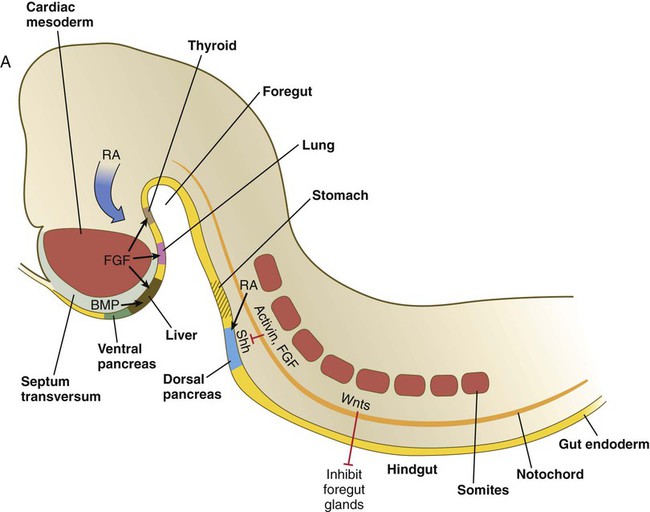
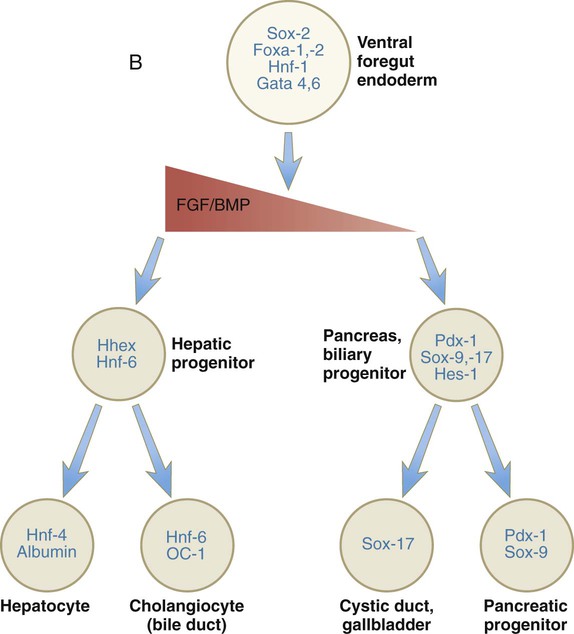
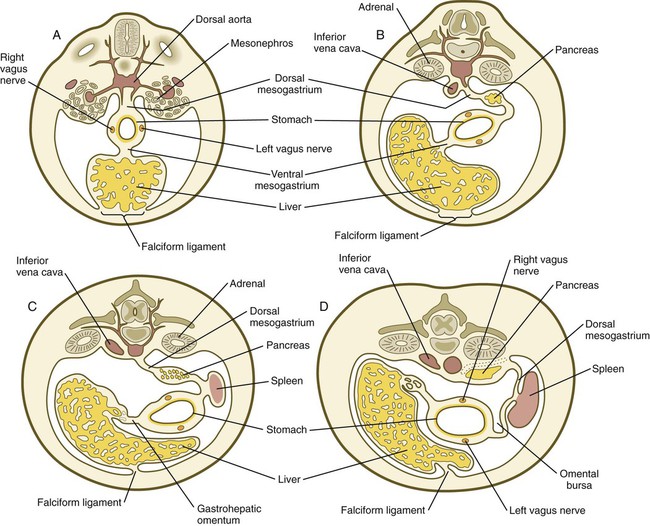
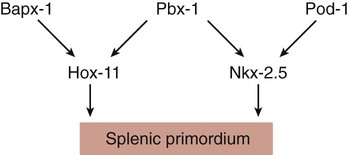
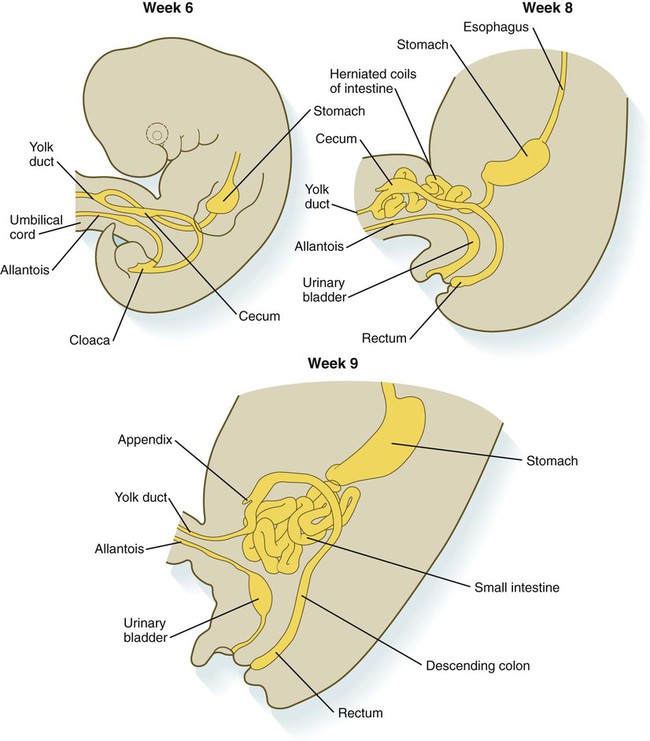
Chapter 15 The initial formation of the digestive system by the lateral folding of the endodermal germ layer into a tube is described in Chapter 6. From its beginnings as a simple tubular gut, development of the digestive system proceeds on several levels, including molecular patterning, elongation and morphogenesis of the digestive tube itself, inductions and tissue interactions leading to the formation of the digestive glands, and the biochemical maturation of the secretory and absorptive epithelia associated with the digestive tract. Clinical Correlations 15.1 to 15.3, later in the chapter, discuss malformations associated with the digestive system. Formation of the respiratory system begins with a very unimposing ventral outpocketing of the foregut. Soon, however, this outpocketing embarks on a unique course of development while still following some of the basic patterns of epithelial-mesenchymal interactions characteristic of other gut-associated glands. Initially, the digestive and respiratory systems form in a common body cavity, but functional considerations later necessitate the subdivision of this primitive body cavity into thoracic and abdominal components. Later in the chapter, Clinical Correlation 15.4 presents malformations associated with the respiratory system, and Clinical Correlation 15.5 discusses malformations related to other body cavities. Chapter 6 describes the formation of the primitive endodermal digestive tube, which is bounded at its cephalic end by the oropharyngeal membrane and at its caudal end by the cloacal plate (see Fig. 6.20). Because of its intimate relationship with the yolk sac through the yolk stalk, the gut can be divided into a foregut, an open-bottomed midgut, and a hindgut. Patterning of the broad foregut area occurs through the inhibition of Wnt signals (Fig. 15.1). The foregut domain is then marked by the expression of the transcription factors, Sox-2, Hhex, and Foxa-2. In contrast, a mix of activity by Wnts, FGFs, and bone morphogenetic proteins (BMPs), along with retinoic acid, represses foregut identity and maintains the regional identity of the hindgut. This is marked by the expression of the transcription factor Cdx-2 throughout the broad hindgut and the later expression of Pdx-1 in the midgut as this region emerges as a separate entity. Cdx-2 acts upstream of a broad range of Hox activity (Fig. 15.2) that is expressed throughout the gut. The activity of specific signaling molecules is associated with important transition points along the gut. FGF-4 is strongly expressed near the foregut-midgut boundary (around the duodenal-jejunal junction), and FGF-10 is associated with the establishment of the cecum. Largely through the action of Cdx-2, the orderly expression of homeobox-containing genes then takes over in the regional patterning of the digestive system (see Fig. 15.2). By the end of the first month, small endodermal diverticula, which represent primordia of the major digestive glands, can be identified (Fig. 15.3). (Development of the pharynx and its glandular derivatives is discussed in Chapter 14.) The digestive glands and respiratory structures grow in complex branching patterns, resembling fractals, as the result of continuous epithelial-mesenchymal interactions. These interactions also occur in the developing digestive tube itself, with specific regional mesenchymal influences determining the character of the epithelium lining that part of the digestive tract. During neurulation, as the head bends sharply to create the foregut, the ventral foregut endoderm is closely opposed to two mesodermal masses: the cardiac mesoderm and the primordium of the septum transversum (see Fig. 15.4). High levels of FGF, secreted by the cardiac mesoderm, and also retinoic acid induce the formation of the liver, lung bud, and thyroid (see Fig. 15.4). BMP-4 from the mesoderm of the septum transversum is also required for liver induction. By contrast, endodermal movements carry the preventral pancreas cells far enough from the cardiac mesoderm to expose them to a low level of FGF, thus allowing the ventral pancreas to develop. For the dorsal pancreas to develop, locally produced sonic hedgehog (shh) must be inactivated by activin and FGF emanating from the notochord. In addition, retinoic acid from the somitic mesoderm is needed for induction of the dorsal pancreas. Meanwhile, in the hindgut, the actions of Wnt and other signaling molecules repress the expression of genes, such as Hhex and Pdx1, which are essential for formation of the liver and pancreas, respectively. Induction of these organs is marked by the activation of transcription factors specific for the organ and stage of development of that organ. Some of these factors are schematically represented in Figure 15.4B. Just caudal to the most posterior pharyngeal pouches of a 4-week-old embryo, the pharynx becomes abruptly narrowed, and a small ventral outgrowth (lung bud) appears (see Fig. 6.20). The region of foregut just caudal to the lung bud is the esophagus. This segment is initially very short, with the stomach seeming to reach almost to the pharynx. In the second month of development, during which the gut elongates considerably, the esophagus assumes nearly postnatal proportions in relation to the location of the stomach. Although the esophagus grossly resembles a simple tube, it undergoes a series of striking differentiative changes at the tissue level. In its earliest stages, the endodermal lining epithelium of the esophagus is stratified columnar. By 8 weeks, the epithelium has partially occluded the lumen of the esophagus, and large vacuoles appear (Fig. 15.5). In succeeding weeks, the vacuoles coalesce, and the esophageal lumen recanalizes, but with multilayered ciliated epithelium. During the fourth month, this epithelium finally is replaced with the stratified squamous epithelium that characterizes the mature esophagus. The cross-sectional structure of the esophagus, similar to that of the rest of the gut, is organized into discrete layers. The innermost layer (mucosa) consists of the epithelium, derived from endoderm, and an underlying layer of connective tissue, the lamina propria (see Fig. 15.5C and D). A thick layer of loose connective tissue (submucosa) separates the mucosa from the outer layers of muscle (usually smooth muscle, with the exception of the upper esophagus). This radial organization is regulated by the epithelial expression of shh, acting through its receptor, patched, and BMP-4. Shh inhibits the formation of smooth muscle in the submucosal layer of the esophagus. Farther from the source of the endodermal shh, smooth muscle can differentiate in the outer wall of the intestine. How the developing smooth muscle layer of the mucosa (muscularis mucosae) escapes this inhibitory influence is also unclear. Intestinal mesenchyme can spontaneously differentiate into smooth muscle in the absence of an epithelium (which produces shh). Because in humans the muscularis mucosae differentiates considerably later than the outer muscular layers, it is possible that inhibitory levels of shh are reduced by that time. Very early in the formation of the digestive tract, the stomach is recognizable as a dilated region with a shape remarkably similar to that of the adult stomach (see Fig. 15.3). The early stomach is suspended from the dorsal body wall by a portion of the dorsal mesentery called the dorsal mesogastrium. It is connected to the ventral body wall by a ventral mesentery that also encloses the developing liver (Fig. 15.6). When the stomach first appears, its concave border faces ventrally, and its convex border faces dorsally. Two concomitant positional shifts bring the stomach to its adult configuration. The first is an approximately 90-degree rotation about its craniocaudal axis so that its originally dorsal convex border faces left, and its ventral concave border faces right. The other positional shift consists of a minor tipping of the caudal (pyloric) end of the stomach in a cranial direction so that the long axis of the stomach is positioned diagonally across the body (Fig. 15.7). During rotation of the stomach, the dorsal mesogastrium is carried with it, thus leading to the formation of a pouchlike structure called the omental bursa (bursa is a Latin word meaning “sac” or “pouch”). Both the spleen and the tail of the pancreas are embedded in the dorsal mesogastrium (see Fig. 15.6). Another viewpoint suggests that the right pneumatoenteric recess, a projection from the pleural cavity into the dorsal mesogastrium, persists as the omental bursa. As the stomach rotates, the dorsal mesogastrium and the omental bursa that it encloses enlarge dramatically. Soon, part of the dorsal mesogastrium, which becomes the greater omentum, overhangs the transverse colon and portions of the small intestines as a large, double flap of fatty tissue (Fig. 15.8). The two sides of the greater omentum ultimately fuse and obliterate the omental bursa within the greater omentum. The rapidly enlarging liver occupies an increasingly large portion of the ventral mesentery. Clinical Correlation 15.1 presents malformations of the esophagus and stomach. Development of the spleen is not well understood. Initially, two bilaterally symmetrical organ fields are reduced by regression to one on the left. The spleen is first recognizable as a mesenchymal condensation in the dorsal mesogastrium at 4 weeks and is initially closely associated with the developing dorsal pancreas. Initiation of splenic development requires the cooperative action of a basic helix-loop-helix protein (Pod-1) and a homeobox-containing protein (Bapx-1), acting through another transcription factor, Pbx-1. Such a combination is emerging as a common theme in the initiation of development of several organs. These substances act on the downstream molecules Nkx 2.5 and the oncogene Hox-11 (T-cell leukemia homeobox-1) in early splenic development (Fig. 15.9). The intestines are formed from the posterior part of the foregut, the midgut, and the hindgut (Table 15.1). Table 15.2 summarizes the chronology of major stages in the development of the digestive tract. Two points of reference are useful in understanding the gross transformation of the primitive gut tube from a relatively straight cylinder to the complex folded arrangement characteristic of the adult intestinal tract. The first is the yolk stalk, which extends from the floor of the midgut to the yolk sac. In the adult, the site of attachment of the yolk stalk is on the small intestine about 2 feet cranial to the junction between small and large intestine (ileocecal junction). On the dorsal side of the primitive gut, an unpaired ventral branch of the aorta, the superior mesenteric artery, and its branches feed the midgut (see Fig. 15.7). The superior mesenteric artery itself serves as a pivot point about which later rotation of the gut occurs. Table 15.1 Derivatives of Regions of the Primitive Gut Table 15.2 Timelines in Development of the Digestive System By 5 weeks, rapid growth of the gut tube causes it to buckle out in a hairpinlike loop. The growth in length results in large part from the effect of FGF-9, which is produced by the epithelium and stimulates proliferation of the fibroblasts in the intestinal walls. The major change that causes the intestines to assume their adult positions is a counterclockwise rotation of the caudal limb of the intestinal loop (with the yolk stalk attachment and superior mesenteric artery as reference points) around the cephalic limb from its ventral aspect. The main consequence of this rotation is to bring the future colon across the small intestine so that it can readily assume its C-shaped position along the ventral abdominal wall (see Fig. 15.7). Behind the colon, the small intestine undergoes great elongation and becomes packed in its characteristic position in the abdominal cavity. The rotation and other positional changes of the gut occur partly because the length of the gut increases more than the length of the embryo. From almost the first stages, the volume of the expanded gut tract is greater than the body cavity can accommodate. Consequently, the developing intestines herniate into the body stalk (the umbilical cord after further development) (Fig. 15.11). Intestinal herniation begins by 6 or 7 weeks of embryogenesis. By 9 weeks, the abdominal cavity has enlarged sufficiently to accommodate the intestinal tract, and the herniated intestinal loops begin to move through the intestinal ring back into the abdominal cavity. Coils of small intestine return first. As they do, they force the distal part of the colon, which was never herniated, to the left side of the peritoneal cavity, thus establishing the definitive position of the descending colon. After the small intestine has assumed its intra-abdominal position, the herniated proximal part of the colon also returns, with its cecal end swinging to the right and downward (see Fig. 15.7). During these coilings, herniations, and return movements, the intestines are suspended from the dorsal body wall by a mesentery (Fig. 15.12). Experimentation has shown that looping of the intestine is caused principally by tension-compression relationships between the intestine and its dorsal mesentery. When the intestine is separated from the mesentery, the normal looping does not occur. As the intestines assume their definitive positions within the body cavity, their mesenteries follow. Parts of the mesentery associated with the duodenum and colon (mesoduodenum and mesocolon) fuse with the peritoneal lining of the dorsal body wall. Starting in the sixth week, the primordium of the cecum becomes apparent as a swelling in the caudal limb of the midgut (see Fig. 15.7). In succeeding weeks, the cecal enlargement becomes so prominent that the distal small intestine enters the colon at a right angle. The sphincterlike boundary at the cecum between the small and large intestines, similar to that in other regions of the gut, is regulated by a high concentration of Cdx-2 and a sequence of Hox gene expression. In mice, deletion of Hoxd4, Hoxd8 to Hoxd11, and Hoxd13 results in the absence of this region. When the overall pattern has been set by combinations of Hox genes, cecal development depends on an interaction between FGF-9 produced by the cecal epithelium and FGF-10 produced by the overlying mesoderm. In the early embryo, the caudal end of the hindgut terminates in the endodermally lined cloaca, which, in lower vertebrates, serves as a common termination for the digestive and urogenital systems. The cloaca also includes the base of the allantois, which later expands as a common urogenital sinus (see Chapter 16). A cloacal (proctodeal) membrane consisting of apposed layers of ectoderm and endoderm acts as a barrier between the cloaca and an ectodermal depression known as the proctodeum (Fig. 15.13). A shelf of mesodermal tissue called the urorectal septum is situated between the hindgut and the base of the allantois. During weeks 6 and 7, the urorectal septum advances toward the cloacal membrane. At the same time, lateral mesodermal ridges extend into the cloaca. The combined ingrowth of the lateral ridges and growth of the urorectal septum toward the cloacal membrane divide the cloaca into the rectum and urogenital sinus (see Fig. 15.13B). Double mutants of Hoxa13 and Hoxd13 result in the absence of cloacal partitioning, along with hypodevelopment of the phallus (genital tubercle). In addition, they lead to the absence of the smooth muscle component of the anal sphincter. According to classic embryology, the urorectal septum fuses with the cloacal membrane, thus dividing it into an anal membrane and a urogenital membrane before these membranes break down (see Fig. 15.13C). Other research suggests that the cloacal membrane undergoes apoptosis and breaks down without its fusion with the urorectal septum. The area where the urorectal septum and lateral mesodermal folds fuse with the cloacal membrane becomes the perineal body, which represents the partition between the digestive and urogenital systems. With the formation of villi, pitlike intestinal crypts form at the bases of the villi. Toward the bottom of the crypts are epithelial intestinal stem cells, which, in response to Wnt signaling, have a high rate of mitosis and serve as the source of epithelial cells for the entire intestinal surface (Fig. 15.14). Despite the presence of four to six stem cells per crypt, it has been shown that each crypt is monoclonal (i.e., all the existing cells are descendants of a single stem cell from earlier in development). Toward the top of a crypt, shh and Indian hedgehog (Ihh) signaling stimulates the activity of BMP. This BMP has two main functions. It counteracts the effects of Wnt and thus keeps proliferation deep in the crypt, and it also facilitates cellular differentiation. Aided in part by an ephrin-eph gradient, progeny of the stem cells make their way up the wall of the crypt as multipotential transit amplifying cells, which, under the influence of the Delta-Notch system, begin to differentiate into the four main mature cell types of the intestinal epithelium (see Fig. 15.14). These cells then both differentiate and migrate toward the tip of the villus, until after about 4 days they die and are shed into the intestinal lumen as they are replaced below by new epithelial cells derived from the crypts. Human intestinal epithelial cells develop the intrinsic capacity for apoptosis by 18 to 20 weeks of gestation. Histodifferentiation of the intestinal tract is not an isolated property of the individual tissue components of the intestinal wall. During the early embryonic period and sometimes into postnatal life, the epithelial and mesodermal components of the intestinal wall communicate by inductive interactions. In region-specific manners, these interactions involve hedgehog signaling (shh for the foregut and midgut and Ihh for the hindgut) from the endodermal epithelium. BMP signaling from the mesoderm is involved in positioning the crypts and villi in the small intestine and the glands of the colon. Interspecies recombination experiments show that the gut mesoderm exerts a regional influence on intestinal epithelial differentiation (e.g., whether the epithelium differentiates into a duodenal or colonic phenotype). When regional determination is set, however, the controls for biochemical differentiation of the epithelium are inherent. This pattern of inductive influence and the epithelial reaction are similar to those outlined earlier for dermal-epidermal interactions in the developing skin (see Chapter 9). Although the intestine develops many functional capabilities during the fetal period, no major digestive function occurs until feeding begins after birth. The intestines of the fetus contain a greenish material called meconium (see Fig. 18.9), which is a mixture of lanugo hairs and vernix caseosa sloughed from the skin, desquamated cells from the gut, bile secretion, and other materials swallowed with the amniotic fluid. As outlined in Chapter 12, the enteric ganglia of the gut are derived from neural crest. Pax-3–expressing cells from the vagal neural crest migrate into the foregut and spread in a wavelike fashion throughout the entire length of the gut. Slightly later, cells from the sacral crest enter the hindgut and intermingle with cells derived from the vagal neural crest. The migratory properties of vagal crest cells are much more pronounced than are those of cells from the sacral crest. Initially, the vagal crest cells migrate throughout the mesenchyme of the gut, but as the smooth muscle of the intestines begins to differentiate, the migrating vagal crest cells become preferentially distributed between the smooth muscle and the serosa, where the myenteric plexuses form. They are absent from the connective tissue of the submucosa because of the inhibiting effects of shh, secreted by the epithelial cells. During migration through the gut, the population of neural crest cells undergoes a massive expansion until the number of enteric neurons ultimately exceeds the number of neurons present in the spinal cord. Glial cells also differentiate from neural crest precursors in the gut, but the environmental factors that contribute to the differentiation of neural crest cells in the gut wall remain poorly understood.
Digestive and Respiratory Systems and Body Cavities
Digestive System
Patterning the Gut
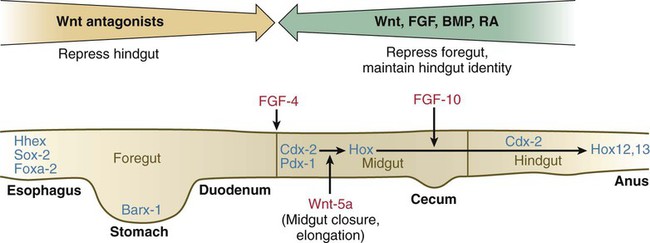
See text for details. Red letters, signaling molecules; blue letters, transcription factors.
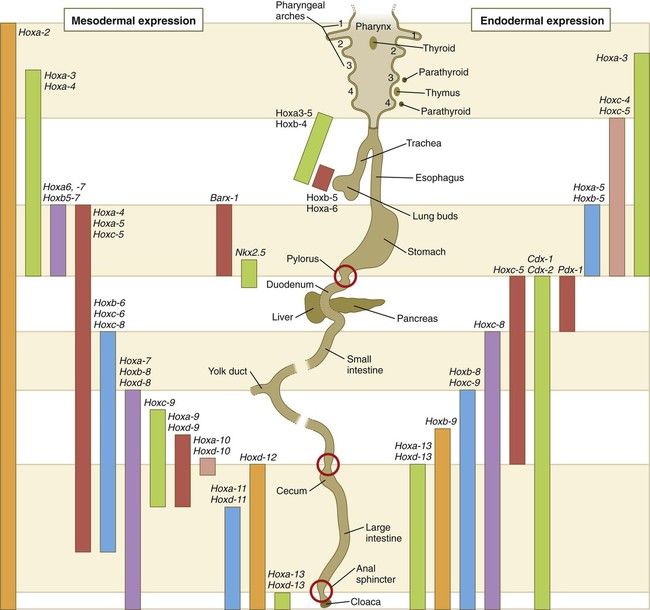
Expression along the gut endoderm (right) and in the gut-associated mesoderm (left). The circles represent areas where sphincters are located.
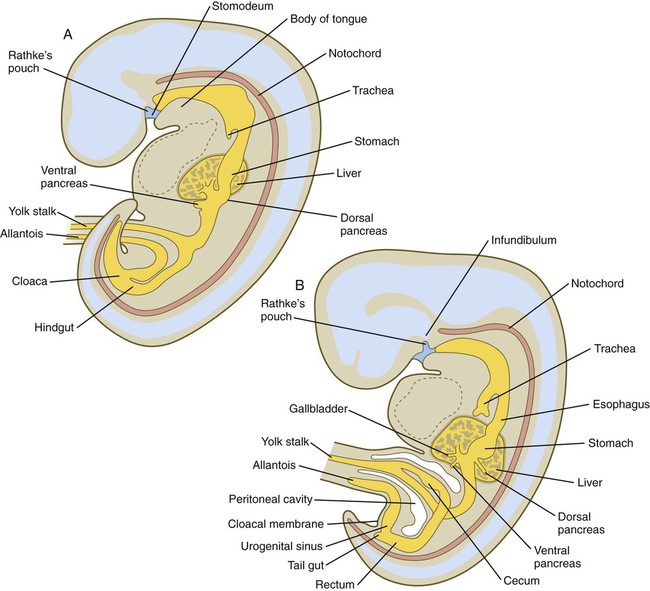
A, Early in the fifth week. B, Early in the sixth week.
Formation of the Esophagus
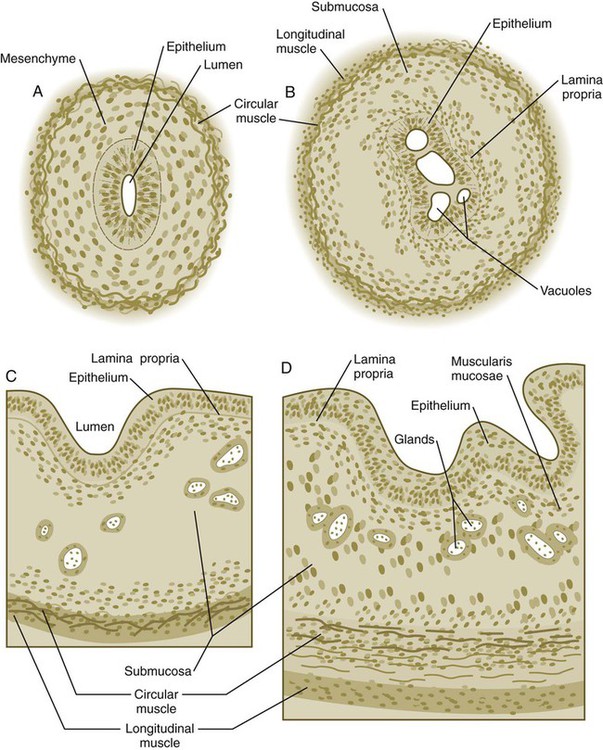
A, At 7 weeks. B, At 8 weeks. C, At 12 weeks. D, At 34 weeks.
Formation of the Stomach
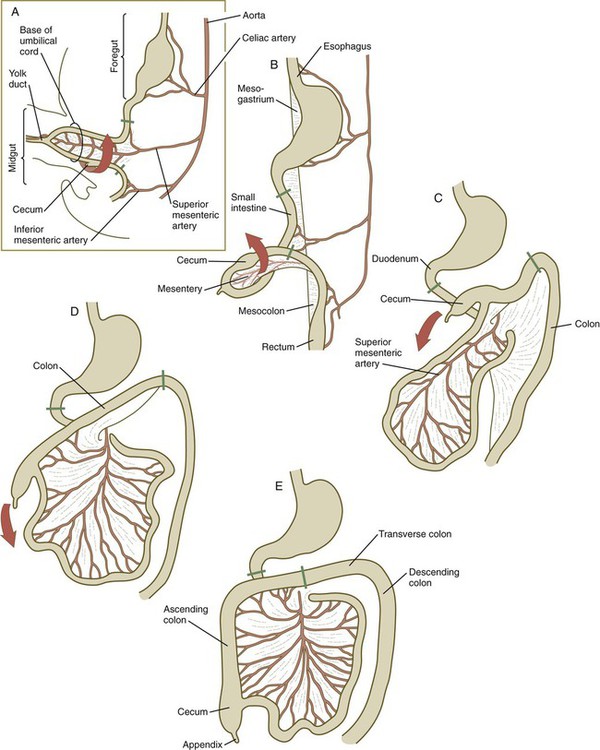
A, At 5 weeks. B, At 6 weeks. C, At 11 weeks. D, At 12 weeks. E, Fetal period. Areas between the green lines represent the midgut, which is supplied by the superior mesenteric artery.
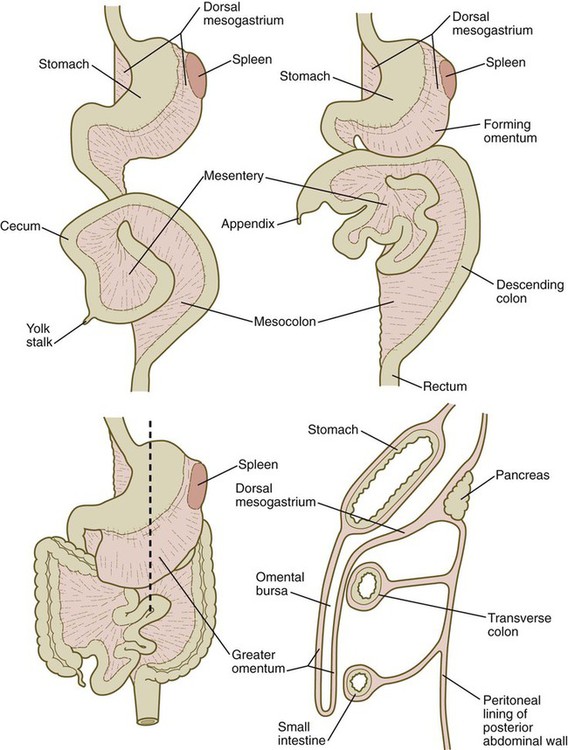
A section through the level of the dashed line (lower left) is shown (lower right).
Development of the Spleen
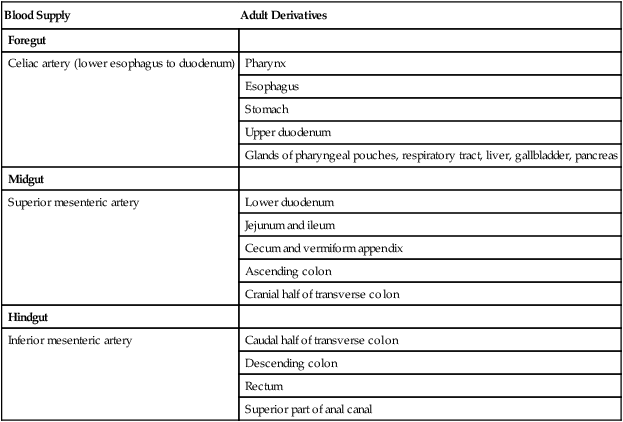
Formation of the Intestines
Blood Supply
Adult Derivatives
Foregut
Celiac artery (lower esophagus to duodenum)
Pharynx
Esophagus
Stomach
Upper duodenum
Glands of pharyngeal pouches, respiratory tract, liver, gallbladder, pancreas
Midgut
Superior mesenteric artery
Lower duodenum
Jejunum and ileum
Cecum and vermiform appendix
Ascending colon
Cranial half of transverse colon
Hindgut
Inferior mesenteric artery
Caudal half of transverse colon
Descending colon
Rectum
Superior part of anal canal
Normal Time (wk)
Developmental Events
3
Tubular gut beginning to form; early induction of major digestive glands
4
Most of gut tubular; primordia of liver, dorsal and ventral pancreas, and trachea visible; rupture of oropharyngeal membrane
5
Expansion and early rotation of stomach; intestinal loop beginning to form; cecum and bile duct evident
6
Rotation of stomach completed, prominent intestinal loop; appearance of allantois and appendix; urorectal septum beginning to subdivide cloaca into rectum and urogenital sinus
7
Herniation of intestinal loop; rapid growth of liver; fusion of dorsal and ventral pancreas; cloacal septation complete
8
Counterclockwise rotation of herniated intestinal loop; recanalization of intestine; early penetration of parasympathetic neuronal precursors from cranial neural crest into gut
9
Return of herniated gut into body cavity; differentiation of epithelial types in intestinal lining
11
Villi appearing in small intestine; differentiation of goblet cells
16
Villi lining entire intestine (including colon)
20
Peyer’s patches seen in small intestine
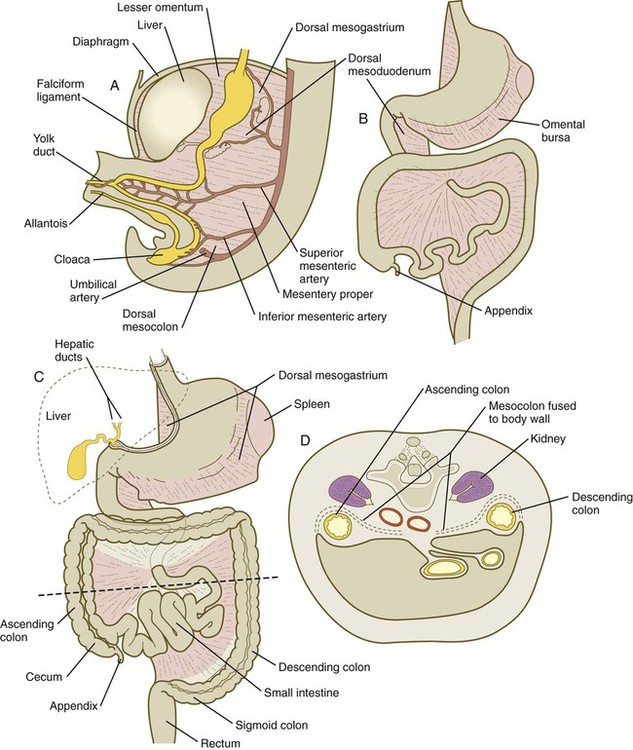
A, At 5 weeks. B, In the third month. C, During the late fetal period. D, Cross section through the dashed line in C. In C, the shaded areas represent regions where the mesentery is fused to the dorsal body wall.
Partitioning of the Cloaca
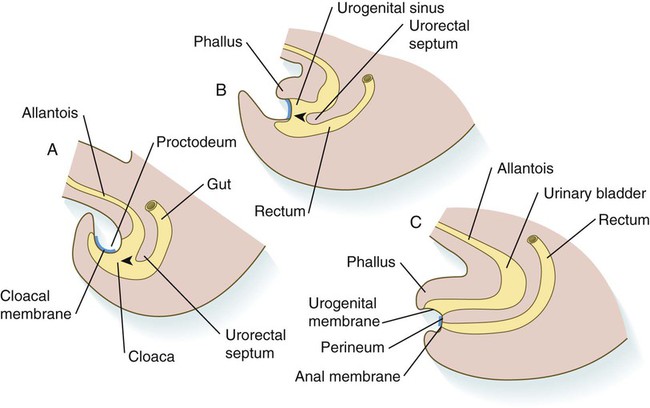
A, In the fifth week. B, In the sixth week. C, In the eighth week. The arrowheads indicate the direction of growth of the urorectal septum.
Histogenesis of the Intestinal Tract
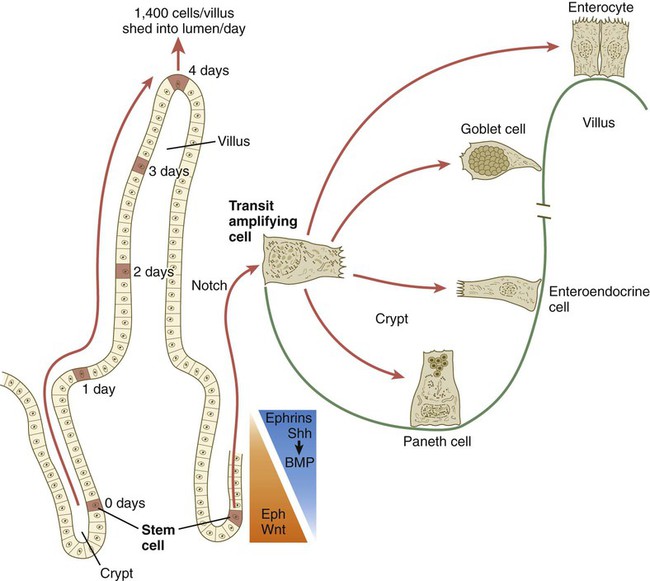
The time scale shows the typical course of migration of daughter cells starting with their generation from the stem cell population to their being shed from the villus into the intestinal lumen.
Formation of Enteric Ganglia
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Digestive and Respiratory Systems and Body Cavities