associated with inactivation of the DNA mismatch repair genes hMLH1 and hMSH2. The significance of this relationship is not yet clear.23 Reported 5-year survival ranges from only 7% to 33% for gastric remnant cancers, but this poor prognosis is most likely due to the fact that these cancers are usually diagnosed at an advanced stage when treatment options are limited.
Hereditary Syndromes
Approximately 5% to 10% of gastric cancer may have a familial component, and 3% to 5% are associated with known inherited cancer syndromes. Hereditary diffuse gastric cancer (HDGC) is an autosomal dominant syndrome that confers a lifetime risk for the development of gastric cancer by age 80 years of 67% for men and 83% for women.24 The average age at diagnosis is 37 years, and cancers tend to be of the diffuse type. Germline mutations in CDH1, a tumor suppressor gene that encodes the cell-to-cell adhesion protein E-cadherin, are found in 25% of patients with HDGC.25 Patients with documented CDH1 mutations and a family history of gastric cancer may be offered prophylactic gastrectomy at a young age. Other familial cancer syndromes associated with an increased risk of gastric cancer include Lynch syndrome, juvenile polyposis syndrome, and Peutz–Jeghers syndrome. Surveillance upper endoscopy may be considered in patients with these syndromes.
Clinical Features
3 The symptoms produced by gastric cancer are nonspecific and can closely mimic those associated with a number of nonneoplastic gastroduodenal diseases, especially benign gastric ulcer (Fig. 47-2). In early gastric cancers, epigastric pain is present in over 70% of patients.26 The pain is often constant, nonradiating, and unrelieved by food ingestion. In a surprising number of patients, pain can be relieved, at least temporarily, by antacids or gastric antisecretory drugs. Anorexia, nausea, and weight loss are present in less than 50% of patients with early gastric cancers but become increasingly common with disease progression. Dysphagia is present in 20% of patients with proximal gastric lesions. Overt gastrointestinal hemorrhage is present in only 5%. Perforation is uncommon (1%).

Figure 47-2. Clinical symptom frequency in benign gastric ulcer, early gastric cancer, and advanced gastric cancer. (After Meyer WC, Damiano RJ, Postlethwait RW, et al. Adenocarcinoma of the stomach: changing patterns over the past four decades. Ann Surg 1987;205:18.)
In most patients with early gastric cancers, physical examination is unremarkable. Stool tests positive for occult blood in one-third. Abnormal physical findings usually reflect advanced disease (Table 47-1). Cachexia, abdominal mass, hepatomegaly, and supraclavicular adenopathy usually indicate metastasic disease.27 There are no simple laboratory tests specific for gastric neoplasms.
Diagnosis and Staging
Fiberoptic endoscopy is the most definitive diagnostic method when gastric neoplasm is suspected. In the initial stages, gastric cancers can appear polypoid, as flat, plaquelike lesions, or as shallow ulcers. Advanced lesions are typically ulcerated. The ulcer border can have an irregular, beaded appearance because of infiltrating cancer cells, and the base is frequently necrotic and shaggy. The ulcer can appear to arise from an underlying mass. Although each of these features suggests a malignant ulcer, differentiation of benign from malignant gastric ulcers can be made definitively only with gastric biopsy. Multiple biopsies of any gastric ulcer should be performed. The sensitivity of a single biopsy for diagnosing a gastric cancer is 70%, but performing multiple biopsies can increase the sensitivity to greater than 98%.28 False-negative results occur in approximately 10% of patients, usually as the result of sampling error or due to the absence of a mucosal abnormality as can occur with linitis plastica; false-positive results are rare. Diagnostic accuracy can be further enhanced by the addition of endoscopic ultrasound (EUS) with fine-needle aspiration biopsy for infiltrative tumors involving the wall of the stomach without obvious mucosal abnormalities.
DIAGNOSIS
Table 47-1 Common Symptoms and Physical Findings in Gastric Cancer


Figure 47-3. Early cancer survival rate in Japan.
Annual mass screening programs have been instituted in some countries (e.g., Japan, Venezuela, Chile) with high incidence of gastric cancer. Whether such programs significantly reduce gastric cancer mortality is unclear. In Japan, compliance with screening has been associated with a 50% decrease in gastric cancer mortality, but most of this benefit is attributable to confounding factors such as baseline general health.29 A large cohort study failed to show any effect of screening on mortality.30 Cancers detected in screened patients tend to be earlier cancers with fewer nodal metastases,31 and patients with resected gastric cancer diagnosed by screening have better survival than those diagnosed after development of symptoms (Fig. 47-3). However, a survival difference between screened and unscreened gastric cancer patients persists even after accounting for stage,31 suggesting that patient selection confounds the effect of screening on mortality. The Japanese findings that early detection and identification of early gastric cancer can improve survival has been confirmed by European investigations, in which patients with early gastric cancers have been shown to have survival rates equivalent to those of patients with benign gastric ulcer (Fig. 47-4).26 Mass screening programs have been found to be cost-effective in high-incidence countries such as Japan and China,32 but they are unlikely to be cost-effective in lower-incidence countries such as the United States. The cost-effectiveness of the Japanese screening program is likely to change given the significant decrease in the rate of chronic H. pylori infection in Japanese under the age of 30 (25% vs. 60% as compared to their parents).33

Figure 47-4. Early cancer survival rate in Europe.
Barium-contrast radiographs have, in the past, been the standard method for diagnosing gastric neoplasm. Single-contrast examinations have a diagnostic accuracy of 80%. This diagnostic yield increases to approximately 90% when double-contrast (air and barium) techniques are used. Typical findings include ulceration, the presence of a gastric mass, loss of mucosal detail, and distortion of the gastric silhouette (Fig. 47-5). Contrast radiography has been largely supplanted by endoscopy because of the ability to obtain biopsy material by the latter technique.

Figure 47-5. Barium-contrast radiograph demonstrating extensive involvement of the gastric body by infiltrating adenocarcinoma (linitis plastica). The gastric silhouette is narrowed (arrows), and the stomach is nondistensible.
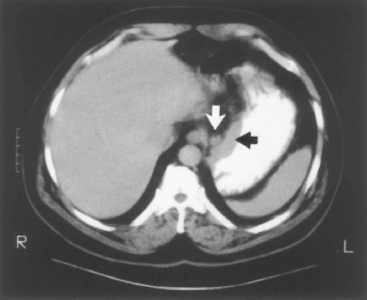
Figure 47-6. Computed tomography scan demonstrating mass along lesser curvature of the stomach (black arrow) and associated lymph node enlargement (white arrow).
Computed tomography (CT) can provide information both about the primary tumor and visceral metastatic disease. Because it is noninvasive and widely available, it is often the first staging modality that is employed in a patient diagnosed with gastric cancer. Patients who are found to have metastatic disease can be spared further, potentially invasive staging studies. When performed with intraluminal and intravenous contrast, CT can demonstrate infiltration of the gastric wall by tumor, gastric ulceration, and the presence of hepatic metastases (Figs. 47-6 and 47-7). CT may overestimate depth of invasion, but serosal involvement can be reliably assessed (sensitivity 83% to 100%, specificity 80% to 97%).34 The technique is less reliable for detection of small peritoneal metastases, which may be missed in 30% of cases.35 Similarly, evaluation of nodal disease by CT is limited, with accuracy of 70% to 80% even with modern CT techniques.36
EUS is another useful method of preoperative evaluation for local staging and diagnosis. EUS can assess subepithelial lesions that may be confused with gastric cancer and guide biopsy of submucosal tumors within the wall of the stomach. Investigation of submucosal masses, infiltrative gastric disorders, and enlarged gastric epithelial folds, as well as differentiation of gastric lymphoma from gastric adenocarcinoma are all aided by EUS. This technique has the ability to assess the depth and pattern of gastric wall penetration by the tumor as well as relationship to adjacent structures, and has good correlation with intraoperative assessment and histologic findings. Perigastric lymph nodes involved with tumor are also reliably identified by EUS, and therefore EUS provides the most accurate assessment of local stage of disease (TN status), with an accuracy of 65% to 90% for staging depth of tumor invasion34 and 50% to 78% for nodal involvement.37 EUS is generally not useful for detecting metastatic disease, but it can help identify patients at risk for radiographically occult metastatic disease (e.g., peritoneal metastases) for staging laparoscopy.35 Therefore, EUS serves as a useful adjunct to cross-sectional imaging and can help guide selection of patients for further staging studies or multimodality therapy.
Metabolic imaging with positive emission tomography (PET) using 18F-fluorodeoxyglucose has been found to be less accurate than cross-sectional imaging and EUS for staging locoregional involvement, but more sensitive for detecting distant metastases in patients with gastric cancer.37 A meta-analysis comparing PET, ultrasound, CT, and magnetic resonance imaging (MRI) found that PET scan was the most sensitive imaging modality for detecting hepatic metastases.38 A separate study found that tumors which responded metabolically on PET to neoadjuvant chemotherapy correlated highly with histopathologic response and improved patient survival.39 Therefore, current recommendations regarding the use of PET for staging gastric cancer are for selective use for patients with locally advanced tumors where the metastatic potential is high, and in cases where neoadjuvant therapy is being considered.37 In these patients, the addition of PET can result in net cost savings by reducing the number of futile surgical procedures.40

Figure 47-7. Computed tomography scans of the upper abdomen showing extensive thickening of the gastric wall (black arrows) caused by infiltrating adenocarcinoma and associated hepatic metastasis (white arrow).
Staging Laparoscopy
The peritoneal lining, omentum, and liver capsule are common sites for gastric cancer metastasis that are difficult to evaluate preoperatively by CT scanning. In prospective studies, diagnostic laparoscopy has been superior to preoperative CT or percutaneous ultrasound in detection of peritoneal, hepatic, or lymphatic metastasis.41 Accurate identification of patients with metastatic disease is important in order to spare them futile, ultimately noncurative surgical procedures. In up to 25% of patients, laparoscopy will detect metastatic disease that precludes curative resection.42–44 Relative to laparotomy, the shorter hospitalization and reduced operative trauma following laparoscopy may both hasten recovery and facilitate earlier initiation of systemic chemotherapy. Most patients with systemic metastasis can be treated without the need for palliative surgical resection.45
In addition to grossly evident intra-abdominal metastatic disease, patients with microscopic metastatic disease are at high risk for early recurrence and death after attempted curative resection.46,47 Based on this finding, patients with positive cytology in peritoneal washings are considered to have M1 disease. Approximately one-fourth of patients subjected to staging laparoscopy prior to planned curative resection of gastric cancer will have positive peritoneal cytology; one-third of these patients will not have grossly apparent metastatic disease. Patients who clear their initially positive peritoneal cytology after systemic chemotherapy have an improved prognosis, but cure remains highly unlikely.46
Diagnostic laparoscopy may be considered in patients being considered for surgical resection without neoadjuvant therapy. In these situations, the procedure can be conducted at the beginning of the planned resection so as to avoid an additional general anesthesia. Patients with locally advanced (T3–T4 or node-positive) tumors who would typically be selected for neoadjuvant therapy should be considered for diagnostic laparoscopy with peritoneal washings prior to initiation of chemotherapy. The finding of positive peritoneal cytology should prompt adoption of a noncurative paradigm of treatment in most cases.
Pathology
Gastric adenocarcinoma occurs in two distinct histologic subtypes—intestinal and diffuse. These subtypes are characterized by differing pathologic and clinical features and by differing patterns of metastatic spread.
In the intestinal form of gastric cancer, the malignant cells tend to form glands. The intestinal form of malignancy is more frequently associated with gastric mucosal atrophy, chronic atrophic gastritis, intestinal metaplasia, and dysplasia. Gastric cancer with the intestinal histologic subtype is more common in populations at high risk (e.g., Japan), and it occurs with increased frequency in men and older patients. Clinical studies suggest that this subtype more frequently demonstrates bloodborne metastases.
The diffuse form of gastric adenocarcinoma does not demonstrate gland formation and tends to infiltrate tissues as a sheet of loosely adherent cells. Lymphatic invasion is common. Intraperitoneal metastases are frequent. The diffuse form of gastric adenocarcinoma tends to occur in younger patients, in women, and in populations with a relatively low incidence of gastric cancer (e.g., the United States). The prognosis is poorer for patients with the diffuse histologic subtype.
Sporadic gastric adenocarcinomas demonstrate a number of chromosomal and genetic abnormalities. Cytometric analysis reveals that gastric tumors with a large fraction of aneuploid cells (with a greater-than-normal amount of nuclear DNA) tend to be more highly infiltrative and have a poorer prognosis. Amplifications of both the HER-2/neu and K-ras proto-oncogenes have been consistently detected in gastric adenocarcinomas, and in a small percentage of tumors a lack of expression of the tumor suppressor gene MKK4 is robustly associated with poor survival.48 The exact mechanisms by which these genetic abnormalities contribute to gastric oncogenesis are currently unclear. Additionally, a number of growth factors, including epidermal growth factor, platelet-derived growth factor, and transforming growth factor-β, are overexpressed in gastric carcinoma cells.49
In the United States, the incidence of proximal gastric cancers has been increasing; such that in 2001 the rate of proximal cancers, defined as cancers arising in the cardia and fundus, exceeded that of distal cancers, defined as cancers arising in the antrum and pylorus. Proximal cancers are more likely to occur in young white men and distal cancers are more likely to occur in Asian, African American, and Hispanic patients within the United States. The proportion of tumors involving the proximal stomach has dramatically increased over the past decades; in the 1960s, only 16% involved this region, and a clear explanation for this rise in proximal disease remains elusive. In 10% of cases, the stomach is diffusely involved at the time of diagnosis.27 Prognosis is poorer for tumors arising from the proximal stomach or for those with diffuse involvement of the organ relative to distal tumors, and these patients are much more likely to need neoadjuvant and adjuvant therapy.9,50

Figure 47-8. Gastric cancer survival by stage. Used with the permission of the American Joint Committee on Cancer (AJCC), Chicago, Illinois. The original source for this material is the AJCC Cancer Staging Manual, Seventh Edition (2010) published by Springer Science and Business Media LLC, www.springer.com
CLASSIFICATION AND STAGING
Table 47-2 Seventh Edition AJCC Staging System for Gastric Cancer

The seventh edition American Joint Committee on Cancer (AJCC) staging system for gastric cancer is presented in Table 47-2.51 The staging system accurately discriminates prognosis based on pathologic factors for tumors located 5 cm distal to the esophagogastric junction (EGJ) and below or arising within 5 cm of, but not crossing, the EGJ (Fig. 47-8). The AJCC recommends that cancers arising within 5 cm of the EGJ that cross into the EGJ or esophagus be staged and treated as esophageal cancers. A consideration of staging data illustrates the high frequency with which lymph node metastases are present at the time of diagnosis in the United States, and the severe impact lymphatic involvement has on survival. Even early gastric cancers (tumors restricted to the mucosa and submucosa) have a 15% prevalence of nodal metastasis.
Curative-Intent Treatment
Surgical resection is the only potentially curative therapy for gastric cancer, but an advanced stage of disease at the time of diagnosis precludes curative resection for most patients.
Since the mid-1990s, the surgical treatment of gastric cancer has continued to evolve, with minimally invasive approaches increasingly pursued for early cancers and increasingly radical operations advocated by some for advanced tumors. Japanese surgeons have reported the largest experience with early gastric cancer. The Japanese Gastric Cancer Association defines early gastric cancer as tumor in which invasion is restricted to the mucosa or submucosa.52 The presence or absence of lymph node metastasis is not considered in this classification. While the presence of lymphatic metastasis cannot be correctly judged by endoscopic findings, it is critically important in prognosis. For tumors confined to the mucosa, lymphatic metastasis is present in 1-3% of cases; with submucosal involvement, the rate of nodal positivity increases to 14-20%.53,54
Endoscopic Resection
For intestinal-type mucosal tumors less than 2 cm in size without ulceration or evidence of lymphovascular invasion, endoscopic mucosal resection (EMR) may be performed. With this approach, postoperative bleeding or perforation has been reported in 5%, and in 17% histologic examination revealed submucosal invasion that required further operative treatment.55 Earlier reports suggested underestimation of tumor invasion in 45% and missed lymphatic metastasis in 9% urged caution before widespread acceptance of this technique.56 However, in experienced centers good results can be obtained. A Japanese series of 131 patients reported disease-free survival of 99% at 10 years.57 Endoscopic submucosal dissection (ESD) is an emerging technique that may allow larger tumors to be endoscopically resected than with EMR. In the absence of randomized trials comparing EMR and ESD against surgical resection, surgical resection remains the gold standard for potentially curative therapy in appropriate-risk patients.
Surgical Resection
4The fundamental principle of surgical resection of gastric cancer is complete extirpation of the primary tumor. The extent of gastric resection is determined primarily by the need to obtain a resection margin free of microscopic disease (R0 resection). Microscopic involvement of the resection margin by tumor cells (R1 resection) is associated with poor prognosis.27 Patients with positive surgical margins are at high risk for development of recurrent disease, and histologically positive margins are strongly correlated with the development of anastomotic recurrence. In the setting of ≥5 positive nodes, however, margin positivity does not impact survival,58 because these patients are at higher risk for systemic recurrence. In contrast to other gastrointestinal malignancies such as colon cancer, gastric cancer frequently demonstrates extensive intramural spread, especially the diffuse type. The propensity for intramural spread is related, in part, to the extensive anastomosing capillary and lymphatic network in the wall of the stomach. Retrospective studies suggest that when performing a subtotal gastrectomy, a margin of 6 cm from the tumor mass proximally and 3 to 5.9 cm distally is necessary to minimize anastomotic recurrence.59 Frozen section evaluation of resection margins may be obtained prior to proceeding with reconstruction in order to improve the probability that R0 resection can be achieved.

Figure 47-9. Surgical options for resection of gastric neoplasms. A: Subtotal gastrectomy with gastrojejunal reconstruction. B: Total gastrectomy with esophagojejunostomy. C: Esophagogastrectomy with anastomosis in cervical or thoracic position.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


