ENERGETICS OF INTERMEDIATE METABOLISM
To sustain life, humans must eat. The chemical free energy of food is the only form of energy humans can use to maintain the structural and biochemical integrity of the body; to perform internal work of circulation, respiration, and muscle contraction; and to perform external work (
1,
2,
3). Our ability to use the chemical free energy of food results from the development of the biochemical, structural, and physiologic apparatus that permits the transformation of chemical free energy into other energy forms essential for life. Part of the energy from food, on the order of 5%, is thermodynamically obligated for conversion to heat because the entropy of the metabolic end products is greater than the initial substances (
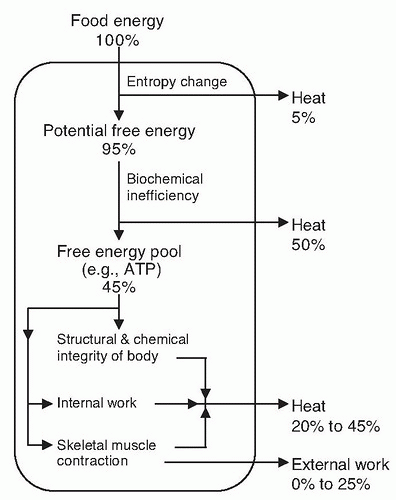
Fig. 5.1). Conversion of food energy into high-energy biochemical compounds is an inefficient process, with approximately 50% lost as heat. Through biochemical transformations, approximately 45% of the energy of food is available to the body, primarily as adenosine triphosphate (ATP). Eventually, all the energy of food is lost from the body in the form of heat or external work.
Energy is provided in the diet by protein, carbohydrate, fat, and alcohol. The energy in foods is expressed as a unit of heat, the calorie. A calorie is defined as the amount of heat required to raise the temperature of 1 g of water by 1°C from 15°C to 16°C. The scientific international unit of energy is the joule (J), defined as the energy expended when 1 kg is moved 1 m by a force of 1 newton. In 1956, an international committee standardized the equivalency of these units as 1 cal = 4.1868 J, but the figure of 4.184 is more commonly used in nutrition studies. For practicality, a kilocalorie (kcal), which is 1000 times the energy of a calorie (cal), is commonly used in nutrition. Hence, 1 kcal = 4.184 kJ, and 1 kJ = 0.0239 kcal. Another, less frequently used unit, is the thermochemical calorie, which is the heat liberated by the combustion of 1 g of pure benzoic acid and is also equivalent to 4.184 J (
1).
The potential energy contribution of food is determined experimentally by measuring the heat evolved in a bomb calorimeter when foodstuffs are completely combusted to carbon dioxide (CO
2) and water (
4). The actual amount of heat evolved per gram of foodstuff varies according to its chemical composition. Average values are 4.1 kcal/g of carbohydrate, 9.3 kcal/g of fat, and 5.4 kcal/g of protein. The body cannot oxidize nitrogen, and therefore energy resulting from the oxidation of the nitrogenous component of protein is unavailable to the body. Consequently, only 4.2 kcal/g protein is potentially available to the body. The physiologic fuel value is compromised further by the apparent digestibility of various foodstuffs that vary among food sources. These factors result in physiologic fuel values of 4 kcal/g for carbohydrate, 9 kcal/g for fat, and 4 kcal/g for protein, also known as the Atwater factors. The physiologic fuel value for alcohol is 7 kcal/g (
Table 5.1).
Substrate oxidation rates are a function of dietary macronutrient intake and level of energy turnover (
5). Protein oxidation is largely determined by protein intake, whereas
the relative contributions of glucose or free fatty acids (FFAs) to the fuel mix are more variable. Glucose oxidation is adjusted to carbohydrate intake to maintain stable glycogen stores. Fat intake, in contrast, does not promote its own oxidation, and under conditions of positive energy balance, some fat will be deposited. Most cells can use the metabolic intermediates of carbohydrates, fats, and proteins interchangeably to regenerate ATP, with a few exceptions. The brain preferentially uses glucose and is able to use ketone bodies after adaptation to starvation, but it does not use FFAs (
6). Red blood cells also depend on glucose. At rest, the brain (20%), internal organs (25% to 30%), and skeletal muscle (20%) account for the majority of energy turnover. During vigorous activity, skeletal muscle overwhelms the utilization of other tissues. In the postabsorptive state, FFAs are mainly oxidized by muscle, whereas during exertion, muscle’s own glycogen reserve is used, with a subsequent shift toward use of FFAs mobilized from muscle fat stores and adipose tissue.
When alcohol is consumed, it promptly appears in the circulation and is oxidized at a rate determined largely by its concentration and by the activity of liver alcohol dehydrogenase. Oxidation of alcohol rapidly reduces the oxidation of the other substrates used for ATP regeneration. Ethanol oxidation proceeds in large part through conversion to acetate and oxidative phosphorylation. Approximately 80% of the energy liberated by ethanol oxidation is used to drive ATP regeneration, and approximately 20% is released as heat (
7). Alcoholic beverages can contribute to weight gain in healthy persons consuming an otherwise adequate diet (
8), in contrast to the pharmacologic effect of excessive ethanol, which can inhibit normal eating and can cause emaciation in persons with alcoholism.
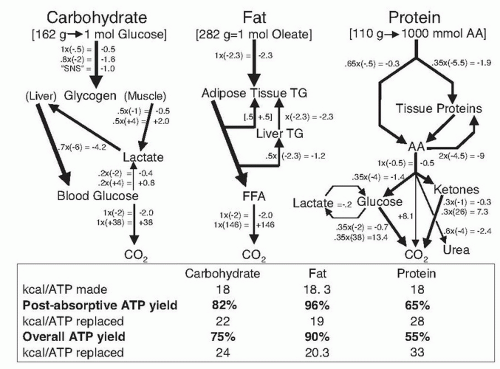
Flatt and Tremblay (
5) computed the ATP yield from oxidation of macronutrients based on the phosphorylationto- oxidation (P:O) ratio and the ATP required to initiate degradation, transport, activation, and handling of the metabolic fuels (
Fig. 5.2). Assuming a P:O ratio of 3:1 for the reoxidation of mitochondrial reduced nicotinamide adenine dinucleotide, the oxidation of 1 mol of glucose produces 38 mol ATP, but 2 mol is used for activation; therefore, the net ATP yield is 95%. Allowing for the costs of recycling through the Cori cycle, the glucose-alanine cycle and gluconeogenesis, the net postabsorptive ATP yield is approximately 82%. Accounting for the postprandial phase of digestion, absorption, and transport, the net ATP yield from dietary carbohydrate is 75%, such that the oxidation of 24 kcal of dietary carbohydrate is required to replace 1 mol of ATP. To calculate the ATP yield from dietary fat, the fatty acid oleate was used as an example. The oxidation of 1 mol of oleate yields 146 mol ATP but expends 5.5 mol ATP in lipolysis/reesterification and activation to oleylcoenzyme A; therefore, the ATP yield for fat oxidation is approximately 96%. Accounting for the postprandial phase, the net ATP yield from dietary fat is approximately 90%.
In the case of proteins, the oxidation of 1 mol of amino acids generates approximately 28.8 mol of ATP or 18 kcal/ mol ATP. The costs of gluconeogenesis, ureagenesis, and protein resynthesis reduce the net postabsorptive ATP yield to 65%. Accounting for the postprandial phase, the overall ATP yield is 55%. Based on these estimates, the transport, storage, recycling, and activation dissipate approximately 10%, 25%, and 45% of the ATP produced in the oxidation of dietary fat, carbohydrate, and protein, respectively. Therefore, the corresponding net ATP yields are estimated to be 90%, 75%, and 55% with dietary fat, carbohydrate, and protein.
Lipogenesis from the conversion of dietary carbohydrate to fat is an inefficient process estimated at 25%. This pathway appears to be of minor importance in humans, because large amounts of dietary carbohydrate expand glycogen reserves, not body fat (
9). Lipogenesis therefore does not account for the higher dissipation of dietary energy by carbohydrate compared with fat. Similarly, the energy dissipation by futile cycles or substrate cycles that dissipate ATP with no net change in the organism also appears to make only a minor contribution to overall energy economy. Futile cycles are thought to account for only a small percentage of total energy expenditure (TEE) (
10).
ENERGY BALANCE
Energy balance is the accounting for the energy consumed in foods, losses in excreta, heat produced, and retention or secretion of organic compounds (
4). Implicit in the delineation of energy balance is that energy is conserved. The energy balance may be expressed as follows:
Eintake − Efeces − Eurine − Ecombustible gas − Eexpenditure = Eretention or Esecretion
Digestible energy is the dietary energy absorbed by the gastrointestinal tract after accounting for loss in feces (
11). Metabolizable energy is that energy available to the organism after accounting for losses in feces, urine, and combustible gases. Metabolizable energy is measured by meticulous energy balance techniques and was determined for human diets by Atwater in the early 1900s. The Atwater factors of 4, 9, and 4 kcal of metabolizable energy per gram of protein, fat, and carbohydrate, respectively, are widely used to express the energy content of foods in food composition tables, including those in the United States (
12). The Atwater factors are applied to the protein estimated from its nitrogen content, fat determined by extraction, and carbohydrates determined by difference, after taking into account the water and ash in the food. In the United Kingdom, the metabolizable energy factors of 4, 9, and 3.75 kcal/g of protein, fat, and carbohydrate, respectively, are used in food composition tables (
13). In this system, the metabolizable energy factor is applied to available carbohydrate, defined as the sum of free sugars, dextrins, starch, and glycogen, and the result is lower estimates of the caloric content of foods than in the Atwater system.
Humans can survive on foods with varying proportions of carbohydrates, fats, and proteins (
14,
15,
16,
17,
18). The ability to shift from carbohydrate to fat as the main source of energy, coupled with substantial reserves of body fat, makes it possible to accommodate large fluctuations in
energy intake and energy expenditure (EE). Energy balance is regulated by a complex set of neuroendocrine feedback mechanisms. Changes in energy intake or in EE trigger metabolic and behavioral responses aimed at restoring energy balance.
MEASUREMENT OF ENERGY INTAKE AND ENERGY EXPENDITURE
Several methods are used to assess dietary intake including weighed or observed diet records, dietary recalls and diaries, and food frequencies. It is now generally recognized that reported energy intakes tend to underestimate usual energy intake (
19). Evidence of underreporting has been substantiated from measurements of TEE by the doubly labeled water (DLW) method (
20,
21). Implausibly low energy intakes have been revealed when TEE was substantially greater than reported usual energy intakes in weight-stable individuals. Underreporting of food intake is pervasive, ranging from 10% to 45% depending on the age, gender, and body composition of the study subjects (
22).
Methods for measuring EE include direct calorimetry, indirect calorimetry, and noncalorimetric methods (
23). Direct calorimetry is the measurement of the heat emitted from the body over a given period (
1,
24). A direct calorimeter chamber measures heat loss by radiation, convection, conduction, and latent heat arising from vaporization of water. Heat sink calorimeters capture the heat produced by liquid-cooled heat exchangers. Gradient layer calorimeters measure heat loss by a network of thermocouples in series surrounding the insulated chamber. Indirect calorimetry estimates heat production indirectly by measuring oxygen consumption ([V with dot above]O
2), CO
2 production (VCO
2), and the respiratory quotient (RQ), which is equal to the ratio of the VCO
2 to [V with dot above]O
2 (
25). Indirect calorimetry arose from the observations of Lavoisier and Laplace that heat production of animals as measured by calorimetry was equal to that released when organic substances are burned, and that the same quantities of oxygen were consumed by the two processes. The RQ reflects substrate utilization. The complete oxidation of glucose results in an RQ equal to 1.0. The complete oxidation of fat and protein results in an RQ averaging about 0.71 and 0.84, respectively, depending on the chemical structure of the foodstuff. Specific RQs for FFAs range from 0.69 to 0.81. RQs of amino acids range from 0.56 to 1.00, with conventional food proteins ranging from 0.81 to 0.87. In mixed diets, the RQ is approximately 0.85. Lipogenesis, the conversion of carbohydrate to fat, can substantially increase the RQ. The conversion of fat to carbohydrate, in contrast, will lower the RQ to less than 0.70.
Substrate utilization can be determined from rates of [V with dot above]O
2, VCO
2, and urinary nitrogen (
23,
25). First, gas exchange must be corrected for the incomplete oxidation of protein. One gram of urinary nitrogen represents the combustion of an amount of protein that would require 5.92 L of oxygen and produce 4.75 L of CO
2. The [V with dot above]O
2 and VCO
2 associated with the protein oxidized are subtracted from the total and are used to compute a nonprotein RQ (NPRQ). The amount of protein oxidized may be calculated directly from urinary nitrogen assuming that 1 g of nitrogen represents 6.25 g of protein. The NPRQ is then used to calculate the proportions of carbohydrate and fat oxidized when the NPRQ is less than 1.00 (
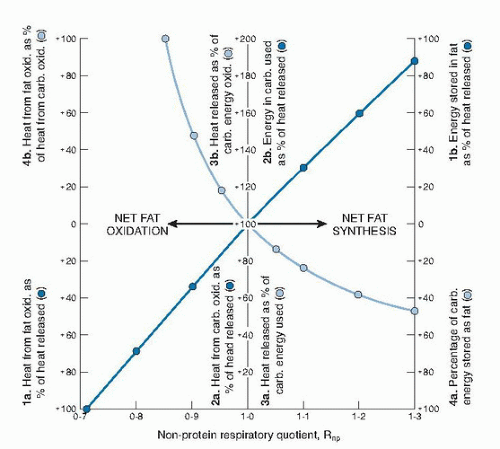
Table 5.2). When the NPRQ is greater than 1.00, net fat synthesis occurs, as shown in
Figure 5.3. When the NPRQ is greater than 1.00, carbohydrate is used both for storage of energy and for oxidation (
25).
Weir (
26) demonstrated that the error in neglecting the effect of protein metabolism on the caloric equivalent of oxygen is 1% for each 12.3% of the total calories that arise from protein. The most widely used equation for the calculation of total heat output is by Weir:
EE (kcal) = 3.941 × [V with dot above]O2 (L) + 1.106 VCO2 (L) − (2.17 × UrN [g]) or EE (kcal) = 3.941 × [V with dot above]O2 (L) + 1 VCO2 (L)/(1 + 0.082 p)
where UrN is urinary nitrogen and p is the fraction of calories resulting from protein. Assuming approximately 12.5% of total calories will arise from protein; therefore, the foregoing equation can be reduced to the following:
EE (kcal) = 3.9 × [V with dot above]O2 (L) + 1.1 VCO2 (L)
Whole body respiratory calorimeters are small rooms in which the subject may reside comfortably for longer periods unencumbered by respiratory gas collection devices. In these rooms, the concentrations of oxygen (O
2) and CO
2 and airflow through the system are monitored continuously. Whole body calorimeters provide a controlled experimental environment to measure TEE and its components. Portable indirect calorimetric systems also have been devised to measure EE in field, clinical, and laboratory settings (
27,
28,
29). The Douglas bag method has been used historically for many measurements of basal and resting metabolism. In this method, all expired air is collected into a nonpermeable bag with a capacity up to 150 L. After a known period, the volume of expired air at standard temperature and pressure dried, and the concentrations of O
2 and CO
2 are measured from which [V with dot above]O
2 and VCO
2 and RQ are calculated. Commercial metabolic carts in laboratory and clinical settings have largely replaced the Douglas bag method. For field measurements, several portable respirometers have been designed with oxygen analyzers as well as gas flow meters and electronics to process and store data. These systems require airtight masks, breathing valves, and nose clips to measure respiratory gas exchange quantitatively. Hood devices and canopies have been designed for more comfort, but they restrict movement.
Other methods to assess EE applicable to field conditions include heart rate (HR) monitoring and DLW. The HR monitoring method is based on the linear relationship between HR and EE (
30). Because of variations resulting from age, sex, body size, fitness, and nutritional status, the relationship must be calibrated on an individual basis. Simultaneous measurements of EE and HR in subjects are performed across a range of activities to calibrate the individual. Other confounding factors such as ambient conditions, time of day, emotional state, hydration status, food and caffeine intake, and smoking may influence the EE:HR relationship. As a result, HR data on individuals are subject to error and may yield unreliable estimates of EE. When applied to groups of individuals, the HR monitoring method provides an acceptable estimate of TEE.
DLW is a stable (nonradioactive) isotope method that provides an estimate of TEE in free-living individuals. The DLW method was originally developed by Lifson et al for use in small animals (
31,
32), and it was later adapted for humans (
33,
34). Two stable isotopic forms of water (H
218O and
2H
2O) are administered to the individual, and their
18O and
2H disappearance rates from the body are monitored for 7 to 21 days, equivalent to one to three halflives for these isotopes. The disappearance rate of
2H
2O reflects water flux, whereas that of H
218O reflects water
flux plus VCO
2, because of the rapid equilibration of the body water and bicarbonate pools by carbonic anhydrase. The difference between the two disappearance rates is used to calculate VCO
2. Assuming an RQ, [V with dot above]O
2 and, hence, EE are calculated. When energy balance prevails, the average RQ may be estimated from the composition of the diet by using the food quotient (
35). If substantial gains or losses of body constituents are known to occur during the period of measurement, appropriate adjustments must be made in estimating the RQ. Under field conditions, this method is accurate within 5% or better. The advantage of this technique is the noninvasive, nonintrusive manner in which it measures TEE. In weight-stable individuals, the DLW method may be used to assess energy requirements. The disadvantages of the method are the high cost of
18O and expensive, sophisticated mass spectrometric equipment and the expertise required to measure
18O and
2H.