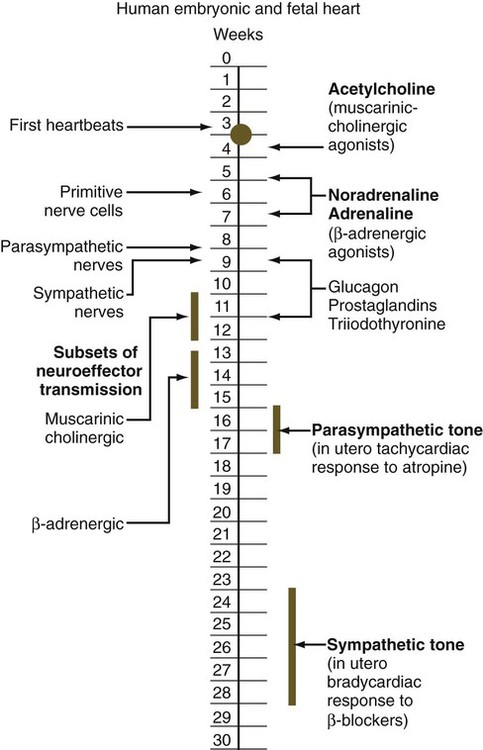
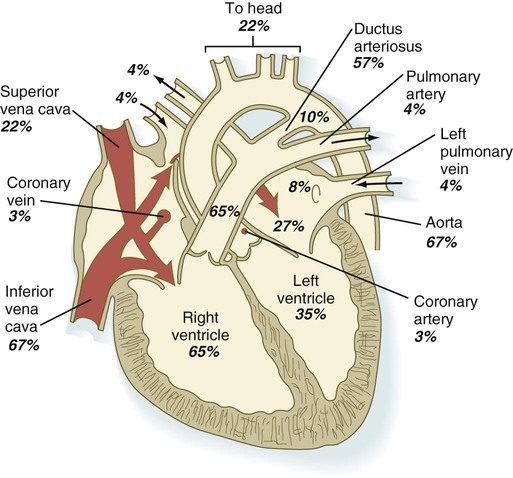
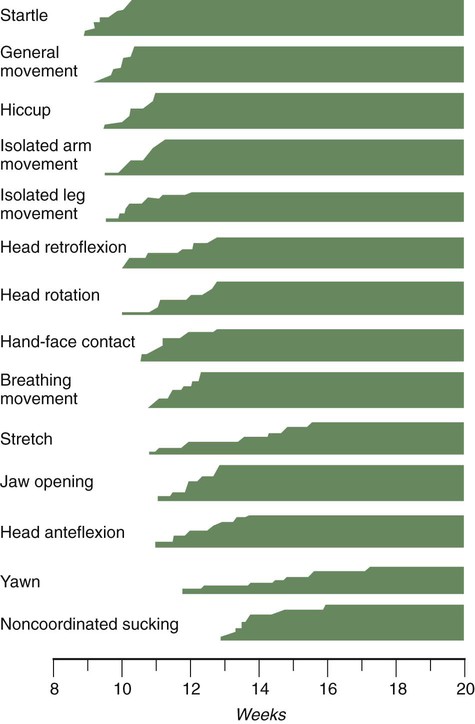
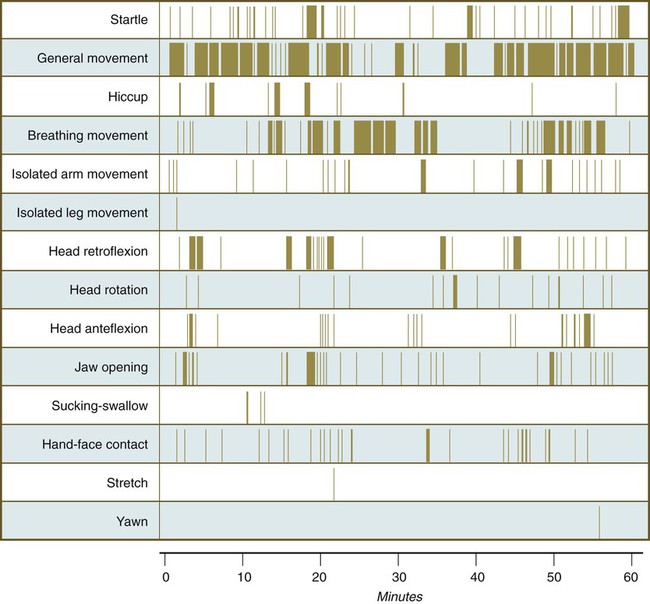
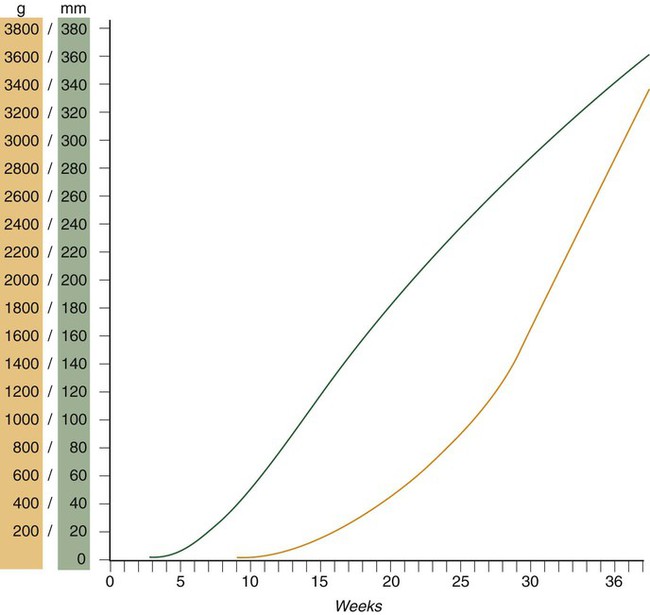
Chapter 18 After the eighth week of pregnancy, the period of organogenesis (embryonic period) is largely completed, and the fetal period begins. By the end of the embryonic period, almost all the organs are present in a grossly recognizable form. The external contours of the embryo show a very large head in proportion to the rest of the body and greater development of the cranial than of the caudal part of the body (Figs. 18.1 and 18.2). This chapter emphasizes the functional development of the fetus and the adaptations that ensure a smooth transition to independent living after the fetus has passed through the birth canal and the umbilical cord is cut. Techniques that are used to monitor the functional state of the fetus are also described in Clinical Correlation 18.1, later in the chapter. Despite the intense developmental activity that occurs during the embryonic period (3 to 8 weeks), the absolute growth of the embryo in length and mass is not great (Fig. 18.3). The fetal period (9 weeks to birth), however, is characterized by rapid growth. The change in proportions of the various regions of the body during the prenatal and postnatal growth periods is as striking as the absolute growth of the embryo. The early dominance of the head is reduced as development of the trunk becomes a major factor in the growth of the early fetus. Even later, a relatively greater growth of the limbs changes the proportions of various regions of the body. During the early fetal period, the entire body is hairless and very thin because of the absence of subcutaneous fat (Fig. 18.4). By midpregnancy, the contours of the head and face approach those of the neonate, and the abdomen begins to fill out. Beginning at around week 27, the deposition of subcutaneous fat causes the body to round out. (Some major developmental landmarks during the fetal period are summarized in the table on pp. xii and xiii.) The circulation of the human embryo can be first studied at about 5 weeks by means of ultrasound. At that time, the heart beats at a rate of approximately 100 beats/minute. This probably represents an inherent atrial rhythm. The pulse rate increases to about 160 beats/minute by 8 weeks and then decreases to 150 beats/minute by 15 weeks, with a further slight decline near term. The pulse rate in utero is remarkably constant, and embryos exhibiting bradycardia (slow pulse rate) often die before term. Near term, the pulse rate varies to some extent if conditions in the uterus change or if the embryo is stressed. This variation is related to the functional establishment of the autonomic innervation of the heart (Fig. 18.5). Quantitative studies have shown a good correlation between blood flow and functional needs of various regions of the embryo. Approximately 40% of the combined cardiac output goes to the head and upper body and supplies the relatively great needs of the developing brain. Another 30% of the combined cardiac output goes to the placenta via the umbilical arteries for replenishment. Figure 18.6 shows the relative amounts of blood that enter and leave the heart via various vascular channels. (The general qualitative pattern of blood flow in a human fetus is presented in Fig. 17.30.) Ultrasonography has revolutionized the analysis of fetal movements and behavior because the fetus can be examined virtually undisturbed (except for an increase in vascular activity induced by the ultrasound) for extended periods. Earlier studies of fetal movements were principally concerned with the development of reflex responses, and the information was obtained largely by the analysis of newly aborted fetuses (see Chapter 11). Although valuable information on maturation of reflex arcs was obtained in this manner, many of the movements elicited were not those normally made by the fetus in utero. The undisturbed embryo does not show any indication of movement until about Continuous ultrasound monitoring for extended periods reveals patterns involving many types of movements (Fig. 18.8). At different weeks of pregnancy, some movements are predominant, whereas others are in decline or are just beginning to take shape. Analysis of anencephalic fetuses has shown that although many movements occur, they are poorly regulated. These movements start abruptly, are maintained at the same force, and then stop abruptly. These abnormal patterns of movements are considered evidence for strong supraspinal modulation of movement in the fetus. Individual epithelial cell types, including Brunner’s glands, which protect the duodenal lining from gastric acid, appear in the small intestine early in the second trimester. Although the presence of most enzymes or proenzymes characteristic of the intestinal lining can be shown histochemically during the midfetal period, the amounts of these substances are generally quite small. Activity of some of the enzymes secreted by the exocrine pancreatic tissue can also be shown between 16 and 22 weeks’ gestation. Meconium, a greenish mixture of desquamated intestinal cells, swallowed lanugo hair, and various secretions, begins to fill the lower ileum and colon late in the fourth month (Fig. 18.9).
Fetal Period and Birth
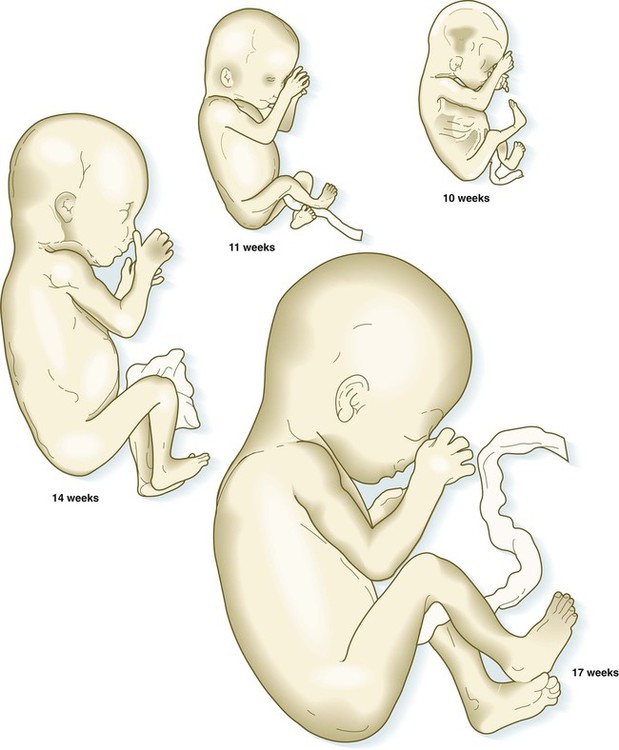
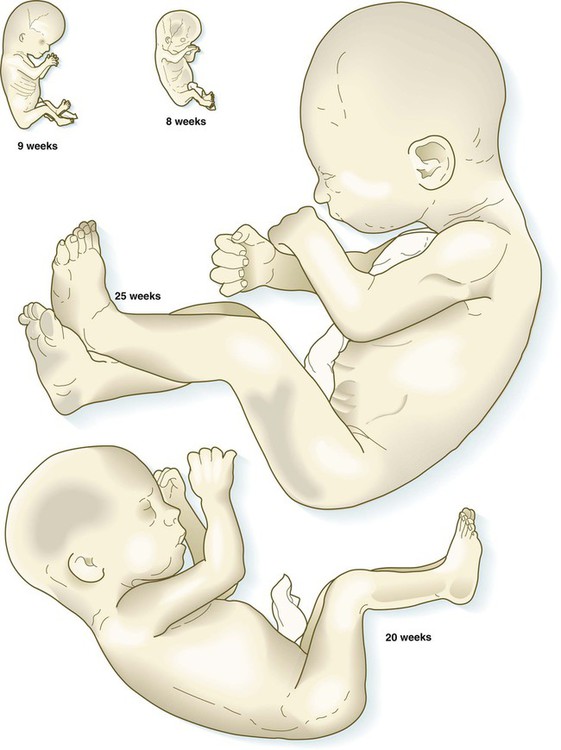
The fetuses from 8 through 17 weeks are drawn actual size. The fetuses from 20 and 25 weeks are drawn two-thirds actual size.
Growth and Form of the Fetus
Fetal Physiology
Circulation
Fetal Movements and Sensations
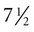 weeks. The first spontaneous movements consist of slow flexion and extension of the vertebral column, with the limbs being passively displaced. Within a short time, a large repertoire of fetal movements evolves. After study by numerous investigators, a classification of fetal movements has been suggested (Box 18.1). The first fetal movements are followed in a few days by startle and general movements. Shortly thereafter, isolated limb movements are added (Fig. 18.7). Movements associated with the head and jaw appear later. Toward the end of the fourth month, the fetus begins a pattern of periods of activity, followed by times of inactivity. Many women first become aware of fetal movements at this time. Between the fourth and fifth months, the fetus becomes capable of gripping firmly onto a glass rod. Although weak protorespiratory movements are possible, they cannot be sustained.
weeks. The first spontaneous movements consist of slow flexion and extension of the vertebral column, with the limbs being passively displaced. Within a short time, a large repertoire of fetal movements evolves. After study by numerous investigators, a classification of fetal movements has been suggested (Box 18.1). The first fetal movements are followed in a few days by startle and general movements. Shortly thereafter, isolated limb movements are added (Fig. 18.7). Movements associated with the head and jaw appear later. Toward the end of the fourth month, the fetus begins a pattern of periods of activity, followed by times of inactivity. Many women first become aware of fetal movements at this time. Between the fourth and fifth months, the fetus becomes capable of gripping firmly onto a glass rod. Although weak protorespiratory movements are possible, they cannot be sustained.
Fetal Digestive Tract
Fetal Period and Birth

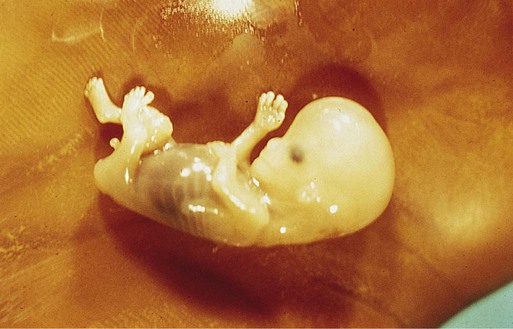
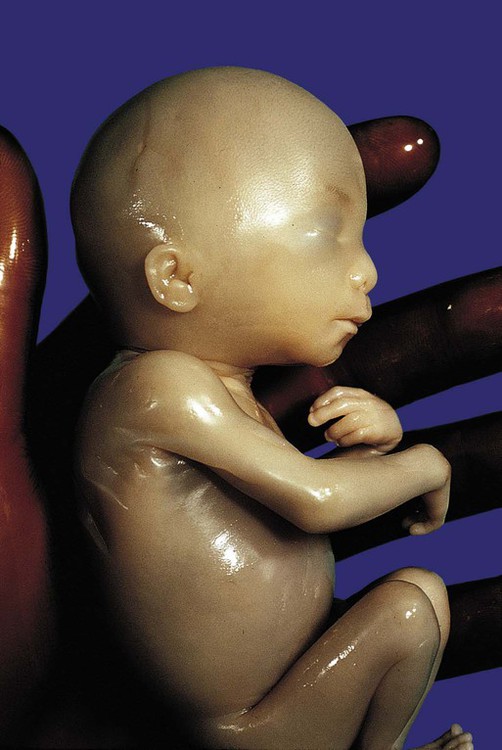
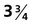 -month-old human fetus (130-mm crown-rump length).
-month-old human fetus (130-mm crown-rump length).